Abstract
Apoptosis in the testis and prostate exposed to disrupters of endocrine function, including diethylstilbestrol (DES), during neonatal or postnatal periods has repeatedly been demonstrated, but not in the mature epididymis. We investigated the effects of DES, a potent and synthetic estrogen, on apoptosis in the adult. Adult male golden hamsters received an SC injection of DES and were then sacrificed to collect epididymides after 1, 4, or 7 days of treatment. A significant decrease in epididymal weight and an increase in apoptotic cells were shown on the first day after DES injection. Flow cytometry showed that DES treatment (1 mg/kg) for 1, 4, or 7 days induced significant apoptosis both in the caput and the cauda epididymides. Greater numbers of apoptotic cells were detected in the caput than in the cauda at a fixed time after DES treatment. Serum levels of testosterone decreased markedly within 24 hr after DES administration, reaching undetectable levels of 0.1 ng/ml at 4 days and thereafter. These results indicate that DES administration can increase epididymal apoptosis with a decrease in serum testosterone levels. Because DES used to be injected into domestic animals, adult males also have a chance to take this substance through food. Our study indicates that exposure to DES in adults is as toxic as that in the perinatal period.
A
The importance of estrogens in male reproduction was recently reinforced by the infertility observed in estrogen receptor (ER)-α knockout mice (Eddy et al. 1996) and aromatase knockout mice (Robertson et al. 1999). The wide distribution of ERs in the reproductive tract, including the epididymis, can account for the fact that estrogens play an important role in male reproductive development and function (Fisher et al. 1997). Various morphological and physiological changes in male reproductive organs resulting from exposure to environmental estrogens, including DES, have repeatedly been reported (Nonclercq et al. 1996; Khan et al. 1998). Among these abnormalities, atrophies in the testis and prostate induced by estrogen have been shown to be due to apoptosis (Nonclercq et al. 1996; Robertson et al. 1996). Inhibition of cell proliferation and an increase in apoptosis were also detected during testis regression after chronic DES administration (Nonclercq et al. 1996).
Not only the fetus and the newborn but also adult males may be exposed to high doses of environmental estrogen, i.e., through food, because DES is sometimes injected into domestic animals. Therefore, we started to examine the effect of DES on adult reproductive organs. On the basis of our previous results showing that polyamine metabolism in the adult epididymis is disturbed by DES treatment (Qiu et al. in press), we have presumed that cell death by apoptosis in the epididymis may be also induced by estrogen administration. In an attempt to examine this hypothesis, DES was tested for its ability to induce apoptosis in adult hamster epididymis. Apoptotic cells present in tissues of both segments of epididymis were shown on paraffin sections by a method based on in situ 3′-OH end-labeling of fragmented DNA (TUNEL method), and the percentage of apoptotic cells was also measured by fluorescence-activated cell sorting (FACS) analysis by Annexin-V-Fluos and propidium iodide (PI).
Materials and Methods
Chemicals
DES was purchased from Sigma (St Louis, MO); methyl green solution, n-butanol, apoptosis in situ detection kit and other reagents were obtained from Wako Pure Chemical Industries (Osaka, Japan). Annexin V-biotin apoptosis detection kit was purchased from MBL (Nagoya, Japan) and fluorescein-conjugated streptavidin was from DAKO Cytomation (Kyoto, Japan). Total testosterone kit was obtained from Diagnostic Products (Los Angeles, CA).
Animals and Treatments
Studies with hamsters were approved by the Animal Care and Use Committee, Dokkyo University School of Medicine. The animals were treated according to the guidelines for the Care and Use of Laboratory Animals of the Committee. In all experiments, 7-week-old male golden hamsters weighing 100–115 g were used. The animals were kept in a temperature-controlled room under a long photoperiod (light:dark 14:10), with free access to laboratory chow and tapwater for 7 days before further treatment.
At first, different doses of DES (0.01, 0.1, 1 mg/kg bw) dissolved in olive oil were given SC (once daily) to three groups of hamster (n=5) for 1 week to decide the dose for further experiment. Then the hamsters received a daily single injection of 1 mg/kg DES unless otherwise noted. Three groups (n=5) of treated animals were sacrificed by decapitation between 0900 and 1000 hr after 1, 4, or 7 days of DES treatment. Epididymides were removed, dissected free of fat, sectioned into two segments (caput and cauda), and weighed. A group of animals (n=5) without DES treatment served as controls. The right epididymides were placed in ice-cold PBS, pH 7.4, for cell suspension preparations. The left epididymides were fixed in 10% buffered neutral formalin for histological detection of apoptosis immediately after the animals were sacrificed.
In Situ Detection of Apoptosis
Apoptotic cells in the epididymal tissues were identified by terminal deoxynucleotidyl transferase-mediated nick end-labeling (TUNEL) assay using the in situ Apoptosis Detection Kit according to the manufacturer's instructions. Briefly, after deparaffinization and rehydration, the 4–6-μm paraffin tissue sections were incubated with protein digestion enzyme solution for 5 min in a 37C water bath, rinsed three times in PBS, and then incubated with digoxigenin-deoxy(d)-UTP and terminal deoxynucleotidyl transferase in a humidified chamber at 37C for 30 min. Sections completely rinsed in PBS were incubated with anti-biotin-peroxidase in a humidified chamber at 37C for 20 min after treatment with 3% hydrogen peroxide in PBS for 5 min at room temperature (RT) to quench endogenous peroxidase activity. Peroxidase activity was then visualized by precipitation of 3,3′-diaminobenzidine. The sections were washed rapidly in distilled water and then counterstained with methyl green solution for 5 min, dehydrated in 50%, 70%, 95%, and 100% ethanol, and cleared in xylene. Apoptotic cells were observed under brightfield optics in a Vanox AHBS-3 microscope (Olympus; Tokyo, Japan) and were shown in each epididymal tubule randomly.
Effects of various doses of DES on the weight of the hamster epididymis, prostate, and testis a
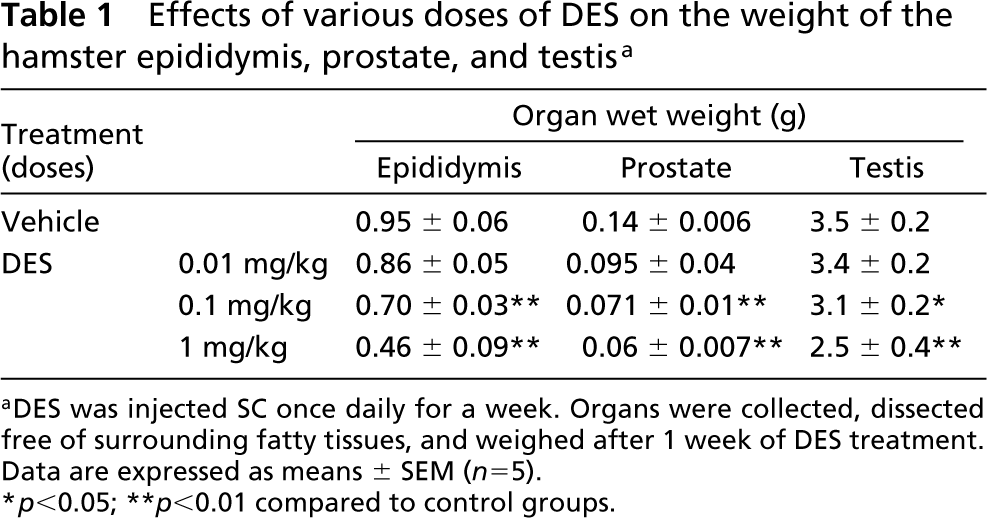
aDES was injected SC once daily for a week. Organs were collected, dissected free of surrounding fatty tissues, and weighed after 1 week of DES treatment. Data are expressed as means ± SEM (n=5).
∗ p<0.05; ∗∗ p<0.01 compared to control groups.
Effects of DES on the weight of hamster epididymis a
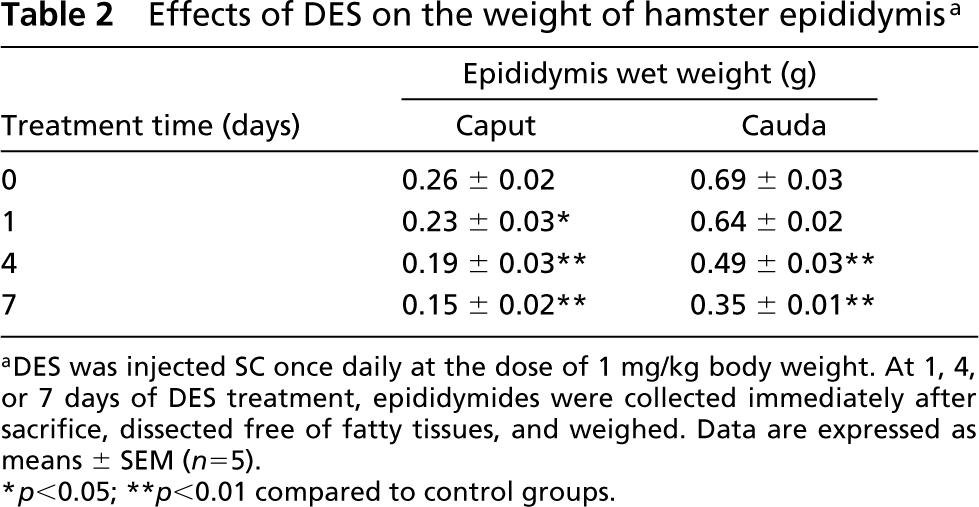
aDES was injected SC once daily at the dose of 1 mg/kg body weight. At 1, 4, or 7 days of DES treatment, epididymides were collected immediately after sacrifice, dissected free of fatty tissues, and weighed. Data are expressed as means ± SEM (n = 5).
∗ p<0.05; ∗∗ p<0.01 compared to control groups.
Fluorescent Flow Cytometry
Epididymal apoptosis was also evaluated by biotin-conjugated annexin-V, a protein that has a strong natural affinity for phosphatidylserine (PS), which translocates from the inner face of the plasma membrane to the surface after initiation of apoptosis. The cell suspensions used for flow cytometry were prepared as described previously (Burikhanov et al. 2000) and the presence of an early stage of apoptosis was evaluated by detecting streptavidin-fluorescein. In brief, the epididymides placed in ice-cold PBS, pH 7.4, were minced with scissors, pressed, and then dissociated by passage through nylon mesh. The cell pellet was centrifuged at 200 × g, gently resuspended in binding buffer, and then incubated with annexin-V-biotin and PI according to the protocol of a commercial kit. The simultaneous application of PI as a DNA stain that is used for dye exclusion testing allows necrotic cells to be discriminated from the annexin V-stained cell clusters. Ten thousand cells were analyzed by FACScan flow cytometry (Becton-Dickinson; Mountain View, CA) (ex 488 nm; em 530 nm) using an FITC signal detector and PI staining by the phycoerythrin emission signal detector. Data were recorded on a logarithmic scale and processed with specific Cell Quest software (Becton Dickinson) for FACScan research.
Serum Testosterone Assay
The total testosterone in serum samples was assayed according to the manual of a commercial kit purchased from Diagnostic Products. The testosterone levels were expressed as ng/ml of serum.
Statistical Analysis
Quantitative data were expressed as mean ± SEM of the individuals in each experimental group. Differences between DES-treated groups and the control animals were compared by one-way analysis of variance (ANOVA), and a p value of less than 0.05 was considered statistically significant.
Results
Effects of DES on Epididymal Weight
The weights of the testis, prostate, and epididymis were decreased by various doses of DES treatment for 1 week. DES at the dose of 0.1 mg/kg significantly decreased the organ weights. The highest dose (1 mg/kg) induced a 28% decrease in the testis, 51% in the epididymis, and 54% in the prostate (Table 1). On the basis of these findings, the dose of 1 mg DES/kg was chosen for further studies. The body weight in DES-treated hamsters was not significantly different from those of controls (data not shown). On the other hand, after DES treatment, a significant decrease in the weight of the epididymal caput and cauda was detected as early as the first day after DES administration (Table 2). At 7 days of treatment, the reduction of organ weight was greatest, 42% in the caput and 49% in the cauda.
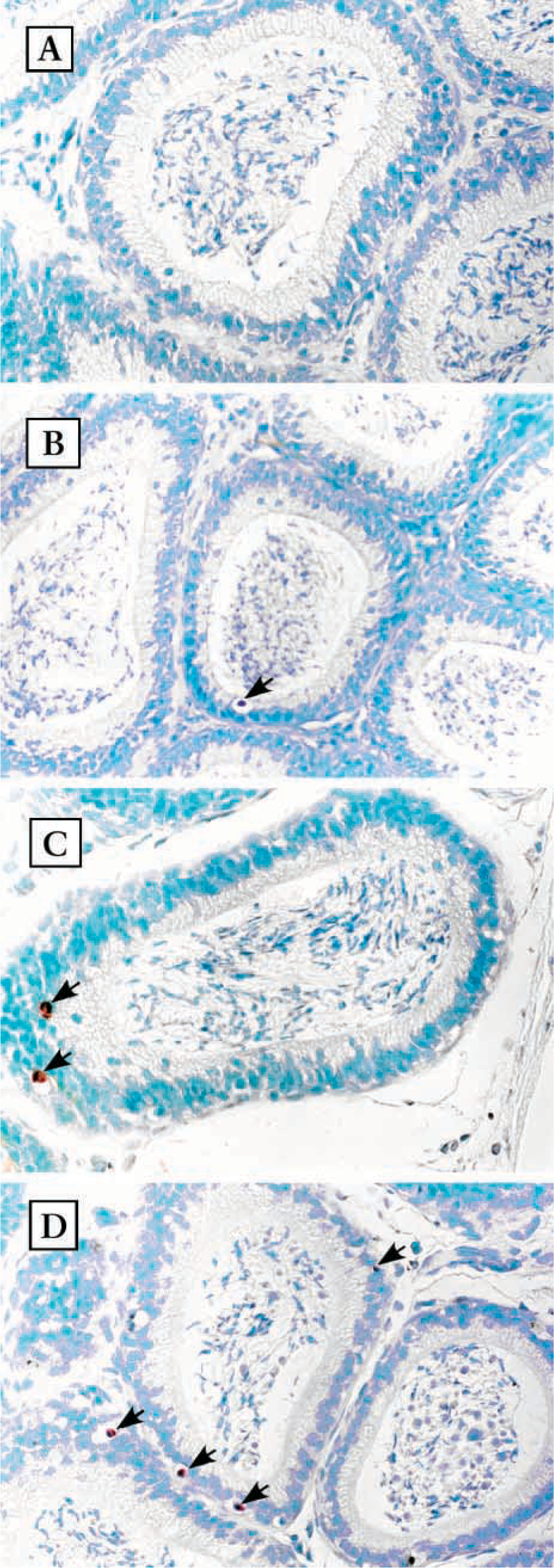
Detection of apoptotic cells in the epithelium of the caput epididymis. Photomicrographs of the caput epididymis after TUNEL staining. Sections were also stained with methyl green. (
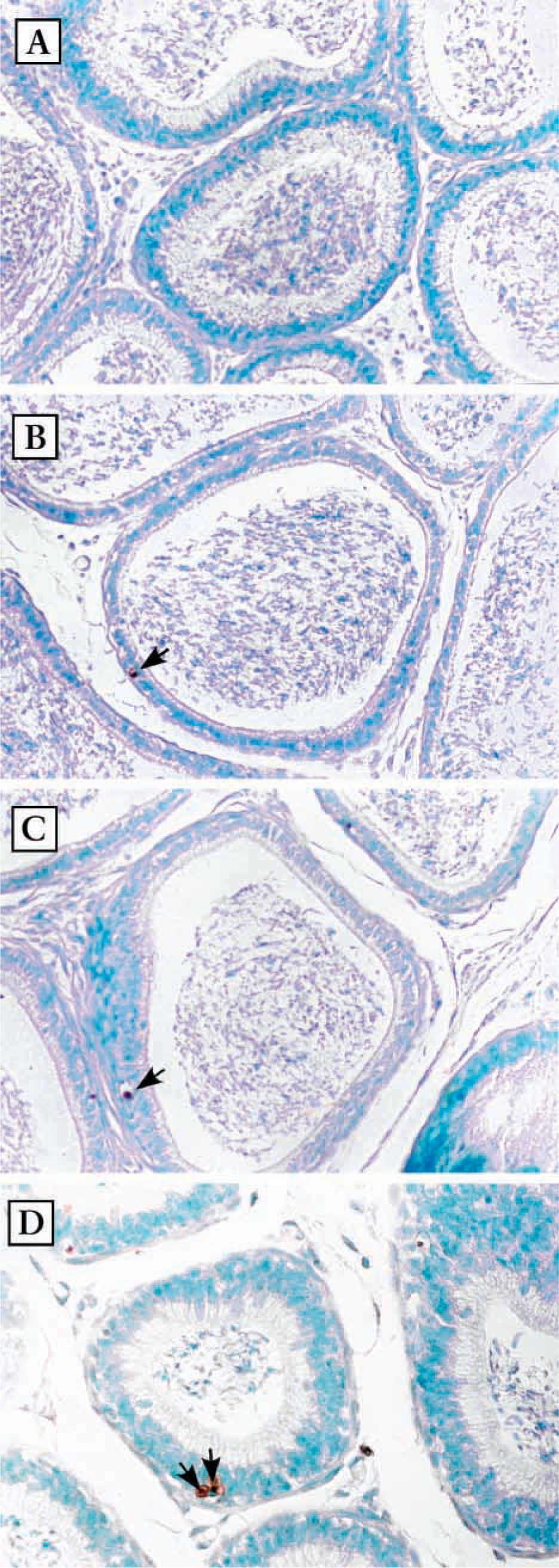
Detection of apoptotic cells in the epithelium of the cauda epididymis. Photomicrographs of the cauda epididymidis after TUNEL staining. (
Histological Examination of Apoptotic Cells in Epididymis Exposed to DES
Apoptotic cells were shown on paraffin sections using TUNEL staining. Positive cells were stained in brown, as shown in Figures 1 and 2. In the normal epididymis, very few apoptotic cells were observed. Less than one apoptotic cell per section was detected in caput and cauda epididymidis (Figures 1A and 2A). In epithelium of the epididymis, apoptotic cells were constantly seen in all sections starting at 1 day after DES administration. However, no marked increase was observed (Figures 1B-1D and 2B-2D). In both segments, apoptotic cells were generally surrounded by an open space, possibly resulting from shrinkage of nuclei and cytoplasm. Apoptotic bodies at an advanced stage of apoptosis and apoptotic nuclei within the lumens were also observed occasionally.
Flow Cytometric Analysis
To study further the occurrence of apoptosis in the hamster epididymis after DES injection, we used FACS analysis to examine the percentage of apoptotic cells by Annexin V-FLUOS and PI. A flow cytometric analysis for epithelial cells of epididymis is shown in Figure 3. Increased annexin-V combined cells in Figure 3 indicate that significant apoptosis of epithelial cells started as early as the first day after DES administration. The percentage of apoptotic cells recorded by this method reached its peak on the fourth day, 33% and 20% in caput and cauda epididymidis, respectively. At 7 days of DES injection, the numbers of apoptotic cells decreased in both the caput and cauda. However, the percentage of apoptotic cells was always slightly less in the cauda than in the caput.
Serum Testosterone Concentrations
The concentrations of circulating testosterone dropped significantly at 24 hr after DES treatment, as shown in Figure 4. The levels decreased rapidly within 24 hr and reached undetectable levels of below 0.1 ng/ml at 4 days and thereafter.
Discussion
The results of our present study show that a dramatic increase of apoptosis in adult hamster epididymis was induced by high-dose estrogen exposure. After injection of DES, a decrease in reproductive organ weights and an increase in apoptotic cell numbers were detected simultaneously in the epididymis. Therefore, the decrease in the weight of reproductive organs could be induced, at least in part, by apoptosis, which was associated with the decrease in circulating testosterone levels.
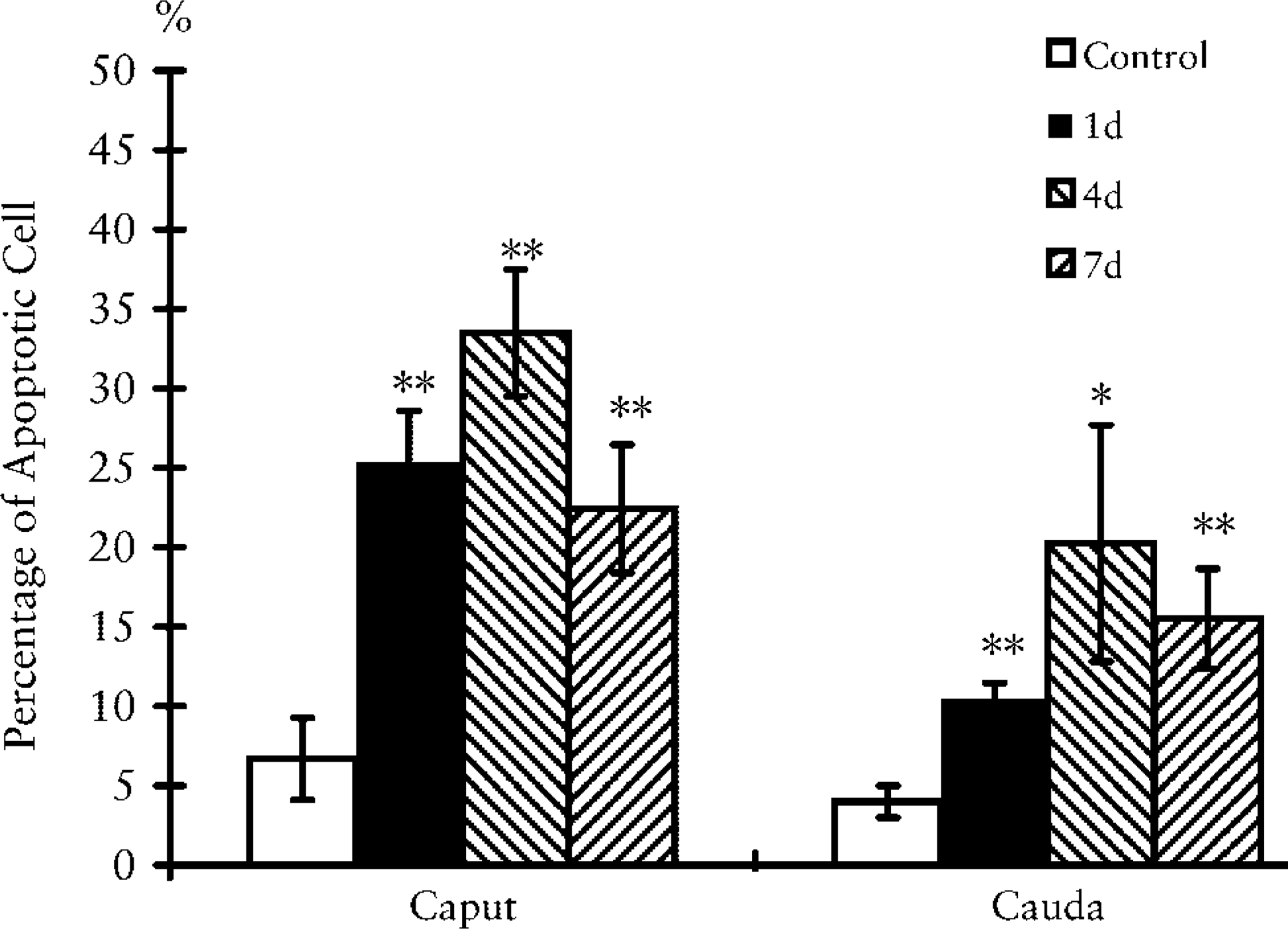
Histograms of percentage of apoptotic cells in epididymal epithelium of untreated (control) and treated hamsters at 1, 4, and 7 days after DES exposure. Epithelial cells of epididymis were processed and analyzed as described in Materials and Methods. This procedure gave a better discrimination of necrotic and apoptotic cells. Data are expressed as means ± SEM (n = 5). ∗ p<0.05; ∗∗ p<0.01 compared to control groups.
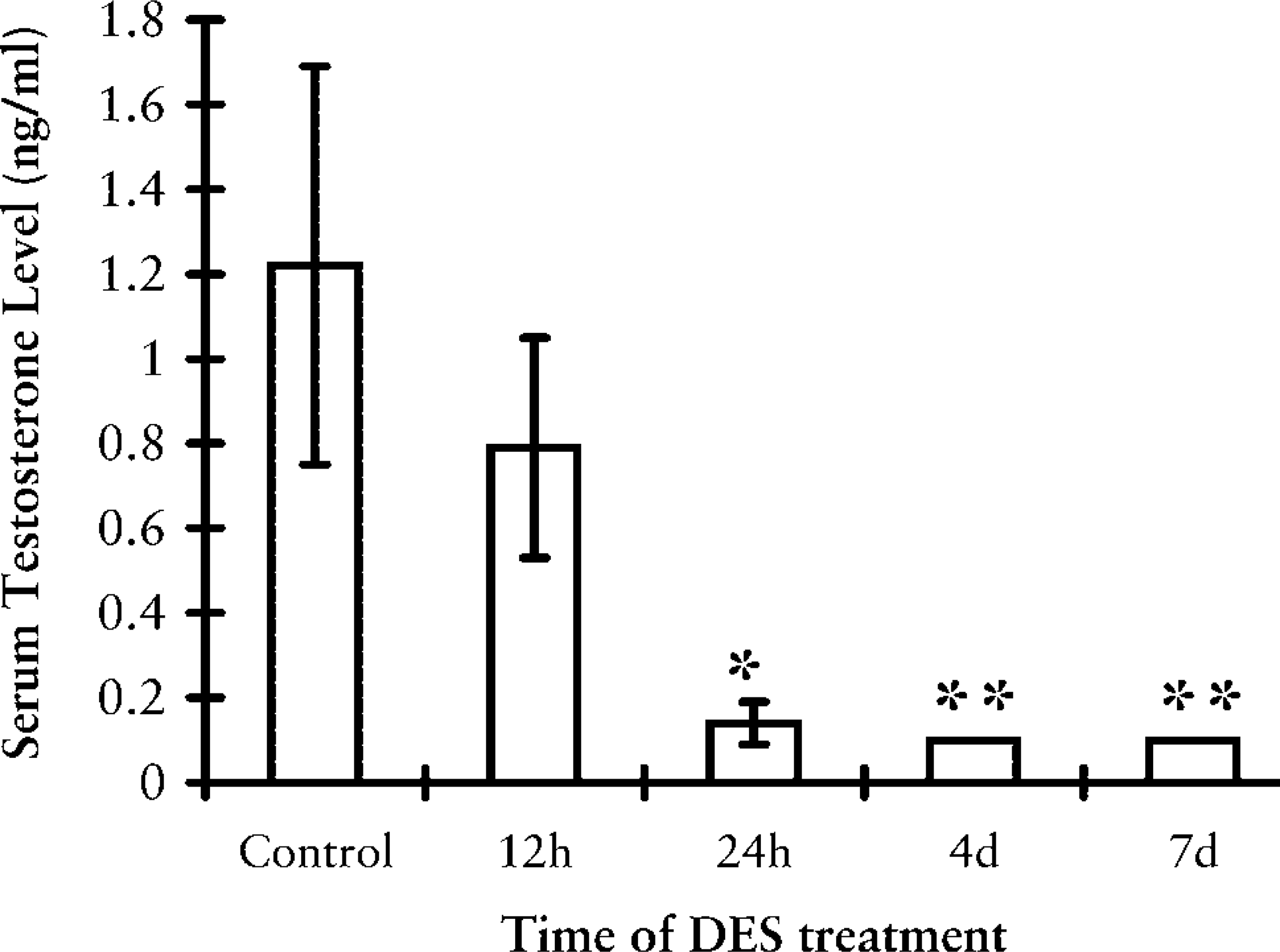
Serum testosterone levels in the hamster during the course of DES treatment. Data are expressed as means ± SEM (n = 5). ∗ p<0.05; ∗∗ p<0.01 compared to control groups.
We have shown here that the regression of caput and cauda epididymides started at 1 day after DES injection and that a severe decrease in organ weight was detected at 7 days. Organ regression by apoptosis induced by estrogen was demonstrated in the testis (Nonclercq et al. 1996) and prostate (Robertson et al. 1996), but is still unclear in epididymis. Our present study confirmed that the adult epididymis also underwent apoptosis to increase rates of cell death after DES treatment. The data showed that essentially no apoptotic cells were detected by the TUNEL method in the intact epididymis, which is in agreement with low cell turnover in the epididymis (Clermont and Flannery 1970).
The results of flow cytometry showed that more cells underwent apoptosis after DES in the caput than in the cauda. Such a region-specific difference of apoptosis in epididymis was also observed after castration in rats (Fan and Robaire 1998) and after increased epididymal temperature in mice (Jara et al. 2002). The cause of such a difference is not clear. One possible mechanism might be differential expression of ERs. Although ERα is distributed evenly throughout epithelial cells in the epididymis, ERβ protein appears to be more abundantly expressed in the caput than in the cauda (Sar and Wetsch 2000). Such differential expression of ERβ may have caused the difference of the effect of DES.
The role of estrogen in male reproduction has attracted more attention than it did 10 years ago, probably because of the reported decrease in human sperm counts that may result from exposure to environmental estrogens (Sharpe and Skakkebaek 1993; Toppari et al. 1996). Programmed cell death plays a significant role in testicular germ development, and appears to be, at least in part, responsible for male infertility caused by exposure to toxicants, including environmental estrogens (Richburg 2000). Not only DES but also many other environmental estrogens may influence male reproductive organs (Akingbemi and Hardy 2001). The results of our present study have shown that such chemicals may affect the function of the epididymis as well as other male reproductive organs, even in adults.
In conclusion, the results of our present study suggest that the weight and the apoptosis in adult hamster epididymis can be affected by DES, an exogenous synthetic estrogen. DES also exerts its effects by decreasing the serum testosterone level, which may affect many organs. Because DES and other estrogen compounds are sometimes injected into domestic animals, adult males as well as the fetus and newborn, might be exposed to DES through food. Our result has clearly shown that adult male reproductive organs are also sensitive to environmental estrogens.
