Abstract
After 14 weeks of topical application of 0.1% all-
K
This regulation is dependent on the interplay of a number of growth factors and morphogenetic molecules, among which all-
The gap junctions of human keratinocytes comprise at least two distinct connexins. Cx43 is abundantly expressed within the interfollicular epidermis (Guo et al. 1992; Wingelbus et al. 1992; Salomon et al. 1994), whereas Cx43 and Cx26 have been shown to be variably distributed within skin adnexae (Salomon et al. 1994). This distinct and partially overlapping distribution is believed to impart specific characteristics to the gap junctions of different skin regions, which presumably account for the differential patterns of communications that are observed between different subsets of human keratinocytes (Salomon et al. 1988; Kam and Hodgins, 1992). In turn, it is assumed that these differential communications underlie different physiological functions of gap junctions in various skin regions.
A first objective of this study was therefore to investigate whether the two connexins that are normally expressed in normal skin are similarly affected by an RA treatment that markedly alters the differentiation program of human keratinocytes. Because such an alteration is known to be associated with the appearance of proteins that are not natively expressed by interfollicular keratinocytes, e.g., keratin K6 (Rosenthal et al. 1990, 1992), we have also assessed whether RA could induce the expression of connexins that are usually undetectable in control human skin.
Materials and Methods
RA Treatment and Tissue Sampling
The in vivo treatment was performed in agreement with the guidelines of our institutional committee for clinical investigation and with the informed consent of seven volunteers (four women and three men, 35–52 years old).
Each volunteer applied once a day, over 14 weeks, 0.2 ml of vehicle solution to an area of approximatively 6 cm2 on one side of the nape and a similar volume of a 0.1% all-
For Northern blot analysis, three of the volunteers (one woman and two men, 35–39 years old) applied RA as indicated above on their buttock. This region was chosen because of its low density in hair follicles and the possibility of obtaining dermatome skin samples of constant thickness (0.3 mm) without esthetic prejudice.
Immunostaining
For immunofluorescence, sections about 5-μm thick were cut with a cryomicrotome (1720 digital MGW; Leitz, Oberkochen, Germany), collected on gelatin-coated slides, and fixed for 3 min in −80C acetone. All slides were then rinsed in cold (4C) PBS containing 0.5% bovine serum albumin and were processed for indirect immunofluorescence staining.
For localization of gap junction proteins, sections were first incubated for 2 hr at room temperature with one of the following antibodies: (a) affinity-purified rabbit polyclonal antibodies against liver Cx32, diluted 1:100 (Dermietzel et al. 1984); (b) affinity purified rabbit polyclonal antibodies against residues 108–122 of liver Cx26, diluted 1:500 (Goliger and Paul, 1995); (c) affinity-purified rabbit polyclonal antibodies against residues 101–119 of liver Cx26, diluted 1:100 (Kuraoka et al. 1993); (d) affinity-purified rabbit polyclonal antibodies against mouse liver carboxy terminal portion of Cx26 (Traub et al. 1989); (e) polyclonal rabbit antiserum against residues 252–271 of heart Cx43, diluted 1:100 (Beyer et al. 1989); (f) affinity-purified rabbit polyclonal antibodies against residues 314–322 of heart Cx43, diluted 1:100 (El Aoumari et al. 1990); (g) mouse monoclonal antibodies against 19 amino acids of the carboxy terminal portion of Cx43, diluted 1:500 (Zymed Laboratory; So. San Francisco, CA); (h) affinity-purified rabbit polyclonal antibodies against residues 313–330 of Cx40 (Bruzzone et al. 1993); or (i) affinity-purified rabbit polyclonal antibodies against residues 266–281 of Cx37 (Haefliger; submitted for publication). Sections were then rinsed in PBS and incubated for 1 hr at room temperature with either fluorescein-conjugated anti-rat or anti-rabbit antibodies, whichever applicable, diluted 1:200. After further rinsing, sections were stained with a 0.03% Evans Blue solution, covered with 0.02% paraphenylenediamine in PBS–glycerol (1:2, v/v), and photographed on a Axiophot microscope (Zeiss) fitted with filters for fluorescein detection.
A similar protocol was followed to localize keratin 6, using either an affinity-purified rabbit polyclonal serum, diluted 1:200, (kindly provided by Dr. E. Fuchs) or mouse monoclonal antibody D5/16B4 (Boehringer; Mannheim, Germany), diluted 1:50. Accordingly, the second incubation was performed with fluorescein-conjugated anti-rabbit or anti-mouse antibodies, diluted 1:200.
In all immunostaining experiments, negative controls included exposure of sections during the first incubation to either pre-immune serum, normal rabbit or mouse serum, or fluorescein-conjugated anti-rabbit or anti-mouse antibodies, whichever applicable. Cryosections of rat liver, pancreas, and heart and of mouse skin were incubated in parallel with the human samples and used as positive controls for the different anti-connexin antibodies tested.
Morphometric Evaluation
Sections of RA- and vehicle-treated samples from each volunteer were incubated with an identical batch of affinity-purified antibodies against either Cx43 or Cx26, to quantitate immunostaining within the interfollicular epidermis. To this end, color slides of each section, taken at a magnification of × 157, were projected at a final magnification of × 2440 on a graphics tablet connected to an IBM personal computer for area measurements and event counting. In each slide, we scored the number of keratinocytes and immunofluorescent spots and measured the area of epidermis. Data were collected both cumulatively for the entire epidermis (excluding the stratum corneum, which never showed specific immunolabeling) and, separately, in the basal layer of the tissue, in the uppermost layer of the granular region, and in one layer in the middle of the spinous region. Controls showed that the latter layer was representative of the entire spinous region, as far as Cx labeling was concerned (not shown). On the basis of these data, we calculated the number of fluorescent spots per layer of keratinocytes at different stages of differentiation.
Aldehyde-fixed skin from RA- and vehicle-treated samples was embedded in Lowicryl K4M resin and sectioned at about 1-μm thickness using a Reichert OM10 ultramicrotome. Sections were stained with methylene blue and photographed as described above. Color slides of each section were projected at a final magnification of × 1840 and were used to evaluate the number of keratinocytes per epidermal field (defined as the area of epidermis photographed in each slide) and the mean area of keratinocyte profiles in the basal, spinous, and granular layers of interfollicular epidermis. All areas were measured by planimetry.
Data were analyzed by analysis of variance and compared by the median, the Kolmogorov–Smirnov, and the Mann–Whitney tests, using programs ANOVA and NPAR TESTS of the SPSS-X statistics package (SPSS; Chicago, IL).
Plasmid Constructions
Human connexin cDNAs for Cx43 (Fishman et al. 1990) and Cx26 (Lee et al. 1992) were subcloned in plasmid Bluescript II KS. Connexin anti-sense probes were synthesized by in vitro transcription of these cDNAs, which were then linearized with Xho I and EcoR I (for Cx43) and Hind III and EcoR I (for Cx26) restriction site enzymes (Boehringer).
Identification of Connexin Transcript
Samples were homogenized in 2.5 ml 0.1 M Tris-HCl, pH 7.4, containing 1.3 M β-mercaptoethanol and 4 M guanidium thiocyanate. After addition of solid CsCl (0.4 g/ml), the homogenate was layered on a 2-ml cushion of 5.7 M CsCl-0.1 M EDTA (pH 7.4) and centrifuged at 35,000 rpm at 20C for 20 hr. Pelleted RNA was resuspended in 300 ml 10 mM Tris-HCl, pH 8.1, suppplemented with 5 mM EDTA and 0.1% SDS, extracted twice with phenol–chloroform, precipated in ethanol, and resuspended in water. For Northern blots, total cellular RNA was denatured by 1 M glyoxal in 0.01 M phosphate buffer containing 50% DMSO, electrophoresed in a 1% agarose gel (5–10 μg total cellular RNA/lane), and transferred overnight onto nylon membranes (Hybond N; Amersham International, Poole, UK). Filters were exposed for 30 sec to 302-nm light and stained with methylene blue. Radiolabeled cRNAs were synthezised by in vitro DNA transcription in a Tris buffer supplemented with 0.05 mg/ml BSA, 16 mM DTT, 1 U RNAse inhibitor, 3 NTP with RNA polymerase. Filters were prehybridized for 2 hr at 65C in a 0.05 M Pipes-buffered (pH 6.8) solution of 50% formamide which was supplemented with 2 mM EDTA, 0.1% SDS, 0.1 mg/ml salmon sperm DNA, 0.8 M NaCl, and 2.5 × Denhardt's solution. Filters were then hybridized for 18 hr at 65C with 2.106 cpm/ml solution 32P-labeled probe, washed twice at 65C in 3 × SSC and 2 × Denhardt's solution, followed by three washings at 70C in 0.2 × SSC, 0.1% SDS, and 0.1% sodium pyrophosphate. Filters were then exposed to film (XAR-5; Eastman Kodak, Rochester, NY) between intensifying screens at −80C for 1 to 4 days.
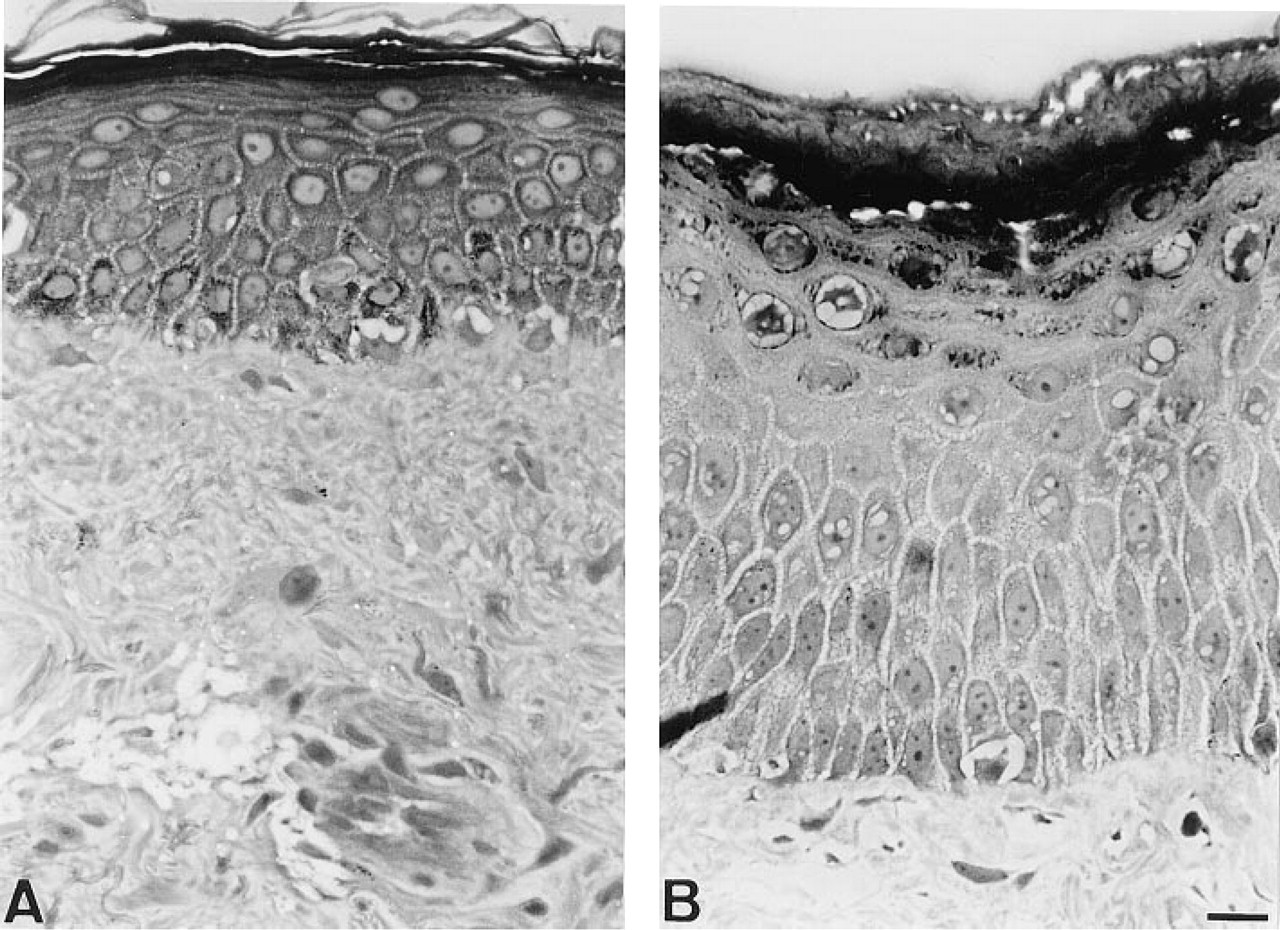
RA treatment increased the thickness of epidermis.
Results
RA Treatment Affected the Differentiation of Interfollicular Keratinocytes
Compared to controls, all samples of RA-treated skin showed a striking increase in epidermal thickness, which was on average 2.5-fold (
Morphometric evaluation of interfollicular epidermisa
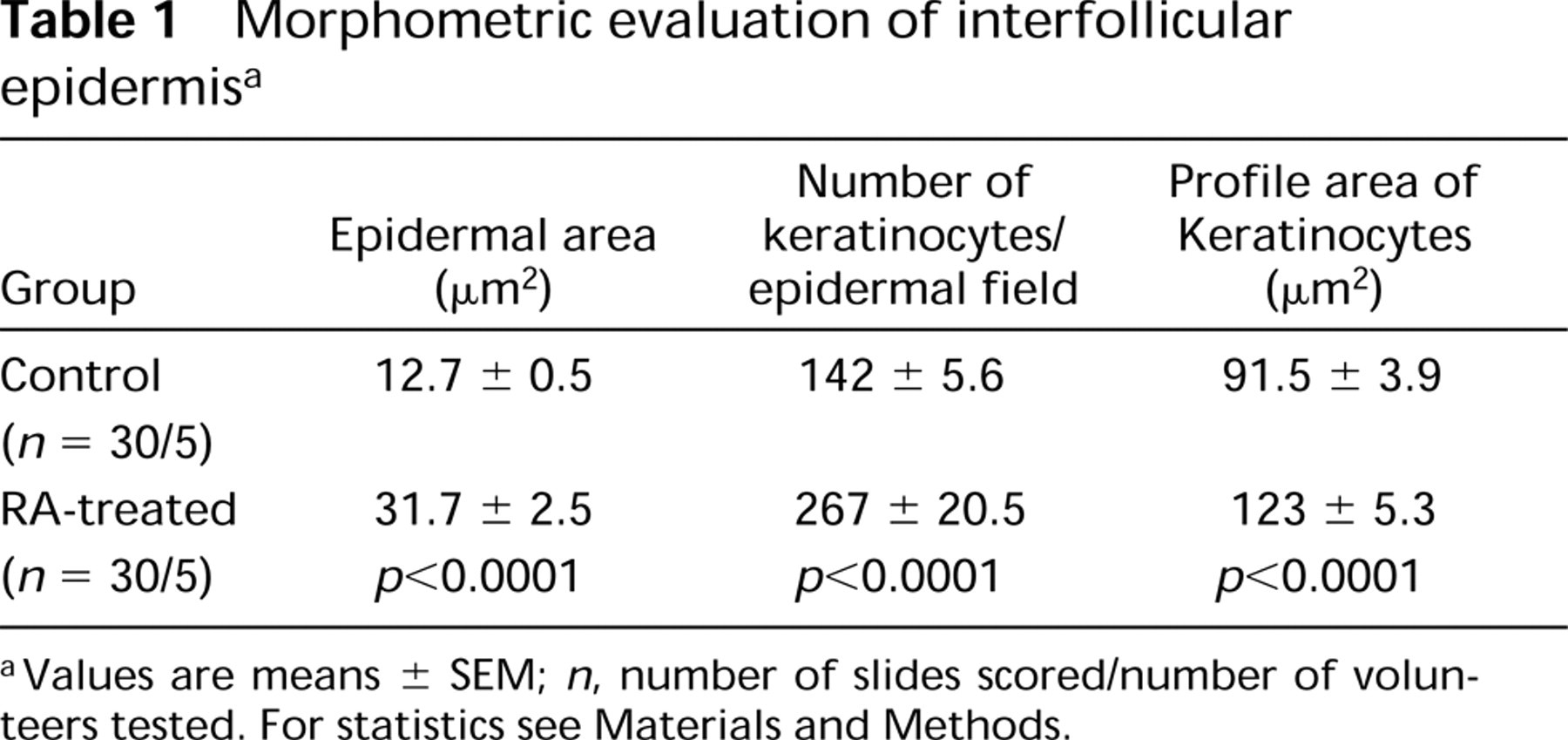
aValues are means± SEM;
RA Treatment Modulated the Expression of Cx43
Immunostaining of cryosections for Cx43, using one affinity-purified and one monoclonal antibody, revealed this gap junction protein in the interfollicular epidermis of control and RA-treated samples (Figure 2). Qualitatively, the distribution of Cx43 appeared similar in all cases. In both control and RA-treated skin, immunolabeling of Cx43 was weak in the basal layer of keratinocytes, strong throughout the spinous layers, weak again in the granular layers, and absent in the stratum corneum (Figure 2). Quantitative comparison of sections that had all been incubated with the same affinity-purified serum (f in the list reported in the Material and Methods section), revealed a significant (
Analysis of skin adnexae failed to detect changes in their immunolabeling for Cx43. This connexin was detected in hair follicles, sebaceous glands, and ducts of eccrine sweat glands in both control and RA-treated skin samples (Table 2).
RA Treatment Induced the Expression of Cx26
Immunostaining of cryosections of control skin, using three different antibodies against Cx26, usually failed to detect this protein in interfollicular epidermis (Figure 3), except in some keratinocytes forming the upper granular layers. In contrast, when applied to sections of RA-treated skin, the same antibodies resulted in an abundant punctate staining of interfollicular keratinocytes of spinous and granular layers (Figure 3). Quantitative comparison of sections that had all been incubated with the same affinity-purified serum against Cx26 (d in the list reported in the Material and Methods section) revealed that this change predominated in keratinocytes of spinous and granular layers (
Contrasting with the major alteration observed within the interfollicular epidermis, no change in Cx26 immunolabeling was detected in skin adnexae. In both control and RA-treated skin, this connexin was readily detected in hair follicles and ducts of sweat glands, but not in sebaceous glands (Table 2).
RA Treatment Did not Induce Expression of Cx32, Cx37, and Cx40
Antibodies that identified Cx32, Cx37, and Cx40 in other tissues (human liver for Cx32, human heart for Cx37 and Cx40) failed to detect these gap junction proteins in the interfollicular epidermis of both control and RA-treated human skin (Table 2).
Discussion
Human keratinocytes are connected by many gap junctions (Salomon et al. 1988, 1993; Watt 1992; Wolff and Schreiner 1968). It is now established that these membrane domains allow direct cell-to-cell exchanges of multiple cytoplasmic constituents (Lowenstein and Rose 1992), but the role these exchanges may serve in skin remains uncertain. In the interfollicular epidermis of normal human skin, gap junctions and their major constitutive protein Cx43 are barely detectable between basal keratinocytes, sharply increase in the suprabasal layers where keratinocytes begin to acquire differentiated characteristics, decrease abruptly in the granular layers as differentiation progresses, and disappear in the uppermost layers of the epidermis, where full differentiation is achieved (Salomon et al. 1994). This heterogeneous distribution suggests that gap junctions may be involved in keratinocyte differentiation. To test this possibility, we have assessed whether connexin expression is affected by conditions that alter the differentiation program of adult epidermis. To this end, we have studied the effects of a long-term topical application of all-
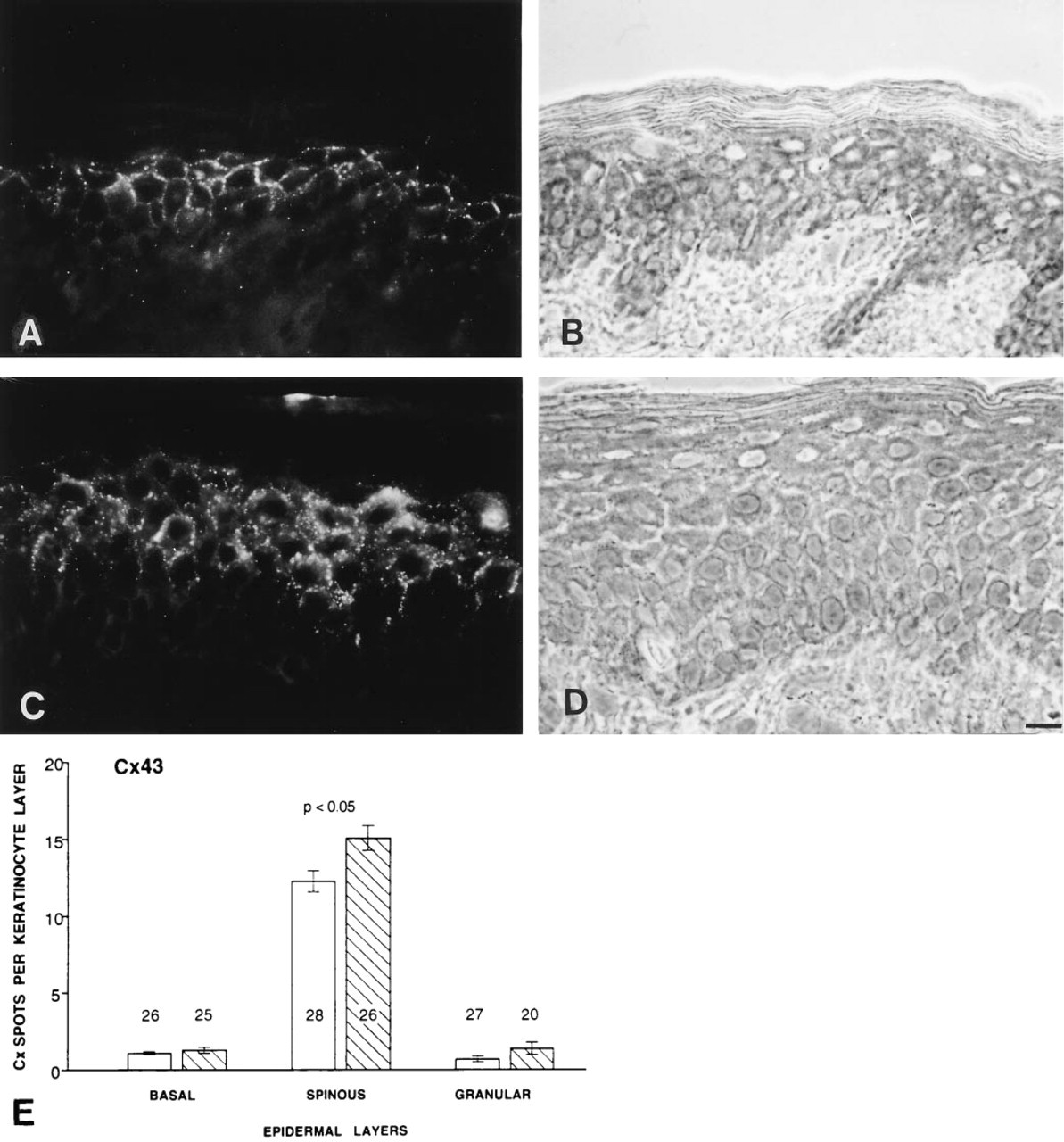
RA treatment modulated the expression of Cx43.
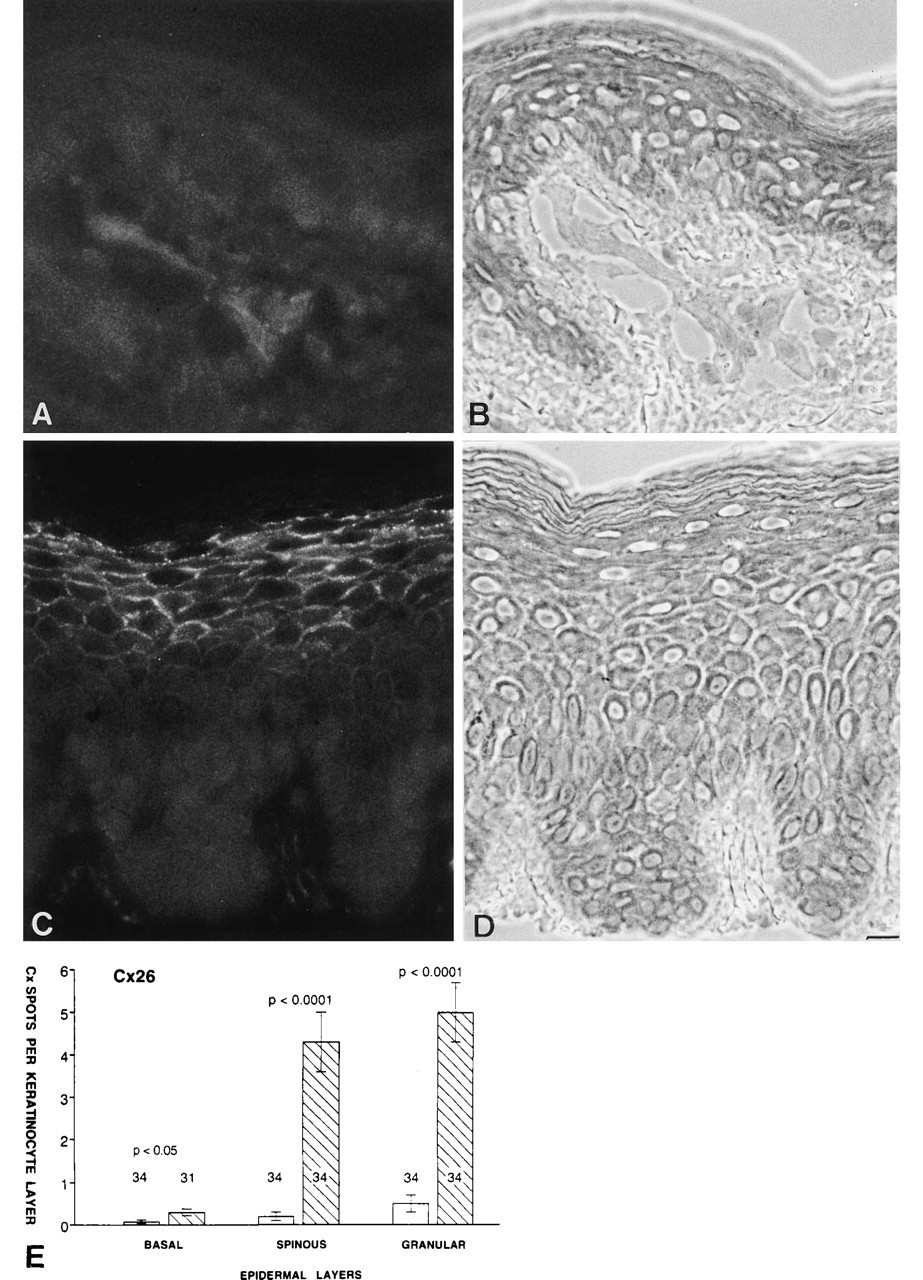
RA treatment induced the expression of Cx26.
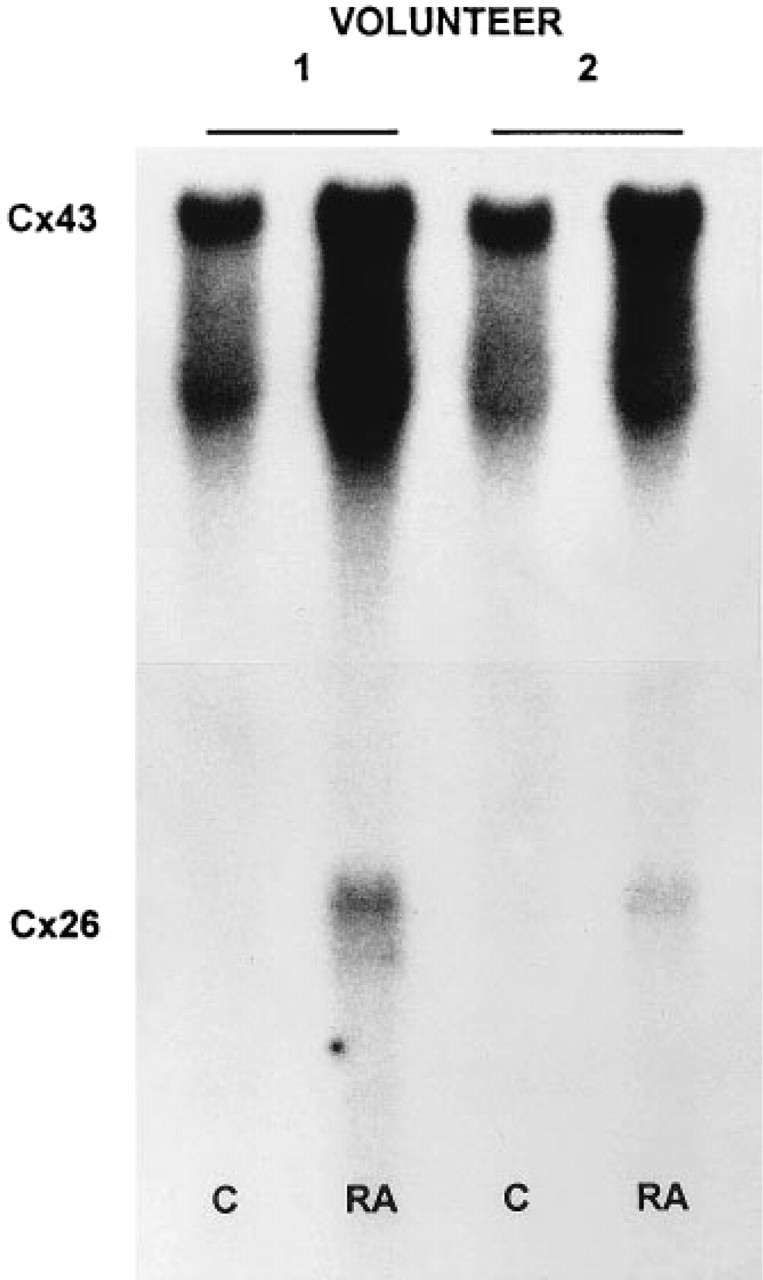
RA treatment increased differentially the expression of Cx43 and Cx26 transcripts.
We have first confirmed that, in normal volunteers, this treatment induces a thickening of interfollicular epidermis (Rosenthal et al. 1990, 1992). Our quantitative analysis showed that this change resulted from both hyperplasia and hypertrophy of keratinocytes of spinous and granular layers. We also observed that these morphological alterations are associated with changes in the pattern of keratins synthesized by these cells (Rosenthal et al. 1990, 1992), as indicated by a massive expression of keratin 6. In humans, this protein is not detected in normal interfollicular epidermis but it is extensively induced in a variety of hyperproliferative skin diseases (Fuchs 1990) as well as after exposure to retinoids (Rosenthal et al. 1992).
Under these conditions of altered differentiation, we have first found that interfollicular keratinocytes increase by about 1.7-fold the expression of Cx43, a connexin that they natively express under control conditions. This increase was paralleled by moderate upregulation in the expression of Cx43 mRNA, analogous to that previously reported for another protocol of topical retinoid treatment (Guo et al. 1992). Our quantitative analysis reveals now that Cx43 increased specifically in the spinous layers of epidermis, in parallel with the increase in the size and number of differentiating keratinocytes, so as to maintain constant its immunoreactive amount per cell. In the very same samples we also found a much more pronounced increase in the expression of Cx26, which resulted in at least a 12-fold increase in the levels of this protein within the epidermis. At variance with what is seen in rodent skin (Risek et al. 1992; Kamibayashi et al. 1993; Butterweck et al. 1994; Goliger and Paul 1994; Sawey et al. 1996), Cx26 is not expressed in the basal and spinous layers of normal human epidermis (Guo et al. 1992; Salomon et al. 1994), although occasionally it is barely detectable between some keratinocytes of the granular layers. Quantitative analysis showed that after RA treatment Cx26 increased, mostly in keratinocytes of spinous and granular layers, i.e., in the same cells that also showed increased levels of Cx43. Previously, in human keratinocytes, the two connexins had been shown to coexist only in vitro (Dupont et al. 1993) in hair follicles and sweat ducts (Salomon et al. 1994) and in skin carcinomas (Wingelbus et al. 1992).
Distribution of gap junction proteins in human skina

a+ and - indicate the presence and absence of connexin immunolabeling, respectively. Values in parenthesis show the numbers of volunteers tested/number of volunteers showing the indicated connexin score.
This finding shows that keratinocytes may express different connexins, not only depending on the species and the region of the epidermis but also as a function of the differentiation program of the tissue. Under the conditions studied here, differentiating keratinocytes of spinous and granular layers shifted their connexin pattern in vivo from the virtually sole expression of Cx43 to an increased expression of both Cx43 and Cx26. Clearly, the levels of these two connexins were not modified to the same extent. Hence, multiple connexins of a same keratinocyte subpopulation may be differentially regulated in vivo, as has been shown in vitro (Dupont et al. 1993). In different in vitro systems, Cx43 and Cx26 appear unable to interact with each other to form functional cell-to-cell channels (White et al. 1994). Therefore, their simultaneous expression by the same cell probably implies the formation of at least two distinct sets of gap junction channels. Because the channels made up of different connexins have distinct conductance and permeability characteristics (Bennett et al. 1993; Brissette et al. 1994), keratinocytes expressing two different connexins could modulate the gap junction-mediated exchange of ions and molecules much more precisely than keratinocytes that express a single connexin.
As yet, the signals crossing gap junctions in vivo remain undetermined, as is the biological function of their cell-to-cell passage. However, the present experiments indicate that a relationship should exist between the control of connexin expression and that of a specific differentiated phenotype. Therefore, chronic exposure to RA, which induces the keratinocytes of spinous and granular layers to enlarge and to express a keratin that is usually retricted to keratinocytes of skin adnexae (Heid et al. 1988), also induces the expression of Cx26, a connexin that is found between keratinocytes of skin adnexae. The differentiation program of keratinocytes comprising characteristics of epidermal adnexae may therefore require the exchange of signals through channels formed by Cx26.
The mechanism by which retinoic acid modulates the differentiation of the epidermis is still uncertain but appears to be related to selective changes in connexin expression (Guo et al. 1992), presumably resulting in a modulation of gap junction-mediated cell-to-cell communication (Pitts and Finbow 1986; Hossain et al. 1989; Mehta et al. 1989). Future studies should investigate the molecular link between the RA stimulus, the connexins, and the differentiation changes.
Footnotes
Acknowledgements
Supported by grants from the Swiss National Science Foundation (32-42539.94 and 32-34086.95), the European Union (BMH4-CT96-1427), and the Research and Inventive Technologies Interstate Commission, Generalitat de Catalunya, Spain (BE 91–134 and BE 92–24).
We thank P. Carraux, A. Charollais, F. Cogne, J.-P. Gerber and E. Sutter for excellent technical assistance. We also thank Drs J. Goliger, Y. Shibata, O. Traub, and E. Beyer for providing us with anticonnexin antibodies, and Drs G. Fishman and S. Lee for human connexin cDNAs.
