Abstract
Tissue function is regulated by the extracellular microenvironment including cell basement membranes, in which laminins are a major component. Previously, we found that laminin-1 promotes differentiation and survival of pancreatic islet cells. Here we characterize the expression pattern of laminins and their integrin receptors in adult pancreas. Although they are expressed in the basement membrane of acinar cells and duct epithelium, no laminin chains examined were detected extracellularly in the pancreatic islets. In contrast to laminin β1- and γ1-chains, the α1-chain, unique to laminin-1, was not detected. Laminin-10 (α5β1γ1) was expressed in acinar tissue, whereas laminins-2 (α2β1γ1) and -10 were expressed in the blood vessels. The laminin connector molecule, nidogen-1, had a distribution similar to that of laminin β1 and γ1, whereas fibulin-1 and -2, which compete with nidogen-1, were mostly confined to blood vessels. Integrin subunits α6 and α3 were detected in acinar cells and duct epithelial cells, but α6 was absent in islet cells. Integrin α6β4 was detected only in duct cells, α6β1 in both acinar and ductal cells, and α3β1 in acinar, duct, and islet cells. These findings are a basis for further investigation of the role of extracellular matrix molecules and their receptors in pancreas function.
T
Integrins, one type of receptor for laminins, comprise a family of heterodimeric cell adhesion glycoprotein molecules composed of non-covalently bound α (120–180 kD)- and β (90–110 kD)-subunits, of which there are 16 and at least 22 isoforms, respectively. In addition to mediating cell-matrix and cell-cell interactions, integrins transduce extracellular signals. All known epithelial integrin receptors for laminins comprise isoforms of α6-, α3-, β1-, and β4-subunits (Belkin and Stepp 2000), α3 and α6 being the only subunits with significant (40%) identity (Hogervorst et al. 1991).
The expression and localization of laminins and integrins in the pancreas are poorly documented, despite the potential importance of these molecules in pancreatic development and function. As a baseline for further investigations, we analyzed the expression of laminins and associated molecules, and of integrins, in adult mouse pancreas.
Materials and Methods
Animals and Tissues
Adult (8–12-week) CBA mice bred under specific pathogen-free conditions were sacrificed by cervical dislocation and their pancreases harvested and snap-frozen in liquid nitrogen.
Primary Antibodies
Primary antibodies available for use for indirect immunofluorescence, immunoblotting, and immunoprecipitation are listed in Table 1.
Indirect Immunofluorescence Microscopy
Cryostat sections (8 μm) were air-dried for 40–60 min and fixed in cold (−20C) acetone for 10 min before indirect immunofluorescence staining, or were stored at −80C if not used immediately. Before antibody staining, nonspecific protein binding was blocked by incubation for at least 30 min with warm MT-PBS containing 2% bovine serum albumin or 2% normal rabbit serum. Controls were performed by replacing the first antibody with isotype control antibodies [for monoclonal antibodies (MAbs)] or with pre-immune serum from the appropriate species (for polyclonal antibodies). Tissues were incubated with primary antibodies for 90 min at 25C, followed by three thorough washes with MT-PBS. Fluorescein isothiocyanate (FITC)-conjugated rabbit anti-mouse or anti-rat immunoglobulins (DAKO; Glostrup, Denmark) were incubated for 30 min at 25C, followed by three thorough washes. Slides were observed and photomicrographed under a Zeis Axiophot fluorescent microscope.
Primary antibodies
Immunoprecipitation and Immunoblotting
To further characterize integrin expression in islet cells, immunoprecipitation and immunoblotting analysis was undertaken. A β-cell line, rat insulinoma RIN A12 cells (Gazdar et al. 1980), reported to express several integrin subunits (Kantengwa et al. 1997), was grown in RPMI + 10% FCS almost to confluence, washed with MT-PBS, and collected by scraping. The cell pellet was resuspended in lysis buffer (20 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, 1 mM EDTA, 10 mM Na2HPO4 with the protease inhibitors 1 mM PMSF, 1 μg/ml pepstatin A, and 0.15 U/ml aprotinin) and incubated on ice for 15 min. The lysate was centrifuged at 13,000 rpm for 15 min at 4C and the supernatant pre-cleared with protein G-Sepharose beads. The pre-cleared supernatant was mixed with protein G beads that had been pre-bound with anti-α6 or -α3 integrin antibodies and rotated at 4C for 2 hr. The beads were washed five times with the lysis buffer, resuspended in reducing SDS sample buffer, and boiled for 3 min. The lysate was electrophoresed in a 10–20% SDS-PAGE gel (Novex; Invitrogen, Carlsbad, CA) and transferred to nitrocellulose membrane. After blocking for 1 hr in 5% milk powder in MT-PBS, the membrane was incubated with anti-β1 or -β4 integrin antibodies diluted in 1% milk powder in MT-PBS with 0.05% Tween-20 (PBS-T) for 2 hr at RT, washed five times for 10 min with PBS-T, and then incubated with secondary antibodies diluted in 1% milk powder in PBS-T for 1 hr at RT. After five washes with PBS-T, the immunoblot was developed for 5 min with Lumi-Light Western Blotting Substrate (Roche; Indianapolis, IN) and exposed to Hyperfilm MP (Amersham Pharmacia Biotech; Poole, UK).
RT-PCR Analysis of Laminin RNAs
Total RNA from mouse pancreas or RIN cells was extracted with RNAzol B (Cinna/Biotecx; Houston, TX) and treated with DNase I (Gibco BRL; Gaithersburg, MD). mRNA was reverse-transcribed with Superscript II reverse transcriptase (Gibco BRL) in 1 X transcription buffer containing 0.5 μM oligo (dT)16–18 primer (Gibco BRL) and 400 μM dNTPs. Aliquots of cDNAs were amplified by PCR in 1 X PCR buffer (Roche) containing 200 μM dNTPs, 1 μM of each primer pair, and 1 U Taq polymerase. The primers for laminin α1 (5’ GACCGCCATGCCGATTTAGC 3', 5’ GACCGCCGTGTTGTTGATGC 3'), laminin α5 (5’ CCCTGGGGCCTTGAACTTCTCCTACTC 3', 5’ GCATTGCGCCGATCCACCTCAG 3'), laminin β1 (5’ ACCAGACGGGCCTTGCTTGTGAAT 3', 5’ AGTTGTGGCCCGTGGTGTAGTCCTG 3') and for the control “housekeeping” gene β-actin (5’ GTGGGCCGCCCTAGGCACCA 3', 5’ CTCTTTGATGTCACGCACGATTTC 3') all yielded PCR products of 530 bp. PCR reactions were performed for 35 cycles (95C, 30 sec; 60C, 30 sec; 72C, 30 sec), and amplified products were separated in 1.5% agarose gels.
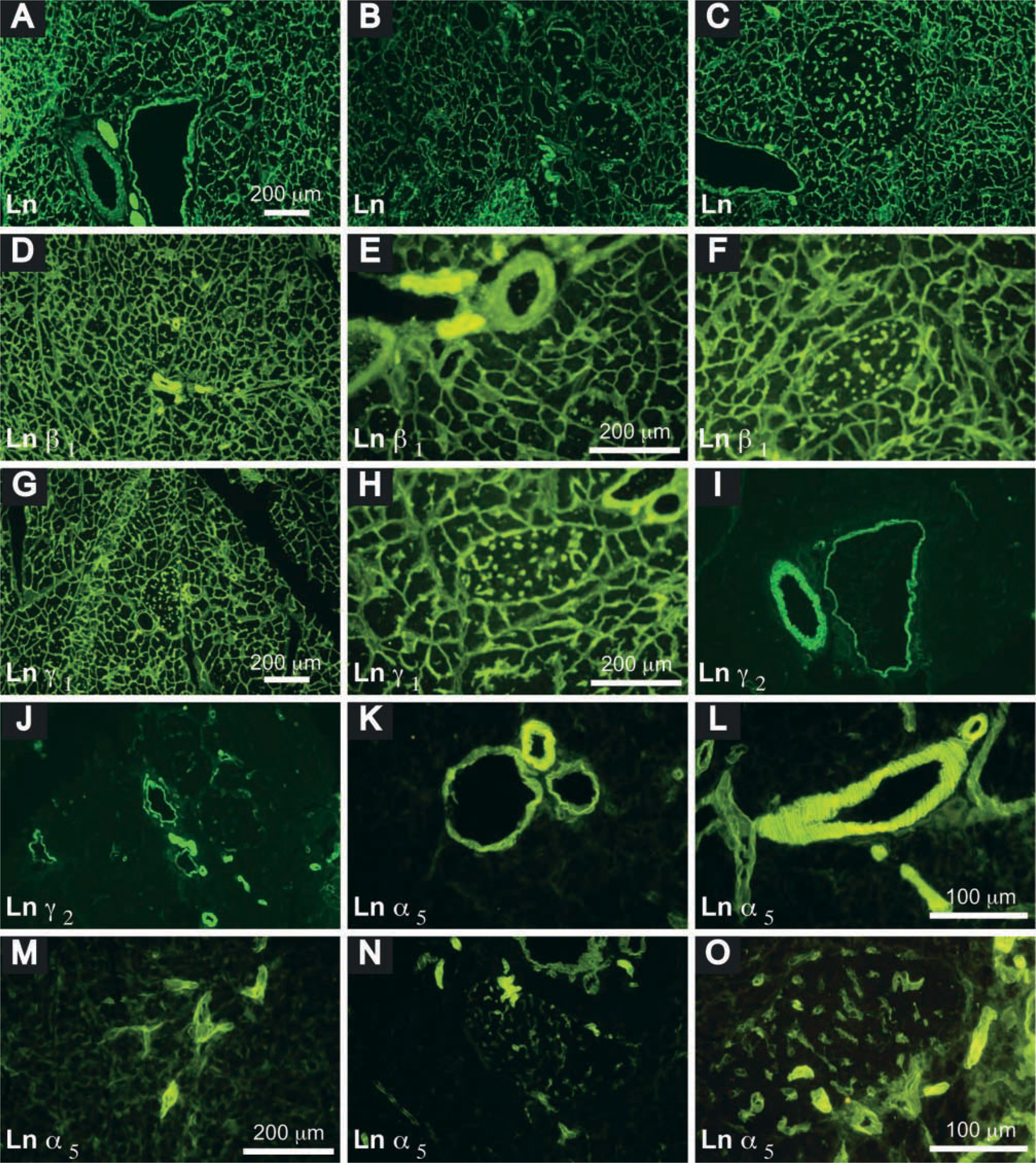
Distribution of laminin chains in the adult mouse pancreas. Laminin, detected by rabbit anti-mouse laminin IgG and fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit immunoglobulins (Ig), was extensively distributed in the ECM, in arteries, and in veins (
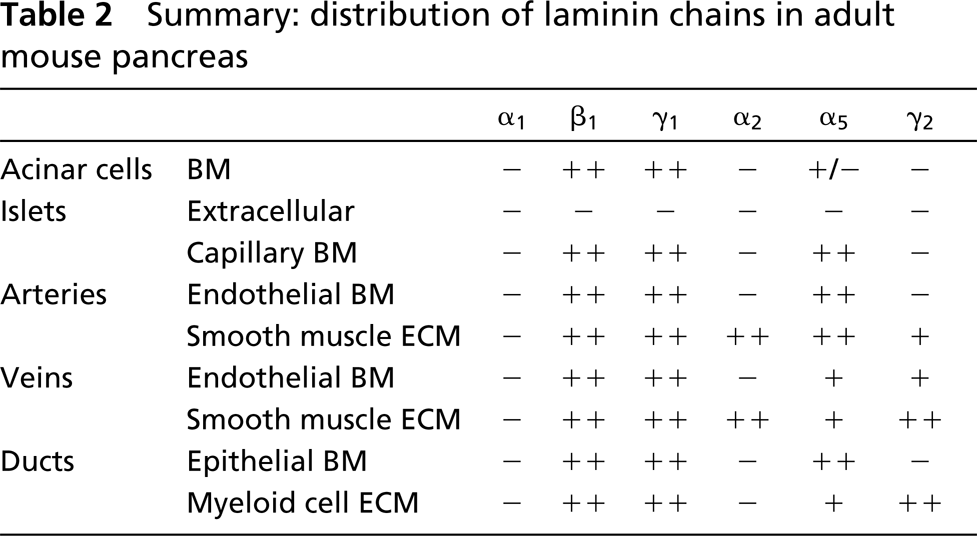
Summary: distribution of laminin chains in adult mouse pancreas
Results
Laminin Expression
Laminin, detected with polyclonal rabbit anti-laminin antibodies, was expressed in the basement membrane (BM) of several tissues in the pancreas. In the basal lamina of exocrine cells, intra-islet capillary plexuses, duct epithelia, and smooth muscle of arteries and veins (Figures 1A-1C). This antibody, generated to murine Engelbreth-Holm–Swarm tumor BM, identifies α1-, β1-, and γ1-chains (Ferletta and Ekblom 1999) and detects all laminin isoforms, except for laminin-5 (α3β3γ2), reported to be preferentially expressed in BM of squamous and transitional epithelia (Burgeson et al. 1994).
Laminin chains detected with specific monoclonal antibodies (MAbs) had a unique distribution pattern in the pancreas (Table 2). Laminin α1, specific for laminin-1 (α1β1γ1), was not detected in the adult mouse pancreas, consistent with our RT-PCR analysis (see below) and with previous reports (Falk et al. 1999; Virtanen et al. 2000). The expression patterns of laminins β1 and γ1 were the same as those revealed with the rabbit anti-laminin antibody (Figures 1D-1H). Laminin α2, a component of laminin-2 (α2β1γ1), laminin-4 (α2β2γ1), and laminin-12 (α2β1γ3), and laminin γ2, which is present in laminin-5, were detected only in the basal lamina of smooth muscle cells in arteries and veins (Figures 1I and 1J). Laminin α5, a component of laminin-10 (α5β1γ1) and laminin-11 (α5β2γ1), was richly distributed in the BM of endothelia, in the basal lamina of smooth muscle cells in arteries and veins and of myoid cells in interlobular ducts, consistent with other reports (Sorokin et al. 1997; Maatta et al. 2001). It was spirally distributed along arteries, being less orderly in the veins and irregular in ducts (interlobular and intralobular). Laminin α5 also appeared in the peripheral islet and penetrated into the islets along intra-islet capillaries (Figures 1K-1O). The pattern of nidogen-1 expression (Figures 2A-2D) was the same as that of laminins β1 and γ1. The distributions of fibulin-1 and -2 were similar (Figures 2E-2H), resembling that of laminin α5.
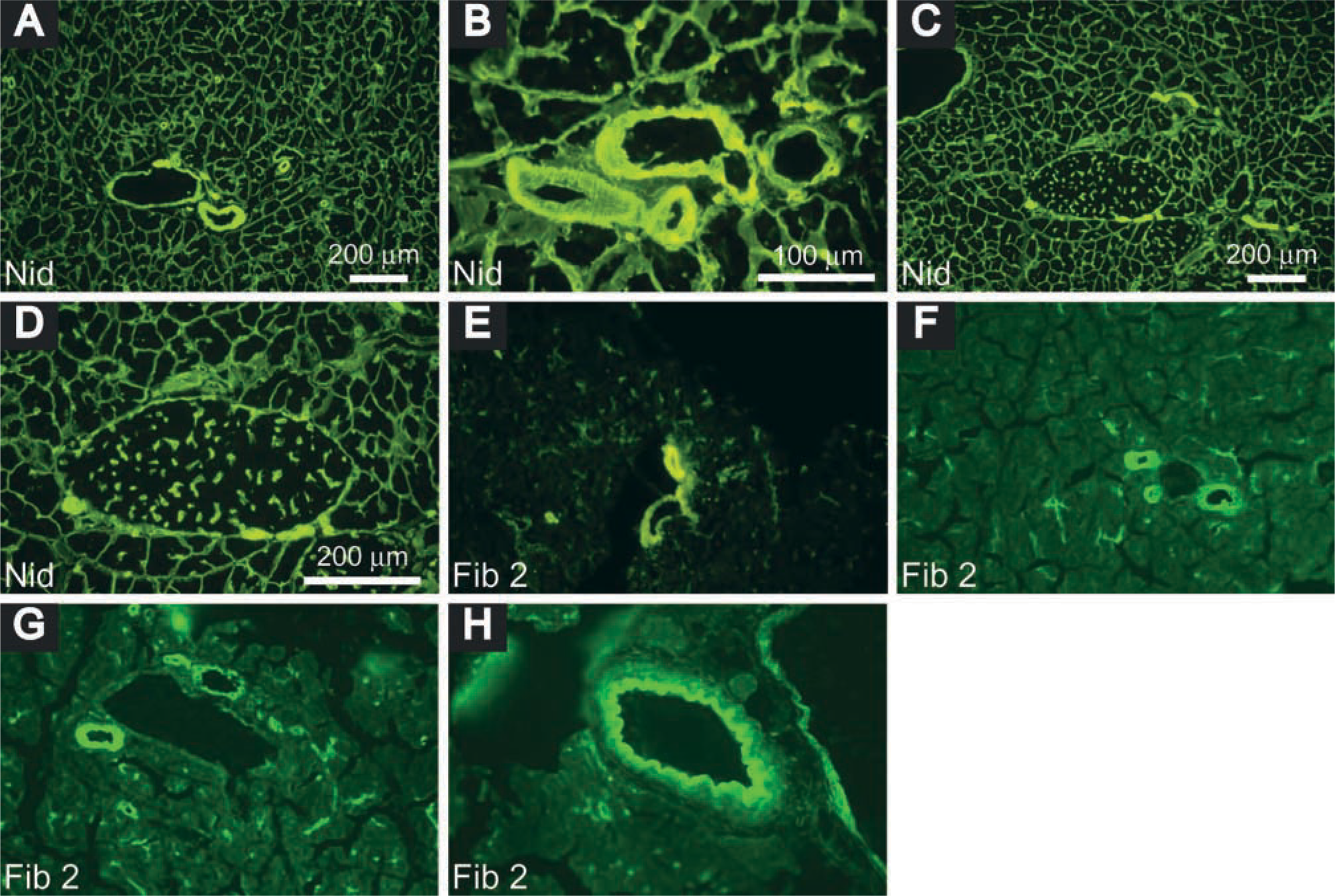
Distribution of nidogen and fibulin in adult mouse pancreas. Nidogen, detected by rabbit anti-mouse nidogen Ig and FITC-conjugated goat anti-rabbit Ig, was expressed in interlobular blood vessels (
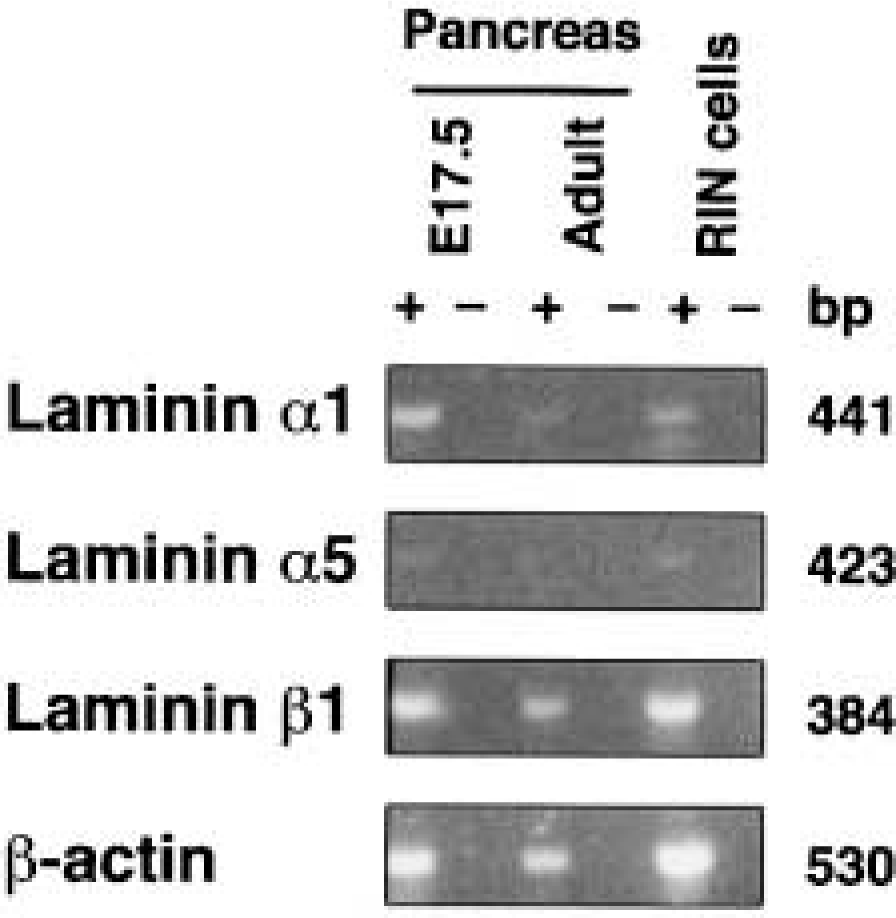
RT-PCR analysis of laminin chains in adult mouse pancreas. mRNAs were incubated with (+) or without (-) reverse transcriptase and subjected to PCR with specific primers as described in Materials and Methods.
To provide additional evidence that the laminin α1-chain is not expressed in adult pancreas, RT-PCR analysis of mRNAs was performed. Consistent with the immunofluorescence studies, laminin α1 transcripts were barely detectable, whereas those for laminin β1 were readily detected (Figure 3). mRNA for laminin α5 was detected only in E17.5 pancreas and RIN cells (Figure 3).
Summary: distribution of integrins in adult mouse pancreas

Integrin Expression
Integrin subunits exhibited a striking cell-specific expression pattern (Table 3). Integrin α6-subunit was strongly expressed basally in acinar cells, where it interacts with laminin, but was not detected in interlobular arteries or in veins and ducts. It also appeared to be expressed in epithelial cells of various orders of pancreatic ducts from interlobular to intercalated ducts, and in endothelial cells (Figures 4A-4C). However, islet cells clearly did not express this integrin subunit (Figure 4D). Integrin α3 was expressed weakly in acinar cells but strongly in arteries, veins, and intralobular ducts (Figures 4K and 4L). Integrin β1 was expressed in various compartments of the pancreas, with strong expression in interlobular duct epithelial cells and endothelial cells (Figures 4E and 4F), and in islet cells (Figure 4G), but with weak expression in acinar cells. β4 appeared to be expressed in epithelial cells in various orders of the duct system (Figures 4H and 4I), including within islets (Figure 4J) but not in acinar cells.
To provide additional evidence that the absence of the integrin α6-subunit in mouse islet cells was not species-specific, a rat β-cell line, RIN-A12, was analyzed by immunoprecipitation-blotting. Neither α6β1 nor α6β4 integrin was detected in the RIN cells (Figure 5), supporting the findings of our immunocytochemical studies. Immunoprecipitation-blotting further demonstrated that the α3-subunit is probably associated with β1 to form α3β1-integrin in mouse islet cells.
Discussion
This study has characterized the expression pattern of laminins and their integrin receptors in the adult pancreas. Laminin isoform expression varied with tissue compartments in the pancreas but was not detected extracellularly in islets. Integrin receptor subunit expression had a cell-specific pattern. Laminin-1 was undetectable in the adult pancreas. We previously showed that laminin α1, unique for laminin-1, is expressed in the BM of developing pancreatic duct epithelium between E13.5 and E17.5 (Jiang et al. 1999). However, laminin α1 was not detected in any compartment of the adult pancreas, consistent with our RT-PCR analysis and a recent report (Virtanen et al. 2000). These data support the current view that laminin α1 is expressed in the epithelial BM during early development but not in the adult (Miner et al. 1997; Virtanen et al. 2000), and imply that laminin-1 may be involved in the proliferation (through α6 integrins) and differentiation (via α-dystroglycan) of the pancreatic precursor cells (Jiang et al. 2001). Strong staining of laminin β1- and γ1-chains and weak staining of laminin α5-chain indicated the existence of laminin-10 (α5β1γ1) in the BM of acinar cells, consistent with the notion that laminin-10 is an adult isoform of epithelial laminin (Ferletta and Ekblom 1999). Laminin α2, α5, β1, γ1 and γ2 were detected in blood vessels, especially arteries, indicating that laminin-2 (α2β1γ1), -8 (α4β1γ1), and -10 are expressed in the BM of these tissues. The functional significance of these laminin isoforms expressed in the blood vessels remains to be determined.
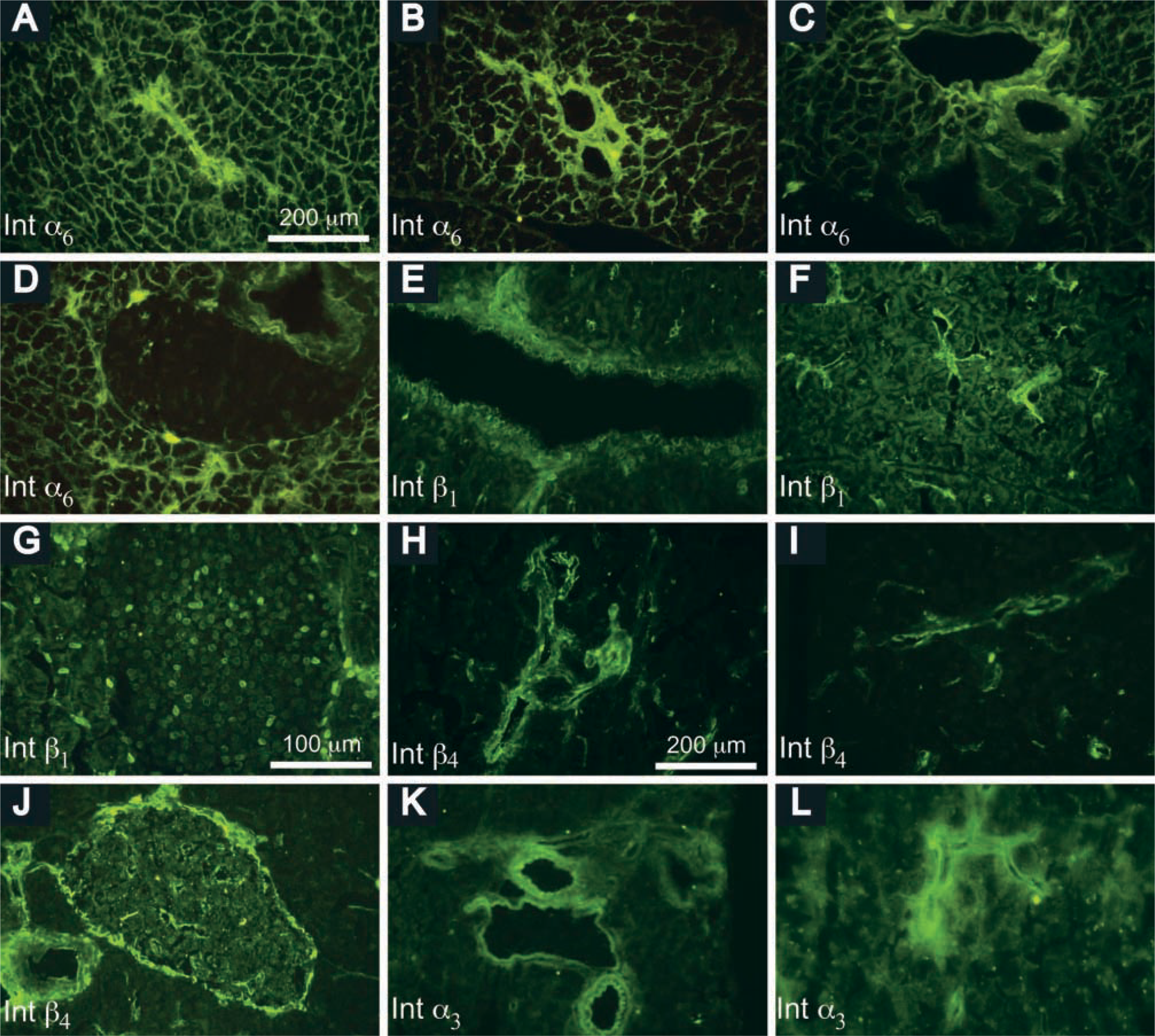
Distribution of integrin subunits in the adult mouse pancreas. Integrin α6-subunit, detected by rat anti-mouse integrin α6 MAb IgG2a and FITC-conjugated rabbit anti-rat Ig, was expressed in acinar cells and intercalated (
Our results show that major ECM proteins are absent extracellularly between islet cells, as are nidogen-1 and fibulin-1 and -2. Similarly, a range of collagen isoforms—fibronectin, vitronectin, and elastin—were not detected between islet cells (van Deijnen et al. 1992; Meyer et al. 1997).
These studies provide molecular evidence to support a previous electron microscopic study (Like et al. 1978) that a BM structure is not found around islet cells. This feature may be related to the diffusion of secreted hormones into intra-islet capillary networks. Lack of ECM proteins between islet cells indicates that the function of islet cells in vivo is regulated mainly by soluble factors and cell-cell interactions mediated, for example, by integrins (see below). However, in an artificial environment, the function of dispersed islet cells can be partially maintained by attachment to ECM in vitro. Thus, insulin secretion can be boosted by attachment to laminin-5 secreted by a rat carcinoma cell line (G804) (Baker et al. 1996; Bosco et al. 2000). Similarly, proinsulin, insulin content, and stimulated insulin release in cultured rat islets can be partially preserved by attachment to collagen II and fibronectin (Wang et al. 1999).
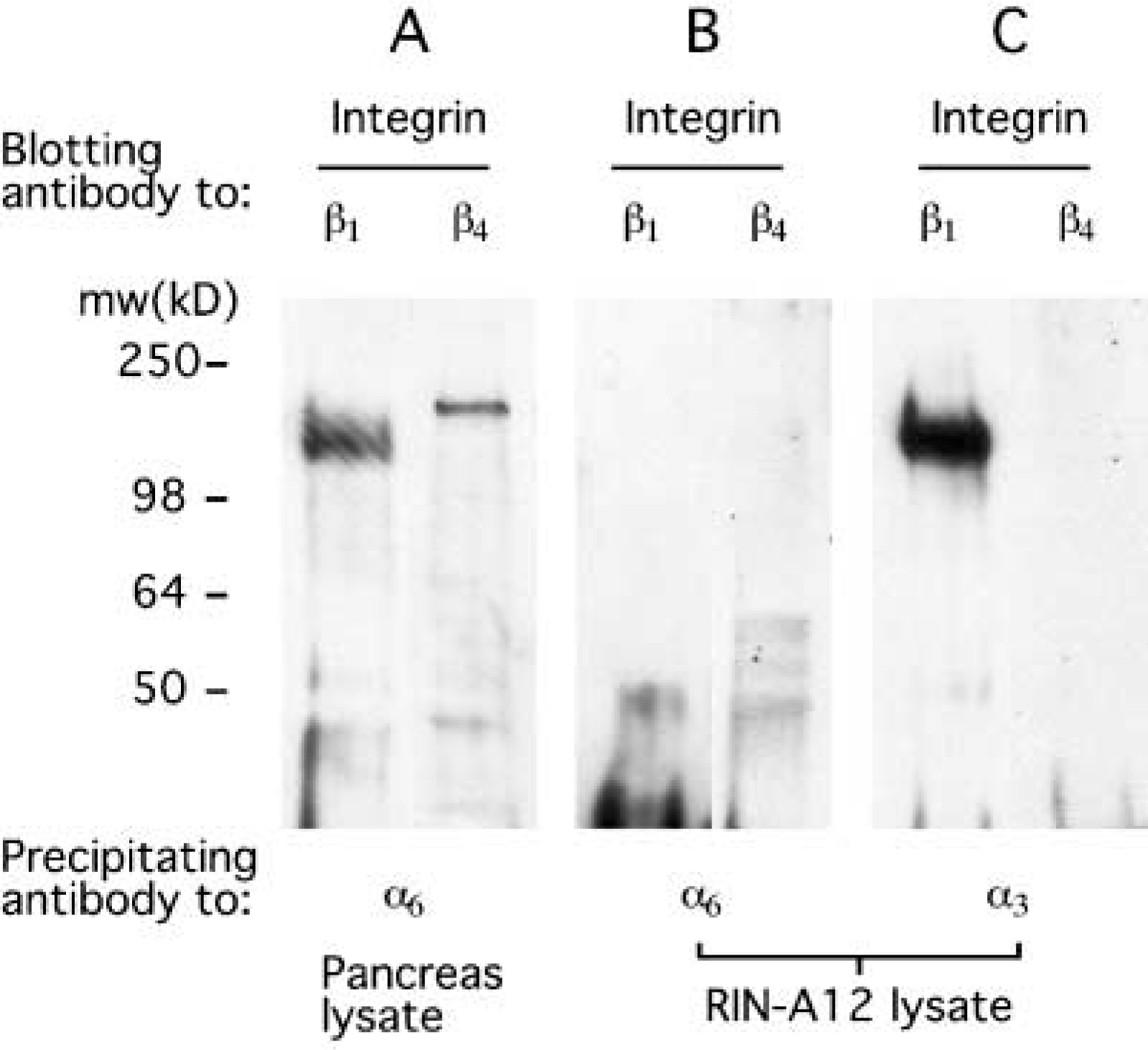
Expression of integrin subunits in RIN A12 β-cells. Pancreas cell lysate (
Only a limited amount of information on the expression pattern of integrin subunits in the adult pancreas has hitherto been available. We found that the integrin α6-subunit was undetectable in adult mouse islet cells by indirect immunofluorescence and immunoprecipitation, consistent with previous immunochemical studies in several species, including humans (Terpe et al. 1994; Kantengwa et al. 1997; Wang et al. 1999). This suggests that α6 integrin signaling is not required for adult β-cell function. Because α6 integrins transduce a moderate proliferation signal in the developing pancreas (Jiang et al. 2001), absence of α6 integrin expression in the adult islet cells may, at least partially, be attributed to the low proliferative potential of these cells. However, our observation contrasts with a report (Bosco et al. 2000) that the α6-subunit is presen on primary rat β-cells. This discrepancy may be partially due to differences in experimental techniques and antibody used.
What is the dimerization partner for the integrin β1 subunit on islet cells? There are six known members of the β1 integrin subfamily: α1β1, α2β1, α3β1, α6β1, α7β1, and α9β1. Our study has demonstrated that the α6β1 integrin is absent on mouse islet cells. However, we observed that the α3-subunit was weakly detected on islet cells, suggesting the existence of α3β1 integrin. This was supported by our immunoprecipitation—blotting experiments on RIN cells and by the findings of others on primary and transformed rat islet cells (Kantengwa et al. 1997; Wang et al. 1999). α3β1 integrin may also be at least partially responsible for binding to laminin-5 secreted by 804G cells, a rat carcinoma cell line, and for increasing proliferation of human fetal islet cells (Beattie et al. 1996). In addition, the function of α3β1 integrin may partially overlap with α6 integrins because the islet cell lineage develops normally in homozygous α6 gene knockout (α6 −/-) mice (Jiang et al. 2001), but not in double α6 −/-/α3 −/- mice (De Arcangelis et al. 1999).
Integrin β4-subunit expression is primarily restricted to cells of ectoderm- and endoderm-derived tissues, such as skin and intestine (Belkin and Stepp 2000). This subunit is unique among integrins in that it has a very large cytoplasmic domain (∼1000 amino acid residues compared to less than 50 residues in other integrin β-subunits). The expression of α6β4 in epithelial cells of various orders of pancreatic ducts may be associated with hemidesmosome formation along the basement membrane (Dowling et al. 1996; van der Neut et al. 1996).
In conclusion, laminin and its integrin receptors exhibit a selective pattern of expression in the adult pancreas. Understanding the functions of these molecules may facilitate the engineering of specific pancreatic cells such as insulin-producing β-cells.
Footnotes
Acknowledgements
Supported by a Juvenile Diabetes Research Foundation Fellowship and a Diabetes Australia Research Grant (2001) (F-XJ) and by the National Health and Medical Research Council of Australia (LCH).
We thank Dr L.M. Sorokin (Institute of Experimental Medicine, University of Erlangen-Nurnburg, Germany), who kindly provided rat monoclonal anti-mouse laminin α1 (clone 198), α2 (clone 4H8), and α5 (clone 4G6) antibodies and rabbit anti-nidogen-1 (#913 and 914), fibulin-1 (#1034), and fibulin-2 (#1028) antibodies. We also thank Catherine O'Shea for secretarial assistance.
