Abstract
There are marked changes in vascular dynamics during prolonged periods in the cold, entrance into hibernation, and arousal to euthermy. Cell-to-cell communication through gap junction channels plays a pivotal role in the control of vasomotor function. Multiple gap junction proteins are expressed in blood vessels, including connexins 37 (Cx37), 40 (Cx40), 43 (Cx43), and 45 (Cx45). Using immunolabeling techniques combined with confocal microscopy, we quantitated the levels of these connexins in coronary arterioles and the thoracic aorta of the golden hamster in four physiological conditions: normal control animals at euthermy; cold-exposed animals (before entrance into hibernation); during hibernation; and after 2-hr arousal from hibernation. In all groups, Cx37 was localized between endothelial cells of the aorta and Cx40 was observed between endothelial cells of coronary arterioles and the aorta. Cx43 was confined to smooth muscle cells of the aorta. Labeling for Cx45 was detected in the endothelium of the ascending aorta. The expression of Cx37 was significantly reduced in cold-exposed, hibernating, and aroused animals. Immunolabeling for Cx40 was increased in the coronary arteriolar endothelium of the cold-exposed group compared with normal controls, hibernating, and aroused animals, perhaps to facilitate intercellular communication during the prolonged circulatory changes to vascular dynamics required to maintain core temperature during cold adaptation. Cx40 expression was unchanged in the aorta. Cx43 immunoexpression in the aorta remained constant under all conditions examined. These changes in connexin expression did not occur during the rapid circulatory changes associated with arousal from hibernation.
M
The change of vascular tone in response to neural and endothelial vasoactive factors is achieved by synchronization of changes in membrane potential through gap junction channels coupling endothelium, smooth muscle, and/or endothelial and smooth muscle cells (Segal and Beny 1992; Segal 1994; Chaytor et al. 1998). Gap junction channels have been shown between endothelial cells (Larson and Sheridan 1982), between smooth muscle cells (Uehara and Burnstock 1970), and between smooth muscle and endothelial cells (Spagnoli et al. 1982). Gap junctions are intercellular channels connecting the cytoplasmic compartments of adjacent cells that permit direct exchange of ions and small molecules between cells (Kumar 1999; Willecke et al. 2002). Each cell of adjacent pairs contributes one hemichannel (connexon), which is composed of six protein subunits called connexins (a multigene family of proteins). The sequencing of the human genome has identified 20 human connexins, which thus far have 19 murine homologues (Willecke et al. 2002).
Although there are variations in the reported connexin detection patterns between different animal species and vessel types, Cx37, Cx40, and Cx43 remain the major gap junction proteins expressed in blood vessels. There is a recent report of Cx45 expression in smooth muscle cells of rat aorta (Ko et al. 2001). Previous reports have demonstrated that Cx37, localized in the aorta, pulmonary, and coronary arteries, is restricted to the endothelium (Yeh et al. 1997,1998; Gabriels and Paul 1998; Ko et al. 1999; Severs 1999; van Kempen and Jongsma 1999). Cx40 (and the chick equivalent Cx42) has been observed between endothelial cells in large arteries and microvessels (Bruzzone et al. 1993; Gourdie et al. 1993; Little et al. 1995; Yeh et al. 1997,1998; Gabriels and Paul 1998; Ko et al. 1999; Severs 1999; van Kempen and Jongsma 1999) and smooth muscle cells of resistance arterioles, coronary arteries, and aorta (Little et al. 1995; van Kempen and Jongsma 1999). In addition, Cx43 has been detected in the endothelium and/or smooth muscle of various arteries (Bruzzone et al. 1993; Gourdie et al. 1993; Little et al. 1995; Yeh et al. 1997,1998; Hong and Hill 1998; Severs 1999; van Kempen and Jongsma 1999).
There is an increase in Cx43 gap junction protein expression in ventricular cardiomyocytes of cold-exposed and hibernating hamsters (Saitongdee et al. 2000). In view of the marked changes in vascular dynamics during cold exposure, hibernation, and arousal, in this study we investigated whether there are changes in gap junction expression in the vasculature under these physiological conditions. We examined the expression of Cx37, Cx40, Cx43, and Cx45 in the coronary arterioles (terminal branches of arterial tree) and aorta (a major distribution vessel) in hamsters during hibernation and after 2-hr arousal from hibernation compared to cold-exposed control (cold control) and normal control animals. The expression of gap junction proteins was determined by indirect immunofluorescence techniques combined with quantitative laser scanning confocal microscopy.
Materials and Methods
Animals
We used adult male golden hamsters (Mesocricetus auratus) aged 12 weeks and weighing 140–150 g. Breeding, maintenance, and sacrifice of the animals used in this study followed principles of good laboratory animal care and experimentation in compliance with the UK Animals (Scientific Procedures) Act, 1986. Hamsters were induced to hibernate in the laboratory by shortening the photoperiod and reducing the external ambient temperature, as previously described (Saitongdee et al. 1999a). Hamsters were allowed to hibernate for 8 weeks. During this time they entered bouts of hibernation, lasting about 3–4 days, with alternate periods of wakefulness, lasting 1–2 days. Hibernating animals were sacrificed after a minimum of 3 days into a hibernation bout, while still in deep hibernation. Even when maintained in conditions that induce hibernation, not all hamsters enter hibernation. Hamsters that did not hibernate under these conditions were used as cold control animals. These are well-matched controls because they were maintained under exactly the same conditions of daylight hours, temperature, and nourishment as those that hibernated.
A group of hibernating hamsters were aroused from hibernation by being transferred to room temperature (22C) in daylight for 2 hr. Age-matched hamsters that had not been induced to hibernate were used as normal control animals. All experimental animals were weighed, and cheek pouch and rectal temperatures were measured.
Tissue Preparation
Six hamsters from each of the four experimental groups (normal controls, cold controls, hibernating, and aroused) were sacrificed by CO2 asphyxiation and death was confirmed by cervical dislocation according to Home Office (UK) regulations covering Schedule 1 procedures. The left ventricle, thoracic aorta, and ascending aorta were quickly dissected out. The outer wall of the left ventricle was separated from the aorta; the thoracic and ascending aortas were separated and orientated for transverse sectioning, embedded in OCT, precooled in isopentane, then frozen in liquid nitrogen until sectioning. Cryosections were cut at a thickness of 10 μm (Reichert cryostat) and mounted on albumin-coated slides.
Antibodies
The immunolabeling procedures for quantitative assessment of connexin expression used polyclonal antibodies against Cx37, Cx40, Cx43 and Cx45. The details of the antibodies against the four connexins are shown in Table 1.
Details of connexin antibodies
To confirm the distribution of labeling, normal control hamster vessels were immunostained using another antibody to Cx40 raised in guinea pig (see Table 1) and two additional antibodies to Cx43 from different sources [Gap15 (rabbit) and Gap1A (mouse monoclonal)] (see Table 1). Normal rat (Sprague-Dawley, male 250 g) left ventricle and thoracic aorta were also processed alongside these hamster tissues.
Immunolabeling
To detect gap junction proteins, indirect immunofluorescence was performed on cryosections of left ventricle and aorta according to the protocol described by Saitongdee et al. (2000). In brief, tissue sections were fixed in acetone for 7–10 min, then blocked with PBS containing 0.1 M
To confirm the total labeling pattern of each connexin, we used a whole-mount immunostaining technique. Freshly dissected aortas were collected in Hank's buffered saline solution (HBSS) and then cut into small rings before processing for immunofluorescence as for the cryosections. In the last step, the rings of aorta were carefully cut and spread on the slide, endothelial layer face up, then mounted with Citifluor.
To confirm the labeling of connexin antibody in gap junctions, we used immunoelectron microscopy. The perfusion-fixed specimens (for 10 min) were continuously immersed in the same fixative (2% paraformaldehyde) for 30 min, then washed three times in PBS. The fixed specimens were dehydrated in 50% methanol for 15 min, followed by 70% and 90% methanol for 45 min each. Infiltration with LR Gold (Agar Scientific; Stansted, UK) was carried out using a 1:1 dilution of resin and then pure resin twice for 30 min, 1 hr, and overnight, respectively. The specimens were embedded in fresh LR Gold and polymerized with UV light at −20C for 24 hr.
Immunogold Labeling Techniques
The thin sections were picked up on nickel grids and incubated at room temperature successively in 1% BSA in PBS for 10 min, 1% gelatin in PBS for 10 min, and 0.02 M glycine in PBS for 3 min. Sections were immunolabeled as follows. The sections were incubated in the primary antibody, Cx40 antibody raised in rabbit, at a dilution of 1:100 with 1% BSA-Tris-buffered saline (BSA-TBS) blocking medium overnight at 4C. After washing with TBS, the sections were labeled with goat anti-rabbit-10-nm gold complex (British BioCell International; Cardiff, UK) 1:70 in TBS for 1 hr, then washed with TBS and distilled water. The labeled sections were dried and stained with uranyl acetate and lead citrate for 5 min each. Negative controls included either omission or replacement of the primary antibody with pre-immune serum.
Quantitation Techniques
In each of the four groups, sections of the left ventricle and aorta from six individual animals were used. For each animal, four or five coronary arterioles (diameter 20–100 μm) were selected from ventricular sections and five selected fields of aorta were optically sectioned in five 1-μm steps. Sections were scanned under identical parameters of imaging (pinhole), objective, filter, and laser power. Levels of PMT gain and offset were set according to procedures standardized to ensure that the image collected displayed a full range of gray level values from black (0 pixel intensity level) to peak white (255 pixel intensity level) and kept constant. Each image was signal-averaged (line average = 8) during acquisition to improve image quality by reducing noise. The entire Z-series was projected as a single composite image by superimposition. Quantification of gap junction plaque expression was performed according to an adaptation of the methods in Gourdie et al. (1991) and Haefliger et al. (1997) as described in detail in Saitongdee et al. (2000). In brief, the final image was thresholded to form a binary image. This image was then analyzed using NIH image software 1.62, which generated information on the size and number of individual gap junction plaques in a given area. Five optical fields for six animals of each group were analyzed. Confocal microscope Z-series of aorta were rotated to obtain optimal cross-sections of the tissue. An area of smooth muscle from the internal to the external elastic lamina was then demarcated digitally for quantitative analysis of connexin staining. Smooth muscle gap junction plaques were analyzed in a number of ways: first, as the total combined gap junction area/1000 μm2 (“area density”) and second as the total number of individual plaques/1000 μm2 (“numerical density”).
In the aortic endothelium, gap junction plaque area densities and numerical densities were determined per length of circumferential lining and expressed per 1000 μm2. In the coronary arterioles, however, this was not possible because nuclear staining showed that most sections were tangential. Therefore, it was necessary to demarcate the endothelial cell layer digitally for quantitative analysis and to express the plaque area and numerical densities per 1000 μm2.
Statistical Analysis
Data are expressed as the mean ± SEM of six animals in each group. Statistical analyses and tests were carried out using Minitab software (Minitab Inc. and GB-Stat School Pak). Statistical differences among the four groups were determined by analysis of variance (ANOVA) followed by Tukey's test; p<0.05 was considered significant.
Results
Animals
There were six hamsters in each experimental group. Tissues from these animals have been used in a previous report on connexin expression in cardiomyocytes during hibernation (Saitongdee et al. 2000). The body weights (g) of animals from the hibernating (104.1 ± 5.2) and aroused (98.6 ± 2.0) groups were significantly (p<0.05) lower than the cold control (136.6 ± 4.6) and normal control (165.4 ± 7.2) groups. The cold control group weighed significantly less (p<0.05) than the normal controls.
The cheek pouch and rectal temperatures (C) of the hibernating group (9.6 ± 0.5, 9.8 ± 0.5, respectively) were significantly lower (p<0.05) than those of normal control (35.1 ± 0.2, 32.2 ± 0.5), cold control (35.0 ± 0.5, 32.1 ± 0.4) and arousal (34.6 ± 0.4, 31.5 ± 0.5) groups, which were not statistically different from each other.
Immunostaining for Cx37
There was no positive immunostaining for Cx37 in the coronary arterioles of the four experimental animal groups.
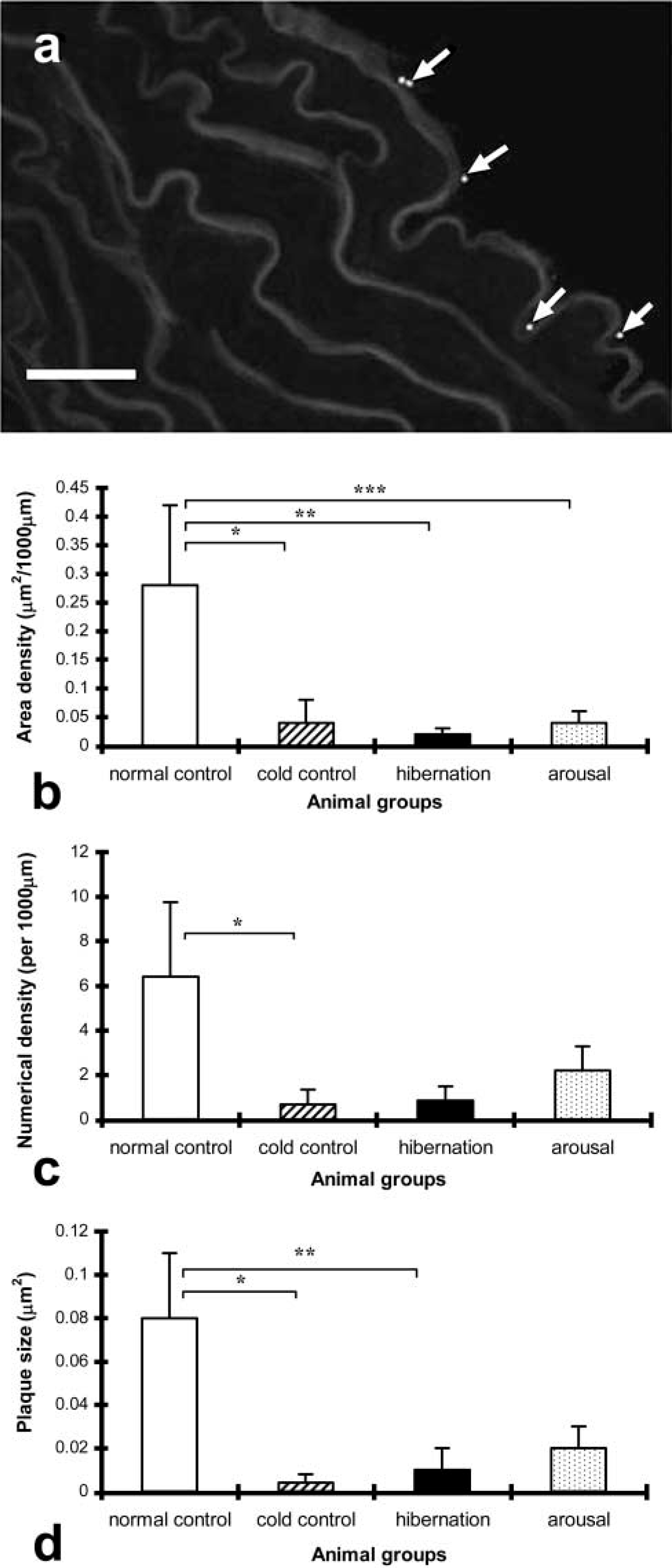
In the aorta, positive immunolabeling of Cx37 was observed between endothelial cells in all four groups with no staining in the media, as shown for the normal control hamster seen in Figure 1a. Histograms showing quantification of Cx37 gap junction plaque area, numerical densities, and plaque size in the aortas of the four animal groups are shown in Figures 1b–1d. There was a significant reduction of the Cx37 area density in the cold controls, hibernation, and arousal animal groups compared with the normal controls (p<0.05). The Cx37 numerical density and plaque size were significantly reduced in the cold controls compared with the normal controls for numerical density and cold control plus hibernating for the plaque size. There was a trend for the hibernating and aroused to be similarly reduced, although statistical significance was not reached.
(
Immunostaining for Cx40
Positive Cx40 immunolabeling was confined to the endothelium of coronary arterioles, with no staining in the media in all groups examined. Cx40 staining was prominent and appeared as bright fluorescent puncta in the luminal aspect of the coronary arterioles, and was consistently seen as a comb-like pattern along the intercellular membrane of adjacent endothelial cells (Figures 2a–2d). The localization of Cx40 immunostaining on endothelial cells, rather than on smooth muscle cells, was verified by co-labeling with anti-myosin followed by secondary antibodies conjugated to Cy3 (results not shown).
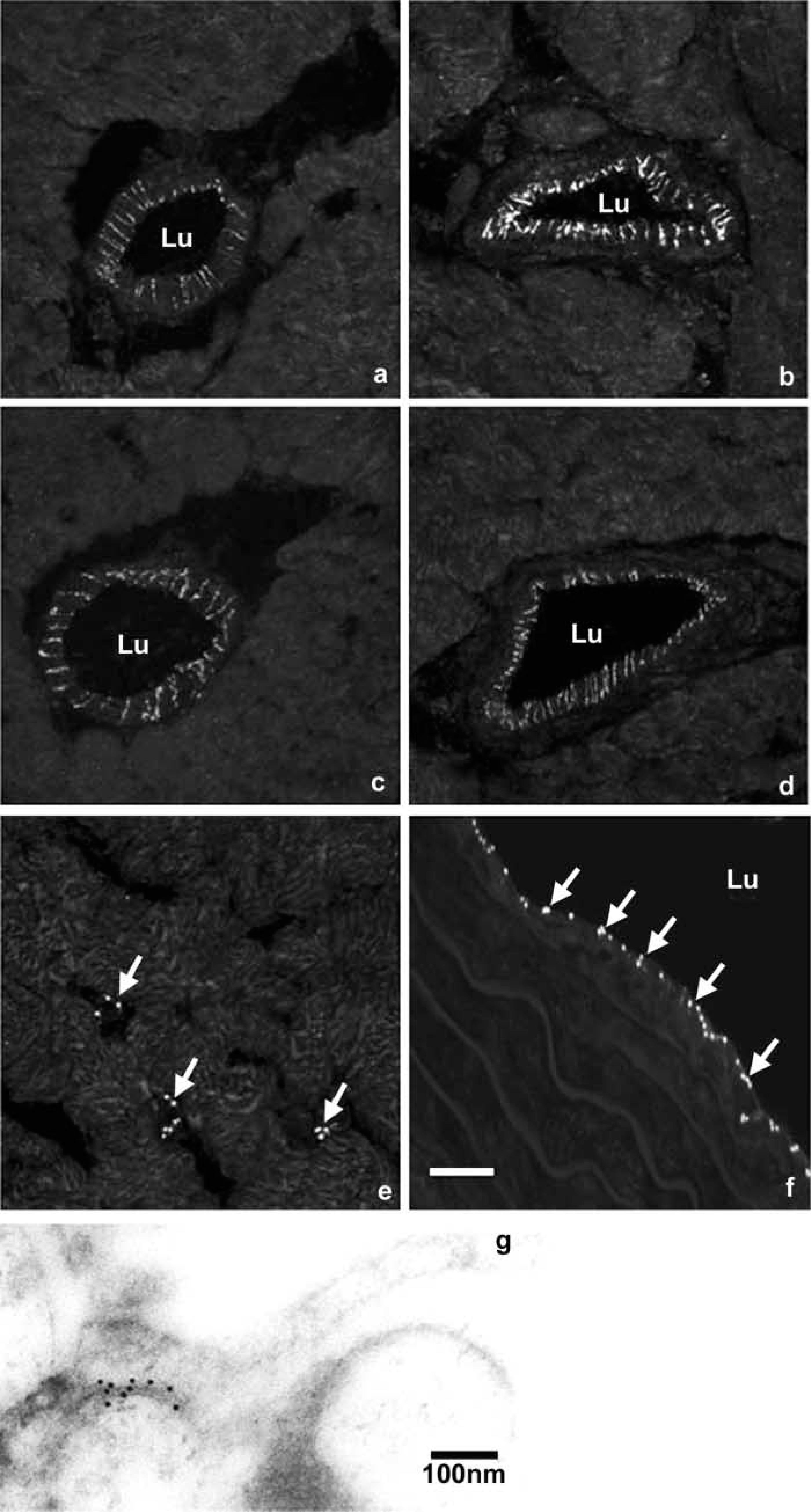
Laser scanning or projection confocal micrographs constructed from five optical sections showing positive Cx40 immunostaining in hamster left ventricle and thoracic aorta. These examples are the median expression levels from each group. (
The cold control animals (Figure 2b) exhibited consistently higher levels of Cx40 staining compared with the normal control (Figure 2a), hibernating (Figure 2c), and aroused (Figure 2d) animal groups. Although this may not always be immediately obvious to the eye, in the median specimen from each group shown in Figure 2, a clear difference was evident when the signal was quantified in all of the animals. There was also clear labeling of Cx40 in capillaries in the ventricular tissue (Figure 2e) in all animal groups examined; nuclear staining revealed that this was between the endothelial cells.
There was no fluorescent signal when the primary antibody was substituted with non-immune serum (not shown). Cx40 immunostaining using a different source of antibody (Gap17) showed the same distribution of staining as above, which was similar to that of the rat coronary arteriole.
Histograms showing the results of quantification of Cx40 gap junction plaque area, numerical densities, and plaque size in the coronary arterioles of the four animal groups are shown in Figure 3. Cold control animals showed an increase in Cx40 area density compared with the other groups, which was significant compared with levels in the hibernation group (p<0.05). There were no significant differences between the normal control, hibernation, and arousal groups. There were no significant differences in the numerical density and plaque size of Cx40 among the four animal groups.
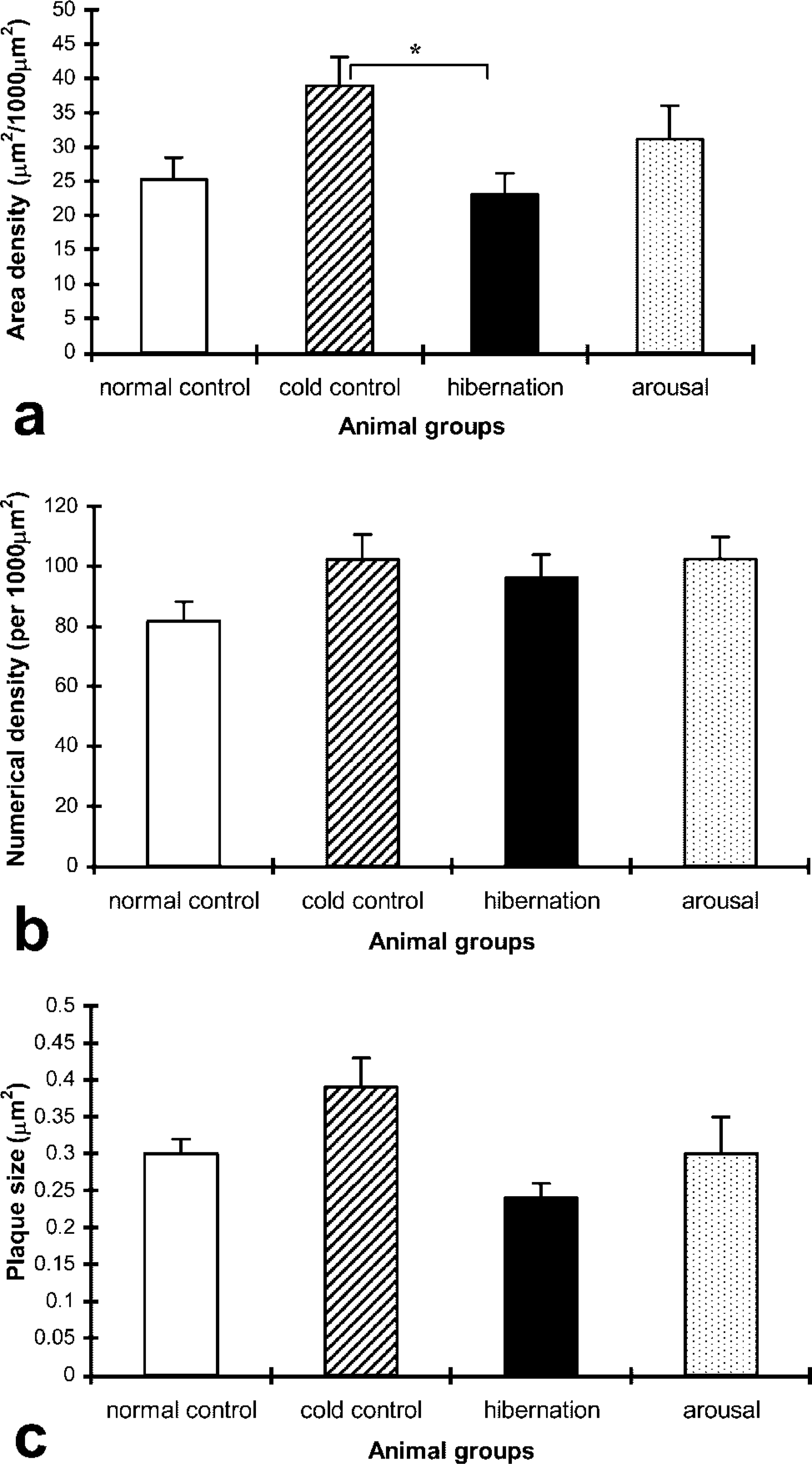
Histograms showing results of quantitative analysis of Cx40 gap junction (
In the aorta of the four experimental animal groups, immunostaining for Cx40 was detected in the endothelium. Prominent fluorescent puncta of Cx40 staining were seen evenly distributed in the tunica intima (Figure 2f). Nuclear labeling revealed that this staining was restricted to the luminal monolayer of endothelial cells.
Histograms showing the quantitation of Cx40 gap junction plaque expression in endothelial cells of the aorta are shown in Figure 4. There were no significant differences in any parameters of endothelial cell Cx40 staining among the four animal groups.
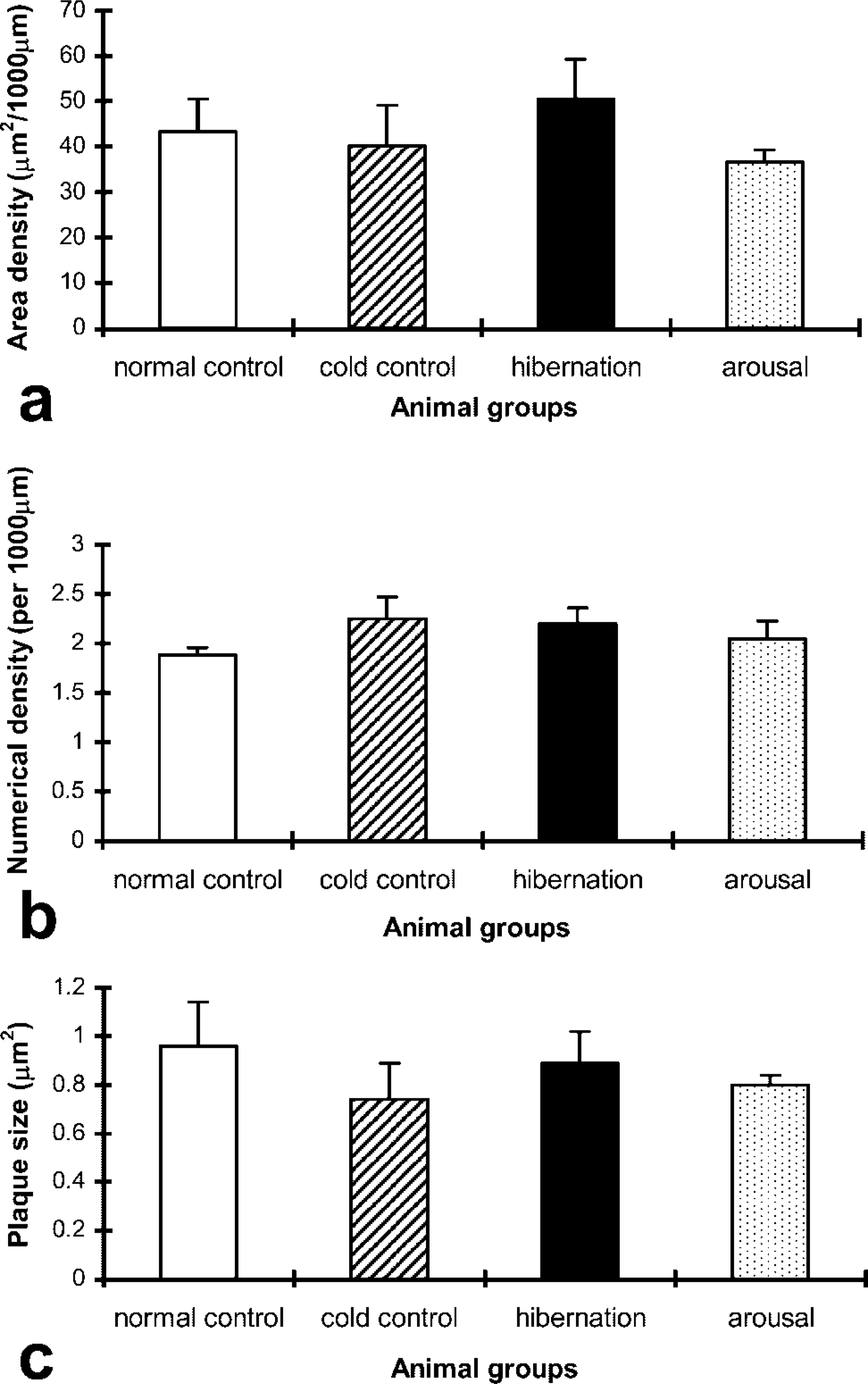
Histograms showing the results of quantification of Cx40 gap junctional (
The normal control mean Cx40 plaque size in the aortic endothelium (0.96 ± 0.18 μm2) was approximately three times that of the coronary arteriole endothelium (0.30 ± 0.02 μm2).
Thin sections of LR Gold-embedded aorta revealed endothelium gap junctions formed by the adjacent cell membranes of neighboring cells (Figure 2g). The Cx40 labeling was decorated with gold particles specifically along the junctional membranes, with minimal background labeling elsewhere in the section. Negative controls consistently showed no labeling.
Immunostaining for Cx43
In all animal groups there was no positive staining for Cx43 throughout the coronary arteriolar wall. However, in sections of left ventricle supplied by coronary arterioles, Cx43 staining was detected at intercalated disk structures between cardiomyocytes (Figure 5a). Some sections were processed alongside sections of aorta that showed positive staining for Cx43, thus confirming that the lack of detection of these Cxs in coronary arterioles was reliable and not due to procedural factors.
Laser scanning or projection confocal microscopy constructed from five optical sections of Cx43 immunostaining in transverse sections of hamster left ventricle and thoracic aorta of the normal control. (
In the aortas of the four experimental animal groups, immunostaining for Cx43 was confined to the smooth muscle cells (Figure 5b), with no labeling in the endothelium. No discernible differences in staining were observed among the four groups of animals. Quantitative measurements of aortic smooth muscle Cx43 plaque area density, numerical density, and size revealed no significant differences among the animal groups, as shown in the histogram (Figure 6).
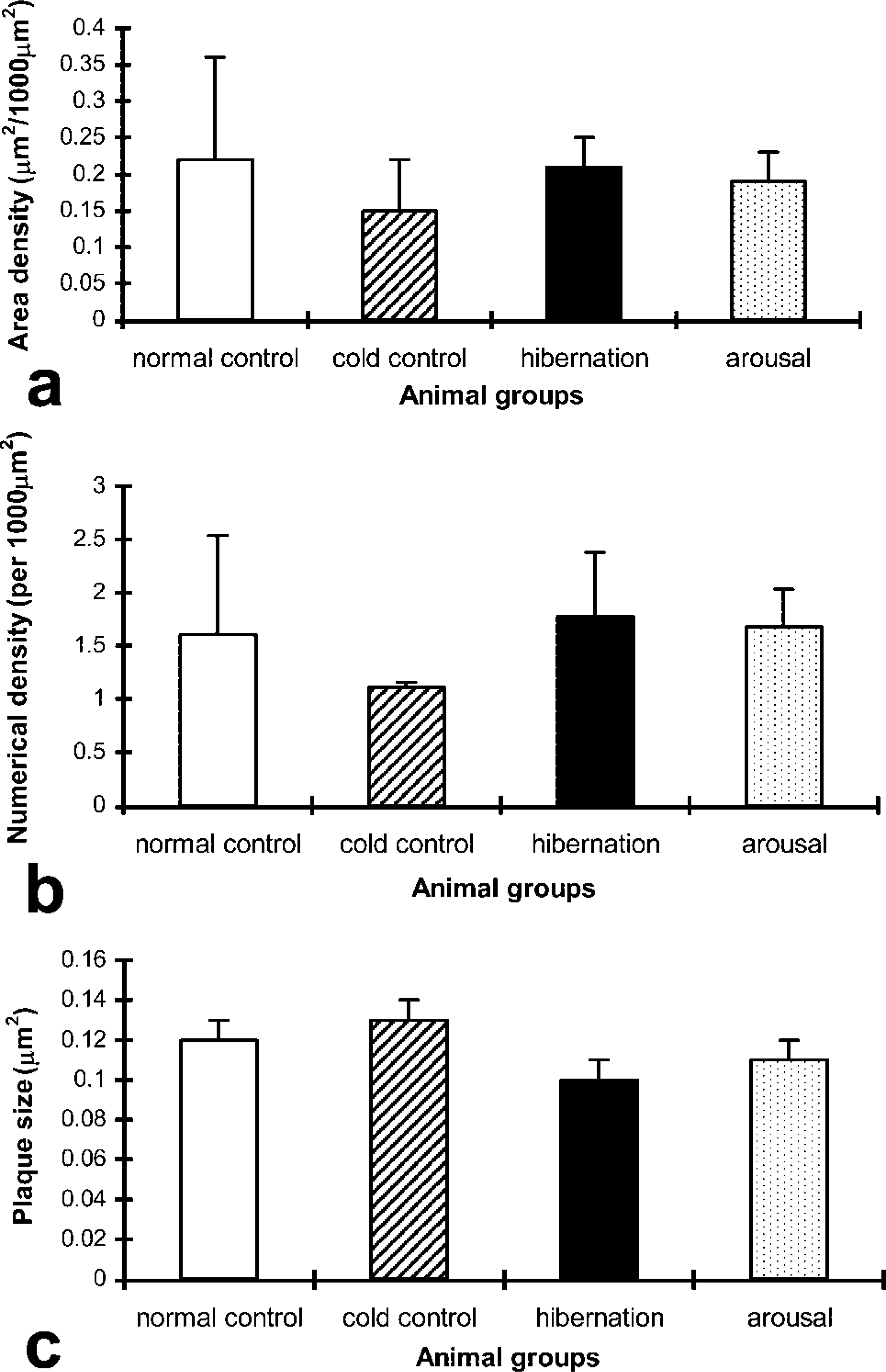
Histograms showing the results of quantification of Cx43 gap junctional (
No immunostaining was detected when the primary antibody was substituted by non-immune serum or, in the case of Cx43, with anti-Cx43 preabsorbed with Cx43 peptide (Figure 5c). Cx43 immunostaining using two different sources of antibody (Gap15 and Gap1A) showed the same distribution of staining as above, which was similar to that of the rat coronary arteriole.
Immunostaining for Cx45
There was no positive Cx45 labeling in the coronary vasculature of any of the animal groups. Similarly, there was no positive staining for Cx45 throughout the arterial wall of the thoracic aorta in any of the animal groups. However, in sections of ascending aorta, sparse Cx45 staining was found at the intimal surface of the vessel (Figure 7).
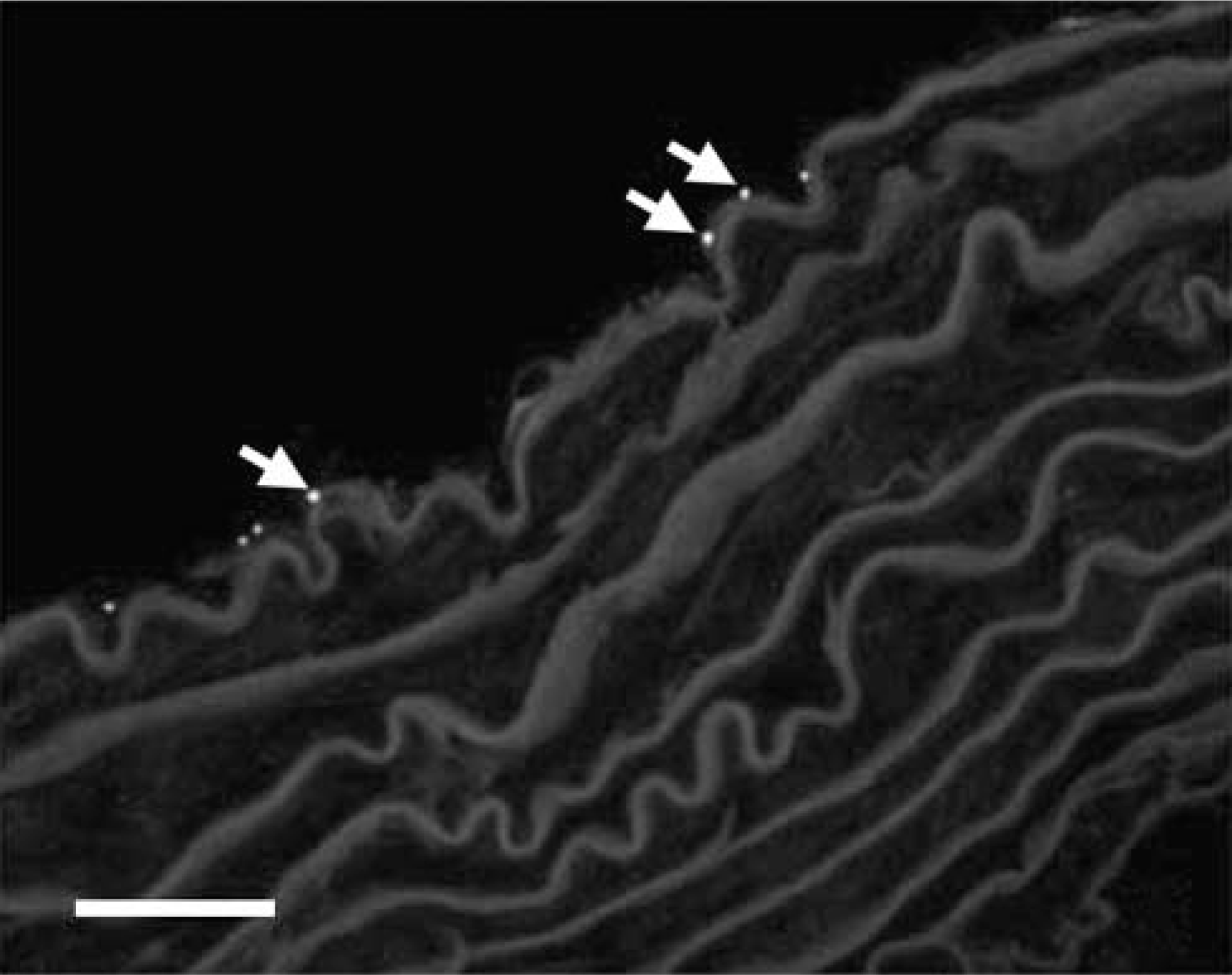
Cx45 immunostaining in the ascending aorta; very little labeling was detected at the endothelium (arrows). To enable optimal Cx45 immunostaining, glutaraldehyde was omitted from the fixation process. Therefore, ultrastructural integrity was not maintained at the normal standard for an electron micrograph. This was unavoidable. Bar = 25 μm.
Distribution of Whole-mount Immunostaining in Aorta
Small fluorescent puncta of Cx37 staining were detected in adjacent endothelial cells (Figure 8a). In contrast, labeling for Cx40 was abundant, with immunofluorescent puncta noted between endothelial cells (Figure 8b), the fluorescent spots defining the border of individual endothelial cells. Cx43 labeling was seen as tiny fluorescent spots between the smooth muscle cells (Figure 8c). Cx45-positive labeling in the hamster ascending aorta was very sparse and was associated with neighboring endothelial cells (Figure 8d) and smooth muscle cells (Figure 8e).
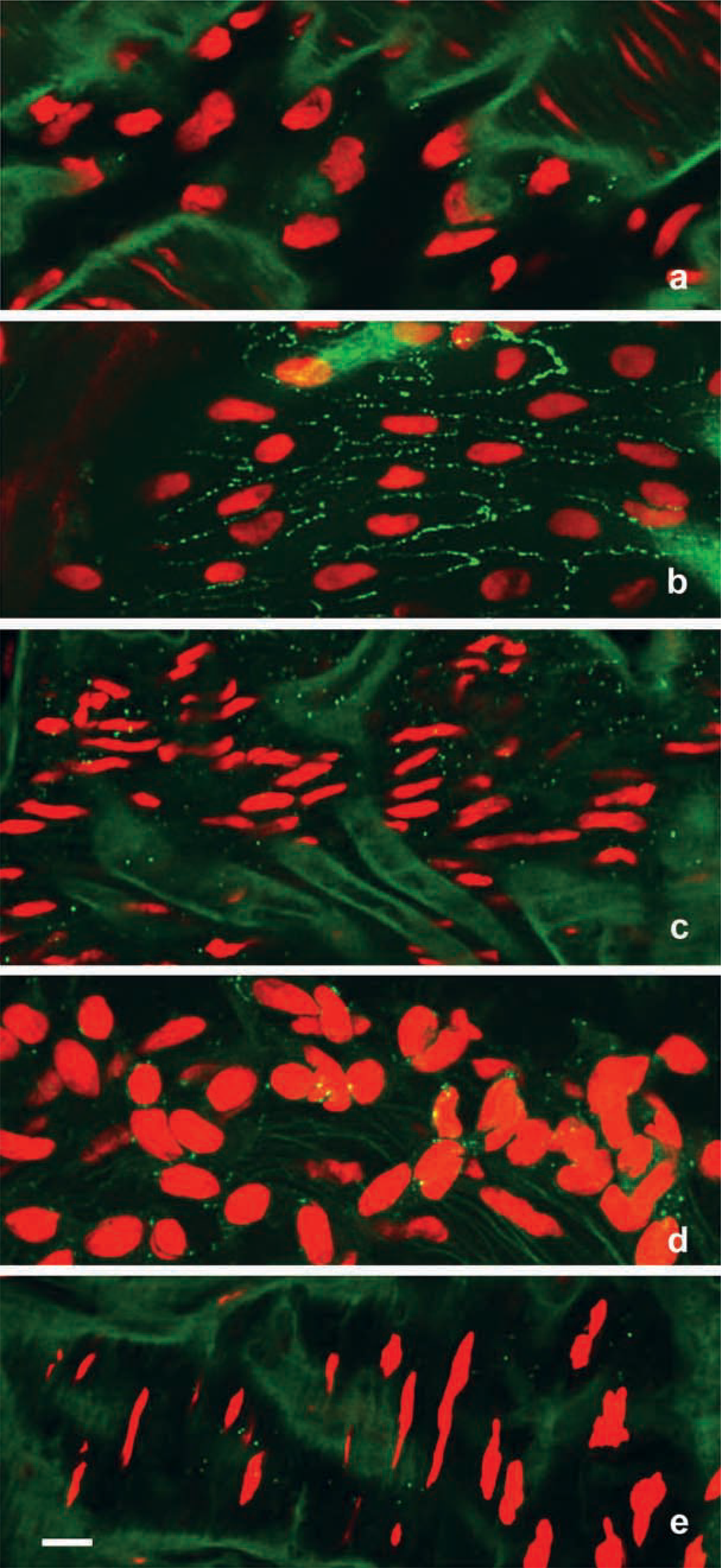
Laser scanning confocal micrographs showing nuclei in red with green positive Cx immunostaining in hamster thoracic aorta (
Discussion
In hamster coronary arterioles and aorta, gap junction proteins were seen by immunofluorescence staining as fluorescent puncta between endothelial cells and/or smooth muscle cells, as previously described in the rat and rabbit (Bruzzone et al. 1993; Carter et al. 1996; Gros and Jongsma 1996; Haefliger et al. 1997; Yeh et al. 1997; Hong and Hill 1998; van Kempen and Jongsma 1999; Ko et al. 2001).
Using selective antibodies to Cx37, Cx40, Cx43, and Cx45, we found a consistent localization of Cx37 in hamster aortic endothelial cells, Cx40 in coronary arteriole and aortic endothelium, and Cx43 in smooth muscle cells of the aorta, whereas there was no positive staining for Cx45 in either coronary arterioles or thoracic aorta from any of the experimental groups. With these same antibodies, the distribution of staining was similar to that in the rat.
The Cx37 labeling was distinct between endothelial cells of the aorta but not in coronary arterioles, a finding in agreement with previous reports in the rat (Yeh et al. 1997,1998; van Kempen and Jongsma 1999). However, Cx37 immunoreactivity has been reported in endothelial and smooth muscle cells of rat coronary artery and cheek pouch arterioles of the hamster (Little et al. 1995; van Kempen and Jongsma 1999). Cx40 expression in endothelial cells of coronary arterioles and aorta is consistent with previous reports (Bruzzone et al. 1993; Yeh et al. 1997,1998; Gabriels and Paul 1998; van Kempen and Jongsma 1999). Cx43 expression in aortic smooth muscle cells but not endothelium has also been described in the rat (Bruzzone et al. 1993), although others have localized Cx43 in the endothelium but not smooth muscle (Haefliger et al. 1997; Yeh et al. 1997,1998; Hong and Hill 1998; van Kempen and Jongsma 1999). Recently, there has been a report of Cx45 labeling in the smooth muscle layer of many areas of rat aorta (Ko et al. 2001), but in our study the expression of Cx45 was different, in that positive labeling was present in both the smooth muscle and endothelium but only in the ascending part of the aorta. This suggests that connexin expression is species- and site-specific.
The main findings of this study are that there are selective changes in endothelial expression of Cx37 in the aorta and Cx40 expression in coronary arterioles among the groups tested. There was a significant decrease in the level of Cx37 in the aortic endothelium of cold-exposed animals, with a trend towards a return to the normal control level after arousal from hibernation. In addition, Cx40 gap junction plaque area in coronary arteriole endothelium of cold-exposed hamsters was increased. During hibernation, the expression of Cx40 gap junction plaques was comparable with that in normal control animals and remained constant during arousal. This finding is remarkable because, during hibernation and arousal from hibernation, there are profound changes in vascular dynamics (Lyman and Chatfield 1955; Lyman 1965). The changes are likely to reflect adaptations in vascular physiology required either to enter a hibernation state or to survive cold exposure. Similarly, the effect on area density is unlikely to reflect contraction of endothelial cells due to cold-induced vasospasm because this effect was not observed in other tissues investigated.
We have previously shown that chronic cold exposure induces an increase in Cx43 expression in ventricular cardiomyocytes in the hamster, which is maintained during hibernation and reverts to normal levels on arousal, which may contribute to tolerance to ventricular fibrillation in hibernators as body temperature drops (Saitongdee et al. 2000).
Hibernators and non-hibernators respond to cold acclimation in a similar way to maintain core temperature (Pohl and Hart 1965). During chronic cold exposure, heart rate and stroke volume increase concomitant with subcutaneous vasoconstriction under sympathetic control (Pohl and Hart 1965). Rats exposed to the cold for several weeks become hypertensive (Fregly et al. 1991; Sun et al. 1997) and in aortic smooth muscle from hypertensive rats, Cx43 expression and plaque size are increased (Berry and Sosa-Melgarejo 1989; Watts and Webb 1996; Haefliger et al. 1997,1999). In the cold, coronary arteriolar vasodilatation ensures adequate nutrient supply to the heart muscle via increased blood flow (Rosaroll et al. 1996). It has been shown that Cx43 mRNA expression is upregulated in rat cultured aortic endothelial cells exposed to laminar shear stress and in smooth muscle cells exposed to stretch (Cowan et al. 1998). The Cx43 expression is localized to sites of turbulent shear stress, whereas Cx37 and Cx40 are distributed uniformly in the aorta (Gabriels and Paul 1998). Shear stress- and stretch-enhanced coupling of cells via gap junctions may amplify signal propagation and influence vasomotor reactivity at sites of increased metabolic activity, i.e., at sites of increased blood flow (Cowan et al. 1998). The prominent Cx40 plaques in all groups examined and the increase in expression in cold-exposed hamsters may reflect the response to increased coronary blood flow and hence increased shear stress at the vessel lumen. Although we cannot comment on the functional integrity of gap junction channels from our studies, increased gap junction plaque formation between endothelial cells may serve to increase the efficiency of interendothelial communication to ensure sustained widespread vasodilatation (Segal and Duling 1986,1989).
Increased blood flow stimulates endothelial cells to release vasoactive agents, which can act via nitric oxide (NO) to bring about smooth muscle relaxation and vasodilatation (Milner et al. 1990). There is evidence that NO modulates gap junctional conductance. In interneurons of the retina, NO reduces the overall open probability of gap junctions by activation of guanyl cyclase and protein kinase G (Lu and McMahon 1997). Recent reports have demonstrated that heterocellular gap junctional (myoendothelial) communication contributes to NO- and prostanoid-independent vasorelaxation attributed to endothelium-derived hyperpolarizing factor (Chaytor et al. 1998).
Another factor that may lead to an increase in gap junction density during cold exposure is reduced degradation of connexins by proteasomal and lysosomal enzymes. Heat shock proteins are increased in cold stress, and it has been shown that HSP70 protects against connexin degradation (Laing et al. 1998).
On entrance into hibernation there was a reversal of the increased Cx40 immunoexpression in coronary arteriole endothelium to normal control levels. On arousal from hibernation there were no significant changes in gap junction protein expression compared to expression during hibernation. In hibernating animals there are several changes in the circulating blood that may influence the endothelium: arterial pH drops from 7.39 to 7.01 due to respiratory acidosis (Malan et al. 1988); plasma glucose levels are reduced (from 142 mg% to 96 mg%) as glucose consumption exceeds glycogenolysis (Galster and Morrison 1970; Atgie et al. 1990); and blood flow is drastically reduced (at least 90%). Because all these factors are reversed on arousal, when gap junction protein levels do not change, it is unlikely that they are involved in the selective reduction of gap junction expression as animals enter hibernation after prolonged periods in the cold.
Cx40 gap junctional protein was far more abundant between endothelial cells of the aorta than was Cx37. These two connexins may form heteromeric connexons [Cx37 and Cx40 form one connexon of gap junction hemichannels (Brink et al. 1997)] and/or heterotypic gap junction channel [each connexon of one channel is formed from different connexins (Wolosin et al. 1997)]. In in vitro studies, connexons composed of Cx37 channels form functional channels with both Cx40 channels and Cx43 channels (Elfgang et al. 1995; Brink et al. 1997).
Different gap junction connexin proteins are functional during the various phases of the healing process (Yeh et al. 2000). Altered expression of Cx37 occurs in the arterial wall in response to artherogenesis in the dog during hemodynamic changes, including increased shear stress (Cai et al. 2001). Our results showed that, in aortic endothelium, only Cx37 expression was reduced during exposure to the cold. This suggests that regulation of expression of Cx37 may be involved in the modulation of the response of the aorta to altered physiological demands. Cx37 gap junctions in the hamster aorta may be involved in dynamic processes associated with adaptation to the cold.
In summary, the increased density of Cx40 gap junctional protein in the endothelial cells of coronary arterioles of cold-exposed animals may reflect increased intercellular communication during prolonged periods of increased blood flow and pressure during cold stress. These changes are reversed during hibernation, when the heart rate and blood flow decrease. Reduced levels of Cx37 gap junctional protein in the hamster aorta during cold exposure may reflect physiological responses in preparation for hibernation. The rapid changes in circulatory dynamics associated with arousal do not appear to involve gap junction protein expression.
Footnotes
Acknowledgements
We are very grateful to Dr C. Orphanides for excellent editorial assistance.
