Abstract
Whatever the protocol used, electron microscopic immunogold detection still suffers from a lack of sensitivity. In rat supraoptico-posthypophyseal neurons, unlabeled secretory granules are always detectable after electron microscopic immunocytochemistry, and their real status remains questionable. To improve the sensitivity of this approach, we assessed a protocol to visualize either one or the other of co-localized neuropeptides, i.e., vasopressin or galanin, after two successive rounds of immunogold with the same primary antibody performed on both faces of the grid. The use of different-sized gold particles enabled us to visualize the respective contribution of each face of the section to the final labeling. Our results showed a moderate but significant increase in both the proportion of labeled granules and the labeling intensity. Although limited, this improvement of immunogold detection strengthens the relevance of quantitative studies at the electron microscopic level, likely to reveal fine variations of the neuron peptidergic content. However, this enhancement depended on the peptide studied. The present data confirmed a progressive decrease of vasopressin immunoreactivity, already suggested by the single-staining procedure, all along the hypothalamo-posthypophyseal tract. In contrast, labeling intensity for galanin remained steady. Finally, our double-face labeling supported a preferential routing of galanin-containing secretory granules towards dendrites.
M
Most of the many authors who have used post-embedding immunogold methods on endocrine or neuroendocrine materials, such as the hypothalamo-posthypophyseal tract, attempted to refine the immunodetection procedures. Current protocols now allow immunolabeling of individual secretory granules and study of their intragranular content. Improving the sensitivity is essential for the reliability of quantitative analysis, which is a prerequisite to the study of dynamic cell biology processes. In particular, neuropeptide maturation mechanisms and pathways analysis require an accurate assessment of secretory granule content in various subcellular compartments. In addition, qualitative aspects, particularly the lack of labeling of some neurosecretory granules, would also profit from an increase in method sensitivity. Are these granules negative because of limited method sensitivity or because of an actual lack of antigen? Although their presence has rarely been discussed, unlabeled granules have been occasionally described, in particular by Merighi (1992) and Bendayan (1995), and technical problems were then postulated. Alternatively, El Majdoubi et al. (1996) also proposed several hypothesis (see Discussion).
Because gold-conjugated antibodies act as surface markers on resin-embedded tissue, one must first consider the access of the granules to the surface of the section (Bendayan 1995). Although granules have diameters larger than the thickness of ultrathin sections, some of them do not reach the surface of the section, because of their random distribution within the tissue (see Figure 1). Consequently, these granules remained unstained after a single immunogold application (Figure 1I) but, according to Bendayan (1982), they could be marked by a second incubation on the other side of the section (Figure 1II) if they do contain the target antigen. Such a double-face labeling might increase labeling intensity and would minimize errors in quantitative study of the proportion of labeled granules and the labeling intensity. Finally, this method is likely to precisely reveal weak variations of neuropeptide amounts related to the physiological state or to the subcellular localization.
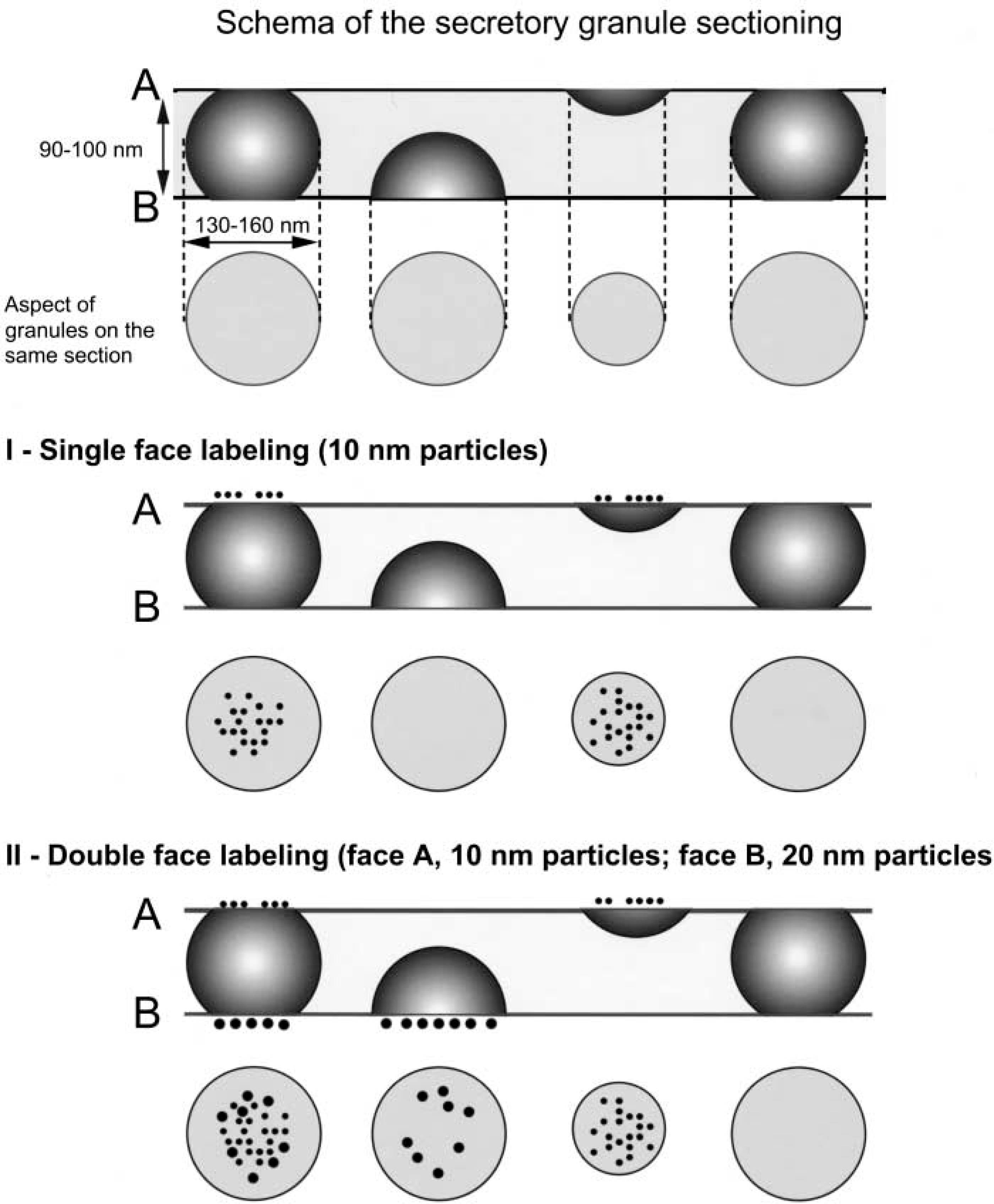
Schema of the neurosecretory granule sectioning. (
In the hypothalamo-neurohypophyseal model, we have routinely used colloidal gold as a marker for two neuropeptides of the rat hypothalamic supraoptic nucleus (SON): vasopressin (AVP) and galanin (GAL). A quantitative study of their distribution was recently reported on magnocellular cell bodies and dendrites in the SON and in terminals in the neural lobe of the pituitary (Landry et al. 2003). The main result of this study was to demonstrate a differential routing for these co-localized peptides. In this previous work, some unlabeled secretory granules were also found. In the present study we have addressed the question of secretory granule content by using a double-face immunogold detection of AVP or GAL. Both the presence of the major neurohormone AVP in all neuro-secretory granules and possible variations of labeling intensity for AVP and GAL all along the hypothalamo-neurohypophyseal tract were considered.
Materials and Methods
Animals and Tissue Preparation
Ten male Sprague-Dawley rats (250–300 g) received food and tapwater ad libitum. They were deeply anesthetized with pentobarbital and were perfused through the ascending aorta first with 50 ml Tyrode solution and then with 300 ml of an ice-cold fixative containing 4% paraformaldehyde and 0.1% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4 (PB). The brains were rapidly dissected out, postfixed in the same fixative without glutaraldehyde for 2 hr, and rinsed in PBS, 0.01 M, pH 7.4. Free-floating sections (400-mm thickness) were cut on a Vibratome and then permeabilized using a previously described freeze-thaw protocol (Landry and Hökfelt 1998).
Vibratome sections were embedded at −30C in hydrophilic Lowicryl K4M resin (Sigma; St Louis, MO) by the progressive temperature-lowering technique using a Reichert AFS system (Leica; Vienna, Austria) according to the instruction manual.
Immunoelectron Microscopy
Ultrathin sections (60–100-nm) from Lowicryl blocks were mounted on gold grids (300-mesh) and submitted to the postembedding immunogold procedure for the detection of vasopressin (AVP) or galanin (GAL) as described elsewhere (Ozawa et al. 1994; Vila-Porcile and Barret 1996). Briefly, the grids were successively floated on drops of 50 mM ammonium chloride in TBS (Tris-HCl buffer 50 mM, NaCl 150 mM, pH 7.45) for 15 min and of TBS supplemented with 1% bovine serum albumin (TBS/BSA) for 1 hr. Grids were then incubated overnight at 4C on drops of the primary antibody (anti-AVP; Chemicon, Temecula, CA, diluted 1:4000 in TBS/BSA, or anti-GAL (a gift from Dr. G. Tramu, diluted 1:200 in TBS/BSA). After several washings on large drops of TBS/BSA, the grids were incubated for 1 hr at RT on goat anti-rabbit colloidal gold conjugate (GAR 10 or 20 nm, Biocell, Cardiff, UK; diluted 1:50–1:100 in TBS/BSA). They were then rinsed once with TBS/BSA, twice with TBS, and twice with distilled water (5 min each) and air-dried.
After complete drying, the procedure was repeated on the other side of the grid (Bendayan 1982) using the same specific primary antibody and a colloidal gold conjugate of either the same or a different size. Finally, a rapid (10–12 sec) staining was performed on the grid side supporting the sections with 2% uranyl acetate in 50% ethanol.
As controls for immunogold specificity, primary antibodies were omitted or replaced by normal rabbit serum, gold conjugates being applied as usual.
Two types of double-labeling experiments were performed with the same anti-AVP (or anti-GAL) antibody, using either the same 10-nm gold particles on both sides of the grids or two differently sized particles (10 and 20 nm), to detect “newly” labeled granules, i.e., granules labeled only after the second labeling round.
Grids were observed under a JEOL 100 CX II electron microscope at a ×20,000 magnification.
On standard micrographs (×40,000), the diameters of a sample (n = 280) of labeled and unlabeled neurosecretory granules were measured in the three neuron compartments.
Quantification
Quantification of labeled neurosecretory granules in the different compartments of magnocellular hypothalamic neurons (i.e., perikarya, neurites, and posterior pituitary nerve endings) was performed on printed micrographs (×40,000) overlaid with a squared transparent sheet. Labeled granules were counted manually by point counting. Results were expressed as percentages of labeled granules related to the total number of granules (± SEM) in a given compartment. SEM was calculated with the one-way ANOVA program, including Fischer's, Scheffé's and t-tests, p<0.01 being considered significant.
In addition, on the same micrographs, the number of gold particles present on secretory granules was also counted and reported to the total number of labeled granules, thus providing a mean density of labeling (mean number of particles per granule ± SEM) in each neuron compartment. Frequencies of granule labeling within the three compartments were evaluated and expressed as percentages of granules distributed according to the number of gold particles they hold.
A comparison with single-labeled sections was systematically established for each size of gold particles used in the different experiments.
Results
Single- and double-face labelings are illustrated in Figures 2 and 3 for AVP and in Figure 4 for GAL. In both cases, the labeling appeared restricted to the neurosecretory granules in the three subcellular compartments (perikarya, dendrites, and neural lobe terminals), as defined in a previous study (Landry et al. 2003). Whatever the immunogold used and the compartment considered, there was apparently no preferential distribution of the gold particles over the granule matrix. Granule diameters were between 120 and 160 nm. As concerns the general aspect of perikarya, mature granules scattered in the entire cytoplasm were labeled, as well as immature granules in the Golgi area, and even forming ones within Golgi saccules (Figures 2a and 2b).
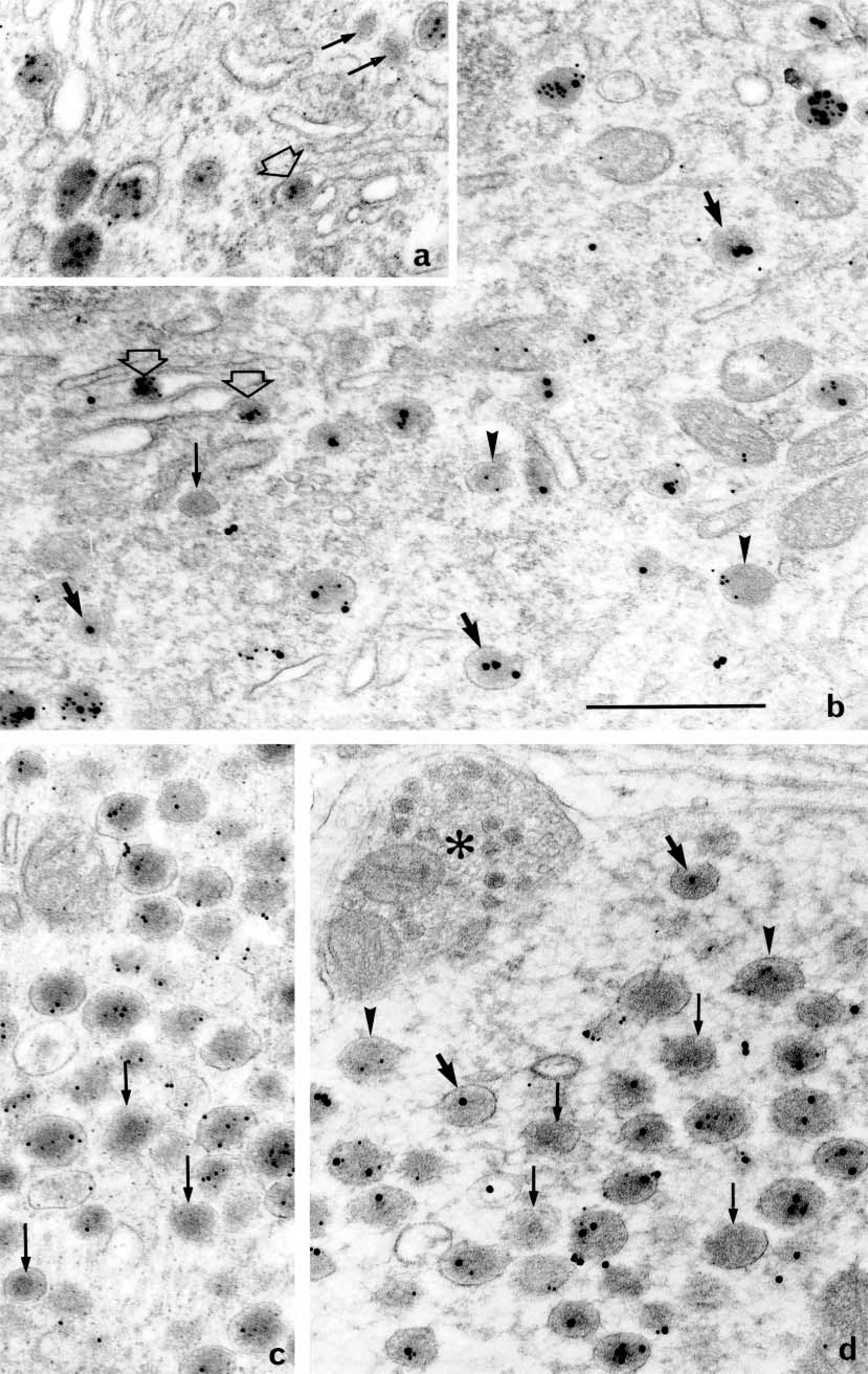
Labeling for AVP in the supraoptic nucleus (×60,000). (
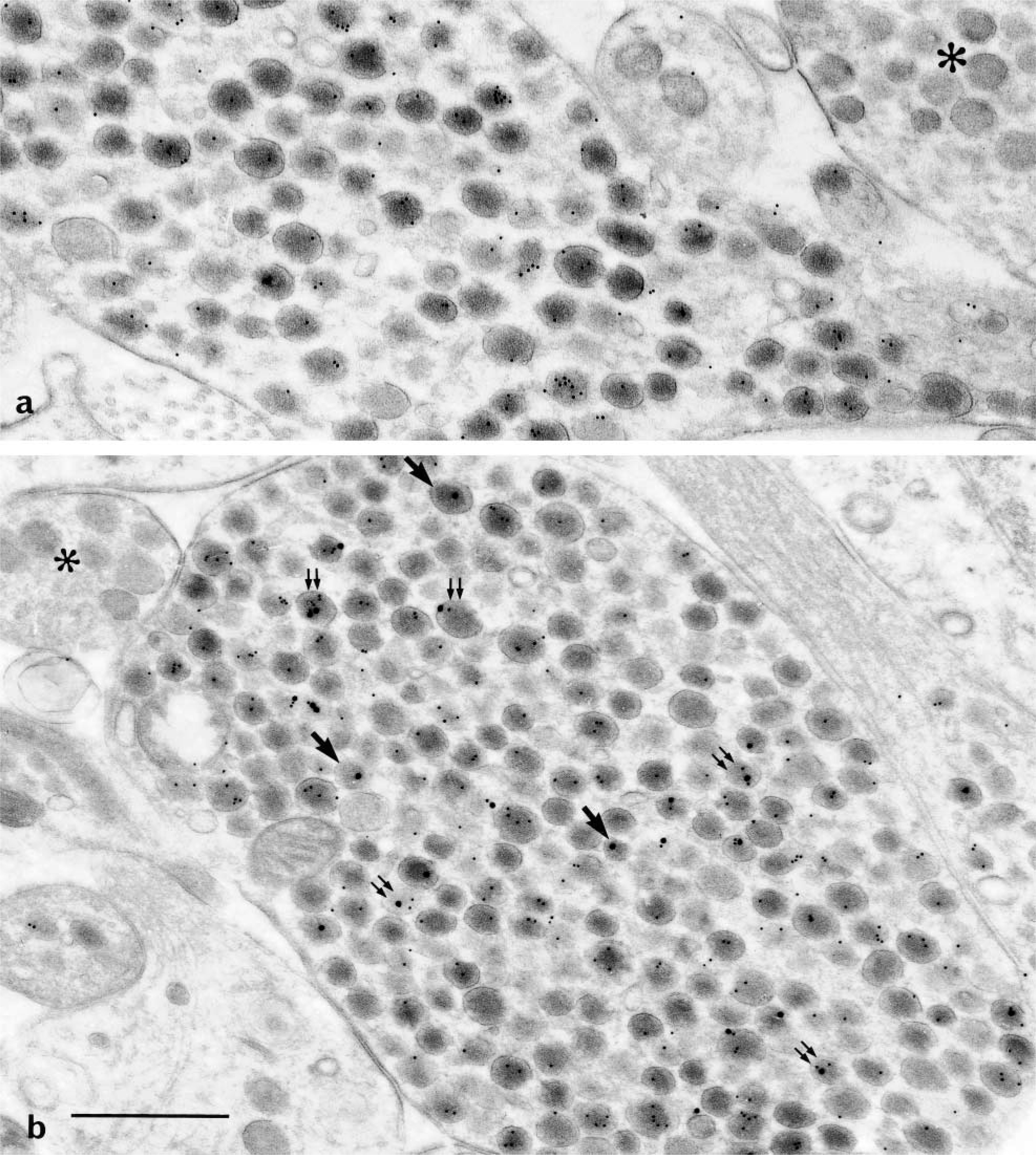
Labeling for AVP in the neural lobe (×50,000). (
Single-face immunodetection of AVP (Figures 2c and 3a) resulted in the labeling of a majority of secretory granules. After double-face detections for AVP (Figures 2a, 2b, 2d, and 3b), stronger labeling was seen. Labeling enhancement was more marked when the same 10-nm gold particles (Figures 2a and 5a) were used on both faces of the sections, owing to a higher sensitivity of the small colloidal gold. After double-face immunodetection, the mean percentage of labeled granules increased by 15–20% compared to single-face staining (Figure 5a). This increase was uniform in all the three subcellular compartments. Such an improvement in AVP immunodetection led to the labeling of almost all secretory granules in perikarya (Figure 5a). In some perikarya, the rate of labeling even reached 100%. However, in other compartments a larger number of granules appeared devoid of gold particles, nevertheless keeping similar features as in cell bodies. Size and electron density were not modified in any of the three compartments of the magnocellular neurons, i.e., perikarya (Figure 2b), dendrites (Figure 2d), and PN (Figure 3b).
Percentages of labeled granules. (
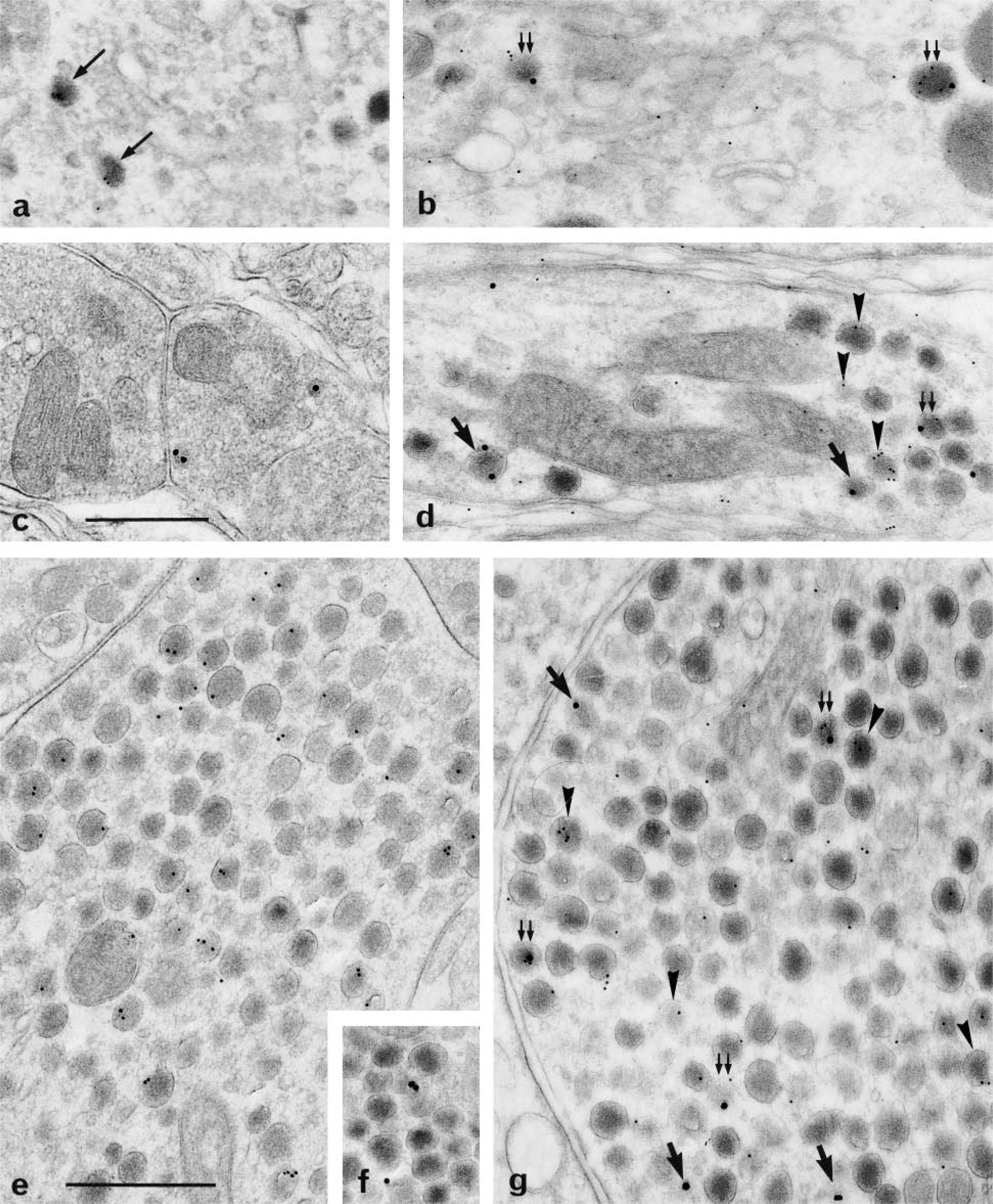
Less than one fifth of the total number of secretory granules (Figure 5b) was immunoreactive after single-face labeling for GAL in perikarya (Figure 4a), dendrites (Figure 4c), and PN (Figures 4e and 4f). Double-face detections for GAL increased the proportion of positive granules (Figures 4b, 4d, and 4g). However, this improvement was restricted to neuron processes but was particularly obvious in dendrites with a 3.5-fold increase (Figure 5b). In all cases, many secretory granules remained unlabeled for GAL.
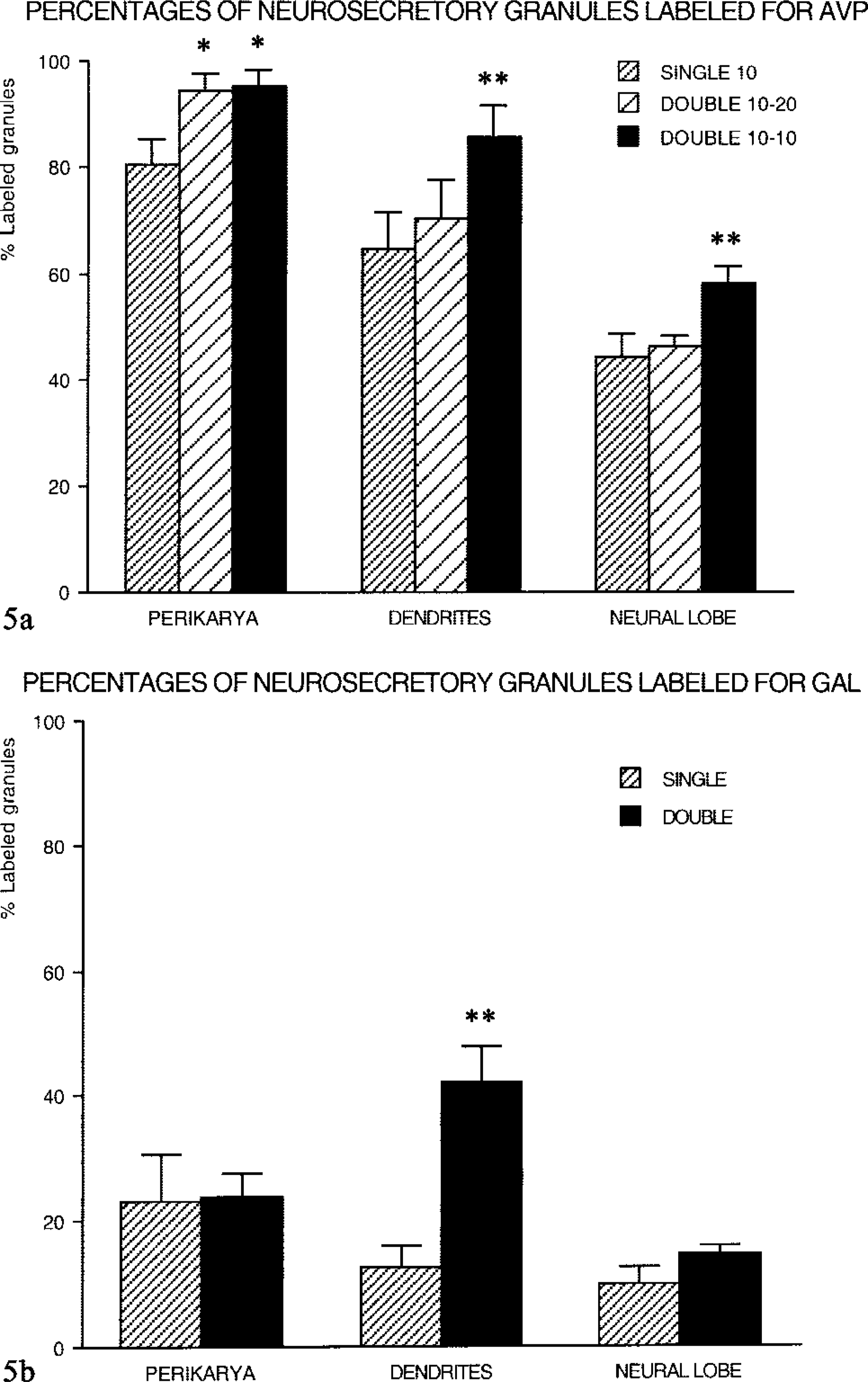
Labeling for GAL (×50,000). (
Results from quantitative analysis indicated that not only the number of labeled granules but also the number of gold particles per granule was higher after double-face labeling for AVP, up to 20–22 gold particles per granule in perikarya. The mean number of gold particles per granule reached 9.2 in perikarya compared to 5.7 after single-face labeling (Figure 6a). The efficiency of double-face labeling appeared slighter in dendrites and PN (Figure 6a). For intensity of gold labeling for GAL, a slight augmentation was observed after double staining in dendrites, and more markedly in the terminals, with respect to single labeling. Moreover, in contrast to the results obtained after single staining, the double labeling revealed that the density of gold particles remained constant, regardless of the subcellular compartment considered, and did not decrease from the perikarya to neural lobe (Figure 6b).
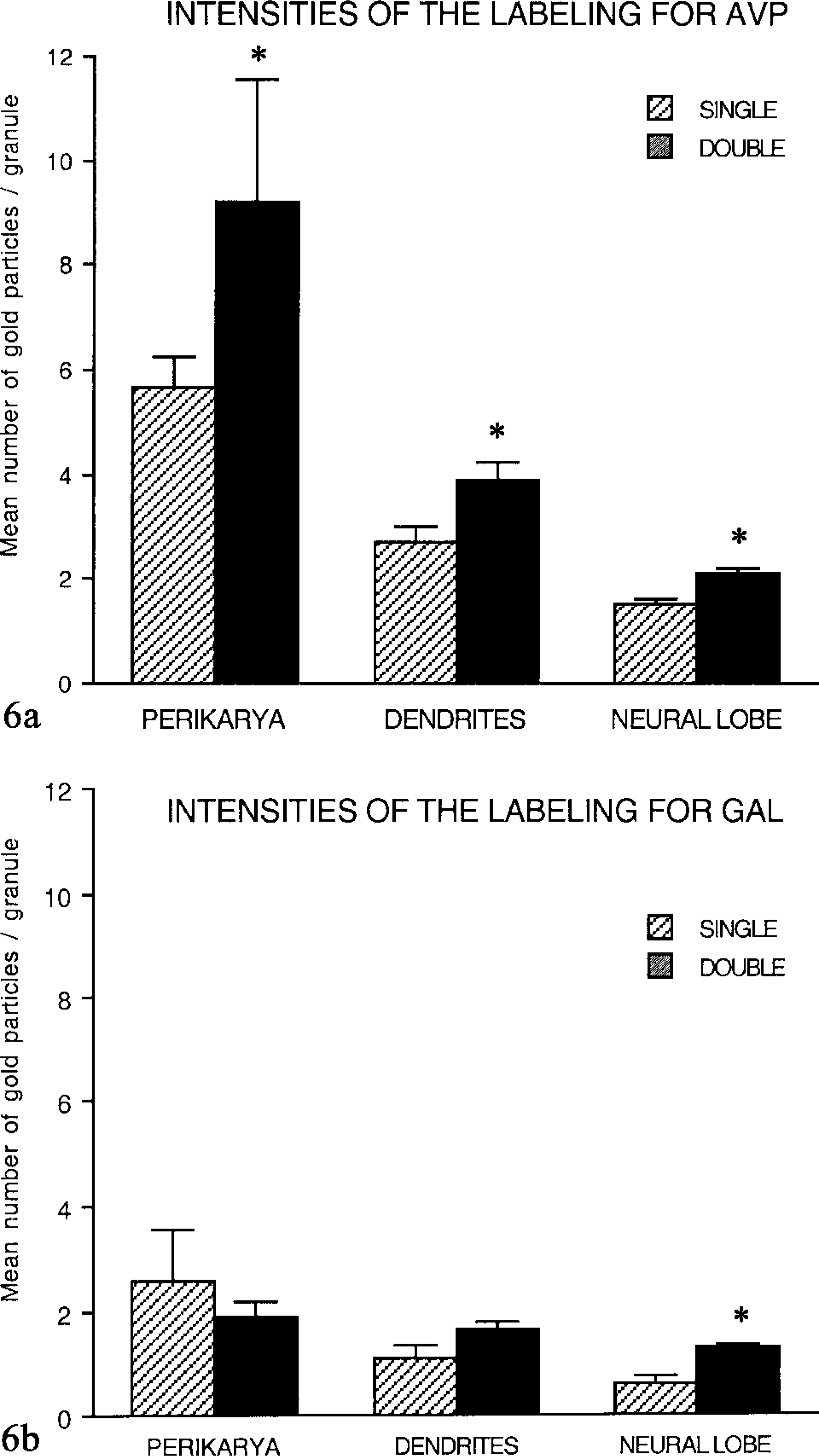
Labeling intensities: Comparison of single (S)- and double (D)-face labeling (pooled data). (
A striking feature of our study is the evident decrease of the labeling intensity for AVP from perikarya to either dendrites or PN terminals, all along the three compartments of the magnocellular neurons, even when the double-side approach was used. Indeed, frequency profiles established for AVP (Figure 7a) showed that, in perikarya, 10% of the labeled granules held five to ten or more particles, whereas in dendrites the major part (70%) of the granules were labeled with one or two particles and none with more than ten. In nerve endings, 85% of the granules were similarly weakly marked. In contrast, no prominent variations in the intensity of GAL labeling could be detected all along the neurosecretory pathway (Figure 7b). However, it should be acknowledged that this intensity remained faint, most of the granules being marked with only one or two gold particles (Figure 7b). Such a labeling was nevertheless considered as significant because the preparations appeared background-free.
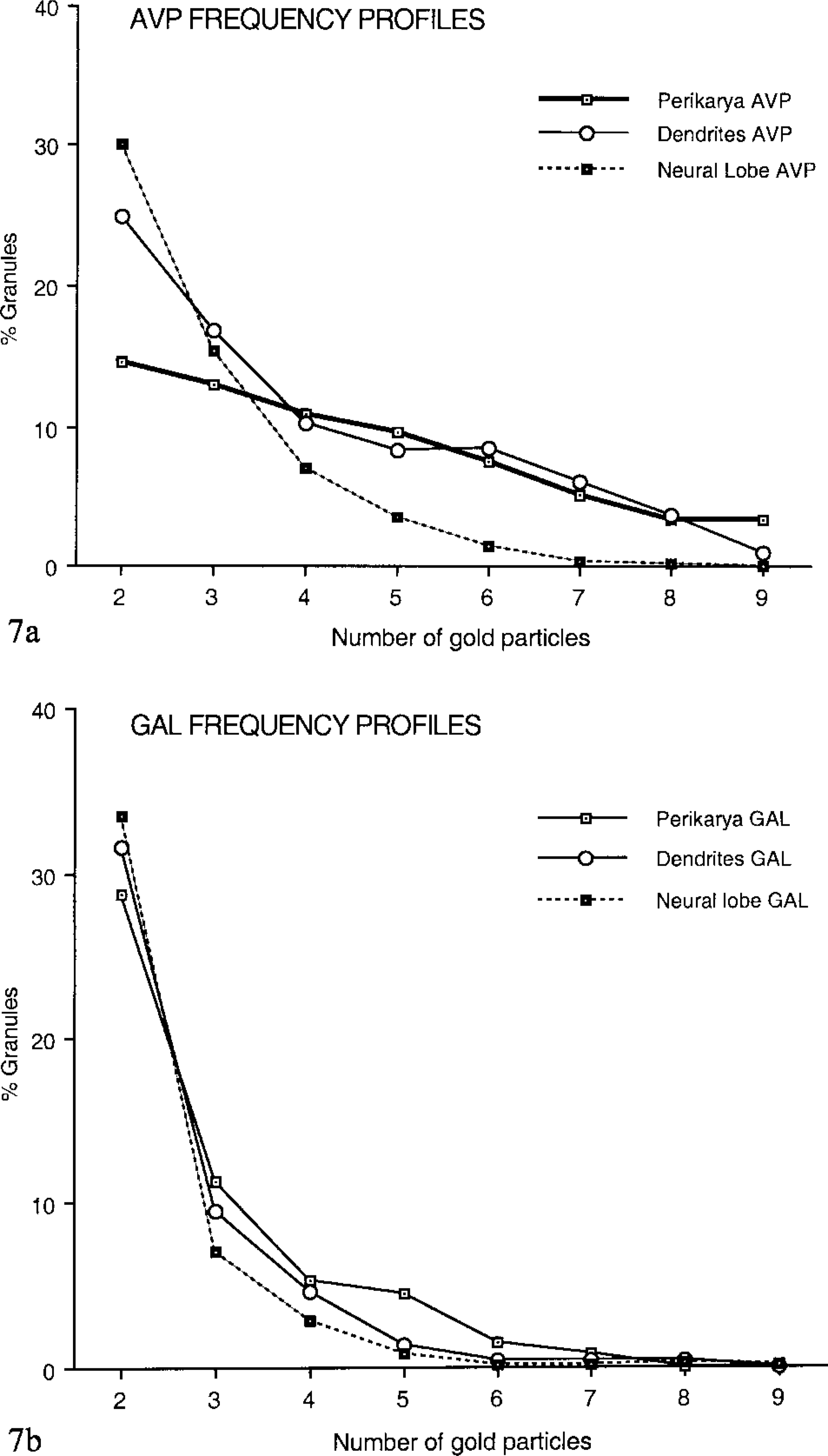
Frequency profiles expressed as the percentages of granules plotted against the numbers of immunogold particles per granule, the latter having been distributed into classes (see Materials and Methods). (
Discussion
The detection at the ultrastructural level of two neuropeptides, vasopressin (AVP) and galanin (GAL), in the magnocellular neurons of the adult rat hypothalamus has confirmed that the immunodetectable molecules are confined to neurosecretory granules. The intragranular random dispersion of gold particles is in contrast to the specific perigranular distribution observed in some other types of neurons, e.g., primary sensory neurons (Zhang et al. 1995). Measurements of granule diameters have provided values in agreement with data reported by other authors (Whitnall et al. 1985b; Pow and Morris 1991; Sonnemans et al. 1996). The mean size of secretory granules was obviously above the thickness of our ultrathin sections, thus excluding the presence of granules completely included within resin and having no access to any surface of the section.
The double-face method used in this study allowed us to improve the immunodetection at two levels, i.e., the number of responsive granules and the number of gold particles over each positive granule. Given the better sensitivity of this approach, the immunodetection threshold was lowered and this method is likely to provide more reliable quantitative results. The resin and the embedding procedure should be carefully evaluated because, in our hands, embedding in acrylic resin (Lowicryl) with a progressive lowering of temperature protocol provided the best compromise between antigenicity and preservation of morphology.
However, the increase was rather moderate both in percentage and intensity and depended on the target peptide and/or the antibody used. It is known that a non-negligible part of immunogold signal is lost during double-labeling procedures (Vila-Porcile and Corvol 1998), even when a postfixation with glutaraldehyde has followed the colloidal gold application. This loss could represent a limiting factor for maximal enhancement of the staining. In our present work, other methodological attempts were performed, i.e., a tyramide signal amplification (Mayer and Bendayan 1999), without improving our final results.
This technique has nevertheless allowed us to detect AVP in all the granules present in some perikarya. Moreover, unlabeled granules were never found within the trans-Golgi saccules, either in these well-labeled cells or in lesser responsive ones. Hence, we can infer that all nascent granules do contain AVP. Only intragranular maturation processes could lead to an under-detection of the peptide. This is in agreement with the decrease in the labeling intensity for AVP.
The better sensitivity of the double-face approach demonstrates that a higher percentage of GAL-labeled granules was detected in dendrites, in agreement with our previous immunodetections.
Because all experiments were performed on the same material with the same set of techniques, all differences in the results of the double-face protocol do not rely on methodological hindrance. They might instead reflect specific biological features of the molecules or compartments under investigation.
The lack of changes in GAL labeling intensity suggests that this particular neuropeptide remains uniformly detectable in all compartments. In that case, the double-face protocol appears well suited to assess the preferential localization of GAL-positive granules in the dendritic compartment, as previously reported for this peptide (Landry et al. 2003). It further supports the hypothesis of a preferential routing of GAL towards dendritic processes and suggests possible dendritic release of GAL within the SON (Landry et al. 2003). Such a somato-dendritic release has been described in the same hypothalamic neurons for the major neurohormones (Pow and Morris 1989; Ludwig and Landgraf 1992; Morris and Pow 1993; Morris et al. 1998). Neuropeptide dendritic release in the hypothalamus is calcium-dependent (Di Scala-Guénot et al. 1987) and is stimulated by intracellular calcium stores (Ludwig et al. 2002). Moreover, this process is regulated by the hypothalamic neuropeptides themselves (Lambert et al. 1994) and was recently shown to depend on physiological conditions, e.g., lactation (de Kock et al. 2003).
Despite the use of a double-face approach, unlabeled granules were nevertheless observed in most ultrathin sections. This lack of labeling confirms that AVP remains undetectable in a subpopulation of secretory granules of the vasopressinergic magnocellular neurons. In the same hypothalamo-hypophyseal model, El Majdoubi et al. (1996) have found neuro-secretory granules not responsive to chromogranin A (CGA) detection. They have raised several hypotheses to elucidate the apparent absence of CGA, i.e., the absence of peptide or its very low amount, or even its “extensive processing” into derivatives no longer recognizable by their antibody. These hypotheses could also be applied to AVP and GAL peptides.
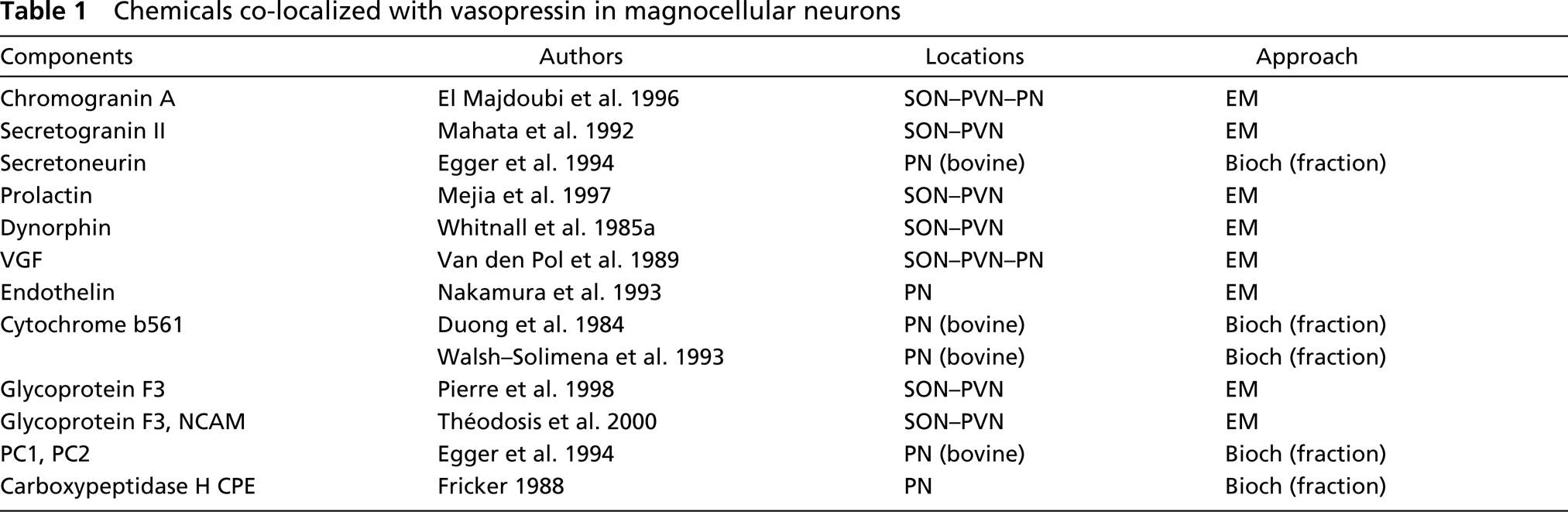
A complete absence of AVP cannot be ruled out in some neurosecretory granules. The unreactive granules may contain only some unrelated molecules, such as GAL. Indeed, in addition to AVP, its precursor and its breakdown products (Pow and Morris 1991), many non-vasopressin-related components were demonstrated to be localized in neurosecretory granules of vasopressinergic magnocellular neurons. In addition to GAL, previously demonstrated to be partly co-localized with AVP in these neurons (Melander et al. 1986; Gayman and Martin 1989; Landry et al. 2003), some of these granular constituents are listed in Table 1. They were shown to be co-localized with AVP in granules of well-identified vasopressinergic neurons either by immunogold or by co-fractionation. In addition, the previously shown existence of granules exclusively labeled for GAL is a potent argument for the possibility that such non-vasopressin-related components may constitute the core of these still vasopressin-negative granules. However, actual technical difficulties or immunoreaction hindrance must also be considered. All molecules concentrated in structures as small as secretory granules could build a mesh so tight that antibodies may have no access to the target antigen, even though the neuropeptide might still be present.
Chemicals co-localized with vasopressin in magnocellular neurons
The notion of extensive processing should be taken into account to explain the progressive decrease in labeling for AVP observed from cell bodies to nerve endings. Nordmann and Morris (1984) have demonstrated that, in hypothalamic neurons, only aged but not newly formed granules contained protein species of smaller molecular weight, which appeared to result from continued intragranular proteolysis. The intra-granular presence of all the enzymes necessary to complete processing of the precursor into vasopressin was already assumed in the 1980s (Brownstein et al. 1980). Furthermore, other maturation processes occur during the transport of secretory granules towards dendrites or nerve terminals. Peptide condensation is ongoing during transport and AVP might become less accessible to antibody. In the present study, the decrease in the number of labeled secretory granules is in agreement with such possible processing of AVP peptide from the Golgi to the neuron extremities. Moreover, the progressive decrease in the density of gold particles all along the hypothalamo-posthypophyseal tract further confirms continuous processing.
Various processes of aggregation/condensation can be considered, depending on the studied peptide. This aggregation of non-related components might occur in addition to the possible aggregation of AVP mentioned above. Compared to AVP, the intensity of GAL labeling seems to remain rather stable from NSO to dendrites or PN. This suggests the absence of GAL proteolysis and aggregation through specific mechanisms of maturation for GAL.
In conclusion, the double-face approach used in this study offers an improvement of neuropeptide-immunogold detection while confirming the lack of immunoreactivity in some secretory granules. Such an approach could be useful for fine quantitative studies at the electron microscopic level. Although moderate, the signal increase has enabled us to demonstrate specific variations of peptidergic content along the secretory pathway of magnocellular neurons, providing new clues to possible sites of release and action of neuropeptides.
Footnotes
Acknowledgements
We are grateful to the SEROMI, University of Bordeaux, for technical support.
