Abstract
Paneth cells are located at the base of the intestinal glands. The origin, composition, and function of these cells have not been well established. The sharing of a common pathway of development with the goblet cells has been suggested. The aim of the present study was to explore the cytochemical composition of rat Paneth cells and to discuss a possible developmental relationship between goblet and Paneth cells. Lectins (WGA, LTA, UEA-I, AAA, and HPA) were used as a precise tool for the ultrastructural localization of carbohydrates. Several procedures were performed in combination with lectin cytochemistry: β-elimination, a reaction that specifically removes O-linked oligosaccharides (typical of mucin-type glycoproteins of goblet cells); and treatment with peptide N-glycosidase F, an enzyme that removes N-linked oligosaccharides from glycoproteins. Secretory granules of Paneth cells showed a biphasic nature composed of an electron-lucent peripheral halo containing O-linked oligosaccharides with GalNAc and GlcNAc residues and N-linked oligosaccharides with GlcNAc residues (only sparse Fuc residues were scarcely identified in O-linked oligosaccharides), and an electron-dense core containing N- and O-linked oligosaccharides with Fuc residues. Neither GlcNAc nor GalNAc was identified. The occurrence of O-linked oligosaccharides in the Paneth cells and the biphasic nature of the secretory granules, similar to that of transitional cells intermediate between mucous and serous cells of other tissues, favor the hypothesis of a common lineage for goblet and Paneth cells.
Keywords
P
The functions of the Paneth cells have not been clearly established. However, it has been demonstrated that Paneth cells produce and secrete antibacterial agents (lysozyme, cryptidin, and immunoglobulin A), hydrolases, lipases, and growth factors and modulators (Desai et al. 1991; Quellette and Lualdi 1990; Saito et al. 1988; Lechene de la Porte et al. 1986; Poulsen et al. 1986; Senegas-Balas et al. 1984; Erlandsen et al. 1974, 1976; Erlandsen and Parsons, 1973). The granules also contain a zinc-binding protein (Sawada et al. 1994). It has been suggested that goblet and Paneth cells share a common pathway of development (Kedinger et al. 1988; Lopez-Lewellyn and Erlandsen 1980; Lopez-Lewellyn 1979).
Lectins are proteins or glycoproteins that bind specifically to carbohydrate groups (Goldstein and Hayes, 1978). They have been widely used in combination with enzymes for in situ characterization of the carbohydrates of glycoproteins (Danguy et al. 1994; Menghi and Matterazzi 1994; Spicer and Schulte 1998, 1992). Oligosaccharide composition of the glycoproteins of the intestinal epithelial cells has been investigated by lectin histochemistry, with special attention to goblet and absorptive cells (Brinck et al. 1995; Madrid et al. 1989; Calderó et al. 1988; Skutelsky et al. 1987; Etzler and Branstrator 1974). Although Paneth cells share ultrastructural characteristics typical of serous cells (large apical secretory granules and abundant basal rough endoplasmic reticulum), early histochemical studies revealed that they contain a mucoid material (Herzog 1937). Paneth cells are reactive to PAS and Alcian blue techniques (Sheahan and Jervis 1976; Selzman and Liebelt 1962) in a similar manner to goblet cells. Paneth cells have also been demonstrated by lectin histochemistry to contain carbohydrates (Evans et al. 1994; Madrid et al. 1989).
The aim of the present study was to determine the oligosaccharide composition of the glycoproteins of the secretory granules of Paneth cells. We have investigated both the nature of the carbohydrates and the nature of the linkage between oligosaccharide chains and protein core, to obtain information that could illustrate the possible common origin of Paneth and goblet cells. To obtain these data, lectin staining was combined at the light and electron microscopic levels with the following methods: (a) β-elimination (chemical deglycosylation), which removes protein-carbohydrate linkage of the O-glycosidic type (Ono et al. 1983); and (b) peptide N-glycosidase F (PNGase-F), which removes N-linked oligosaccharides (Lucoq et al. 1987).
Materials and Methods
Reagents
Polyethylene glycol (mw 20,000), sodium citrate, potassium carbonate, sodium ascorbate, and tetrachloroauric acid were obtained from Merck (Darmstadt, Germany). Bovine serum albumin (BSA), 3,3'diaminobenzidine (DAB); gold-labeled (10-nm) trypsin inhibitor (ovomucoid), GlcNAc, N-acetyl-galactosamine (GalNAc), fucose (Fuc), Helix pomatia agglutinin (HPA), Ulex europaeus agglutinin-I (UEA-I), Triticum vulgaris agglutinin (WGA), Lotus tetragonolobus agglutinin (LTA), horseradish peroxidase labeled (HRP) HPA, UEA-I- HRP, LTA-HRP, and WGA-HRP were purchased from Sigma (Poole, Dorset, UK). Endo-β-N-acetylglucosaminidase F/peptide N-glycosidase F from Flavobacterium meningosepticum, Aleuria aurantia agglutinin (AAA)-digoxigenin (DIG)-labeled lectin, anti-DIG sheep antibody, anti-DIG mouse antibody, anti-DIG-HRP-labeled goat antibody, and DIG antibody labeling kit were from Boehringer Mannheim (Barcelona, Spain). Goat anti-mouse IgG + M-gold complex (15 nm) and donkey anti-sheep IgG-gold complex (15 nm) were from Biocell (Cardiff, UK). The carbohydrate binding specificity of the lectins is summarized in Table 1.
Tissue Sample and Preparation
Seven adult Sprague-Dawley rats were sacrificed under ether anesthesia and a portion of the ileum was resected. The guidelines from the Ministry of Agriculture, Fishing and Alimentation of Spain for care and use of laboratory animals were followed.
For light microscopy, tissue samples were fixed in 10% formalin in PBS, pH 7.4, for 6 hr and embedded in paraffin.
For conventional electron microscopy, specimens were immediately immersed in ice-cold fixative containing 1.25% glutaraldehyde and 4% paraformaldehyde in 0.1 M cacodylate buffer (pH 7.4), for 5–6 hr, at 4C. Then the tissue blocks were postfixed in a 1% osmium tetroxide solution in the cacodylate buffer for 90 min, washed in PBS, and embedded in Epon 812. For ultrastructural cytochemistry, tissue samples were immersed in 2% glutaraldehyde in PBS for 2 hr (Roth 1983a) and embedded in Lowicryl K4M at - 30C (Carlemalm et al. 1982). Ultrathin sections were mounted on formvar-coated nickel grids.
Preparation of Lectin-Gold Complexes
Monodisperse colloidal gold solutions with a mean particle diameter of 14 nm were prepared according to Frens (1973). Mean particle diameter was checked by an IMCO 10 image analyzer computer (Kontron Bildanalyse; Eching, Germany) with Microm Image Processing Software (Microm; Barcelona, Spain). HPA- and UEA-I-gold complexes were prepared as reported previously (Martínez-Menárguez et al. 1993: Madrid et al. 1990).
Preparation of Lectin-DIG Complexes
HPA-, UEA-I-, and LTA-DIG complexes were prepared according to the DIG antibody labeling kit from Boehringer Mannheim Biochemica. The complexes were prepared by chemical coupling of DIG-NHS (digoxigenin-3-O-succinyl-∊-aminocaproic acid-N-hydroxysuccinamide ester) to the amino group of the lectins. DIG-NHS was mixed with HPA, UEA-I, or LTA. The DIG-lectin complexes were purified with a Sephadex G-25 column and samples with a higher absorbance at 280 nm were selected (Martínez-Mernárguez et al. 1993).
Lectin affinity of the granules of Paneth cells. a

a Intensity of staining: ++++, very strong; +++, strong; ++, moderate; +, low; -, negative.
bThe carbohydrate binding specificity of each lectin is indicated in parentheses.
Cytochemical Labeling
Light Microscopy. Histochemical staining with HRP-labeled lectins (WGA, HPA, UEA-I, and LTA) was performed as reported previously (Madrid et al. 1989, 1990). AAA-DIG-labeled lectin was also used according to Sata et al. (1990) and Martínez-Menárguez et al. (1993). The sections were incubated with the DIG-labeled lectin and then with anti-DIG-HRP-labeled goat antibody. Finally, in the two methods, HRP was developed with DAB and H2O2.
Electron Microscopy. Colloidal gold was the marker selected for the ultrastructural studies. For lectin cytochemistry one-step, two-step, and three-step methods were used. (a) The one-step method was used for HPA- and UEA-I-gold complex as previously described (Madrid et al. 1990, 1994; Martínez-Menárguez et al. 1993). The dilution of the lectin gold complex was 1:25 for HPA-gold and 1:8 for UEA-I- gold complex. (b) The two-step method was used for WGA (Castells et al. 1992; Madrid et al. 1990; Roth 1983a,b; Geoghegan and Ackerman 1977). As second layer, ovomu-coid-gold complex was used (dilution 1:50). The concentration of the lectin was 25 μg/ml. (c) The three-step method has been previously reported (Castells et al. 1994; Madrid et al. 1994; Martínez-Menárguez et al. 1993). The DIG-labeled lectins used were AAA (20 μg/ml), UEA-I (1:4), HPA (1:4), and LTA (undiluted). The second layer was either unlabeled anti-DIG mouse antibody (5 μg/ml for HPA; 20 μg/ml for LTA) or unlabeled anti-DIG sheep antibody (7 μg/ml for UEA-I; 5 μg/ml for AAA). The corresponding third layers were goat anti-mouse IgG+M-gold conjugates (1:20 for HPA; 1:5 for LTA) and donkey anti-sheep IgG-gold complex (1:8 for UEA-I; 1:10 for AAA).
Controls. The following controls were used: (a) substitution of conjugated and unconjugated molecules (WGA-HRP, HPA-HRP, LTA-HRP, UEA-I-HRP, AAA-DIG, WGA, HPA-DIG, LTA-DIG, UEA-I-DIG, HPA-gold, UEA-I-gold, ovomucoid-gold, anti-mouse IgG + M-gold, anti-sheep IgG-gold, anti-DIG-HRP and unlabeled anti-DIG antibodies) by the corresponding buffer; and (b) preincubation of the lectins with the corresponding hapten sugar inhibitor (GlcNAc for WGA, GalNAc for HPA, and Fuc for AAA, LTA, and UEA-I) used at a concentration of 0.4 M.
Chemical Treatments
Chemical Deglycosylation (β-elimination). Paraffin sections were treated with 0.5 N NaOH in 70% ethanol at 4C for 7 or 14 days according to Ono et al. (1983). Sections from rat testis were used as control. After an adequate β-elimination procedure, HPA labeling was rendered negative on sections of rat testis (Martínez-Menárguez et al. 1993).
Acid Hydrolysis. Sections were immersed in 0.1 M HCl for 2–3 hr at 82C to remove sialic acid residues (Madrid et al. 1994). Human gallbladder sections were used as control. Acid hydrolysis rendered PNA labeling positive in the secretory granules of principal cells (Madrid et al. 1994).
Enzyme Treatment
The endo-β-acetylglucosaminidase F/peptide N-glycosidase F (Endo F/PNGase F) pretreatment was performed at light and electron microscopic levels as reported previously (Martínez-Menárguez et al. 1993; Lucoq et al. 1987). The tissue sections were incubated with 6 U/ml of Endo F/PNGase F diluted in 0.1 M Tris, 150 mM NaCl, 2.5 mM EDTA, pH 9, for 3 days. At this pH, PNGase-F activity predominated over Endo F activity. In this way, cleavage of β-aspartyl glycosyl linkage (GlcNAc β-1-N-Asn) was produced. Rat testis sections were used as control. PNGase-F activity renders rat testis negative to AAA (Martínez-Menárguez et al. 1993).
Results
Conventional Electron Microscopy
A previous conventional electron microscopic study was realized to observe the ultrastructure of secretory granules of Paneth cells. These granules showed a variable morphology, including bizonal and electron-lucent granules. Bizonal granules were the most common. They were composed of an electron-dense core and an electron-lucent peripheral halo. This halo was usually narrow, except in a pole of the granule where it was enlarged, originating a cap-like structure (Figure 1). Granules lacking this cap (with only a narrow electron-lucent peripheral halo) were also observed. These could represent granules with a different sectioning plane. Electron-lucent granules were usually smaller and might represent sections through the cap.
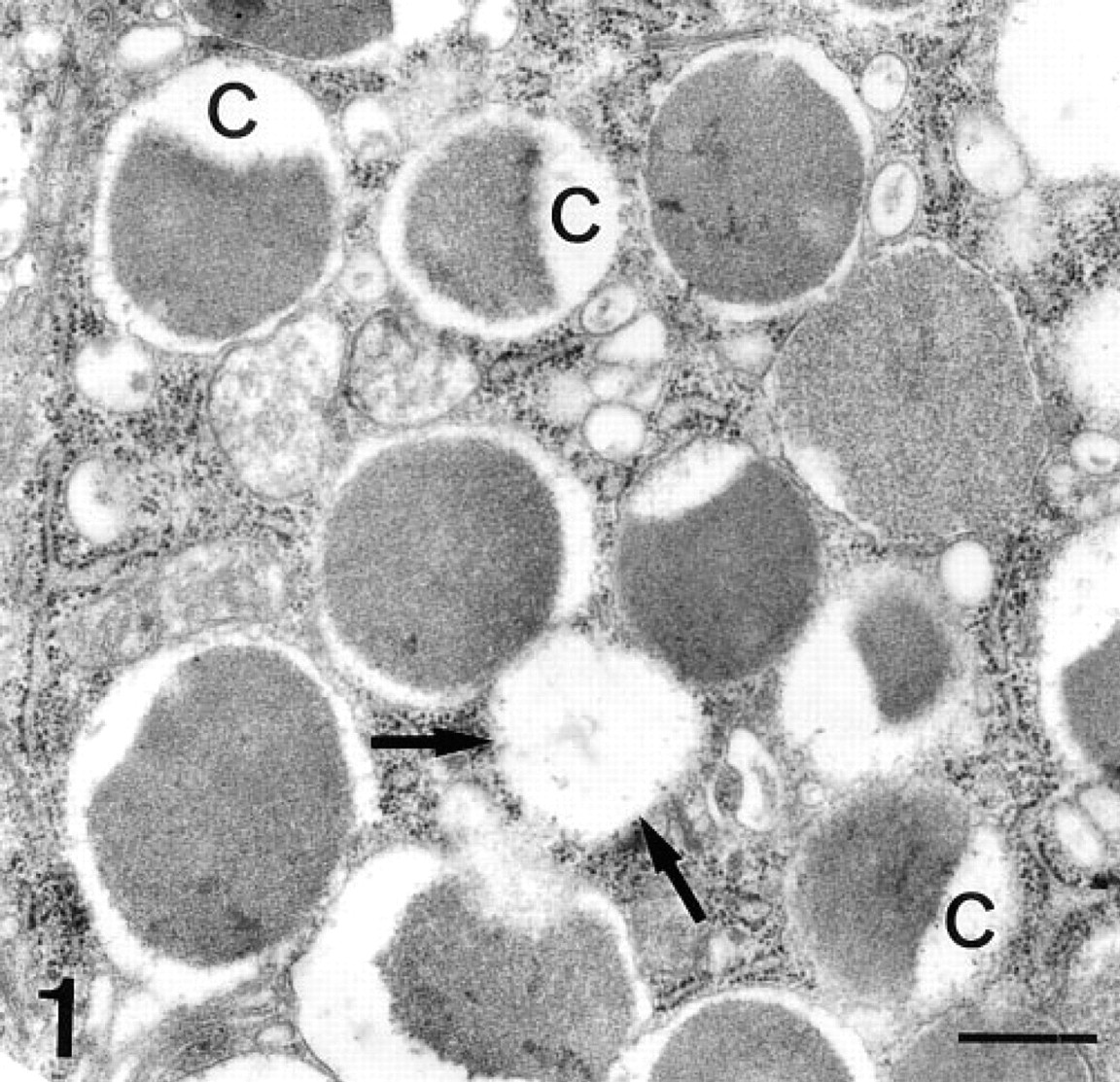
Paneth cell granules. Most granules show a bizonal structure, with an electron-dense core and an electron-lucent peripheral halo. Some granules show an enlarged electron-lucent area, which is a cap-like structure (c). Electron-lucent granules are also observed (arrows). These figures could represent granules sectioned through the cap of the bizonal granules. Bar = 1 μm.
Histochemistry
N-Acetylgalactosamine. At the light microscopic level, Paneth cells were moderately labeled by HPA (Figure 2a). After β-elimination, HPA staining was low or negative (Figure 2b). An increase in staining was observed after PNGase-F pretreatment (Table 1; Figure 2c). Combination of β-elimination and PNGase-F abolished the staining. Microvilli of the absorptive cells stained intensely with HPA. PNGase-F predigestion abolished this reactivity and was considered as an internal control of the activity of the enzyme.
At the ultrastructural level, labeling was observed in the electron-lucent peripheral halo (Figure 2d). Gold granules were mainly located close to the granule membrane or to the interface between the electron-lucent and electron-dense regions (Figure 2d).
Fucose. At the light microscopic level, Paneth cells stained intensely with UEA-I (Figure 3a). After PNGase-F or β-elimination pretreatment, UEA-I staining decreased (Table 1; Figure 3b). Low reactivity was observed with LTA, whereas AAA moderately stained Paneth cells. No modification of LTA and AAA reactivity was observed after PNGase-F pretreatment. After β-elimination, LTA was rendered negative and AAA staining decreased. The three lectins were unreactive when the same sections were pretreated with the β-elimination procedure and PNGase-F.
At the electron microscopic level, UEA-I labeled the electron-dense core of secretory granules of Paneth cells (Figure 3c). At this level, a decrease of labeling was observed after PNGase-F pretreatment (Figure 3d). Sparse labeling observed with LTA was detected in the electron-lucent peripheral halo (Figure 4). AAA labeled the electron-dense cores of secretory granules (Figure 5).
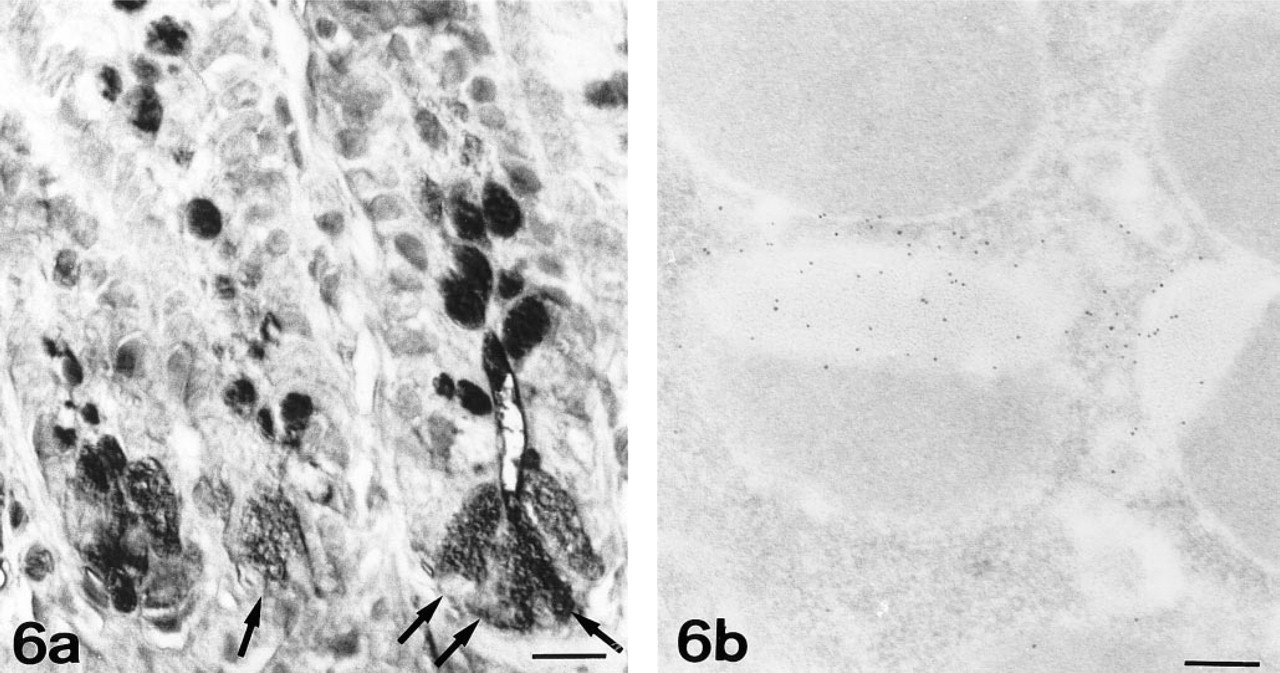
N-Acetylglucosamine. At the light microscopic level, WGA showed a strong affinity for Paneth cells (Figure 6a). No modification of the WGA binding pattern was observed after acid hydrolysis, thus indicating that WGA recognized GlcNAc. A slight decrease in staining was observed after β-elimination or PNGase-F pretreatment. The combination of the two procedures completely abolished this staining (Table 1).
At the ultrastructural level, WGA labeling was observed in the electron-lucent peripheral halo of secretory granules (Figure 6b).
Discussion
GalNAc residues interact specifically with HPA (Piller et al. 1990; Laboisse and Bogomoletz 1989; Roth 1984). Reactivity to HPA was concentrated in the electron-lucent peripheral halo of secretory granules. The β-elimination procedure almost abolished HPA labeling. This suggests that GalNAc residues are located at the non-reducing terminus of O-linked oligosaccharides. However, pretreatment with PNGase-F increased the labeling. It can therefore be assumed that removal of N-linked oligosaccharides with PNGase-F increases the accessibility of the lectin to the GalNAc residues of O-linked oligosaccharides.
Fuc residues were detected with UEA-I, LTA, and AAA (Osawa and Tsuji 1987; Yamashita et al. 1985; Sugii and Kabat 1982; Pereira and Kabat 1974). LTA labeling was restricted to the electron-lucent peripheral halo of secretory granules. Because β-elimination pretreatment abolished LTA staining, Fuc residues contained in the clear halo might be included in O-linked oligosaccharides. Fuc residues were also identified in the electron-dense core of the granules with UEA-I and AAA. The decrease in UEA-I labeling after β-elimination and/or PNGase-F pretreatment suggests that Fuc residues are contained in both N- and O-linked oligosaccharides. The β-elimination decreased AAA labeling, thus indicating the presence of Fuc in O-linked oligosaccharides. The staining was abolished when the two pretreatments were performed before AAA labeling (Table 1), suggesting that Fuc residues identified by AAA are also located in N-linked oligosaccharides. PNGase-F pretreatment did not modify AAA labeling. This finding indicates that the removal of N-linked oligosaccharides (which contain terminal Fuc residues) with PNGase-F could increase the accessibility to Fuc residues of the remaining O-linked oligosaccharides. The reasons for the different binding patterns of AAA and UEA-I with respect to LTA in Paneth cells cannot be clearly explained with the present information on their carbohydrate binding affinities reported by biochemical studies (Osawa and Tsuji 1987; Yamashita et al. 1985; Sugii and Kabat 1982; Pereira and Kabat 1974).
WGA has affinity for GlcNAc and sialic acid (Osawa and Tsuji 1987; Debray et al. 1981). Acid hydrolysis removes sialic acid from the tissue (Madrid et al. 1994; Schauer 1982). This biochemical procedure was preferred to neuraminidase predigestion because of its broader spectrum of action. Neuraminidases have different preferences for the different linkages of sialic acid, depending on the origin of the enzyme. In previous studies we have not found differences between acid hydrolysis and neuraminidases from Clostridium perfringens and Arthrobacter ureafaciens (Madrid et al. 1994). WGA staining was not modified when acid hydrolysis pretreatment was performed, indicating that, in Paneth cells, WGA binds to GlcNAc residues. WGA labeling was decreased with β-elimination or PNGase-F pretreatment and was abolished when the two procedures were used before lectin labeling. These data indicate that GlcNAc residues are contained both in N- and O-linked oligosaccharides. These oligosac-charides with terminal GlcNAc residues are localized in the electron-lucent peripheral halo. Affinity of Paneth cells to pokeweed lectin, which preferentially binds to (GlcNAc)n (Yokoyama et al. 1978), has been previously reported (Evans et al. 1994) at the light microscopic level.
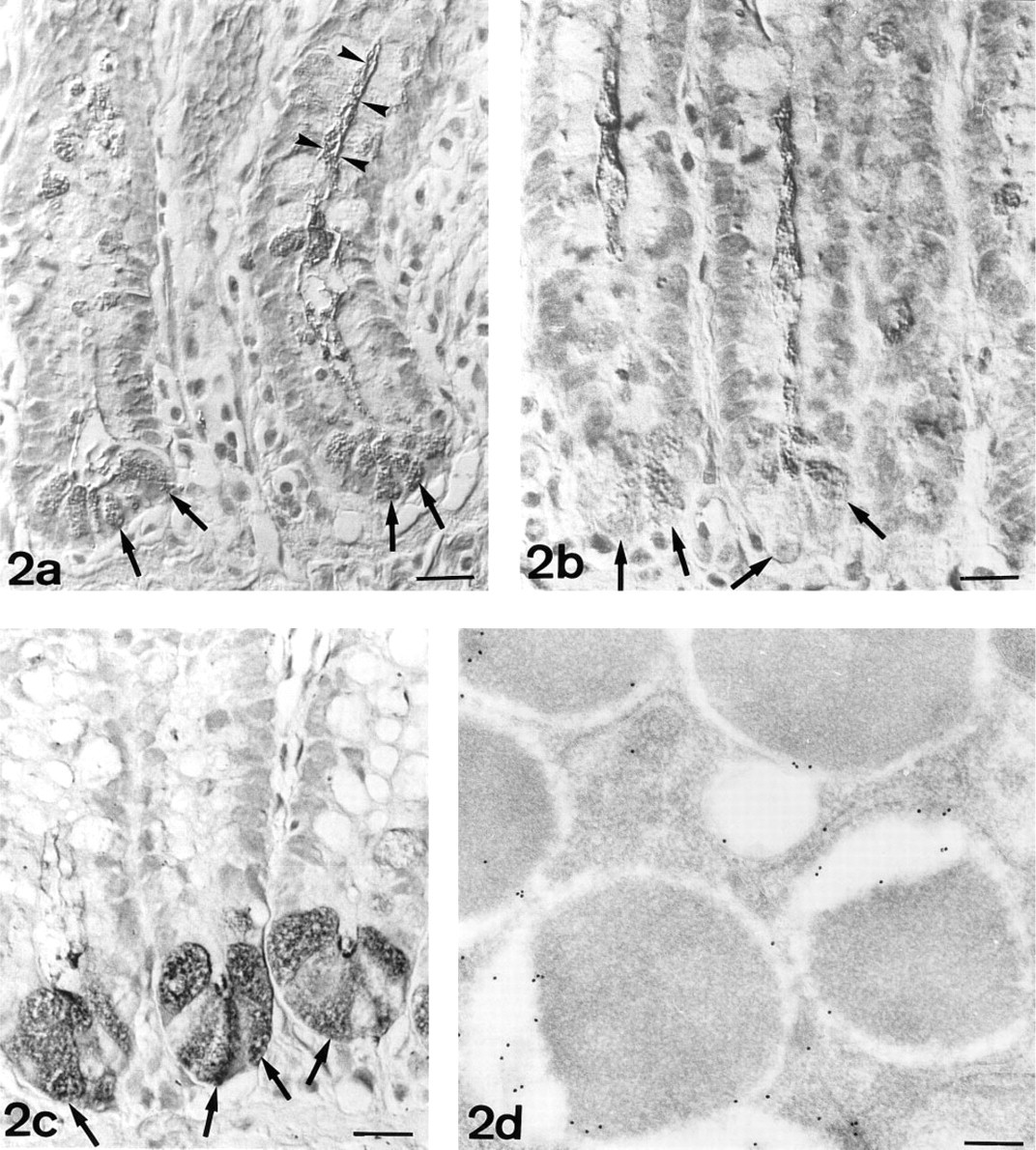
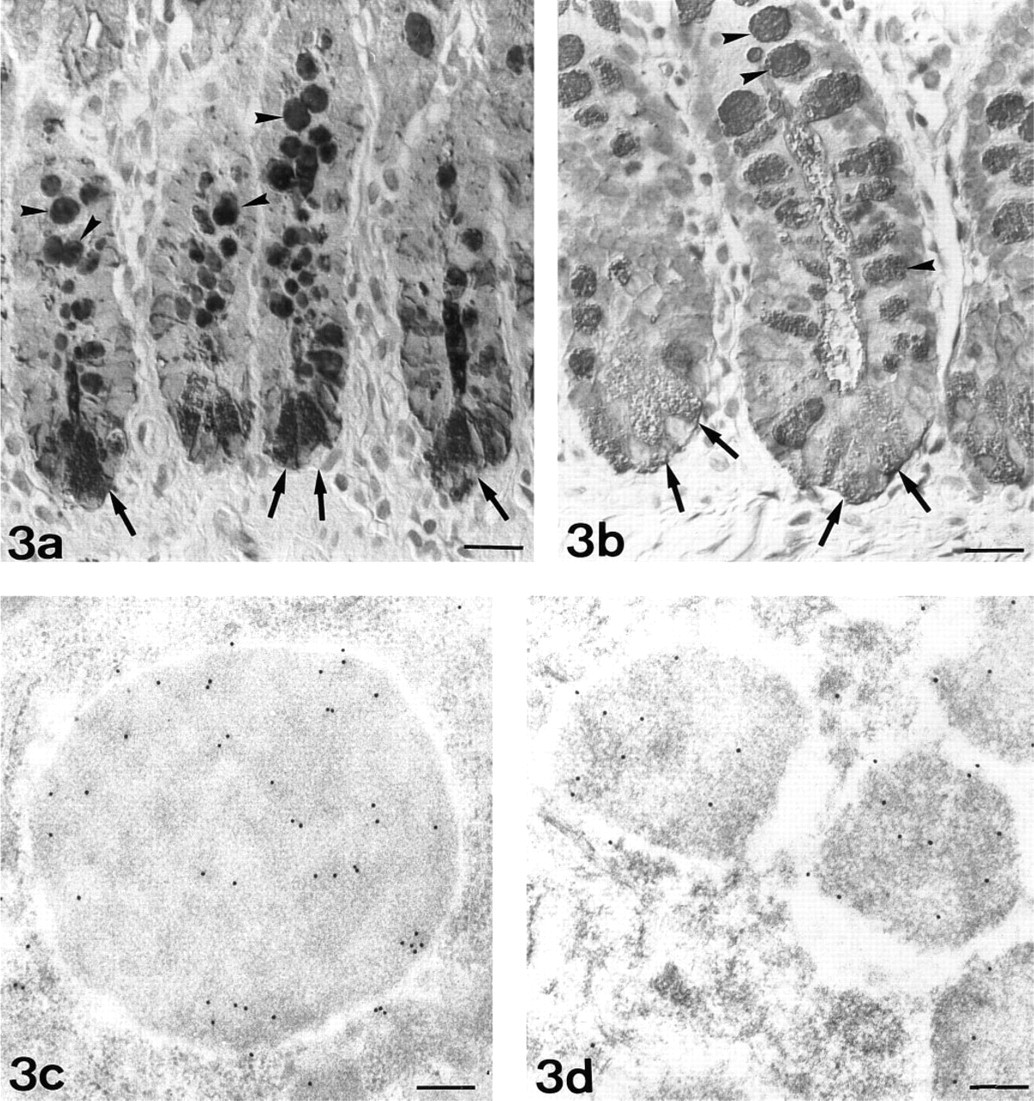
UEA-I-HRP. Paneth cells (arrows) are strongly reactive. Intense staining is also observed in goblet cells (arrowheads). Normarski interference optics.
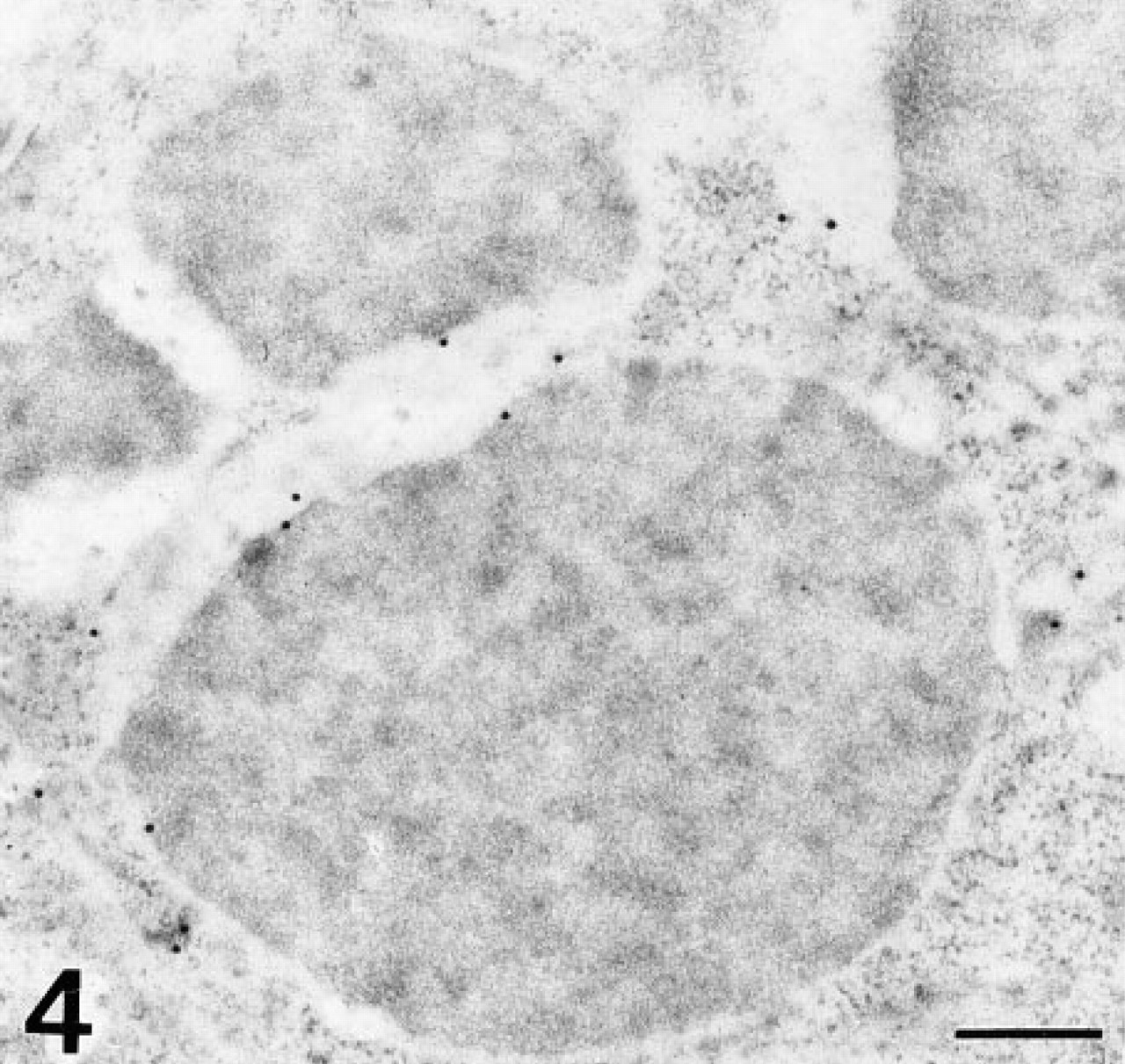
LTA-DIG/mouse anti-DIG/anti-mouse-gold. The electron-lucent peripheral halo of the granules is weakly labeled. No reaction is detected over the electron-dense area of the granules. Bar = 0.2 μm.
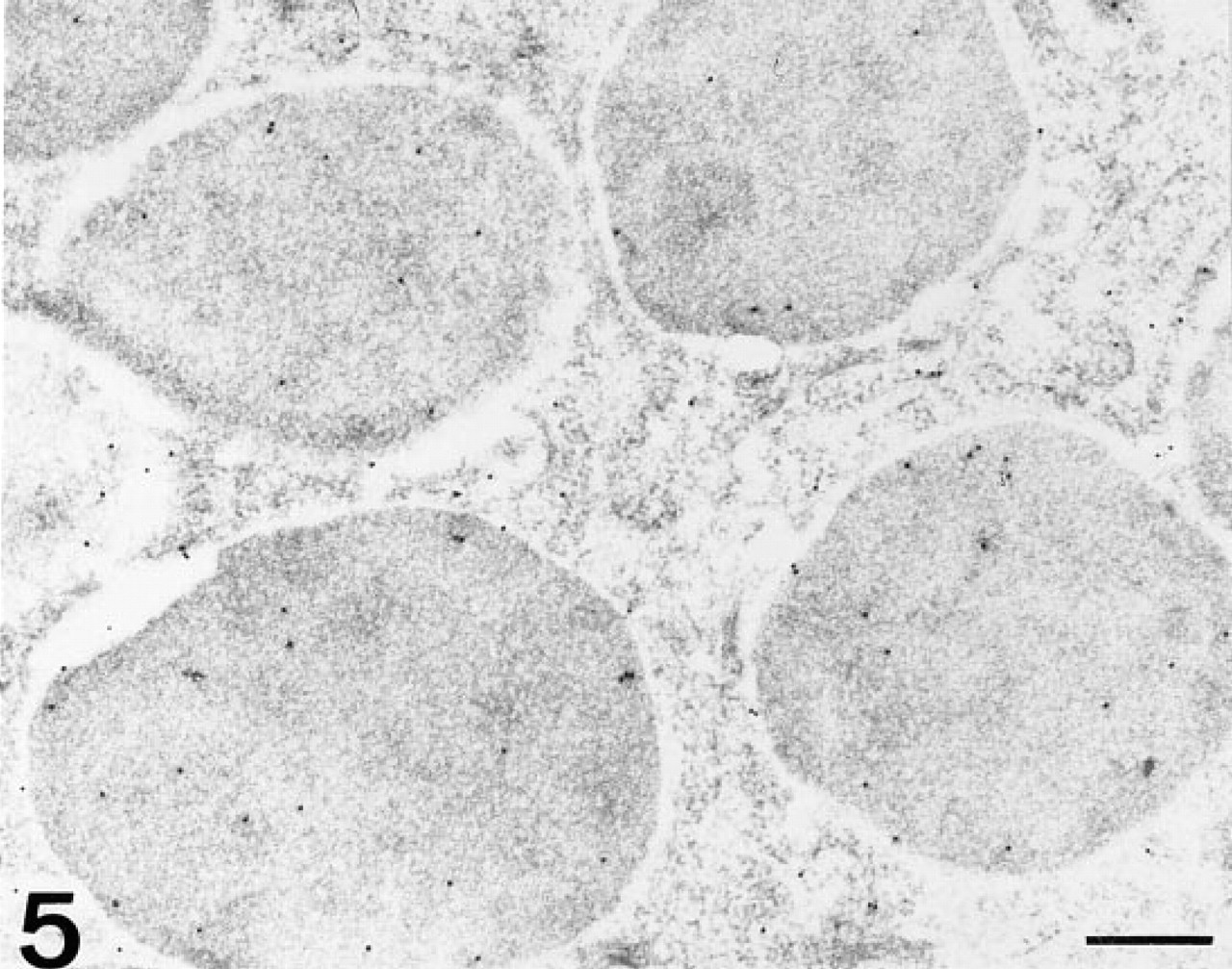
AAA-DIG/sheep anti-DIG/anti-sheep-gold. The electron-dense cores are moderately labeled. Bar = 0.3 μm.
The precise localization of the different carbohydrates performed in the present study enables us to suggest that the biphasic structure of the secretory granules of Paneth cells is not merely a morphological finding but is also a consequence of its cytochemical composition. We have found different oligosaccharide chains, probably belonging to different glycoproteins, in the two compartments. The electron-dense core contains glycoproteins with N- and/or O-linked oligosaccharides with terminal Fuc residues, whereas the electron-lucent halo contains glycoproteins with O-linked oligosaccharides with terminal GalNAc residues and N- and/or O-linked oligosaccharides with terminal GlcNAc residues. Lysozyme has been the most sought-after component (Mathan et al. 1987; Erlandsen et al. 1974; Erlandsen and Parsons 1973), being largely confined to the electron-dense core. Unfortunately, no information about the localization of most of the components of the secretory granules of Paneth cells is available. However, the existence of two compartments with different compositions within the granules is not unusual. Cells containing biphasic granules with different oligosaccharide composition of the electron-lucent and electron-dense areas have also been reported in both respiratory and digestive tracts (Castells et al. 1992, 1994; Madrid et al. 1990). In these systems, the cells with biphasic granules have been considered to be transitional between mucous and serous cells, sharing characteristics of both. The composition of electron-lucent area resembles that of mucous cells, whereas that of the electron-dense areas is similar to the serous cells (Castells et al. 1992, 1994; Madrid et al. 1990). The validity of the concept of mucous and serous cells has been discussed by Schulte and Spicer (1983), who stated that not only the morphology but also the cytochemical composition should be considered in classification of the cells. Our findings in Paneth cells, typically considered as serous cells, demonstrating the presence of O-linked oligosaccharides [which are typical of mucin-type glycoproteins of the goblet cells (Slomiany et al. 1980)] and the biphasic nature of the granules (in a similar way to that of intermediate cells of other tissues) enables us to suggest the existence of a relationship with goblet cells. Moreover, the occurrence of cells intermediate between Paneth and goblet cells has been reported (Lopez-Lewellyn and Erlandsen 1980; Througton and Trier 1969). These intermediate cells could indicate that goblet and Paneth cells share a common pathway of development (Lopez-Lewellyn 1979). Furthermore, Paneth cells containing mucous granules that are discharged during maturation have also been described (Mathan et al. 1987).
WGA-HRP. Paneth cells are strongly reactive (arrows). Goblet cells and microvilli are also stained.
In summary, we have shown the biphasic nature of the secretory granules of Paneth cells, describing a different glycidic composition in the electron-dense core and electron-lucent peripheral halo by ultrastructural cytochemistry. The electron-lucent peripheral halo contains GalNAc residues in O-linked oligosaccharides, and GlcNAc as terminal residue in both N- and O-linked oligosaccharides. The electron-dense core shows Fuc residues in N- and O-linked oligosaccharides. Sparse Fuc residues have also been demonstrated in the O-linked oligosaccharides of the electron-lucent halo. The results obtained enable us to hypothesize the existence of a common lineage with the goblet cell, which would originate from the stem cells according to the unitarian theory of Cheng and Leblond (1974).
Footnotes
Acknowledgements
We are greatly indebted to Ms C. Otamendi, Ms M.D. López-López, Mr J. Moya, Ms M.C. González, and Mr J.A. Madrid for excellent technical assistance.
Supported by grants PB 93–1123 from the Spanish DGI-CYT and UPV 075.327-EC236/95 from the University of the Basque Country. OL is supported by a fellowship from the Ministerio de Educacion y Ciencia (Spain).
