Abstract
Carbonic anhydrase (CA) isoenzymes have been widely studied in the gastrointestinal tract, where they mediate membrane transport events and pH regulation. However, the esophagus has generally received scant attention. In an immunohistochemical study confirmed by Western blotting, we have detected four CA isoenzymes (CAI, II, III, and IV) in the epithelium of human esophagus. Isoenzymes I, III, and sometimes IV (<10%) were present in the cytoplasm of basal cells and II and IV in the cytoplasm and cell surface membranes, respectively, of suprabasal cells (prickle cells). The localization of CAIV to the plasma membranes was confirmed by electron microscopic immunocytochemistry. CA was effectively divided at the basal-suprabasal interface between low-activity CAI and III (basal) and high-activity CAII and IV (suprabasal). Carbonic anhydrase in esophageal epithelial cells may have several functions: elimination of CO2 and metabolites, participation in membrane transport events during active cell growth, and pH regulation as a protective mechanism against acidic gastric reflux.
Keywords
C
Immunohistochemistry has made a considerable contribution to the localization of CA beyond the erythrocyte. The first immunohistochemical attempts to localize CA isoenzymes were made by Spicer et al. (1979), who confirmed results obtained by the cobalt catalytic method and extended the number of cell sites that exhibit the enzyme.
Studies of the distribution and role of CA in the human gastrointestinal tract have seldom extended to the esophagus, the work of Parkkila et al. (1994) being a notable exception. This is perhaps because of the apparent unspecialized nature of its mucosa in comparison, e.g., to stomach and bowel. Putative and established roles have been assigned to the enzyme in the digestive system, largely on the basis of in vivo and in vitro studies using specific inhibitors (Swenson, 1991). For example, in stomach, CA in parietal cells is involved in the supply of H+ as substrate for H+, K+-ATPase in H+ secretion and K+ resorption. In addition, the enzyme may also participate in gastric cell pH regulation by supplying H+ and HCO3 - for Na+/H+ and Cl-/HCO3 - exchange (Seidler et al., 1989).
The report by Parkkila et al. (1994) described the presence of a weak, positive localization of the CAII isoenzyme in human esophageal epithelium and encouraged us to examine further the presence of the enzyme in this organ. In a study using the cobalt catalytic enzyme histochemical technique (Hansson, 1967), we were able to demonstrate and confirm the localization of CA in the esophageal epithelium and also in the submucosal glands of the organ (Christie et al., 1995).
In this report we describe the immunohistochemical localization of CA isoenzymes I, II, III, and IV in human esophageal epithelium in a study supported by Western blotting and immunoelectron microscopy. CA may have a role in provision of bicarbonate ions for maintenance of mucosal protection against gastric reflux (Tobey et al., 1989,1992). Reflux esophagitis is a common problem in gastroenterological practice (McDougall et al., 1996).
Materials and Methods
Tissue Samples
Pinch biopsies of endoscopically and histologically normal mid-third esophagus were obtained by informed consent from 15 patients undergoing endoscopic examination. The tissues were supported in Tryco-M-Bed (Aerosol Marketing and Chemical; London, UK) on cork disks and quenched in melting 2-methylbutane cooled with liquid nitrogen. Orientation of the tissues to produce transepithelial basal to luminal surface cells was often achieved in spite of the small size and nature of the samples. Sections (10 μm) were cut at − 25°C, mounted on subbed (gelatin- and chromalum-coated) coverslips, and fixed in 3% formaldehyde (generated from paraformaldehyde) in 0.1 M phosphate buffer, pH 7.4, for 10 min at room temperature.
Immunohistochemistry of CAI, II, III, and IV
The sections on coverslips were washed in PBS (five times over 10 min); treated with 0.1 M ammonium chloride in PBS for 10 min; nonspecific sites blocked with 0.5% fish skin gelatin (FSG) (Sigma Chemical; Poole, Dorset, UK) in PBS for 10 min; reacted with the primary polyclonal antibodies raised in rabbits (diluted in the range 1:50–1:200 with FSG in PBS) (antibody sources are identified in the Acknowledgments; the specificity of the antibodies is well documented by their respective donors); washed in PBS (five times over 10 min); secondary antibody applied [goat anti-rabbit FITC (Sigma) 1:100 with FSG in PBS] for 20 min. After a final wash with PBS (five times over 10 min) the sections were mounted in Mowiol 40–88 (Aldrich Chemical; Gillingham, Dorset, UK) and examined on a Zeiss Axioskop epifluorescence microscope fitted with a 495-nm excitation filter. Specificity controls for the antibodies included application of nonimmune sera and reaction with tissues and cells known to give a positive (and negative) response. For example, positive controls included CAII in gastric parietal cells, CAIII in striated muscle, and CAIV in lung and kidney. Photomicrographs were taken on Ilford HP5 ISO 400/27° film.
Immunoelectron Microscopy of CAIV
Small fragments of epithelium were fixed in 4% paraformaldehyde containing 0.1% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4, for 48 hr at 4°C and soaked overnight in 2.3 M sucrose containing 20% (w/v) polyvinyl pyrrolidone (40,000 KD) in PBS. The tissue fragments were then cut and mounted on metal stubs, frozen in liquid nitrogen, and 100-nm sections cut at − 100°C on a Reichert Ultracut E microtome with an FC4 cryoattachment. The sections were collected on 2.3 M sucrose PBS, mounted on pioloform-carbon-coated grids, and then incubated on 0.1 M NH4Cl in PBS (10 min), followed by 0.5% FSG in PBS (10 min). The grids were then floated on 20-μl drops of primary rabbit antisera, followed by washes in PBS (three times over 30 min) and 7-nm protein A-gold (Lucocq, 1993). Both the antibodies and the gold complexes were diluted in 0.5% FSG. After further washes in PBS (three times over 30 min) and distilled water (four times over 4 min), the grids were embedded and contrasted in 1.8% (w/v) methyl cellulose/0.3% (w/v) uranyl acetate as described in Griffiths et al. (1984). Sections were examined in a Jeol 1200EX electron microscope in transmission mode.
Western Blotting of Epithelial Cell Proteins
Because of their different cellular locations, CAIV membrane-associated and I, II, and III-soluble, different regimens for preparation of extracts from the biopsy specimens for Western blotting had to be adopted.
Protein Sample Preparation for CAIV. Pinch biopsies of middle-third esophageal epithelium were chopped into small fragments and homogenized in a Dounce homogenizer in pH 7.4 isolation buffer (Davidson et al., 1990) for 5 min on ice. This was followed by centrifugation at 1000 × g for 10 min and the supernatant extracted. The pellet was then resuspended in isolation buffer and homogenized again, followed by centrifugation at 1000 × g for 10 min. The first and second postnuclear supernatants were combined and centrifuged with an SW40Ti rotor at 36,000 rpm for 110 min. The supernatant was discarded and the pellet resuspended in 200 μl of lysis buffer (Heider et al., 1994) and left at 0°C for 10 min. The lysates were centrifuged at 13,000 X g for 5 min and the supernatant prepared for running on SDS-PAGE gels after protein concentration was determined by the Bradford method (Bradford, 1976). Samples containing 30 μg protein were loaded on a 12% gel and run at 200 V for 1 hr. Transfer was carried out overnight at 30 V onto nitrocellulose membranes (Sigma; 0.45-μm pore). This was followed by immunolabeling with the appropriate primary and secondary (goat anti-rabbit IgG; Sigma) antibodies. Primary antibody dilutions were in the range 1:1000–1:2000; the secondary antibody was used at 1:1500. Bands were revealed by the ECL reaction (Amersham Life Science; Little Chalfont, Bucks, UK).
Protein Sample Preparation for CAI, II, and III. Pinch biopsy samples were chopped into tiny fragments with scissors (0.5–0.25 mm) and added to 200 μl of cold lysis buffer (as above). They were left in the buffer on ice for 30 min, then centrifuged at 13,000 X g for 5 min at 4°C. The supernatant was extracted and the pellet discarded. Lysate protein (30 μg) was loaded onto SDS-PAGE gels and blotting carried out as above.
Results
Immunohistochemistry of Carbonic Anhydrase Isoenzymes
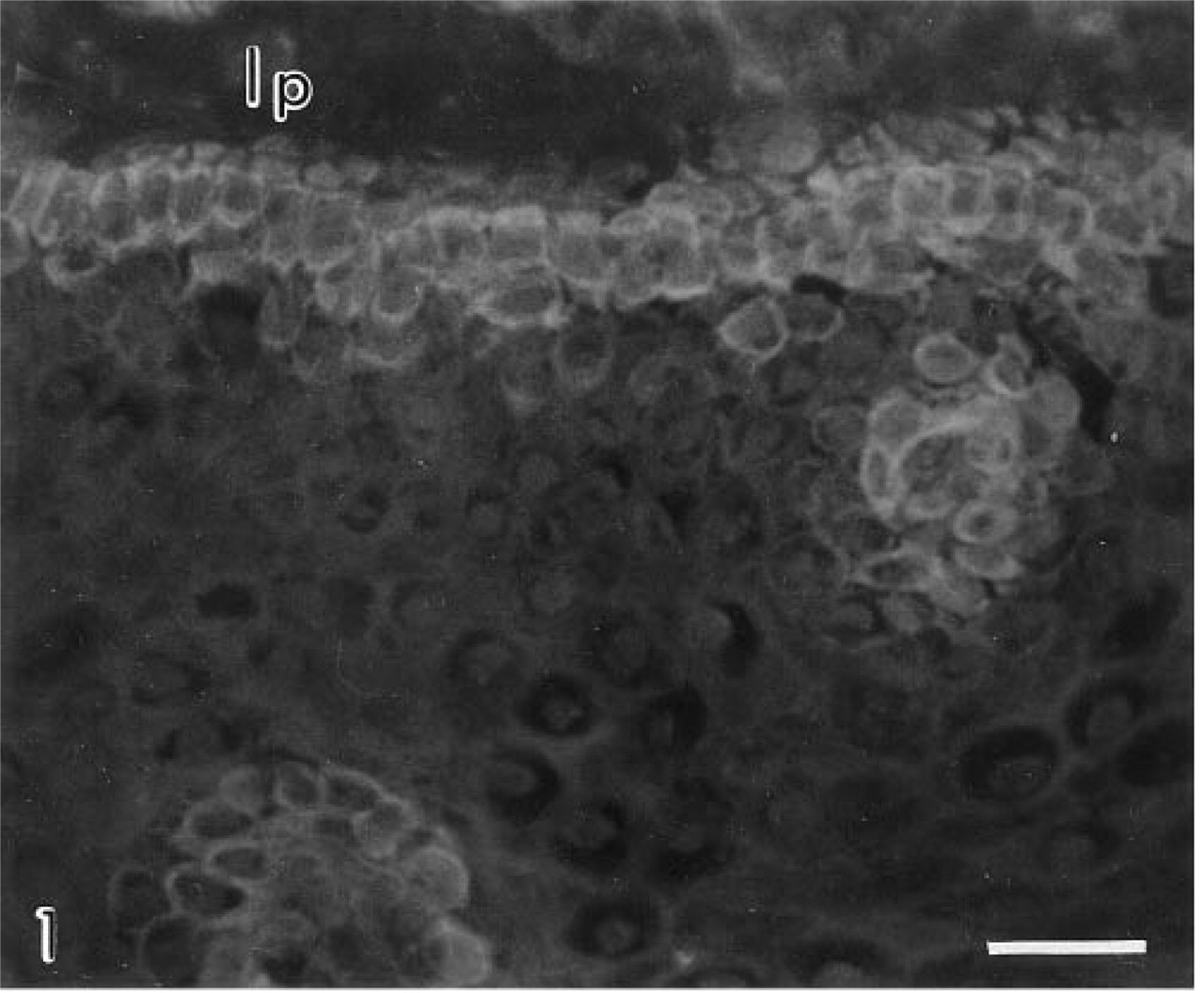
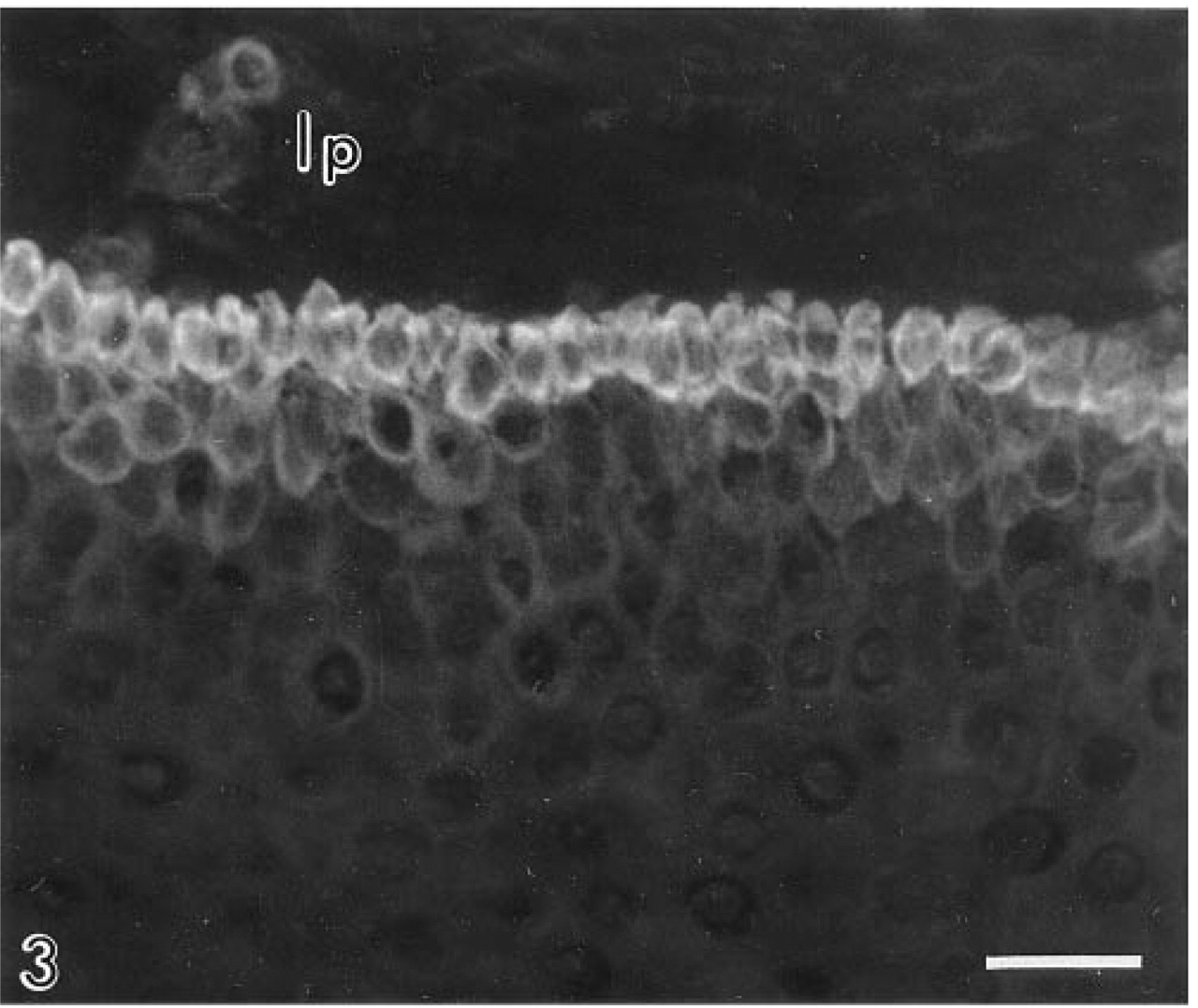
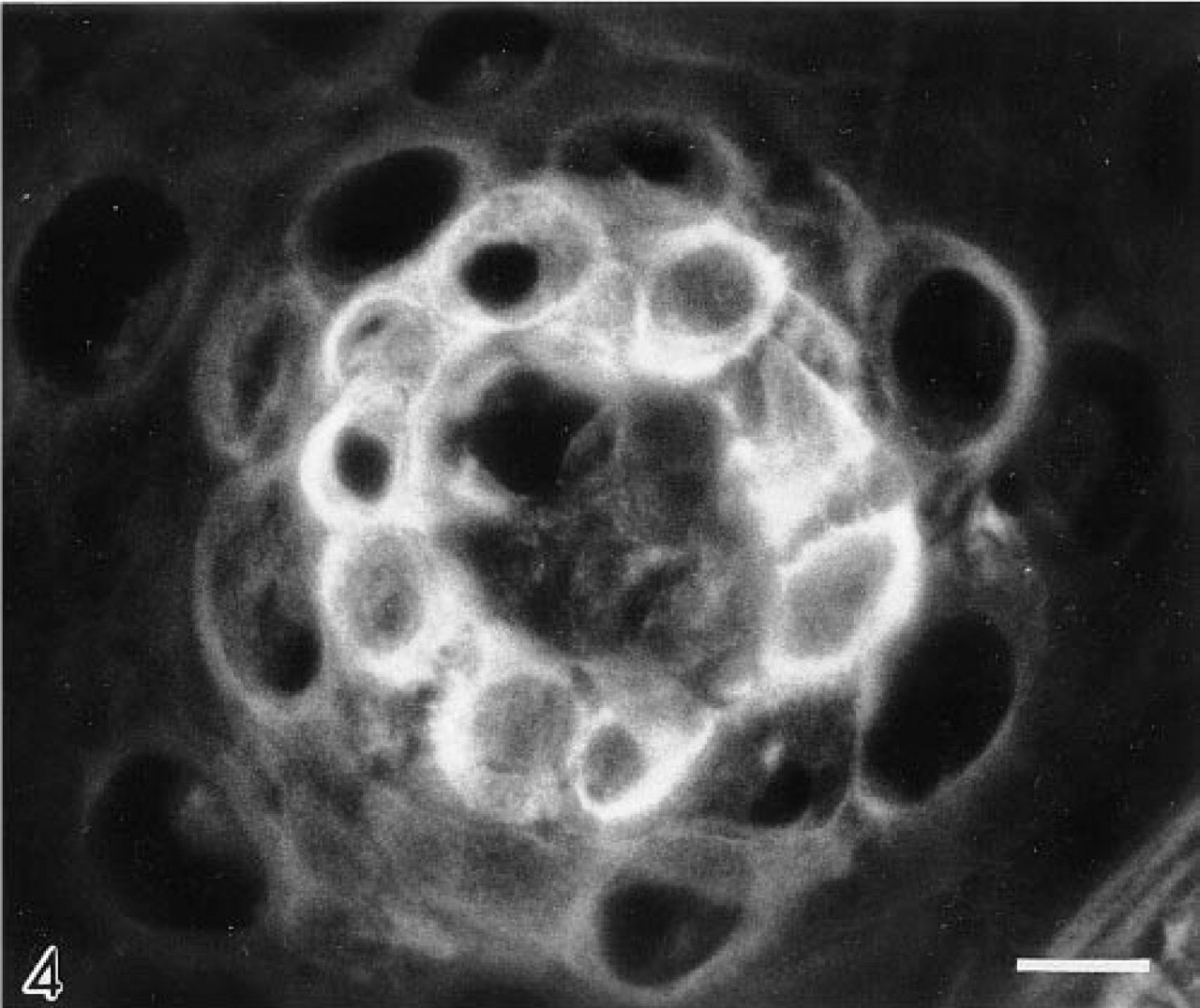
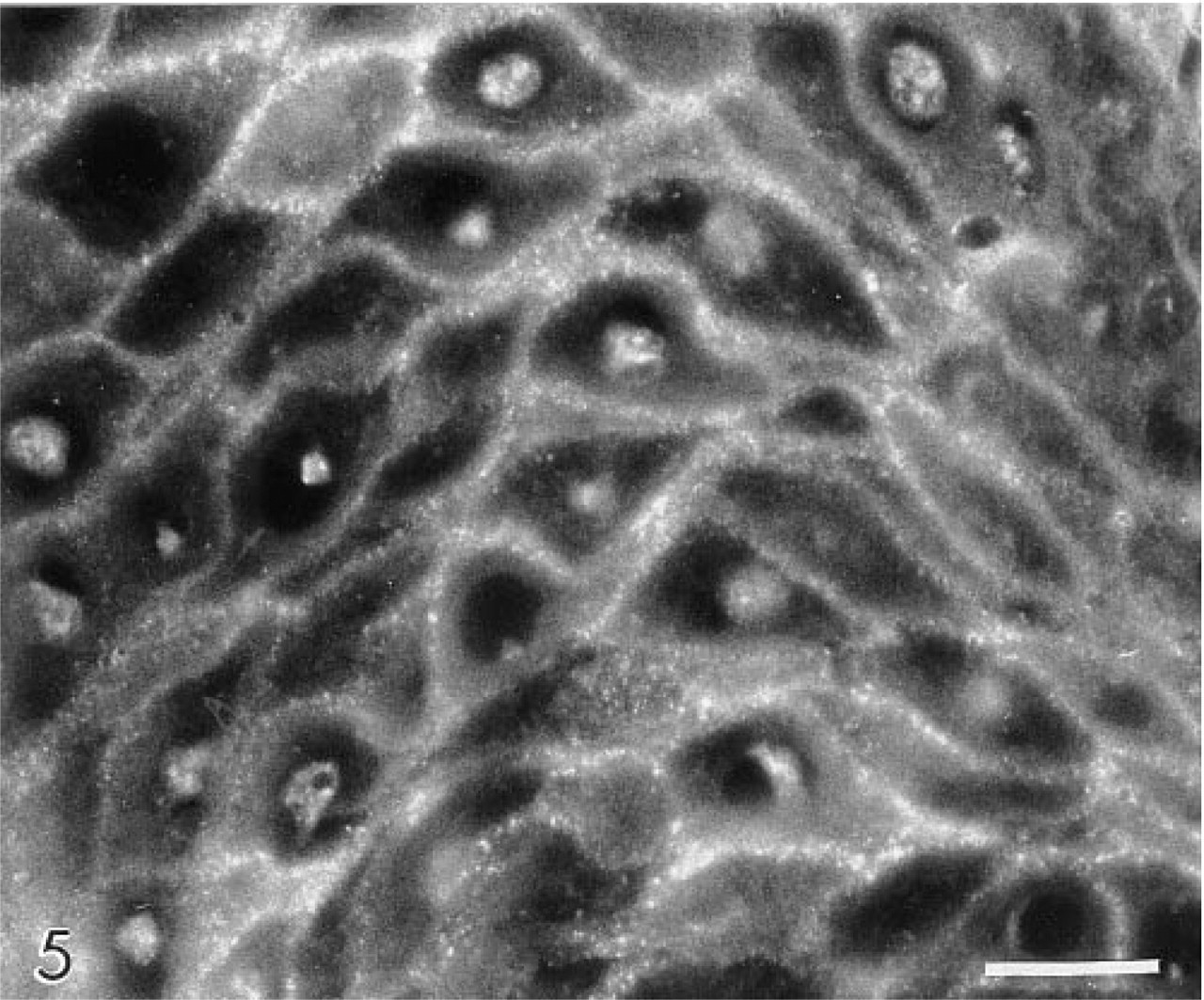
The CAI isoenzyme was confined to the basal cell layer, and of all the antibodies gave the weakest immunolocalization (Figure 1). The CAII isoenzyme was present in the cytosol of suprabasal cells, extending luminally as far as the most superficial three or four cell layers. Large areas of epithelium were frequently devoid of CAII. Reactive cells tended to occur in clusters, with individual cells displaying markedly different levels of immunoreactivity (Figure 2). CAII was never observed in basal cells. CAIII was restricted to basal cells and consistently gave a strong immunolocalization (Figures 3 and 4). CAIV was principally associated with the cell surface membranes of prickle and occasionally basal cells (<10%), where immunostaining appeared as a pericellular halo studded with intensely fluorescent particles, giving the cell peripheries a necklace-like appearance (Figure 5). Nucleus-associated elements were also identified by the CAIV antibody (Figure 5). Like CAII, CAIV tended to be localized to discrete clusters of cells, with large interstitial areas devoid of the isoenzyme. These findings were consistent for all of the biopsies.
Control and test tissue experiments gave results confirming the specificity of the antibodies.
Immunoelectron Microscopy of CAIV
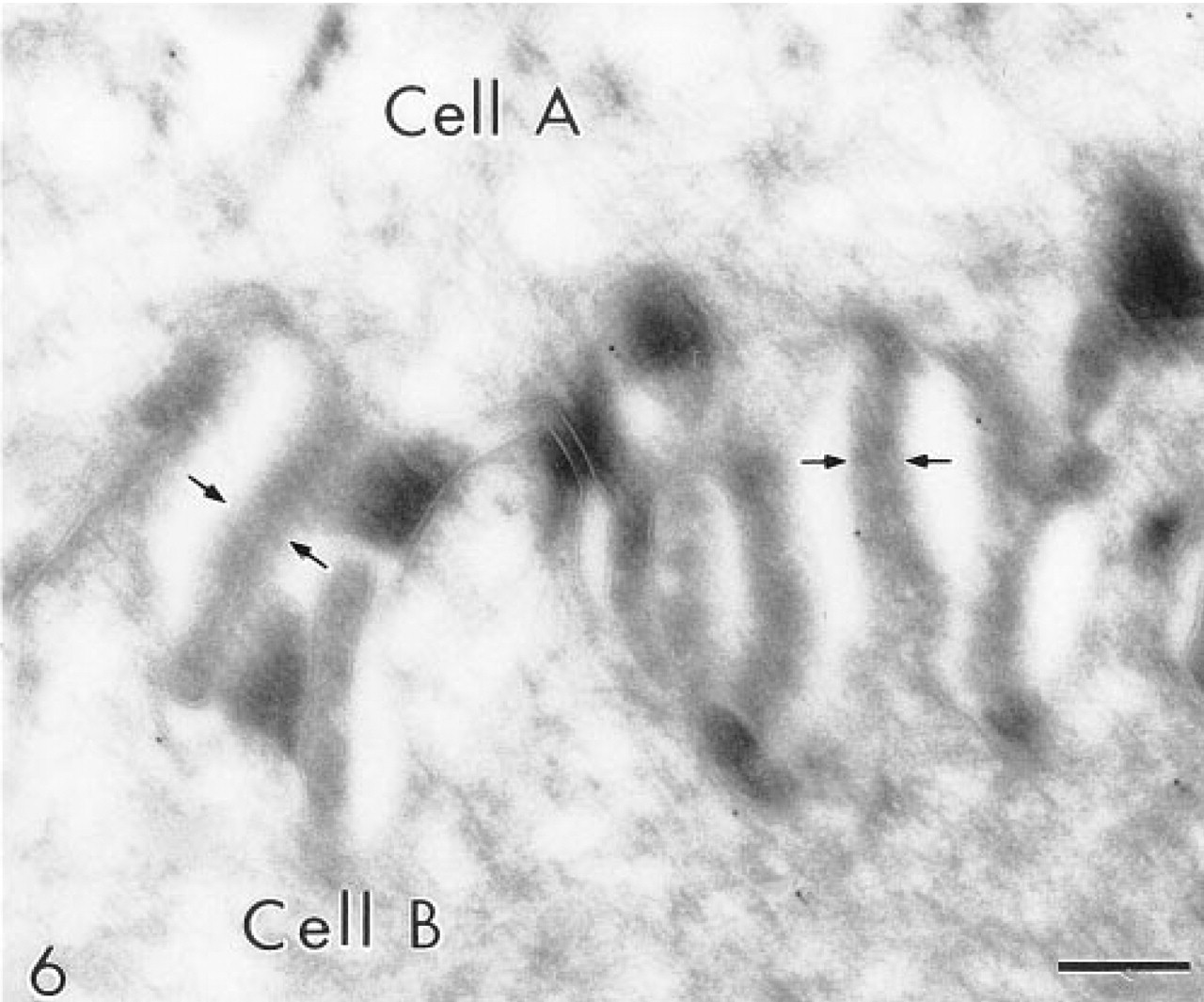
In thawed ultrathin cryosections, immunogold labeling for the CAIV isoenzyme was located principally over the plasma membranes of suprabasal cells (Figure 6). Labeling appeared to be absent over membrane structures present within desmosomes. Plasma membranes of the most superficial epithelial cells were not labeled.
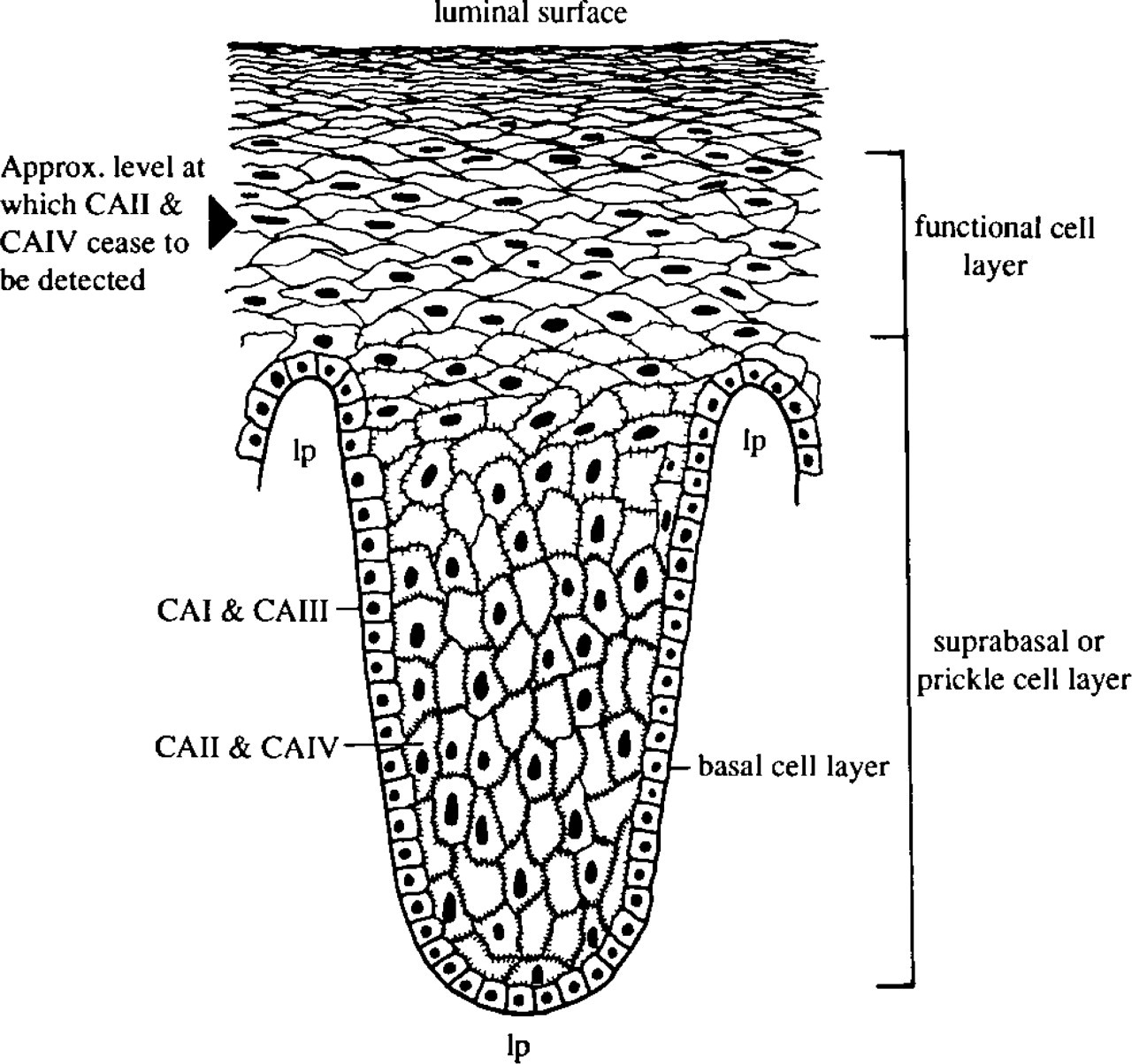
Figure 7 illustrates the different regions of the esophageal epithelium and distribution of the carbonic anhydrase isoenzymes.
Western Blots of CAI, II, III, and IV
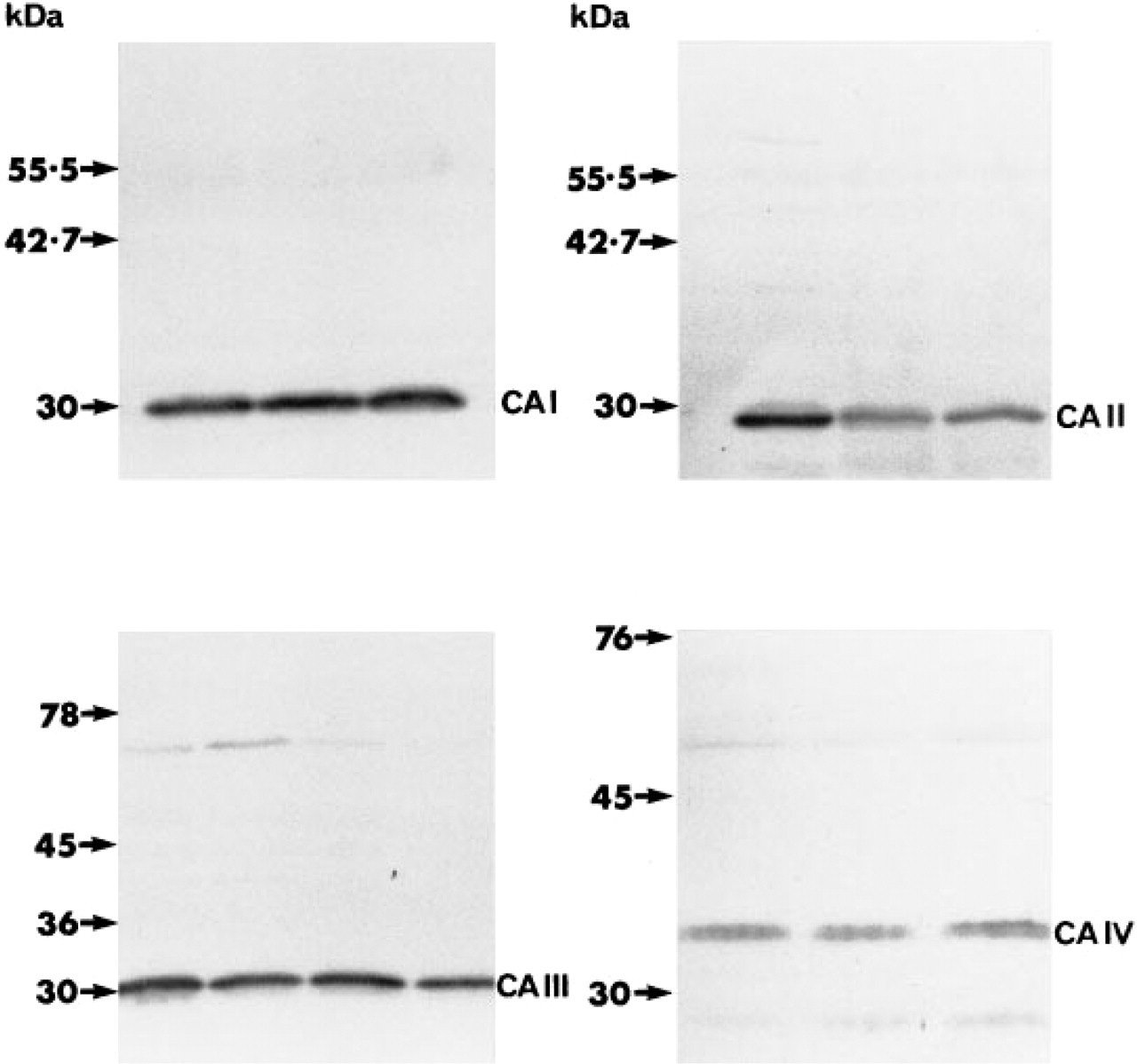
All four isoenzymes were detected in the epithelial protein extracts. CAI, II, and III localized close to 30 KD, with CAIV at or about 35 KD (Figure 8). Additional weakly stained polypeptide bands were consistently seen in CAIV blots in the region of 74–60 KD and 27–20 KD. The former were assumed to be due to nonspecific crossreaction with other esophageal proteins, and the latter to proteolytic cleavage of intact CAIV (advice from Dr. A. Waheed, Edward A. Doisy Department of Biochemistry and Molecular Biology, St Louis University Health Sciences Center, St Louis, MO).
Discussion
Four carbonic anhydrase isoenzymes (CAI, II, III, and IV) have been identified by immunohistochemistry and Western blotting in human esophageal epithelial cells. This supports an earlier study (Christie et al., 1995) in which the enzyme was localized in these cells using the cobalt catalytic enzyme histochemical technique (Hansson, 1967), and confirms the presence of the CAII isoenzyme detected by Parkkila et al. (1994).
In our previous study the cell surface localization of the enzyme was the most prominent feature. We have now confirmed immunohistochemically at the light and electron microscopic levels that this is due to the presence of CAIV on the plasma membranes. However, we have not been able to identify at the electron microscopic level the CAIV-containing elements close to the nuclei that were apparent by light microscopy. The cytoplasmic reaction in the basal and suprabasal cells obtained previously can now be attributed to CAs I, II, and III.
The sharp demarcation at the basal cell layer between CAI and III and II and IV suggests that it is the location of the cells that largely dictates the isoenzymes expressed rather than the specific epithelial cell type. Both basal and parabasal compartments contain stem, committed progenitor, and dividing cells, yet CAI and III were never detected suprabasally, nor was CAII demonstrated in the basal cell layer. Apart from the occasional presence of CAIV in basal cells, the isoenzymes are divided at the basal-suprabasal interface between low-activity CAI and III (basal) and high-activity CAII and IV (suprabasal).
The role(s) of carbonic anhydrase in the esophageal epithelium is open to speculation. However, it is likely to be dictated to some extent by the anaerobic conditions in the epithelium and its need to rid itself of CO2 and H2O produced by oxidative metabolism. Accordingly, CAI and III may facilitate the efflux of CO2 and H2O from the metabolically active basal cells to blood vessels in the adjacent lamina propria. Similarly, as the basal cells mature and move suprabasally, they may require the more efficient CAII and IV to achieve the same end. Another conceivable role may be in cell volume regulation. When esophageal basal cells move into the prickle cell region they undergo a dramatic increase in volume (D. Hopwood, personal communication), and in this situation CAII may supply H+ and HCO3 - for the Na+/H+ and Cl-/HCO3 - exchange during electrolyte gain (Macknight et al., 1994; Strange, 1994).
Finally, and perhaps most importantly, carbonic anhydrase probably plays a significant part in protecting the esophageal mucosa. Bicarbonate derived from saliva and esophageal submucosal glands has been demonstrated to protect against acidic gastric refluxate (Brown et al., 1993,1995; Helm et al., 1987). Tobey et al. (1989), using rabbit esophagus in an Ussing chamber, showed that intercellular bicarbonate ions from adventitial perfusion protected the epithelium from acid injury, which was assessed histologically and by decline in voltage and electrical resistance measured across the esophageal mucosa. In a further in vitro study, Tobey et al. (1992) showed that both basal and prickle epithelial cells possessed a Na+-dependent, amiloride-inhibitable process consistent with a Na+/H+ antiporter which could protect against an acid load, and also that the prickle cells had a greater buffering capacity at acid pH levels than basal cells. This latter feature fits well with our finding of high-activity CA isoenzymes in these cells as opposed to the basal cells. Logically, because prickle cells are closer to the lumen they will require a more rapid response mechanism to acidification. A comparable role in gastric acid neutralization has been assigned to CAII in murine proximal duodenum, where the enzyme is located on the brush border and presumably provides a protective bicarbonate efflux (Spicer, 1993).
Carbonic anhydrase I in human esophageal basal epithelial cells. lp, lamina propria. Original magnification X 350. Bar = 30 μm.
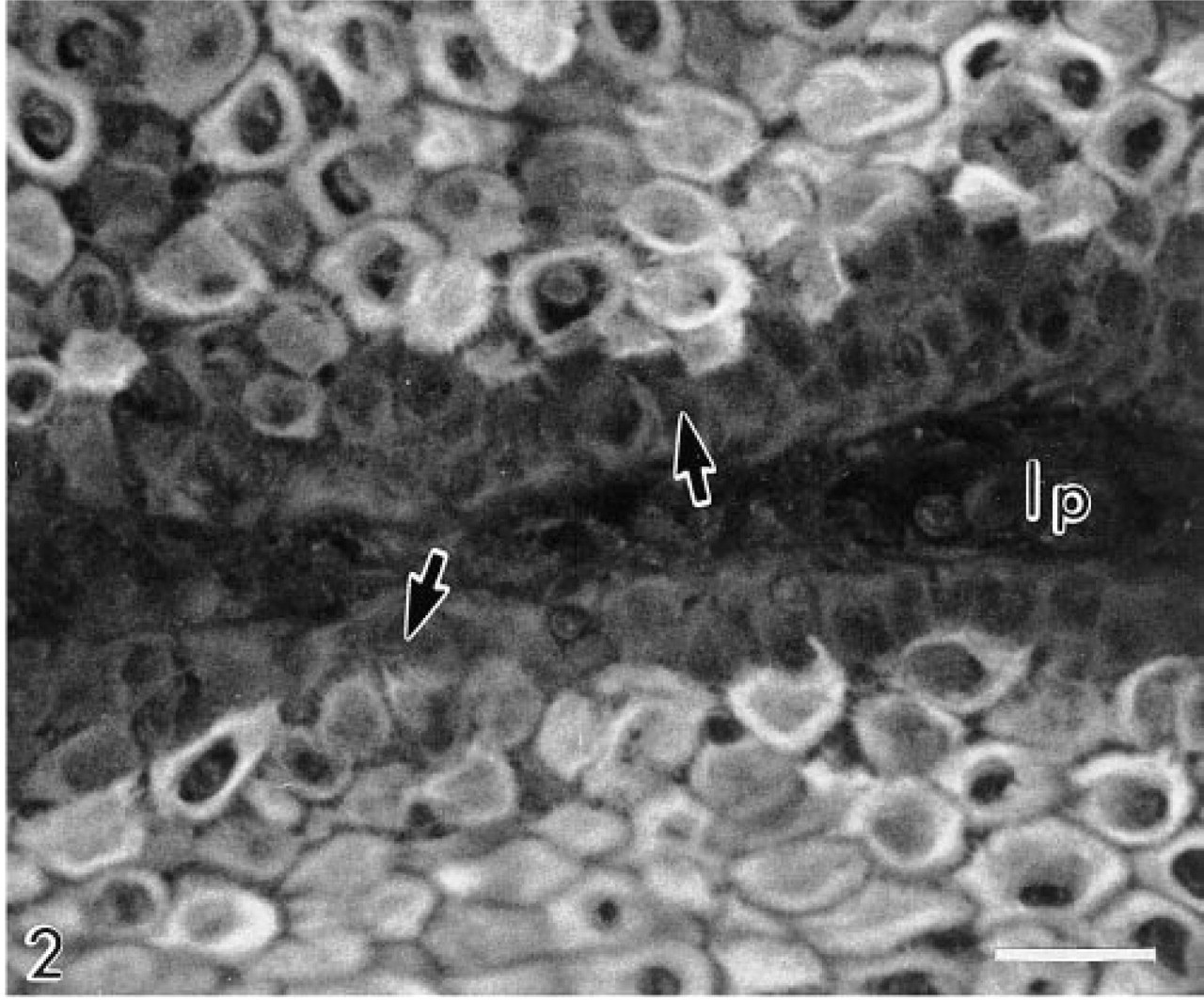
Carbonic anhydrase II in human esophageal prickle epithelial cells. Note the absence of the isoenzyme from the basal cells (arrows) and the variation in immunoreactivity between individual cells. lp, lamina propria. Original magnification X 350. Bar = 30 μm.
Schematic representation of an esophageal epithelial papilla, showing the different cell strata and distribution of CA isoenzymes. lp, lamina propria.
Western blots of solubilized human esophageal epithelial proteins. Antibodies against CAI, II, and III recognize major polypeptide bands at 30 KD and CAIV at 35 KD.
Carbonic anhydrase III in human esophageal basal epithelial cells. The immunoreactivity of this isoenzyme was consistently stronger than CAI in the basal cells. lp, lamina propria. Original magnification X 350. Bar = 30 μm.
Carbonic anhydrase III in a rosette of human esophageal basal epithelial cells surrounding a lamina proprial papilla. Immunoreactive blood vessels and RBCs, which would normally be present in such a section, are absent because the pinch biopsy procedure removes the epithelium from the lamina propria, leaving the vessels behind. Original magnification X 875. Bar = 10 μm.
Carbonic anhydrase IV in human esophageal prickle cells. Individual cells are delineated by a diffuse halo and particulate reaction product. Note the reaction product associated with the nuclei. Original magnification X 550. Bar = 20 μm.
Immunoelectron micrograph showing carbonic anhydrase IV immunolabeling principally over the plasma membranes of adjacent human esophageal epithelial prickle cells. Arrows indicate plasma membranes. Original magnification X 15,000. Bar = 250 nm.
Carbonic anhydrase in esophageal epithelial cells may participate in CO2 and H2O elimination, ion exchange during growth, and may serve as an intrinsic source of bicarbonate, augmenting that of saliva and esophageal submucosal gland secretions in protecting the mucosa against corrosive gastric acid.
Footnotes
Acknowledgements
We gratefully acknowledge the generosity of the following for their donations of carbonic anhydrase antibodies: Dr W.S. Sly, St Louis University Health Sciences Center, St Louis, MO (CAIV); Dr A.-K. Parkkila (CAI and CAII) and Dr H.K. Väänänen (CAIII), both of the University of Oulu, Oulu, Finland. We also thank Ms Jennifer Collie for excellent secretarial assistance in the preparation of the manuscript.
