Abstract
α;1,4-N-acetylglucosaminyltransferase (α4GnT) is a glycosyltransferase that mediates transfer of GlcNAc to βGal residues with α1,4-linkage, forming GlcNAcα1 → 4Galβ→R structures. In normal human tissues, glycoproteins having GlcNAcα1→4Galβ→R structures at non-reducing terminals are exclusively limited to the mucins secreted from glandular mucous cells of gastric mucosa, Brunner's gland of duodenum, and accessory gland of pancreaticobiliary tract. Recently, we have isolated a cDNA encoding human α4GnT by expression cloning. Although α4GnT plays a key role in producing this unique glycan in vitro, the actual localization of α4GnT was not determined. In this study we examined the localization of α4GnT in various human tissues, including gastrointestinal mucosa, using a newly developed antibody against human α4GnT. The specificity of the antibody was confirmed by analyses of human gastric adenocarcinoma AGS cells transfected by α4GnT cDNA. Expression of α4GnT was largely associated with the Golgi region of mucous cells that produce the mucous glycoproteins having GlcNAcα1→4Galβ→R, such as the glandular mucous cells of stomach and Brunner's gland. An immunoprecipitation experiment disclosed that two distinct mucin proteins, MUC5AC and MUC6 present in gastric mucin, carried the GlcNAcα1→4Galβ→R structures. These results indicate that α4GnT is critical to form the mucous glycoproteins having GlcNAcα1→4Galβ→R on MUC6 and MUC5AC in vivo.
(
G
Recently, it was demonstrated that, in the gastrointestinal tract, distribution of the mucins identified by HIK1083 antibody specific for GlcNAcα1→4Galβ→R and that of Class III mucin were identical (Ishihara et al. 1996; Ota et al. 1998). These results strongly suggested (a) that Class III mucin contained GlcNAcα1→4Galβ→R and (b) that PCS identified this particular carbohydrate moiety. To test these hypotheses, we have isolated a cDNA encoding α1,4-N-acetylglucosaminyltransferase (α4GnT) from a human stomach cDNA library by expression cloning and have established that Class III mucin identified by PCS characteristically contained terminal GlcNAcα1→4Galβ→R structures (Nakayama et al. 1999, 2000). We have also demonstrated that this enzyme catalyzes transfer of GlcNAc from UDP-GlcNAc to βGal residues preferentially present in O-glycans with α1,4-linkage, forming GlcNAcα1→4Galβ→R (Nakayama et al. 1999).
Extensive studies have been carried out to determine the tissue localization of glycosyltransferases such as β1,4-galactosyltransferase-I and α1,3-fucosyltransferase-VI using immunohistochemistry (Ichikawa et al. 1999; Schnyder–Candrian et al. 2000). Regarding the tissue distribution of α4GnT, it was demonstrated by Northern blotting analysis that α4GnT was transcribed only in stomach and pancreas (Nakayama et al. 1999), but the actual localization of this enzyme remained to be determined. In addition, mucin core proteins carrying GlcNAcα1→4Galβ→R in gastric mucin were not clarified.
In the present study we have developed monospecific polyclonal antibody I17K against human α4GnT by immunizing rabbits with a synthetic peptide corresponding to the COOH-terminal end of this enzyme. The specificity of I17K antibody was confirmed by analyzing human gastric adenocarcinoma AGS cells transfected by α4GnT cDNA. Using I17K antibody, we have examined the immunohistochemical localization of α4GnT in various human tissues, including gastrointestinal tract. Double immunostaining of gastroduodenal mucosa with I17K antibody together with HIK1083 antibody was also carried out. Finally, we found that MUC5AC and MUC6 immunoprecipitated from gastric mucin carried GlcNAcα1→4Galβ→R structures detected by Western blotting analysis.
Materials and Methods
Production of Polyclonal Antibody Against Human α4GnT
The monospecific polyclonal antibody I17K, directed to the COOH-terminal end of human α4GnT, was generated against a synthetic peptide, IKGPEGSVTGELGPGNK, corresponding to the amino acid residues of 324–340 (Nakayama et al. 1999). The NH2-terminal end of the synthetic peptide was coupled with keyhole limpet hemocyanin, then immunized in rabbits. Primary intradermal injection was given with 0.2 mg of the antigen in complete Freund's adjuvant and 0.2 mg for the following four injections. The polyclonal serum was purified using 5 ml of agarose gel coupled with 5.0 mg of the synthetic peptide used as immunogen.
Establishment of Human Gastric Adenocarcinoma AGS Cells Stably Expressing GlcNAcα1→4Galβ→R
To establish a cell line stably expressing α4GnT, human gastric carcinoma AGS cells were co-transfected with a mammalian expression vector encoding human α4GnT (pcD-NAI-α4GnT) and pSV2neo encoding G418 resistance gene (10:1 by molar ratio) using LipofectAmine (Life Technologies; Grand Island, NY) as described previously (Nakayama et al. 1999). For a control experiment, mock transfection was done by using pcDNAI instead of pcDNAI-α4GnT. After the selection with G418, AGS cells stably expressing GlcNAcα1→4Galβ→R were established by immunofluorescent staining with HIK1083 antibody specific for GlcNAcα1→4Galβ→R (Ishihara et al. 1996), which was kindly provided by Drs. Kazuhiko Ishihara and Kyoko Hotta (Kitasato University School of Medicine; Isehara, Japan).
Laser Confocal Microscopy
The AGS-α4GnT cells as well as mock-transfected AGS cells were grown on coverslips in Dulbecco's modified Eagle's medium to 60% confluence. Then they were fixed with 20% formaldehyde in PBS for 20 min at room temperature. After washing with PBS, these cells were permeabilized with 0.2% saponin as described previously (Ichikawa et al. 1999) and then incubated with a mixture of I17K and HIK1083 antibodies, or I17K and antisera against a Golgi maker, Vti1a (Xu et al. 1998) purchased from BD Transduction Laboratories (Lexington, KY). After rinsing with PBS, these cells were incubated with fluorescence-labeled secondary antibodies. In the double immunostaining with I17K and HIK1083 antibodies, fluorescein isothiocyanate (FITC)-labeled anti-rabbit immunoglobulins (DAKO; Glostrup, Denmark) for I17K, and rhodamine-labeled anti-mouse IgM (Southern Biotechnology Associates; Birmingham, AL) for HIK1083 were used, whereas in the double immunostaining with I17K and antisera against Vti1a, FITC-labeled anti-rabbit immunoglobulins (DAKO) for I17K and rhodamine-conjugated goat anti-mouse IgG (Southern Biotechnology Associates) for anti-Vti1a were used. After washing with PBS, coverslips were mounted with Vectashield (Vector Laboratories; Burlingame, CA) and viewed under a laser confocal microscope LSM510 (Carl Zeiss; Jena, Germany).
Tissue specimens of normal gastric fundic mucosa, duodenal mucosa, and pancreas and gallbladder showing gastric metaplasia were selected from the pathology files of the Central Clinical Laboratories (Shinshu University Hospital; Matsumoto, Japan). These tissue specimens were doubly immunostained with HIK1083 antibody and anti-MUC5AC antibody, CLH2 (Novocastra; Newcastle, UK), or HIK1083 antibody and anti-MUC6 antibody, CLH5 (Novocastra) after microwave irradiation for 25 min in a 1.0 mM EDTA–NaOH solution, pH 8.0 as described before (Kim et al. 1999). The characterizations of CLH2 and CLH5 were described in detail by Reis et al. (1997, 2000). For secondary antibodies, FITC-conjugated anti-mouse IgM (μ-chain specific) (Immunotech; Marseille, France) for HIK1083 antibody and rhodamine-labeled anti-mouse IgG (γ-chain specific) (Southern Biotechnology Associates) for anti-MUC5AC and anti-MUC6 antibodies were used. The localization of these molecules in the gastric mucosa was analyzed using the LSM510 laser confocal microscope.
Western Blotting Analysis
To test whether the I17K antibody specifically detected human α4GnT, cell lysates prepared from 5 × 105 of AGS-α4GnT cells or mock-transfected AGS cells in the presence of a protease inhibitor cocktail Complete (Roche Molecular Biochemicals; Mannheim, Germany) were separated by SDS-PAGE (10% gel) in the presence of β-mercaptoethanol and then transferred onto nitrocellulose membranes. The blots were incubated with I17K antibody, and then with anti-rabbit immunoglobulins conjugated with HRP using an ECL Western blotting analysis system (Amersham Pharmacia Biotech; Piscataway, NJ). The peroxidase reaction was visualized using the same kit.
Immunohistochemistry
Various normal and metaplastic human tissues were selected from the pathology files of the Central Clinical Laboratories (Shinshu University Hospital). These tissue specimens sliced into 3-μm thickness were immunostained with the I17K antibody using a Super Sensitive detection kit (BioGenex Laboratories; San Romon, CA) based on the biotin–streptavidin–peroxidase method (Hsu et al. 1981). Briefly, deparaffinized tissue sections were treated with 0.3% H2O2 in methanol and then, blocked with 1% normal goat serum in Tris-buffered saline (TBS). Sections were incubated with the I17K antibody for 1.5 hr. After washing with TBS, they were incubated with biotinylated anti-rabbit IgG and then with HRP-labeled streptavidin. The peroxidase reaction was developed with a diaminobenzidine (DAB)/H2O2 solution and counterstaining was performed with hematoxylin. In control experiments performed by replacing the primary antibody with preimmune serum or omitting the primary antibody from the staining procedure, no specific staining was found.
For double immunostaining with I17K and HIK1083 antibodies, tissue sections of normal stomach and duodenum were incubated with the I17K antibody, followed by application of alkaline phosphatase-conjugated goat anti-rabbit IgG (Roche Molecular Biochemicals). Visualization was performed with nitroblue tetrazolium and 5-bromo-4-chloro-3-indoyl phosphate (Roche Molecular Biochemicals). After washing with water, sections were incubated with HIK1083 and then reacted with biotinylated secondary antibody, followed by the streptavidin–HRP conjugate using a HistoStain SP kit (Zymed Laboratories; South San Francisco, CA). The peroxidase reaction was visualized with a 3-amino-9-ethyl carbazole/H2O2 solution by using the same kit. After washing with water, sections were mounted with Glycergel (DAKO).
Immunoelectron Microscopy
Ultrastructural immunohistochemistry of α4GnT in the mucous neck cells of gastric mucosa was performed by the pre-embedding method as described previously, with slight modifications (Saito et al. 1988). Biopsy samples of normal fundic mucosa were obtained from a volunteer using a gastroscope after informed consent was obtained. Briefly, the fresh tissue samples were fixed in 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4, for 12 hr at 4C. Then they were washed in increasing concentrations of sucrose (10%-20%) in PBS and immediately frozen in an OCT compound (Sakura Finetek; Torrance, CA) at −80C. Frozen sections sliced at 10-μm thickness were placed on MAS-coated slides (Matsunami Glass; Osaka, Japan) and then air-dried for 30 min. After blocking with 1% BSA in PBS, these tissue specimens were incubated with the I17K antibody overnight at 4C. After washing with PBS, they were incubated with HRP-conjugated goat anti-rabbit immunoglobulins (DAKO) overnight at 4C. Next, the sections were fixed with 1% glutaraldehyde in PBS for 10 min at 4C and then incubated for 30 min at RT with a DAB solution containing 1% dimethylsulfoxide and 0.01 M sodium azide. After developing the peroxidase activity in a DAB solution containing 0.3% H2O2 and 0.01 M sodium azide, sections were fixed with 2% osmium tetroxide in 0.1 M phosphate buffer, pH 7.4, dehydrated in graded alcohol, and then covered with a gelatin capsule (Nisshin EM; Tokyo, Japan) filled with epoxy resin prepared by the method of Luft (1961). After polymerization at 65C overnight, the tissue specimens stuck to the epoxy resin were detached from the glass slides by heating and then trimmed, using a razor blade under a stereomicroscope StereoZoom 6 (Leica; Vienna, Austria). Ultrathin sections were made from these tissue specimens using an ultramicrotome Reichert Ultracut S (Leica) and observed under a transmission electron microscope JEM1010 (JEOL; Tokyo, Japan) at 60 kV accelerating voltage as described previously (Hidaka et al. 2000). A control experiment was performed by omitting the primary antibody, and no specific signal was found.
Immunoprecipitation
Biopsy specimens of the normal fundic mucosa were obtained from a volunteer with a gastroscope after informed consent was obtained. These samples (∼200 mg) were washed with PBS and homogenized in 5 ml of lysis buffer containing 50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1% NP-40, and 0.5% sodium deoxycholate with protease inhibitors, using an Immunoprecipitation Kit (Roche Molecular Biochemicals). Two ml of the lysates were pre-cleared by incubation with 50 μl of a 50% slurry of protein G-Agarose beads for 3 hr at 4C. The extracts were then incubated with anti-MUC5AC antibody (CLH2) or anti-MUC6 antibody (CLH5) for 1 hr at 4C. Immunocomplexes were precipitated with 50 μl protein G-agarose overnight at 4C. After washing with high-salt wash buffer (50 mM Tris-HCl, pH 7.5, 500 mM NaCl, 0.1% NP-40, and 0.05% sodium deoxycholate) and low-salt wash buffer (50 mM Tris-HCl, pH 7.5, 0.1% NP-40, and 0.05% sodium deoxycholate), immunoprecipitates were dissociated from protein G-Agarose beads with electrophoresis sample buffer and then subjected to a 5–15% gradient polyacrylamide gel, followed by transfer to nitrocellulose membrane. The blot was then incubated with HIK1083 antibody for 1 hr at room temperature, followed by visualization with HRP-conjugated goat anti-mouse immunoglobulins using the ECL Western blotting analysis system (Amersham Pharmacia Biotech).
Results
I17K Antibody Specifically Recognizes Human α4GnT Expressed in Transfected AGS Cells
To evaluate the specificity of the I17K antibody, we established a clonal cell line of human gastric adenocarcinoma AGS-α4GnT cells stably expressing GlcNAcα1→4Galβ→R by transfecting with α4GnT cDNA, then doubly immunostained with I17K and HIK1083 antibodies. In parallel, the mock-transfected AGS cells were used as a control. The I17K antibody specifically reacted with dot-like structures dispersed in the cytoplasm of AGS-α4GnT cells (Figure 1A) that expressed GlcNAcα1→4Galβ→R at their cell surface (Figure 1B), whereas no immunoreactivity was found in the mock-transfected AGS cells that did not express GlcNAcα1→4Galβ→R (Figures 1D–1F). It is well known that most of glycosyltransferases reside in the Golgi (Paulson and Colley 1989). Therefore, to determine if the dot-like structures detected by the I17K antibody coincide with the Golgi, we doubly immunostained AGS-α4GnT cells with I17K antibody and antisera against Vti1a that is enriched in the Golgi membrane (Xu et al. 1998). The results clearly demonstrate that α4GnT is largely co-localized with Vit1a (Figures 1G–1I), indicating that α4GnT resides in the Golgi. AGS-α4GnT cells may be unique in that the Golgi apparatus is not confined to the perinuclear region.
In parallel, we analyzed cell lysates made from AGS-α4GnT cells and mock-transfected AGS cells using Western blotting with the I17K antibody. As shown in Figure 2, immunoreactive bands were detected at 40, 42, and 45 kD in AGS-α4GnT cells (Figure 2, Lane 1), but not in the mock-transfected AGS cells (Figure 2, Lane 2). These results, taken together, establish that the I17K antibody specifically recognizes human α4GnT expressed in AGS-α4GnT cells.
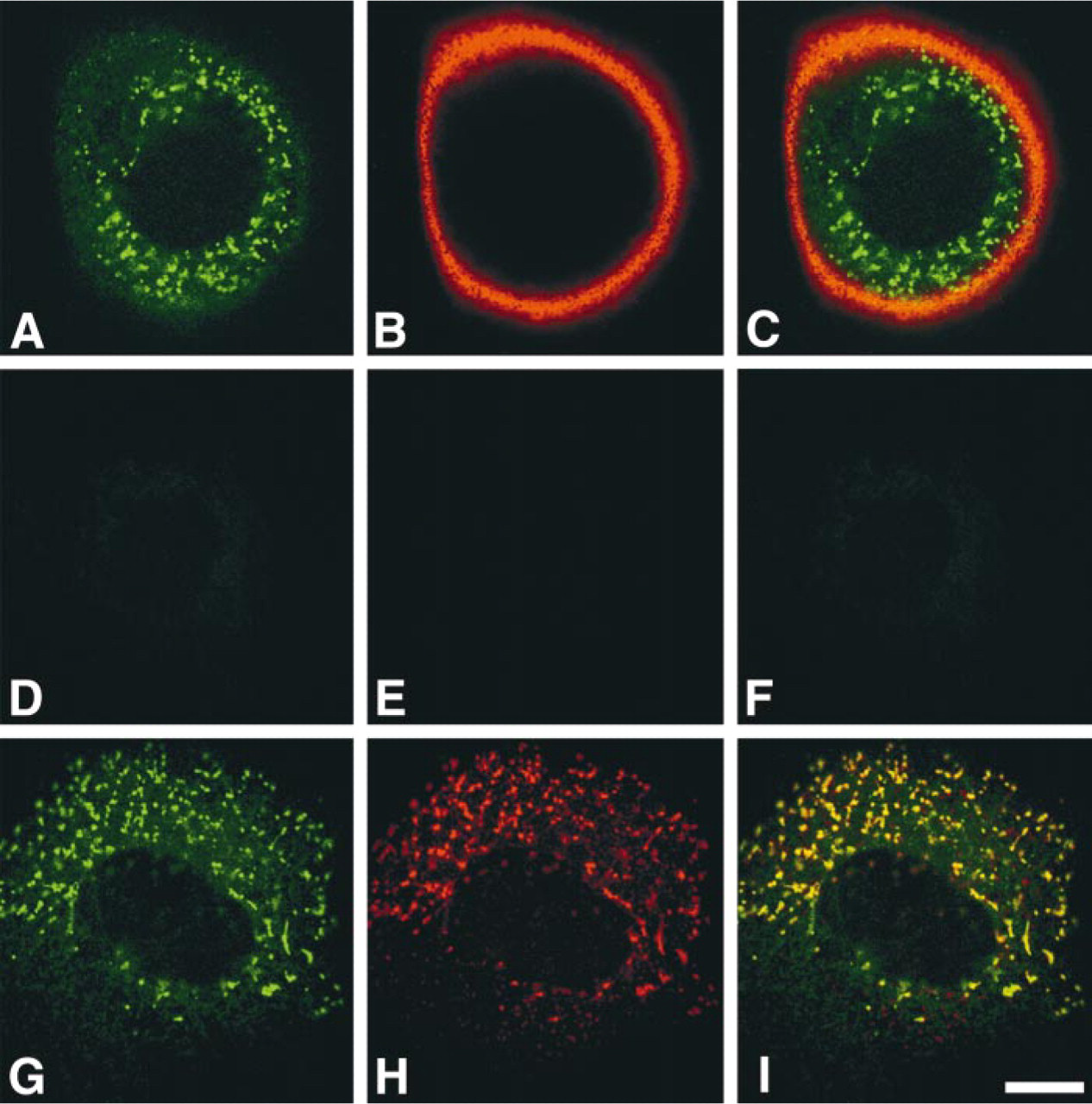
Detection of α4GnT by I17K antibody expressed in AGS-α4GnT cells stably transfected by α4GnT cDNA. (
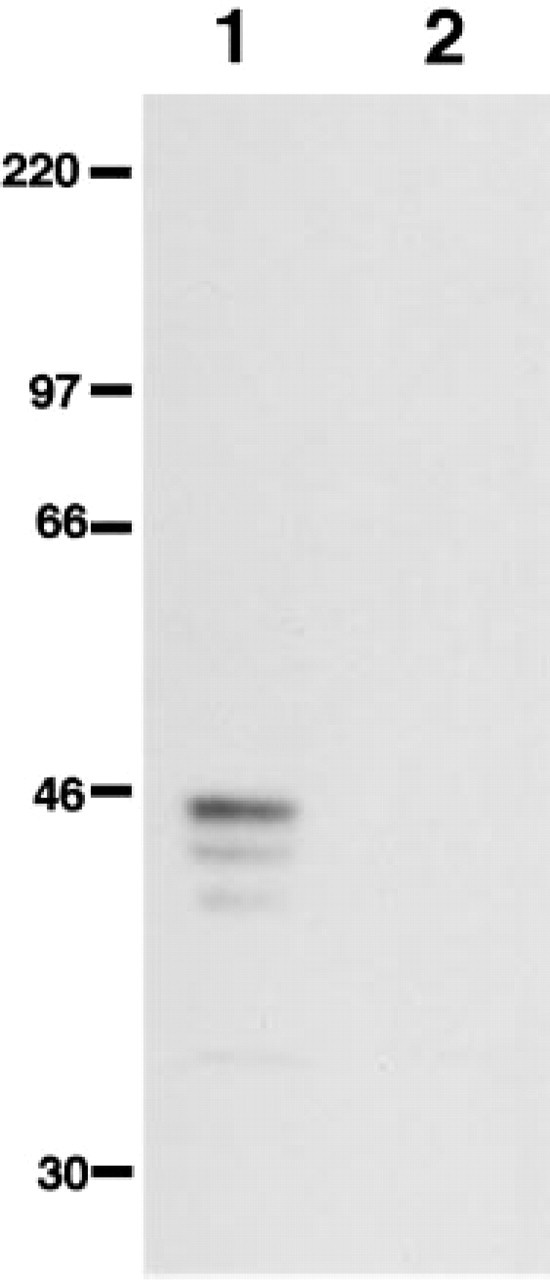
Western blotting analysis of cell lysates from the transfected AGS cells using the I17K antibody. I17K antibody specifically detected α4GnT expressed in AGS-α4GnT cells (Lane 1) but not in mock-transfected AGS cells (Lane 2). Molecular markers are shown in kD.
Distribution of α4GnT in Normal and Metaplastic Human Tissues
We then examined the tissue distribution of α4GnT using I17K antibody specific for this enzyme (Table 1). Among the normal human tissues examined, expression of α4GnT was exclusively limited to the mucous neck cells, cardiac gland, and pyloric gland of gastric mucosa (Figures 3A and 3C–3E), Brunner's gland of duodenum (Figure 3F), and accessory gland of pancreaticobiliary tract, exhibiting a characteristic perinuclear Golgi staining pattern. On the other hand, α4GnT was also detected in gastric metaplasia found in pancreatic duct (Figure 3G) and gallbladder (Figure 3H). However, intestinal metaplasia found in the gastric mucosa did not express α4GnT. Other cells, such as the surface epithelial cells of gastric mucosa (Figure 3B), salivary gland (Figure 3I), submucosal gland of esophagus (Figure 3J), small intestine, and large intestine (Figure 3K) did not express α4GnT. No α4GnT immunoreactivity was detected in non-gastrointestinal tissues, including adrenal gland, Fallopian tube, heart, kidney, lung, lymph node, ovary, placenta, spinal cord, thyroid, and uterus (cervix and body).
We then determined the subcellular distribution of α4GnT in the mucous neck cells of gastric mucosa using immunoelectron microscopy with the I17K antibody. As shown in Figure 4, α4GnT was clearly detected in the medial Golgi of the mucous neck cells.
Human α4GnT Directs the Synthesis of Terminal GlcNAcα1→4Galβ→R Structures In Vivo
To determine if α4GnT expressed in the mucous cells actually produces mucous glycoproteins carrying GlcNAcα1→4Galβ→R, we doubly immunostained gastroduodenal mucosa with the I17K and HIK1083 antibodies. As shown in Figure 5, α4GnT was detected in the mucous cells such as mucous neck cells, pyloric gland, and Brunner's gland that produced GlcNAcα1→ 4Galβ→R. However, the expression level of α4GnT in these mucous cells was variable, and occasionally mucous cells expressing GlcNAcα1→4Galβ→R, but not α4GnT, were found, particularly in the pyloric gland (Figure 5B). In contrast, mucous cells expressing α4GnT but not GlcNAcα1→4Galβ→R were not detected. These results indicate that α4GnT plays a major role in synthesis of GlcNAcα1→4Galβ→R in the gastroduodenal mucosa.
Expression of α4GnT in human gastrointestinal tissues as revealed by immunohistochemistry with the I17K antibody

Terminal GlcNAcα1→4Galβ→R Structures in Gastric Mucin Are Attached to Two Distinct Mucin Core Proteins, MUC6 and MUC5AC
To determine the mucin core proteins carrying the terminal GlcNAcα1→4Galβ→R structures formed by α4GnT, co-expression of MUC5AC and GlcNAcα1→4Galβ→R, or MUC6 and GlcNAcα1→4Galβ→R was analyzed using laser confocal microscopy in gastroduodenal mucosa and in pancreatic duct and gallbladder showing gastric metaplasia. In the fundic mucosa of stomach, MUC5AC was predominantly expressed in the cytoplasm of surface epithelial cells located in the gastric pit (Figure 6A) as described previously (Reis et al. 1999), whereas GlcNAcα1→4Galβ→R structures were detected in the mucous neck cells found in the isthmus and glandular neck of fundic mucosa (Figure 6B). We also detected small but definite numbers of mucous cells doubly expressing MUC5AC and GlcNAcα1→4Galβ→R in the isthmus (Figure 6C). On the other hand, MUC6 was predominantly expressed in the cytoplasm of mucous neck cells located in the glandular base of fundic mucosa (Figure 6D), as shown previously (Reis et al. 1999, 2000), and most of these mucous cells largely co-expressed MUC6 and GlcNAcα1→4Galβ→R (Figures 6E and 6F). Similar results were also found in the pyloric mucosa of stomach and in gastric metaplasia in pancreatic duct and gallbladder, i.e., most of the mucous cells secreting GlcNAcα1→4Galβ→R simultaneously produced MUC6 and, in addition, small numbers of mucous cells actually co-expressed GlcNAcα1→4Galβ→R and MUC5AC (data not shown). Interestingly, MUC5AC was not expressed in the duodenal mucosa, including Brunner's gland (Figures 6G) and accessory gland of pancreaticobiliary tract, and MUC6 was largely co-expressed with GlcNAcα1→4Galβ→R in these mucous cells (Figures 6J–6L). These results, taken together, suggest that GlcNAcα1→4Galβ→R structures could be attached not only to MUC6 but also to MUC5AC in the gastric mucosa, as well as gastric metaplasia found in pancreatic duct and gallbladder, whereas in the Brunner's gland and accessory gland of pancreaticobiliary tract this glycan could be linked to MUC6.
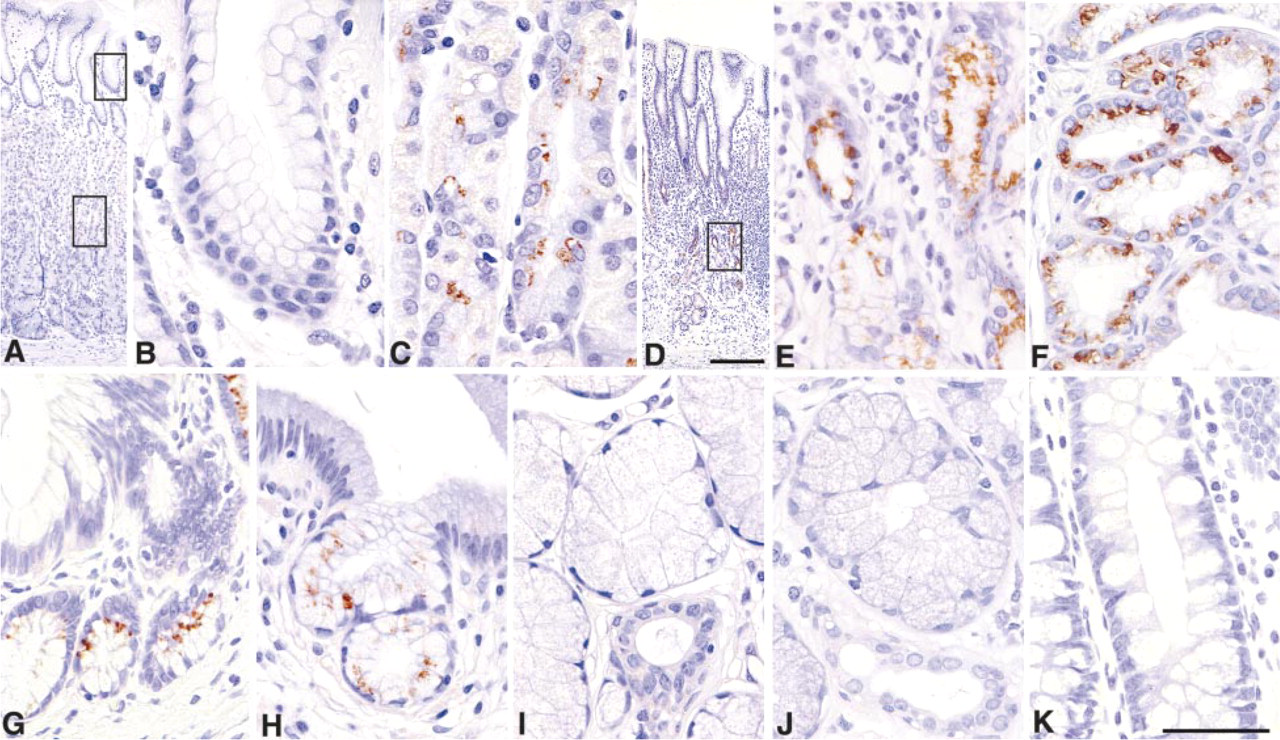
Immunohistochemical demonstration of α4GnT in normal and metaplastic human gastrointestinal tract using the I17K antibody. (
To confirm that the two distinct mucin core proteins, MUC5AC and MUC6, actually carry GlcNAcα1→4Galβ→R, gastric mucin isolated from normal fundic mucosa was immunoprecipitated with anti-MUC5AC or anti-MUC6 antibody and then subjected to SDS-PAGE followed by Western blotting analysis using the HIK1083 antibody. As shown in Figure 7, a broad and high molecular band larger than 220 kD was detected in the immunoprecipitated samples with anti-MUC5AC (Figure 7, Lane 1) as well as anti-MUC6 antibody (Figure 7, Lane 2). These results establish that GlcNAcα1→4Galβ→R structures present in gastric mucin are linked to MUC5AC and MUC6.
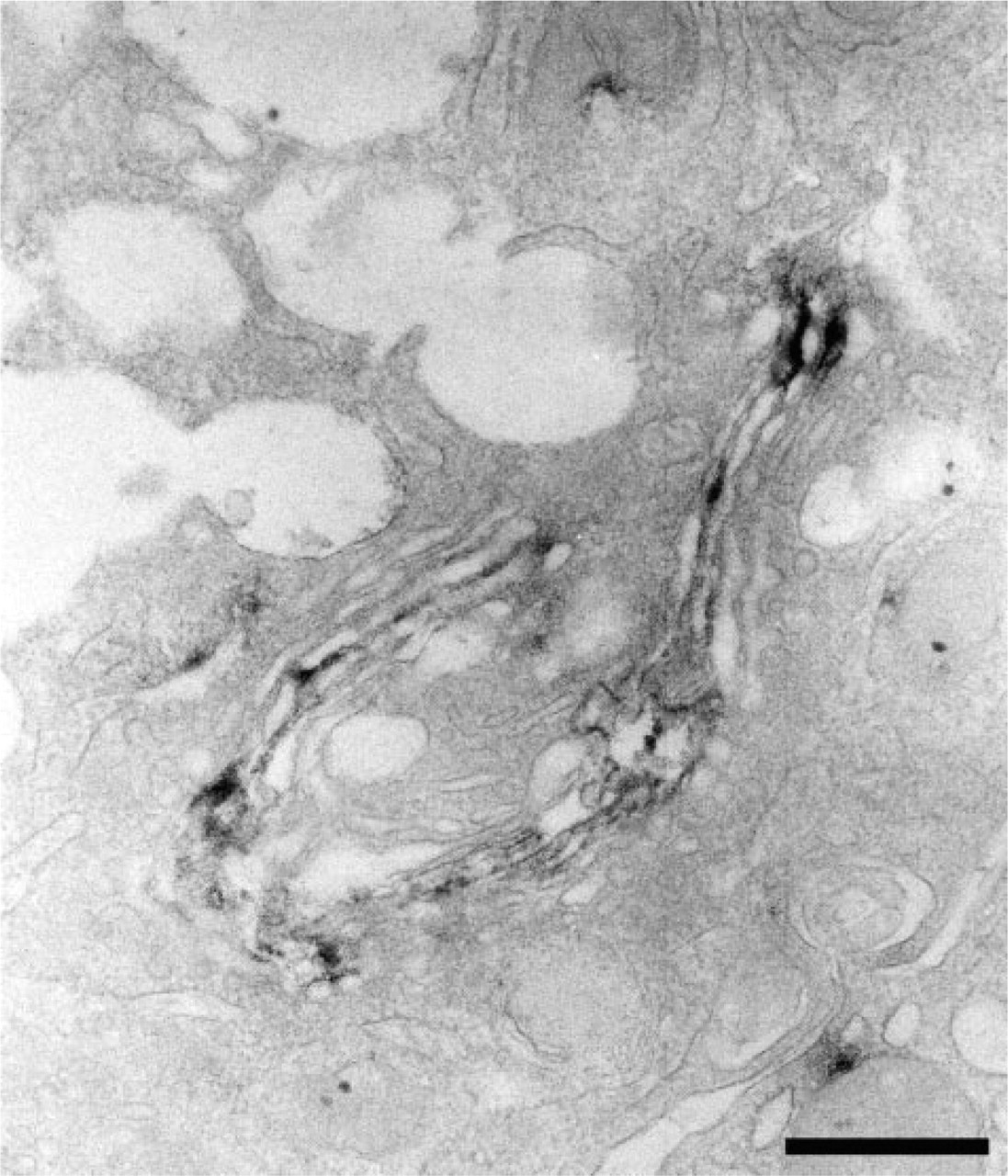
Subcellular localization of α4GnT in the mucous neck cell of gastric fundic mucosa. α4GnT is present in medial Golgi of the mucous cell. Immunoelectron microscopy with the I17K antibody. Bar = 500 nm.
Discussion
In the present study, we have clearly demonstrated that α4GnT, a key enzyme responsible for the biosynthesis of GlcNAcα1→4Galβ→R structures, is expressed in the Golgi of the mucous neck cells and pyloric gland of gastric mucosa, Brunner's gland of duodenum, and pancreatic duct and gallbladder showing gastric metaplasia, using a newly produced polyclonal antibody I17K specific for human α4GnT. In addition, we have also shown that GlcNAcα1→4Galβ→R structures were attached to the two distinct mucin core proteins, MUC5AC and MUC6, present in gastric mucosa.
The antibody I17K was prepared by immunizing rabbits with a synthetic peptide corresponding to the COOH-terminal end of human α4GnT composed of 17 amino acid residues. By using gastric adenocarcinoma AGS-α4GnT cells stably transfected with α4GnT cDNA, we have demonstrated that the I17K antibody specifically reacts with the Golgi of AGS-α4GnT cells. In addition, this antibody recognized the proteins of 40, 42, and 45 kD isolated from these cells. Because the deduced molecular weight of α4GnT is 39.497 kD (Nakayama et al. 1999), four potential N-glycan sites found in the catalytic domain of α4GnT are apparently utilized. Recently we have isolated a cDNA encoding the mouse counterpart of α4GnT and we found that no significant similarity was found in the COOH-terminal end of 17 amino acid residues between human and mouse α4GnTs (unpublished results). This result suggests that the I17K antibody should not recognize mouse α4GnT. In fact, we have shown that this antibody did not react with mouse gastric mucosa (Zhang and Nakayama, personal communication). It is also noteworthy that among the glycosyltransferases cloned thus far, extensive homology at the amino acid level (35% similarity) was found with recently cloned human α1,4-galactosyltransferase (α4GT) responsible for globo-series glycosphingolipid, Gb3 (Galα1→4Galβ1→4Glcβ1→Cer), which is also known as blood group Pk (Furukawa et al. 2000; Keusch et al. 2000; Steffensen et al. 2000). However, the 17 amino acid residues used for the preparation of the I17K antibody were not found in the amino acid sequence of α4GT, indicating that the I17K antibody did not crossreact with α4GT. These results, taken together, indicate that the I17K antibody specifically recognizes human α4GnT.
α4GnT is responsible for the biosynthesis of GlcNAcα1→4Galβ→R structures. In human, mucous glycoproteins having this unique glycan are limited to the mucous cells (cardiac gland, mucous neck cells, pyloric gland) of stomach, Brunner's gland of duodenum, accessory gland of pancreaticobiliary tract, and pancreatic duct and gallbladder showing gastric metaplasia (Nakamura et al. 1998). This enzyme was originally cloned from stomach, and Northern blotting analysis revealed that α4GnT was transcribed in stomach and pancreas (Nakayama et al. 1999), but the actual localization of this enzyme was not known. In the present study, we have for the first time demonstrated the immunolocalization of α4GnT in various human tissues, including the gastrointestinal tract. As expected, α4GnT was significantly expressed in the mucous cells, such as mucous neck cells that produce GlcNAcα1→4Galβ→R, indicating that α4GnT plays a key role in the biosynthesis of GlcNAcα1→4Galβ→R in vivo. Interestingly, double immunolabeling using the I17K and HIK1083 antibodies apparently showed that this enzyme was not always associated with the mucous cells secreting GlcNAcα1→4Galβ→R. On the other hand, GlcNAcα1→4Galβ→R structures can be always found when α4GnT is expressed. These results suggest that α4GnT is not continuously but is intermittently expressed in these mucous cells. However it is also possible that α4GnT-related enzyme(s) might be present in the gastroduodenal mucosa and play a role in the production of GlcNAcα1→4Galβ→R, as exemplified by the fact that polysialic acid is synthesized by two distinct polysialyltransferases, PST and STX (Ong et al. 1998). Future study will be of great significance to address these problems.
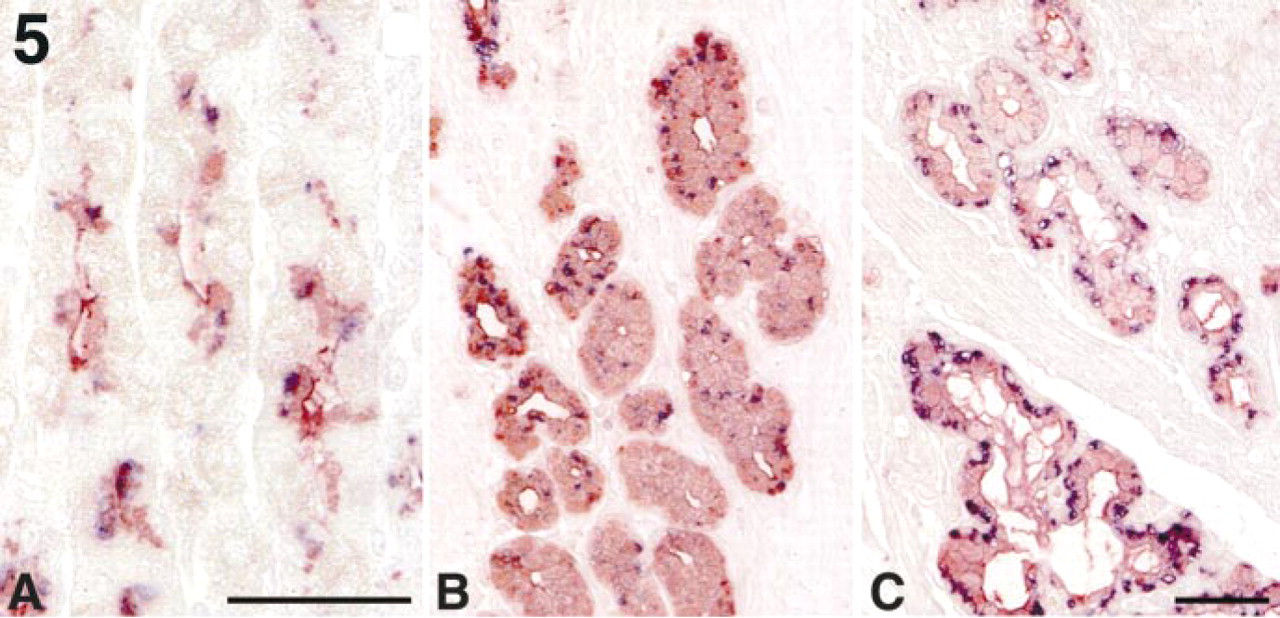
Double immunolabeling for α4GnT and GlcNAcα1→4Galβ→R in gastroduodenal mucosa. In fundic gland (
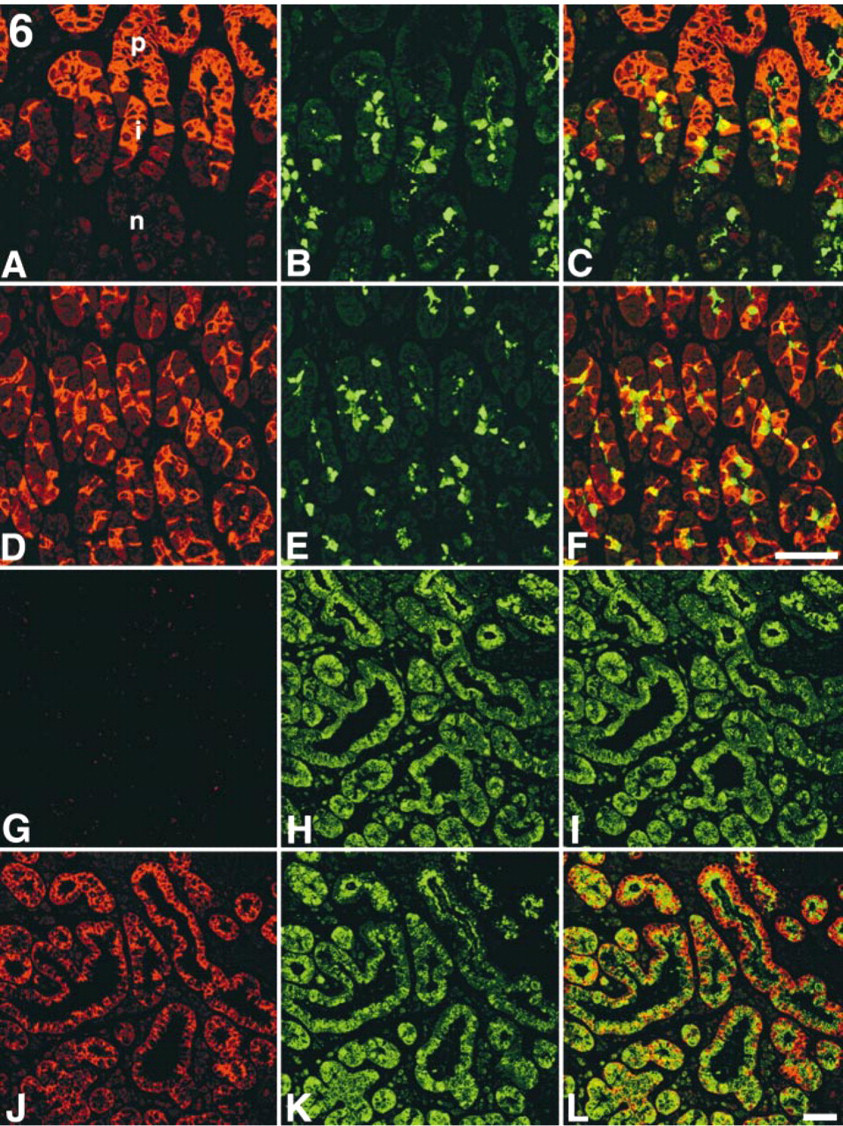
Comparative immunohistochemistry of two mucin core proteins, MUC5AC and MUC6, and GlcNAcα1→4Galβ→R in the fundic mucosa of stomach and Brunner's gland of duodenum. (
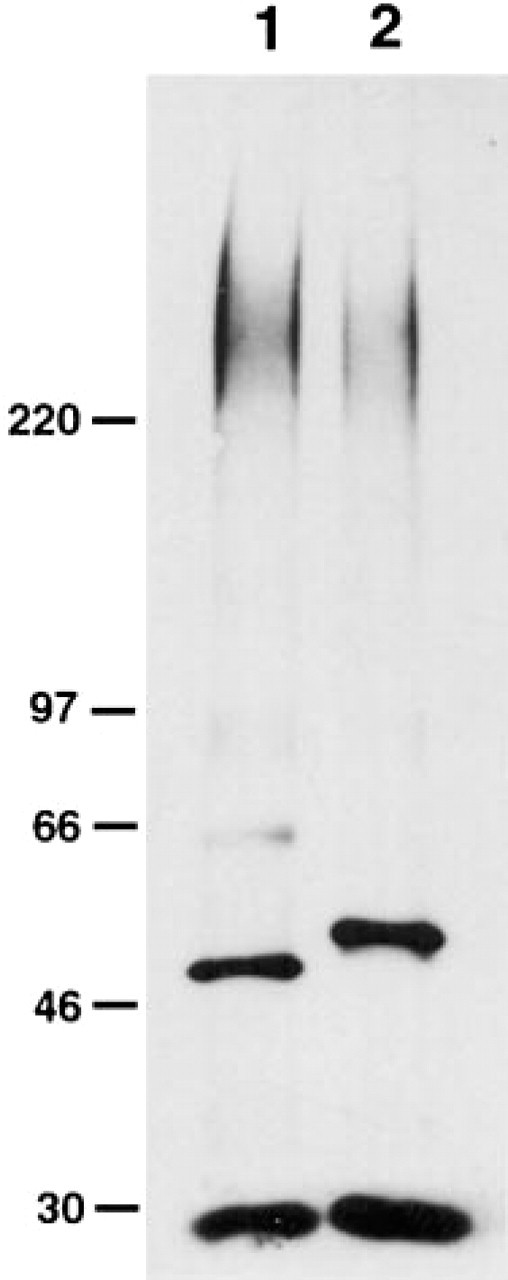
Immunoprecipitation of mucous glycoproteins having GlcNAcα1→4Galβ→R from human fundic mucosa. Homogenized samples made from fundic mucosa were immunoprecipitated with monoclonal anti-MUC5AC antibody (Lane 1) or anti-MUC6 antibody (Lane 2), separated by SDS-PAGE on a 5–15% gel. The blot was then reacted with HIK1083 antibody specific for GlcNAcα1→4Galβ→R. The bands of ∼50 kD and ∼30 kD are immunoglobulin H and L chains, respectively. Molecular markers are shown in kD.
Using immunoelectron microscopy, the subcellular localization of α4GnT in the mucous neck cells of gastric mucosa was determined to be in the medial Golgi. Sugar chains such as O-glycans are synthesized by sequential actions of glycosyltransferases in the Golgi (Paulson and Colley 1989). Therefore, the present result was consistent with the previous report that core 2 β1,6-N-acetylglycosaminyltransferase (C2GnT) resides in cis- to medial Golgi (Skrincosky et al. 1997), because α4GnT most efficiently acts on the core 2 branched O-glycan, GlcNAcα1→6(Galβ1→3)GalNAc, which is synthesized by C2GnT as its acceptor donor (Nakayama et al. 1999).
Among the mucin core proteins, MUC5AC and MUC6 are abundantly expressed in the gastric mucosa (De Bolós et al. 1995; Ho et al. 1995; Reis et al. 1997, 1999, 2000). MUC5AC is predominantly expressed in the surface mucous cells of gastric mucosa, whereas MUC6 is exclusively expressed in the mucous neck cells and in cardiac and pyloric glands. Because the expression pattern of MUC6 and that of mucous glycoproteins having GlcNAcα1→4Galβ→R are quite similar in the gastric mucosa, we considered that MUC6 was a candidate for the carrier protein of this glycan (Nakayama et al. 1999). To test this hypothesis, we examined the fundic mucosa doubly immunostained with anti-MUC6 and HIK1083 antibodies or anti-MUC5AC and HIK1083 antibodies using a laser confocal microscope. As expected, most of the mucous neck cells were found to simultaneously express MUC6 and GlcNAcα1→4Galβ→R. However, mucous cells co-expressing MUC5AC and GlcNAcα1→4Galβ→R were also detectable in the isthmus of the fundic mucosa. These results suggest that not only MUC6 but also MUC5AC can be a carrier protein of GlcNAcα1→4Galβ→R. Indeed, we demonstrate that GlcNAcα1→4Galβ→R structures present in gastric mucin are actually attached to MUC5AC and MUC6 by immunoprecipitation followed by Western blotting analysis (see, Figure 7). De Bolós et al. (1995) reported that Lewisb and sialyl Lewisa are attached to MUC5AC, whereas Lewisy is attached to MUC6. GlcNAcα1→4Galβ→R structures are unique in that they are attached to both MUC5AC and MUC6 in gastric mucin. Regarding the mucous cells found in gastric metaplasia of pancreatic duct and gallbladder, we suggested that GlcNAcα1→4Galβ→R structures could be linked to MUC5AC or MUC6, like gastric mucous cells. In addition, it was also suggested that GlcNAcα1→4Galβ→R structures in Brunner's gland of duodenum and accessory gland of pancreaticobiliary tract could be linked to MUC6 but not MUC5AC (see Figure 6). Future study will be of significance to determine the actual mucin cores carrying this particular glycan in these mucous cells.
Recently, it was shown that mucous glycoproteins having GlcNAcα1→4Galβ→R was produced in gastric and pancreatic cancer (Nakamura et al. 1998). Moreover, mucinous bronchioloalveolar cell carcinoma of the lung and so-called adenoma malignum of the uterine cervix express these particular glycans (Honda et al. 1998; Ishii et al. 1998). Because α4GnT is not expressed in normal lung and uterus, as shown in the present study, it is likely that α4GnT is aberrantly expressed in the lung or uterine cervix, in association with the production of GlcNAcα1→4Galβ→R in these tumor cells. Further study will be of significance to determine whether this enzyme is actually expressed in these cancer cells.
Footnotes
Acknowledgements
Acknowledgments
Supported by Grant-in-Aids for Scientific Research on Priority Area (10178104) from the Ministry of Education, Science, Sports, and Culture of Japan, and for the Ribosome Engineering Project in Organized Research Combination System (to JN), and by National Institutes of Health Grant CA48737 (to MF).
We are grateful to Dr Tsutomu Katsuyama for encouragement and discussion during this study, to Drs Kazuhiko Ishihara and Kyoko Hotta for the generous gift of the HIK1083 antibody, and to Dr Edgar Ong for critical reading of the manuscript.
