Abstract
Integrins are heterodimeric transmembrane receptors, which are expressed in many cells. In vitro experiments have demonstrated that integrins may be important in tumor progression and organ development. The functions of integrins were previously studied in cell cultures and their tissue expression was detected by immunofluorescence or immunoperoxidase in frozen sections. The purpose of this study was to determine the optimal conditions for detection of integrins in formalin-fixed, paraffin-embedded tissues. We utilized microwave heating and enzyme digestion in routinely processed, surgically removed tissues. Our results demonstrate that integrins can be reliably detected in archival material. This approach will facilitate further investigation of the role played by integrins in human malignancies and in developmental processes.
I
Materials and Methods
Antibodies
We have used monoclonal or polyclonal antibodies (Abs) to integrins to include Abs to common β1-chain, several α-integrins, β5, and the αvβ3 complex. Abs identifying integrins that bind primarily basement membrane components (α2, β1, α3, α6) or primarily extracellular matrix proteins (αvβ3, β5) were assayed. These Abs were chosen because in vitro experiments have suggested that integrins with basement membrane or matrix binding specifies are involved in cancer cell invasion and metastases. The integrin specificity and sources of Abs are shown in Table 1.
Tissues
Surgically removed specimens accessioned in our pathology laboratory were used. Non-neoplastic breast (n = 4), normal kidney (
Immunohistochemistry on Formalin-fixed Tissues
All Abs were first tested and titrated in frozen sections. Subsequently, the dilution with the best signal-to-noise ratio was selected and adapted for the formalin-fixed, paraffin-embedded sections from the same tissue. For each experimental slide and for each Ab, a negative control was included in which the primary Ab was omitted during the assay. Immunohistochemistry on frozen sections was performed as previously described (Zutter et al. 1990). For the formalin-fixed sections a modified protocol was applied as described below for the manual and our automated immunohistochemistry system (Microprobe, Fisher). The Microprobe system consists of an incubation chamber (constantly heated at 37C), slide holders, and dishes for washing and counterstaining. Its operation is based on capillary action, for which special wells are provided to be filled with reagents, i.e., Abs or wash buffers, and counterstaining dyes. The slide holders accommodate 10 pairs of slides, which are loaded with tissues facing each other. The slide holders are vertically placed on reagent-containing wells, which are drawn upwards by capillary action.
Slides deparaffinized in xylene and hydrated through 100% and 95% alcohol were placed in a solution of methanol containing 30% hydrogen peroxide in equal volumes (50:50) for 20 min at room temperature (RT) to block endogenous peroxidase. Slides were then thoroughly rinsed in distilled water and then digested with ficin [1:50 dilution of the 2 X suspension preparation from Sigma Chemical (St Louis, MO)] for 20 min at 37C, or slides were placed in Coplin jars filled with citrate buffer (0.01 M, pH 6). Coplin jars were capped with a vented plastic lid and microwaved (Kenmore microwave) for antigen retrieval on full power twice for 3.5-5 min, followed by 20-min incubation each time at RT (Shi et al. 1991). The microwave (MW) temperature was checked by thermometer and was consistently 100C. In some experiments, microwaving was repeated for two additional cycles of 3.5-5 min each. Then slides were placed in PBS. In separate experiments, slides digested with ficin were subsequently microwaved or Ab was applied for 2 hr or overnight without MW heating or ficin pretreatment. For polyclonal Abs, the tissues were blocked with 10% normal serum for 20 min and rinsed before the Ab was applied. Manually processed slides were incubated with 100-150 μl of diluted Ab for 2 hr at RT. Enough Ab solution was added to the slides to cover the entire tissue. Negative controls were placed in PBS. For the Microprobe apparatus, 125 μl of Ab was placed on wells provided by the manufacturer (see above) and drawn upwards by capillary action. Deparaffinized and hydrated slides as described above were digested with ficin or subjected to MW heating before they were loaded on the microprobe. Slides were incubated with Ab for 30 min (at 37C). After the Ab incubation, slide holders were vertically placed on absorbent towels and the solution was removed. Subsequently, slides loaded on slide holders were washed four times with PBS (drawn upwards by capillary action as described above). Secondary Ab (Dako, Glostrup, Denmark; LSAB kit) was applied for 20 min at 37C in the same manner, rinsed in PBS, and followed by label antibody (Dako, LSAB kit) for 20 min and rinsed. Color development was achieved with either diaminobenzidine (DAB) substrate (LSAB kit; Dakopatts, Carpinteria, CA) or alkaline phosphatase substrate kit (Vectastain, Vector Red; Vector Laboratories, Burlingame, CA).
Integrin immunoreactivity in formalin-fixed, paraffin-embedded tissues a
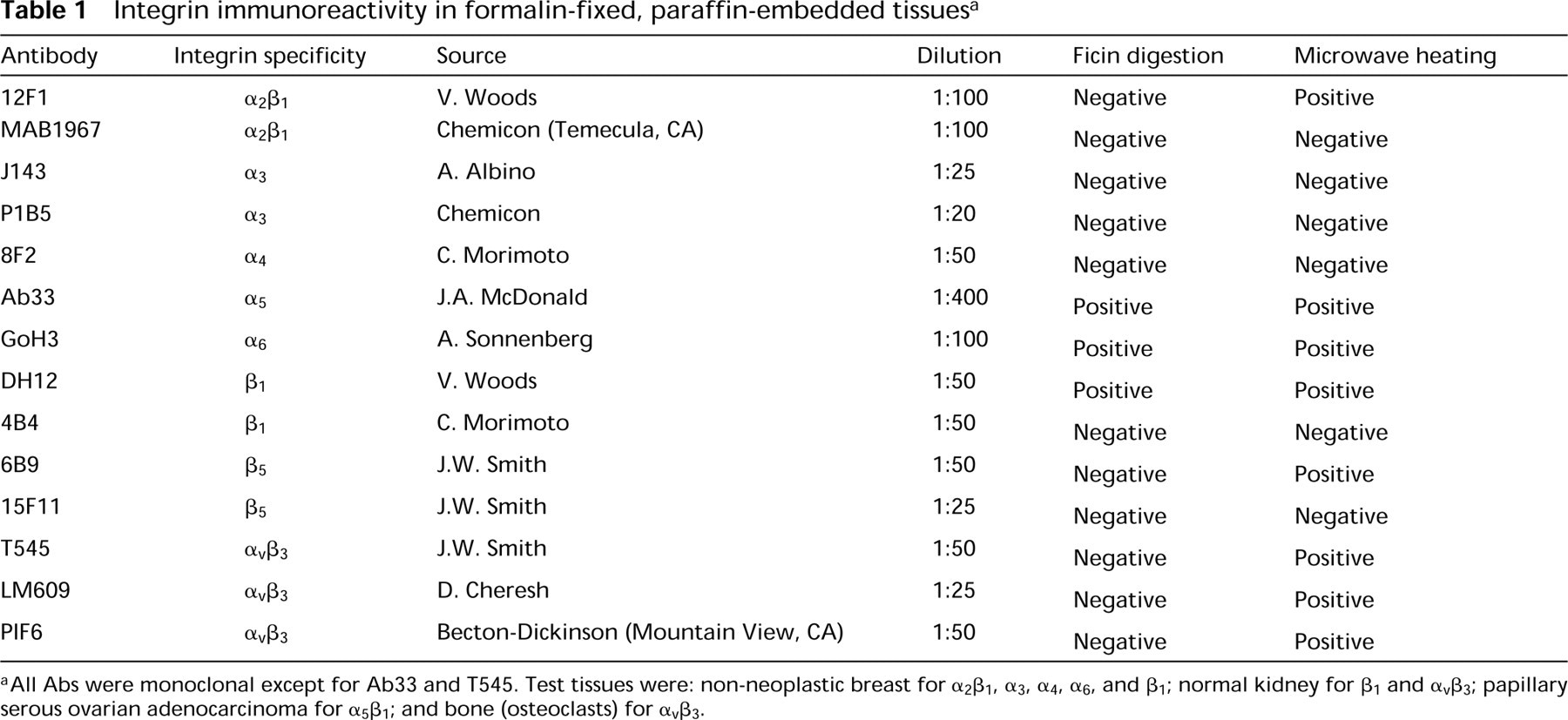
aAll Abs were monoclonal except for Ab33 and T545. Test tissues were: non-neoplastic breast for α2β1, α3, α4, α6, and β1; normal kidney for β1 and αvβ3; papillary serous ovarian adenocarcinoma for α5β1; and bone (osteoclasts) for αvβ3.
(
All experiments were preferably performed on the Microprobe. Positive results were consistent with MW heating when the automated system was used, whereas manual assays at RT were less often successful.
Results
More than half of the Abs we used (8/14) successfully detected integrins in formalin-fixed, paraffin-embedded tissue (Table 1). With the exception of three Abs (Ab33, GoH3, and DH12), integrin detection was achieved only through MW heating before incubation with the primary antibody. GoH3, Ab 33, and DH12 Abs gave equally good detection signal with ficin compared to MW heating. Combination of both ficin and MW treatment did not improve signal detection with any of the Abs used in this study. Experiments without MW or enzyme digestion carried out at RT for 2 hr or overnight at 4C were not successful. Doubling the MW cycles (total of four 3.5-5 min heating cycles) did not prove helpful.
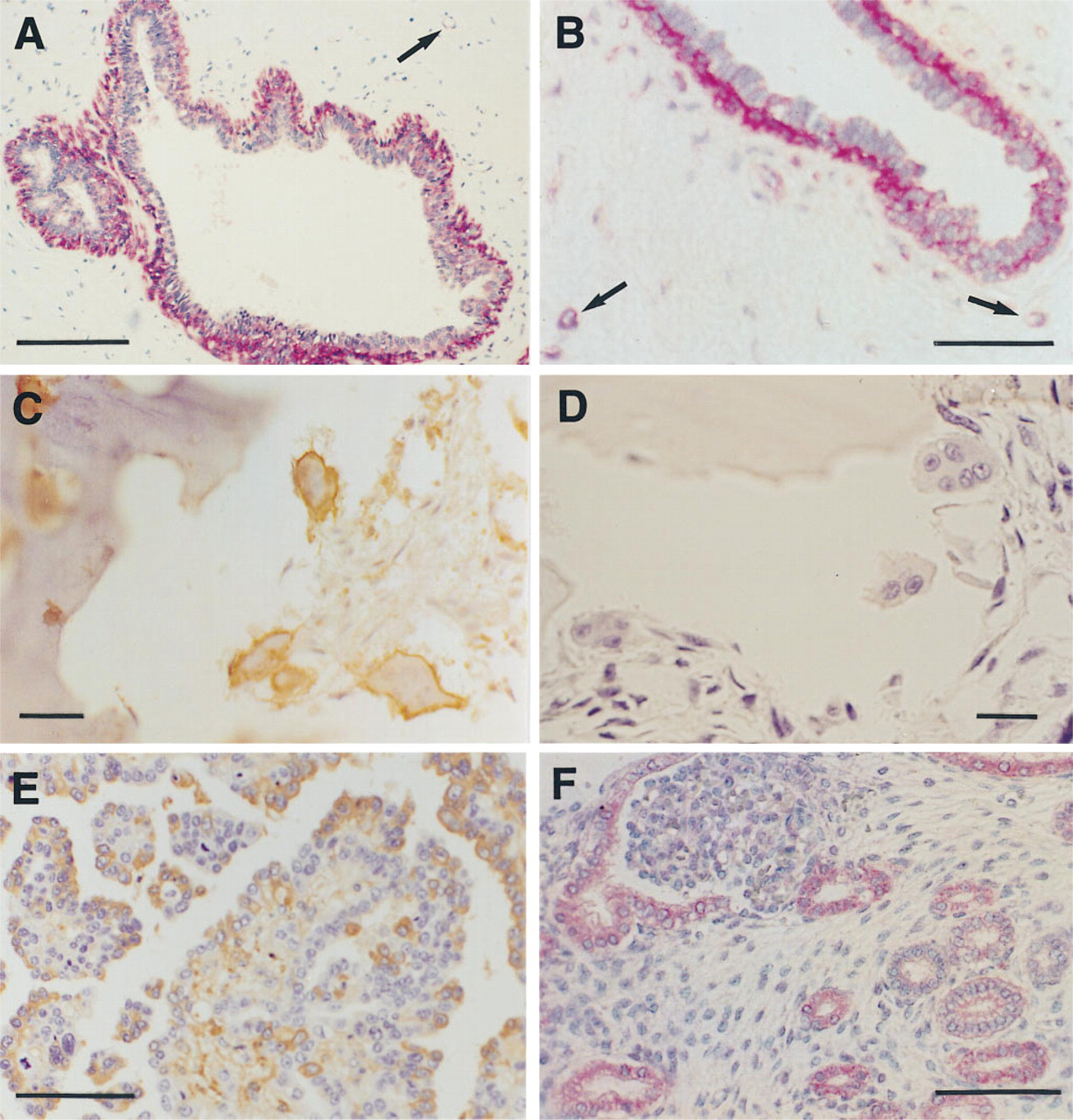
Examples of positive reactions are shown in Figures 1A through 1F. GoH3 ad (Figure 1A) to laminin receptor (α6) strongly stained myoepithelial cells in mammary ducts. Immunoreactivity in side-by-side frozen breast tissue preparations was of equal intensity, as shown in Figure 1B. Stromal vessels demonstrated staining within their thin walls in both the frozen and formalin-fixed tissue (arrows in Figures 1A and 1B). Our results are as previously reported for α6 integrin localization in mammary glands (Berdichevsky et al. 1994). Application of polyclonal Ab T545 to αvβ3 complex showed a plasma membrane type of immunoreactivity within osteoclasts (Figure 1C). αvβ3 integrin was previously detected in vitro by immunofluorescence on osteoclasts at the sealing zone of the osteoclast attachment apparatus to bone, and a role for the αvβ3 integrin in bone resorption has been suggested (Horton and Davies 1989). Parallel sections without Ab (negative control) were entirely negative (Figure 1D). Slightly better was αvβ3 reactivity observed in EDTA decalcified sections compared to acid decalcification. However, the difference, in our opinion, was insignificant. We recently demonstrated abundant β3 mRNA by in situ hybridization in T545-positive osteoclasts of breast cancer metastases to bone (Liapis et al. 1996).
Ab33 Ab to fibronectin receptor showed strong immunoreactivity within cells of papillary serous ovarian adenocarcinoma (Figure 1E). The fibronectin receptor, when genetically overexpressed in Chinese Hamster ovary (CHO) cells, inhibits tumor growth in nude mice (Giancotti and Ruoslahti 1990). We recently found this integrin to be differentially expressed in a series of low malignant potential tumors and ovarian adenocarcinomas (Liapis et al., in press). Ab DH12 detected β1 integrin within tubule epithelial cells and the parietal epithelium of Bowman's capsule in formalinfixed fetal metanephric kidney sections subjected to MW heating (Figure 1F). The results are in agreement with the study of Korhonen et al. (1990), who detected this integrin by immunofluorescence in frozen fetal kidney sections. On frozen kidney sections processed in parallel to formalin-fixed tissue no difference was observed in signal localization (not shown).
Discussion
Formalin fixation has long been known to potentially induce “masking” of antigenic epitopes in tissues. Enzyme treatments to digest aldehyde bonds induced by formalin fixation or, more recently, MW heating is used for detection of a wide range of antigens (Taylor et al. 1994; Leong and Milios 1993; Shi et al. 1991). Previous attempts to utilize MW heating on formalin-fixed tissue for integrin detection have not been successful (Cattoretti et al. 1993), with the exception of a recent report (Gladson et al. 1996) in which the authors utilized three commercially available Abs to integrins αv, β3, and α5β1. As a result, studies of integrin expression in human tissues are limited to frozen material. Even though the exact mechanisms through which microwave heating works are still under investigation (Shi et al. 1995), signal detection for many commonly used Abs is superior compared to other heating methods, e.g., autoclave or waterbath (Tani et al. 1995). However, we were unable to detect specific immunoreactivity with several of the Abs used in this study. It is possible that alternative buffers (Alsbeh and Battifora 1995), varying the buffer pH (Grossfeld et al. 1996), or use of newly developed tissue fixatives (Muller et al. 1996) will be helpful for integrin detection. Manual as well as automated apparatus can be used. In our hands, the automated system gave consistently positive results, perhaps because of continuous heating (37C) within the incubation chamber. This report exemplifies the need for application of the “test battery” approach [proposed by Shi et al. (1995)] in working with Abs to integrins.
The practical aspects of tissue availability, as well as the image superiority of immunohistochemistry or formalin-fixed tissue vs immunofluorescence on frozen sections, cannot be overemphasized. The described modifications provide the potential to study integrin receptors in many common as well as rare neoplasms that might be available only as formalin-fixed, paraffin-embedded blocks.
Acknowledgments
We wish to thank all of the investigators who provided the Abs to integrins listed in Table 1.
