Abstract
Basement membranes (BMs) are thin layers of extracellular matrix (ECM) found at the basal surface of many cell types, including epithelial cells. BMs present growth, differentiation, and anti-apoptotic signals and provide structural support to cells, compartmentalize tissues, and serve as filters. The structure and function of BMs depend on their complement of laminins, a family of αβγ heterotrimeric glycoproteins. We found that laminins containing the α2 and α4 chains are the major laminins in pancreatic acinar BMs. Importantly, these laminins were required for proper basal localization on acinar cells of two laminin receptors, dystroglycan and integrin α6β4.
A
Here we focus on the distribution of laminins in the pancreas. The exocrine pancreas contains acinar cells, epithelia that secrete digestive enzymes. The endocrine pancreas consists of islets of Langerhans, which contain insulin-secreting β-cells and capillaries for delivering insulin to the bloodstream. Many studies in vitro suggest that laminins are involved in pancreatic cell function. To interpret and extend these findings in vivo, it is important to know the distribution of laminins in the pancreas. A recent study concluded that acinar tissue contains laminin-10 (α5β1γ1) but not laminin-2 (α2β1γ1) (Jiang et al. 2002). In contrast, we show here that acinar BMs have little or no laminin α5 but are rich in laminins α2 and α4.
Primary antibodies
(
The primary antibodies used are listed in Table 1. Secondary antibodies were obtained from Chemicon (Temecula, CA). Fresh pancreases were flash-frozen, sectioned, fixed, and stained as described (Patton et al. 2001). Control mice had mixed 129-C57BL/6J backgrounds; similar results were obtained in B6CBAF1 and outbred ICR mice. Lama2 (dy2J and dy3k) and Lama4 mutant mice have been previously described (Xu et al. 1994; Miyagoe et al. 1997; Patton et al. 2001).
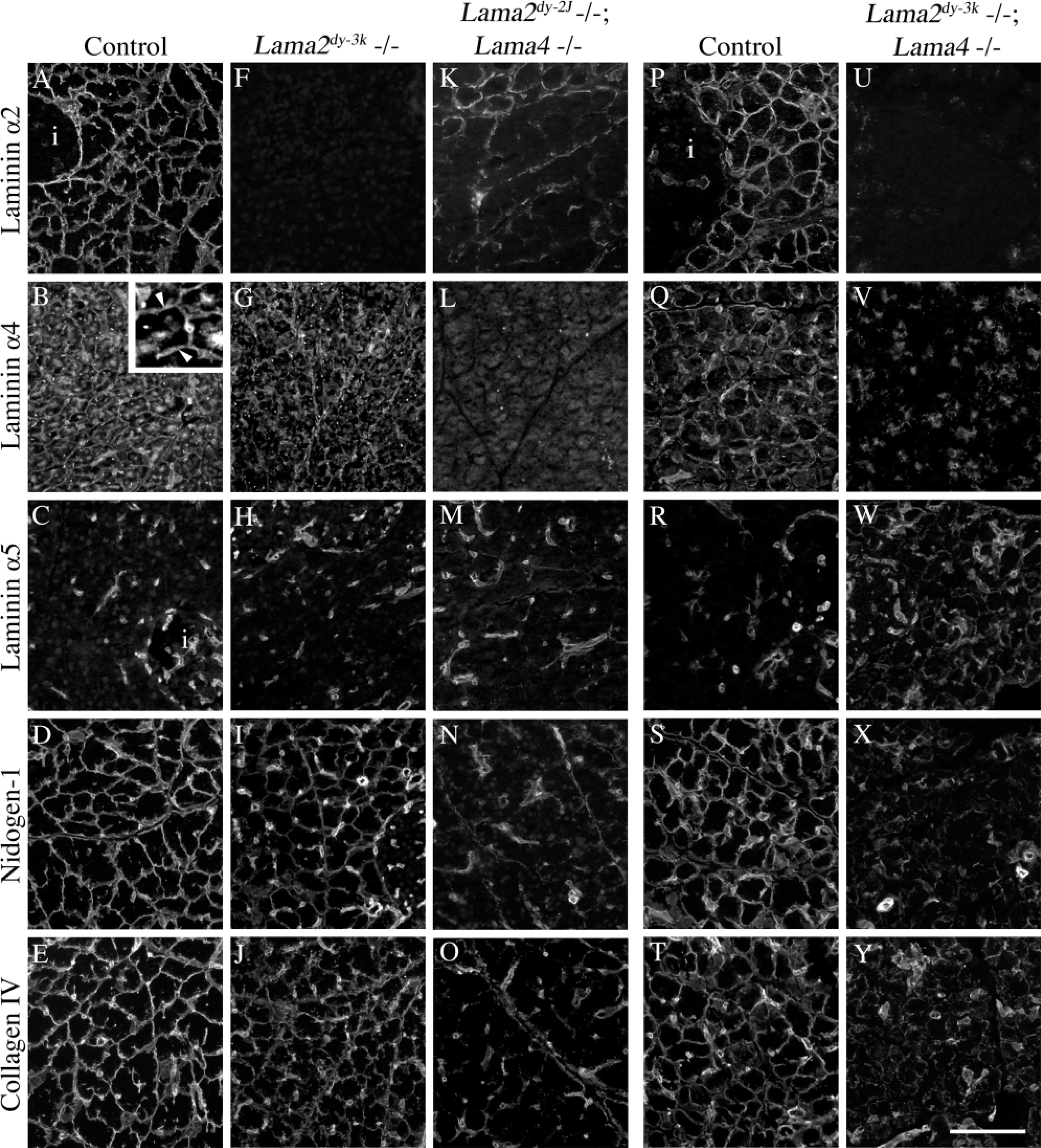
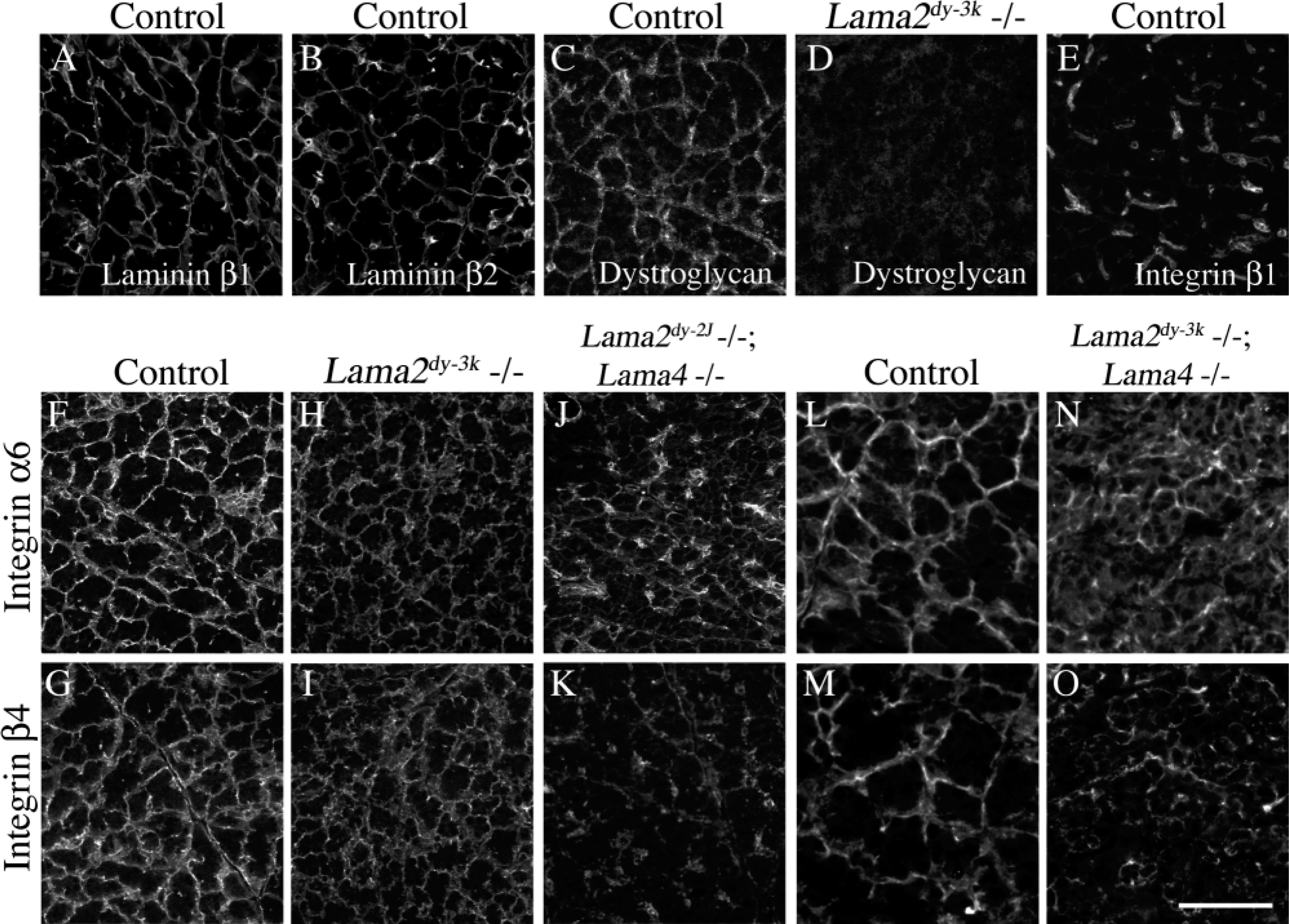
Acinar cell BMs in normal pancreas were labeled uniformly by antibodies to laminins α2 and α4 (Figures 1A and 1B). In contrast, laminin α5 was undetectable in acinar BMs despite intense staining in blood vessels and capillaries (Figure 1C). Laminins β1 and β2 were both present throughout the acinar cell BM (Figures 2A and 2B); laminin γ1 was ubiquitous in pancreatic BMs (data not shown), as reported (Jiang et al. 2002).
(
Laminin α2 was previously reported to be present only in pancreatic blood vessels (Jiang et al. 2002), using MAb 4H8–2 against the NH2-terminal α2 short arm. We found α2 in acinar cell BMs using both 4H8–2 (Figure 1A) and an antiserum directed against the COOH-terminal G-domains (data not shown). Neither antibody stained pancreatic or other tissue from α2-null mutant mice (Lama2dy-3k; Figure 1F, and data not shown), demonstrating their specificity for the α2-chain. Anti-α4 stained α2-deficient acinar BMs weakly (Figure 1G).
To ask if laminins containing α2 and α4 were required for acinar BM formation, we examined mice bearing both α2 and α4 mutations. First, we assessed double-mutant mice harboring a weak Lama2 mutation (Lama2dy-2J) and a Lama4 null mutation. The Lama2dy-2J mutation truncates the α2-chain short arm, prevents polymerization of α2dy2J-containing trimers, and disrupts BM assembly in muscle and nerve (Colognato and Yurchenco 2000). Both antibodies to α2 stained acinar BMs from adult homozygous Lama2dy-2J, Lama4 −/− mutant mice in a weak and interrupted fashion (Figure 1K, and data not shown); α4 was undetectable (Figure 1L), confirming specificity of the α4 antibody. The effect of combined α2–α4 deficiency on overall BM composition was drastic: neither nidogen-1 nor collagen IV was concentrated in a normal linear pattern in acinar BMs (Figures 1N and 1O).
Second, we assessed BM composition in mice homozygous for null mutations in both Lama2 and Lama4, which die before 2 weeks of age (BLP, unpublished data). Pancreases from 12-day-old doubly null mice lacked α2 and α4 immunoreactivity (Figures 1U and 1V), and there was no compensation by other α-chains (data not shown). Moreover, little nidogen-1 or collagen IV accumulated, except in vessels (Figures 1X and 1Y). Therefore, the very existence of acinar BMs in α2;α4 double-null mice is suspect, and H&E staining revealed that acinar cell polarization was impaired (data not shown).
Finally, we asked how changes in laminin composition affected localization of laminin receptors. In normal pancreas, we detected β1-class integrins only in the vasculature (Figure 2E). In contrast, dystroglycan (Figure 2C) and integrin α6β4 (Figures 2F and 2G) were concentrated on acinar cell basal surfaces. Basal localization of dystroglycan was lost in the absence of laminin α2 (Figure 2D), consistent with their direct interaction (Colognato and Yurchenco 2000). Interestingly, the basal localization of integrin α6β4 was maintained in the same cells (Figures 2H and 2I) but was significantly reduced in the absence of both α2-and α4-laminins, compared to controls (Figures 2F-2O). The results suggest selective interactions between laminin α2 and dystroglycan and between laminin α4 and integrin α6β4.
We conclude that laminin-2 (α2β1γ1) and laminin-4 (α2β2γ1), containing the α2-chain, are the major laminins of acinar cell BMs in adult mice. Additional isoforms include laminins-8 (α4β1γ1) and/or -9 (α4β2γ1), containing the α4-chain. However, there is little or no laminin-10 in acinar BMs, previous data notwithstanding (Jiang et al. 2002). We also suggest that dystroglycan and integrin α6β4 are receptors for distinct laminins on acinar cells and that their basal localization is organized by BM composition. The role of cell/matrix interactions in pancreas structure and function remains to be determined, but defects in acinar cell polarization (data not shown) in the apparent absence of basement membrane suggest that they are critical.
Footnotes
Acknowledgements
Supported by grant R01 GM060432 from the NIH and in part by a research grant from the March of Dimes to JHM, and by NIH RO1 NS40759 and a grant from the Muscular Dystrophy Association to BLP.
We are grateful to those investigators who provided antibodies used in this study. We are especially grateful to Drs Yuko Miyagoe-Suzuki, Shin'ichi Takeda, and Karl Tryggvason for providing the α2-null and α4-null mice.
