Abstract
Objectives
Mucin phenotype is a tool to classify gastric cancer, but the relationship between mucin phenotype and its malignancy is still controversial. This study aimed to clarify the relationship between mucin phenotype and the malignant potential of gastric cancer.
Methods
A total of 82 cases of early-stage differentiated adenocarcinoma (submucosal invasion cases) obtained from surgeries were studied by immunohistochemistry. Gastric mucin phenotype and E-cadherin expression were analyzed in the mucosal and submucosal layer. E-cadherin expression was analyzed by using imaging software (ImageJ) for objective data analysis. Furthermore, the mucin phenotypic shift was analyzed from mucosa to submucosa.
Results
We found that: (1) tumors with intestinal mucin phenotype had statistically more venous invasion in the submucosal lesion; (2) tumors with an intestinal phenotype that showed venous invasion in the submucosal lesion had a higher percentage of tumors that showed loss of phenotype; (3) no dominant change in E-cadherin expression was observed from the mucosa to submucosa.
Conclusion
Tumors with loss of phenotype and submucosal intestinal phenotype showed predominantly more venous invasion, so examining the identification of phenotypes and phenotype shifts can be expected to be a factor that influences treatment strategies after endoscopic treatment or after surgical resection.
Introduction
Gastric cancer is classified in tubular adenocarcinoma; papillary adenocarcinoma; poorly cohesive carcinoma, including signet-ring cell carcinoma; mucinous adenocarcinoma; mixed adenocarcinoma; and other rare histological-subtypes. 1 Tubular adenocarcinoma and papillary adenocarcinoma were recognized as intestinal-types. Poorly cohesive carcinoma, including signet-ring cell carcinoma, was recognized as diffuse-type. 1 Tubular adenocarcinoma is the most common subtype in Japan; the relative frequency was 64% in a nationwide study in Japan. 2 In recent years, gastric cancer has been classified by mucin phenotype; the pathological features of differentiated gastric cancer have been evaluated in more detail.3-5 Gastric phenotype tends to infiltrate and metastasize more easily than the intestinal phenotype in the previous papers.6-9 By contrast, the other groups have reported that a phenotype shift from gastric phenotype to intestinal phenotype was observed with the progression of the tumor, suggesting that intestinal phenotype may be aggressive and advanced. 10 According to Wakatsuki et al., 11 intestinal-type of gastric cancer has a poor prognosis and a high risk of postoperative liver metastasis. Kim et al 12 .reported that the prognosis of early gastric cancer with MUC5AC-positive gastric phenotype is better than that of intestinal and null phenotype early gastric cancers. Some other reports also noted that MUC5AC expression was associated with a favorable prognosis. 13 Various opinions have been made about the clinical differences between mucin phenotypes of gastric cancer. This may be partly since these studies have included multiple cases, including those that examined the mucin phenotype of the entire tumor, those that included cases of advanced cancer, and those that included diffuse-type gastric cancer. Thus, no consensus has been obtained. Therefore, we focused on 82 surgically resected specimens of submucosal invasive gastric cancer and analyzed mucin phenotype (CD10, MUC2, MUC5AC, and MUC6) in the mucosa and submucosa. We compared the mucin phenotype with clinicopathological factors (i.e., lymphatic invasion, venous invasion, lymph node metastasis, etc.). We also evaluated the relationship between the cell adhesion factor E-cadherin and mucin phenotype in submucosal invasive gastric cancer. Our analysis focused not only on the comparison of mucin phenotype and clinicopathological factors but also on how mucin phenotype changes as cancer cells invade mucosa to submucosa and how E-cadherin, one of the epithelial-mesenchymal markers, is related to mucin phenotype.
Material and methods
Patient samples
The clinicopathological characteristics of submucosal invasion carcinoma cases.

U: upper part, M: middle part, L: lower part.
Distal: distal gastrectomy, Total: total gastrectomy, Proximal: proximal gastrectomy.
Well: well differentiated adenocarcinoma, Mod: moderately differentiated adenocarcinoma.
Histological and immunohistochemical staining examination
All the lesions of surgical specimens were cut into slices of 5 mm in width. Gastric cancer specimens were fixed in 10% formalin solution (formalin fixation time, 48–120 h, room temperature) and embedded in paraffin. For the histopathological examination, thinly sliced sections (4 μm) were stained with hematoxylin and eosin (H&E: Hematoxylin 20 min and eosin 3 min at room temperature, usually 25°C). Clinicopathological findings such as depth of tumor invasion, histological type, lymph node metastasis, lymphatic invasion, venous invasion, and stage were reviewed according to the Japanese Classification of Gastric Cancer. 14 Immunohistochemical examination was performed on deparaffinized section using the standard avidin-biotin-peroxidase complex method with an automated immunostainer (Benchmark XT; Ventana Medical System, Tucson, AZ, USA). In brief, deparaffinized slides were treated with tris-EDTA buffer (pH 7.8) at 95°C for 44 min. For blocking endogenous peroxides and protein, the slides were treated with 5% non-fat dry milk at 37°C for 15 min. The slides were incubated with primary antibody for 60 min at room temperature. The antibodies used were: MUC5AC (CLH2; Leica, BondTM Ready-To-Use Primary Antibody, Catalog No: PA0052), MUC6 (CLH5; Leica, BondTM Ready-To-Use Primary Antibody, Catalog No: PA0053), MUC2 (Ccp58; Leica, BondTM Ready-To-Use Primary Antibody, Catalog No: PA0155), CD10 (56C6; Leica, BondTM Ready-To-Use Primary Antibody, Catalog No: PA0270), and E-cadherin (NCH-38; Dako, 1:100, Catalog No: M3612).
Immunohistochemical staining evaluation of mucin and mucin phenotype
The results of staining were categorized into positive and negative expressions. When ≧5% of the carcinoma cells in the neoplastic lesion were stained, it resulted in a positive expression of the lesion. When <5% of the carcinoma cells were stained, it resulted in a negative expression of the lesion according to the previous studies.7,15 The phenotypes were classified into four categories (gastric phenotype, intestinal phenotype, mixed phenotype, and null-type) according to the combination of the expression for phenotypic markers (MUC5AC, MUC6, MUC2, and CD10). When MUC5AC and/or MUC6 were positive with negative MUC2 and CD10, it was categorized as gastric phenotype. When MUC2 and/or CD10 were positive with negative MUC5AC and MUC6, it was intestinal phenotype. When MUC5AC and/or MUC6 were positive along with positive MUC2 and/or CD10, it was a mixed phenotype according to the previous studies.12,16,18 Neither gastric nor intestinal phenotype expression was categorized as a null-type. Furthermore, we divided the patients into two categories: the preserved group and the loss group. The preserved group was defined as those lesions which showed no phenotypic shift from the mucosa to the submucosa (i.e., the case of gastric phenotype in the mucosa and gastric phenotype in the submucosa). The loss group was defined as those lesions which showed loss of all or some phenotypic expression in the submucosa (i.e., the case of gastric phenotype in the mucosa and null-type in the submucosa).
Imaging analysis of E-cadherin expression by immunohistochemistry
We showed how to classify layers and select gastric ducts for E-cadherin measurements in Figure 1. We selected representative sections that infiltrate the deepest submucosa layer. We divided the representative submucosal invasion cancer into three layers (Figure 1(a)): the mucosal surface layer (M1), the mucosal deep layer (M2), and the submucosal layer (SM). We divided the mucosal layer into two parts, the surface to halfway from the base side was the surface layer (M1), and the other half from the muscularis mucosae was the deep layer (M2). We selected representative ducts (M1: 5 ducts, M2: 5 ducts, SM: 3 ducts) from each layer at ×1.25 magnification using light microscopy (OLYMPUS BX53). The typical gastric ducts stained with E-cadherin were shown in Figure 1(b)–(d). Gastric ducts were scanned using light microscopy at ×40 magnification (OLYMPUS cell Sens Standard 1.17, Build 16030). The captured image data were saved using the tagged image file format. Color-deconvolution plugin of ImageJ software [Java 1.6.0_24 (64-bit)] was used to make a binary image of the 3-3′-diaminobenzidine stained area of E-cadherin. We binarized the selected gastric ducts using ImageJ software. The binarized image (Figure 1(e)–(g)) was calculated by ImageJ software. Only the cancerous duct was cropped out from the non-cancerous site (Figure 1(h)–(j)). We calculated the average per uniglandular tube of E-cadherin expression (%) in M1, M2, and SM. The submucosal invasion part was so small that we could analyze only three ducts. The analysis of E-cadherin expression area in submucosal invasive gastric cancer and the method for measuring the expression rate of E-cadherin. We show three layers of submucosal invasive gastric cancer in Figure 1(a): the mucosal surface layer (M1), the mucosal deep layer (M2), and the submucosal layer (SM). We selected representative ducts: 5 from M1 (yellow), 5 from M2 (green), 3 from SM (red). E-cadherin immunohistochemical expression in the mucosa surface layer (M1, Figure 1(b)), the mucosa deep layer (M2, Figure 1(c)), and the submucosa layer (SM, Figure 1(d)). The binarization images of E-cadherin immunohistochemical expression in M1 (Figure 1(e)), M2 (Figure 1(f)), and SM (Figure 1(g)). The target ducts are cropped out and calculated using ImageJ software in M1 (Figure 1(h)), M2 (Figure 1(i)), and SM (Figure 1(j)).
Statistical analyses
The relationship between phenotype and clinicopathological features, invasion, and lymph node metastasis was examined by the χ
2
test and Fisher exact probability test. The level of significance was
Results
Representative image of mucin phenotype
We showed representative images of the intestinal-type in Figure 2. Figure 2(a)–(d) shows the typical intestinal-type images of the loss group. MUC2 staining was maintained in the mucosa; however, MUC2 expression was attenuated in the submucosa in the loss group. Figure 2(e)–(h) shows the typical intestinal-type images of the preserved group. MUC2 staining was maintained in the mucosa and submucosa in the preserved group. Representative image of intestinal phenotype for MUC2 expression in the loss group (a–d) and the preserved group (e–h). The typical histological features on H&E stain ×4 (a and e). The typical expression for the MUC2 stain ×1.25 (b and f), MUC2 stain ×4 (c and g), and MUC2 satin ×40 (d and h). MUC2 is positive in the mucosal layers of both the loss group and the preserved group (c and g). MUC2 is decreased for the submucosal layer (SM) in the loss group (d); on the other hand, MUC2 is preserved for SM in the preserved group (h).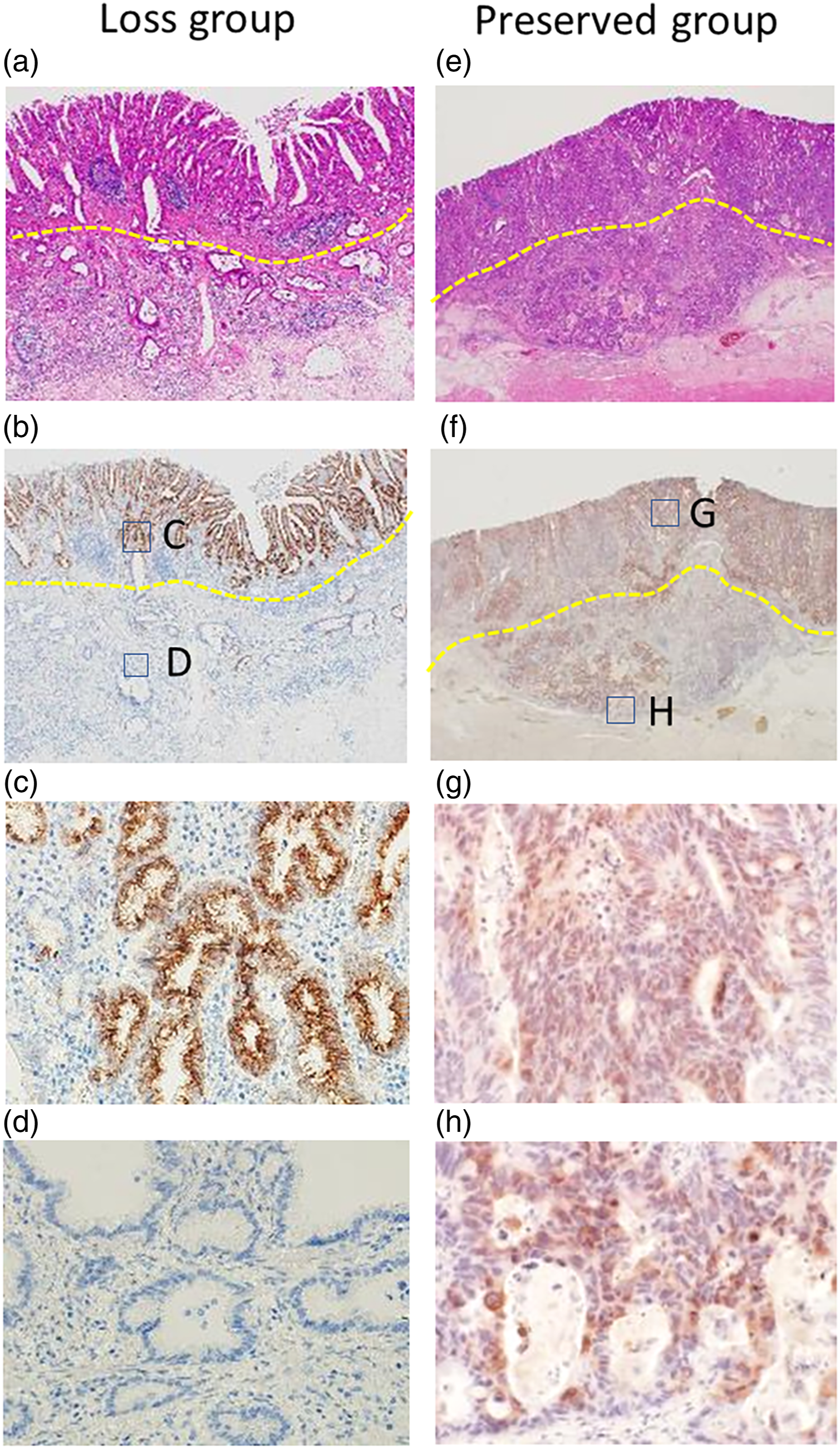
Phenotype of submucosal invasion cancer
The phenotype ratio of the mucosal area and submucosal area.

Phenotypic shift (mucosa to submucosa)
Among the 22 cases of the submucosa intestinal phenotype, 4 cases were mucosal mixed-type, and 18 cases were mucosal intestinal-type. Among the 16 cases of the submucosa gastric phenotype, 9 cases were mucosal mixed-type, and 7 cases were mucosal gastric-type. Among the 3 cases of the submucosa mixed phenotype, all 3 cases were mucosal mixed-type. Among the 41 cases of the submucosa null phenotype, 28 cases were mucosal non-null-type (intestinal or gastric or mixed-type), and 13 cases were mucosal null-type. 41 cases (50%) were in the loss group, 41cases (50%) were in the preserved group. On the other hand, there were no cases that acquired a new mucin phenotype, that is, when the tumor invades the submucosa from the mucosa (Figure 3). Phenotypic shift patterns from the perspective of submucosa phenotype.
Lymphovenous invasion and lymph node metastasis for each submucosa phenotype
Comparison of lymphovenous invasion and lymph node metastasis for each submucosa phenotype.

∗
Venous invasions for the loss group and preserve group from the perspective of submucosal phenotype
The comparison of the number of venous invasions for loss group and preserve group in the perspective of submucosal phenotype.

Immunohistochemical expression of E-cadherin
Immunohistochemical expression of E-cadherin in each layer.

E-cadherin expression of perspective of submucosal phenotype.

M1: mucosal surface layer.
M2: mucosal deep layer.
SM: submucosal layer
E-cadherin expression by submucosal type of each layer between venous invasion group and no venous invasion group.

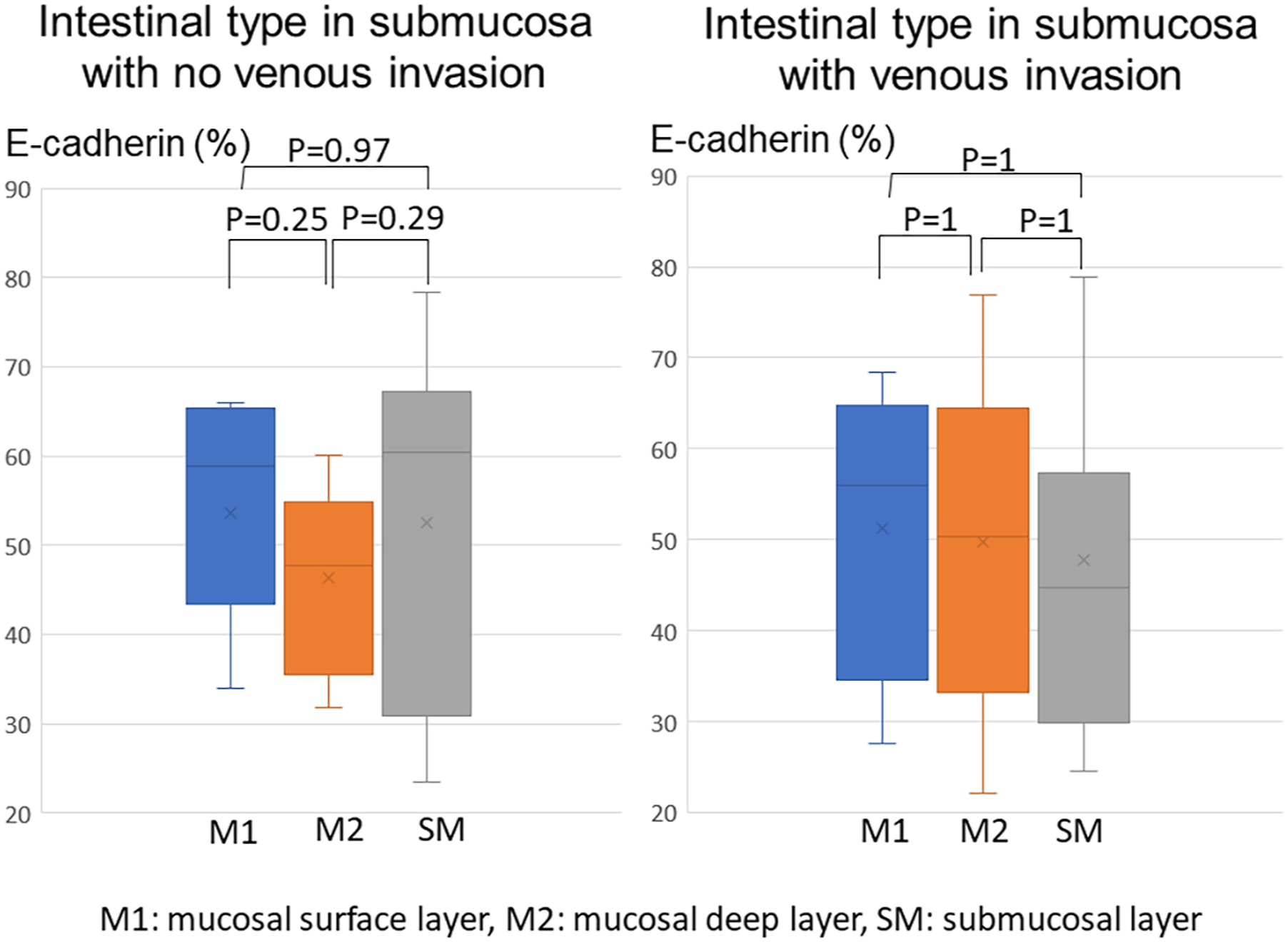
The E-cadherin expression rate of submucosal intestinal-type in venous invasion group and no venous invasion group. We could not get the result of statistically significant differences for E-cadherin expression of the submucosa intestinal-type in venous and no venous invasion group.
Discussion
This study reported changes in the mucosal/submucosal mucin phenotype along with the relationship between lymphatic/venous invasion and mucin phenotype in differentiated early gastric cancer. We also quantitatively evaluated E-cadherin changes, a marker of epithelial-mesenchymal transition in the mucosa and submucosa.
In this study, we found that: (1) tumors with intestinal phynotype had statistically more venous invasion in the submucosal lesion; (2) tumors with intestinal phenotype that showed venous invasion in the submucosal lesion had a higher percentage of tumors that showed loss of phenotype; (3) in differentiated gastric cancer, no dominant change in E-cadherin expression was observed from the mucosa to the submucosa.
Firstly, tumors with intestinal phenotype in the submucosa showed a statistically higher rate of venous invasion. And a large percentage of such cases showed loss of mucin phenotype. Previous studies reported that the gastric phenotype was an independent determinant of lymph node metastasis.6,15,17 Koseki et al. 17 reported that the null-type also showed the highest rate of lymphovascular invasion and lymph node metastasis next to the gastric-type. Nakamura et al. 16 reported that unclassified-type (negative for CD10, MUC2 and Hunan gastric mucin, MUC6) showed a high rate of distant metastasis. Moreover, Han et al 18 .showed that unclassified-type (negative for CD10, MUC2 and MUC5AC, MUC6) gastric cancer has a higher probability of lymph node metastasis, lymphovenous infiltration, and nerve infiltration than the other types. On the other hand, a recent study reported a good prognosis for MUC5AC-positive early gastric cancer, 12 while another study reported a poor prognosis of intestinal type gastric cancer and a high rate of postoperative liver metastasis, 11 so no consensus has been obtained. Many papers that have examined mucin phenotype in gastric cancer have examined the mucin phenotype of the entire tumor and have not analyzed the mucin phenotype of the invasive lesion involved in the tumor’s malignancy.3,4,7-9,11-13,17,18 Some of the previous papers included advanced gastric cancers and poorly differentiated cancers or diffuse-type cancers,11,18 which may have affected the results. However, we are not able to fully explain this discrepancy. Wakatuki et al. reported a correlation between the malignancies of the intestinal form of the mucin phenotype. 11 This result was consistent with our result. They also reported that loss of expression of the runt-related transcription factor 3 genes (RUNX 3) is a poor prognostic factor. In addition, the loss of RUNX3 is associated with intestinal metaplasia of the gastric mucosa19,20 and has been reported to be a poor prognosis in gastric cancer. 21 In this study, we found that the venous invasion rate was higher in intestinal-type cases showing loss of mucin phenotype in the mucosa to the submucosa. These are cases of loss of mucin phenotype from mixed phenotype (gastric and intestinal phenotype) to intestinal phenotype, suggesting that RUNX3 may be involved.
Secondly, in differentiated gastric cancer, no dominant change in E-cadherin expression was observed from the mucosa to the submucosa. We analyzed the expression of E-cadherin in M1, M2, and SM using image analysis software and found no significant difference in E-cadherin expression in M1, M2, and SM in differentiated submucosal invasive gastric cancer as a whole. However, when we analyzed E-cadherin’s expression in cases with and without venous invasion in the submucosa showing intestinal-type, we observed a decreasing trend in E-cadherin expression in M1, M2, and SM, in patients with venous invasion. However, the difference was not statistically significant. In contrast, in patients without venous invasion, a decreasing trend in E-cadherin expression was observed in M2, but no sustained decrease in E-cadherin was observed in SM. According to previous reports, loss of RUNX3 is also involved in transforming growth factor-β-mediated epithelial-mesenchymal transition (EMT), 22 and the persistent decrease in E-cadherin may be related to this phenomenon. Xiangming et al. studied the relationship between E-cadherin expression and its membrane lining protein, α-catenin, and clinicopathological factors in early-stage gastric cancer. In this study, they reported that the attenuation or loss of E-cadherin expression did not correlate with lymphatic invasion and lymph node metastasis, but the attenuation or loss of α-catenin correlated with lymph node metastasis. 23 Since the release of α-catenin initiates E-cadherin’s intercellular binding, it is possible that the weakening of α-catenin is observed before the weakening of E-cadherin expression, and further examination of α-catenin expression may be necessary for this study.
We acknowledge some limitations of this study. First, to examine the expression of E-cadherin, we performed image analysis using ImageJ objectivity. While the evaluation of immunostaining by image analysis provided objectivity, it was difficult to assess staining intensity. In addition, the staining of E-cadherin was non-uniform within the specimens. In the future, it is desirable to find a more objective evaluation method that includes staining ratio and intensity. Second, we did not conduct a prognostic study related to distant metastasis in this study. However, in a previous large-scale study of early-stage gastric cancer, lymph-vascular involvement showed a strong correlation with lymph node metastasis, 24 suggesting that vascular invasion is involved in lymph node metastasis and consequently distant metastasis. Therefore, in this study, it is necessary to accumulate cases and calculated the sample size, conduct prognostic studies, and analyze them in the future.
Conclusion
Our study found that submucosal intestinal gastric cancer in the loss group has a significantly higher potential for venous invasion. Thus, the identification of phenotypes and phenotype shifts can be expected to be a factor that influences treatment strategies after endoscopic treatment or after surgical resection.
Footnotes
Acknowledgments
The authors would like to thank Ms. Yukie Fujita and Ms. Sayumi Kubo for providing technical assistance.
Declaration of conflict of interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and /or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was performed following the Declaration of Helsinki and was approved by the ethics committee of Hirosaki University Graduate School of Medicine (2020-157).
Informed consent
All patients gave written informed consent to participate in this study.
Trial registration
Not applicable.
