Abstract
We have previously reported upregulation of clusterin at the time of islet cell regeneration after β-cell injury. This led us to speculate that clusterin might be involved in the neogenic regeneration of the pancreas. Clusterin expression was examined throughout the process of pancreatic neogenesis in pancreatectomized rats. For in vitro analysis, duct cells were isolated from the rat pancreas and clusterin cDNA was transfected for its overexpression. Clusterin and its mRNA increased significantly in the early phase of regeneration, particularly at 1–3 days after pancreatectomy. Clusterin was transiently expressed in the differentiating acinar cells but faded afterwards. Interestingly, these clusterin cells were negative for PCNA (proliferating cell nuclear antigen), whereas most epithelial cells in ductules in the regenerating tissue showed extensive proliferative activity. Clusterin expression was also detected in some endocrine cells of the regenerating tissue. Transfection of clusterin cDNA into primary cultured duct cells resulted in a 2.5-fold increase in cell proliferation and induced transformation of non-differentiated duct cells into differentiated cells displaying cytokeratin immunoreactivity. Taken together, these results suggest that clusterin may play essential roles in the neogenic regeneration of pancreatic tissue by stimulating proliferation and differentiation of duct cells.
D
Clusterin is a glycoprotein expressed by many cell types in association with a variety of pathophysiological conditions (Murphy et al. 1988; French et al. 1994; Park et al. 1999). We have reported the transient expression of clusterin and its modification along with the islet cell development as well as its upregulation on cytotoxic insult of β-cells (Min et al. 1998; Park et al. 1999). Recently, we also suggested that this protein might be involved in regeneration of pancreatic endocrine cells after various types of tissue injury (Kim et al. 2001). These results led us to propose that clusterin might be involved in the neogenic regeneration and reorganization of pancreatic tissue.
The aim of the present study was to evaluate whether clusterin is involved in the neogenic process of pancreatic tissue. Therefore, we examined clusterin expression as related to the process of pancreatic regeneration after subtotal Px. Furthermore, we induced clusterin overexpression by transient transfection of clusterin cDNA into pancreatic duct cells in culture and examined the mode of cell proliferation and differentiation.
Materials and Methods
Experimental Animals, Tissue Sampling, and Px
Four-week-old male Sprague–Dawley rats (Daehan Experimental Animals; Seoul, Korea) showing normal fasting blood glucose levels were used for Px or sham operation. The animals were fasted for 18 hr before operation and allowed free access to standard diet and water after operation. Blood glucose levels were determined daily with a glucometer (Glucotrend; Roche Diagnostics, Mannheim, Germany). The entire pancreas or what remained after pancreatectomy was removed at 1, 2, or 3 days as well as 1, 2, or 4 weeks after operation. For Px, 85–90% of the pancreatic tissue was removed by gentle abrasion with cotton swabs, leaving the tissue intact between the common bile duct and the loop of the duodenum as described previously (Bonner–Weir et al. 1983). Some rats were injected with BrdU (20 mM in 0.1M NaOH, 5 ml/kg bw) at the day of Px and sacrificed 3 days after operation. For sham operation, the same surgical procedure was carried out without any removal of the pancreatic parenchyma. Anesthesia was achieved by inhalation of gaseous nitrous oxide–oxygen and isofrane. The experimental animals were treated humanely and all surgical procedures were performed according to the Animal Use and Care protocol.
Morphological Examination of Pancreatic Tissues After Px
Pancreatic tissues were fixed with Bouin solution or 4% paraformaldehyde and embedded in paraffin. Immunostaining was carried out by the avidin–biotin–peroxidase complex method (Hsu and Raine 1981). The tissue sections were incubated with the goat anti-clusterin antibody (Santa Cruz Technology; Santa Cruz, CA), mouse anti-insulin (Bio-Genex; San Ramon, CA), rabbit anti-glucagon or anti-somatostatin (Dako; Carpinteria, CA) antibodies. After applying the biotinylated anti-mouse, anti-goat, or anti-rabbit IgG and the avidin–biotin–peroxidase complex (Vector; Burlin-game, CA), the reaction was detected by diaminobenzidine (Sigma; St Louis, MO). To identify clusterin immunoreactivity in proliferating cells, double immunostaining was carried out on the same tissue sections using a mouse anti-PCNA (Zymed; South San Francisco, CA) and clusterin antibodies. For detection of clusterin expression in the postmitotic cells, tissue sections of pancreas obtained from BrdU-injected rats were subjected to double labeling using the peroxidase-conjugated anti-mouse BrdU antibody (Dako) and the clusterin antibody. In double immunolabeling, first and second immunoreactions were detected by DAB and nickel-DAB, respectively. Insulin mRNA expression in the regenerating β-cells was assessed by in situ hybridization as described previously (Kim et al. 2001).
Morphometry for Regenerating Tissue Area
The regenerating tissue consisted of the proliferating area showing extensive PCNA labeling and the differentiating area with clusterin expression. For morphological analysis, the tissue sections double immunolabeled for PCNA and clusterin were photographed at a low magnification (×4) for mosaic image. On microscopic examination, the regenerating tissues were marked on the microphotograph mosaic that displayed the whole area of the pancreas in each section. The images were digitalized and the percentage of the regenerating area to whole pancreatic tissue was calculated using an image analyzing system (Image-Pro Plus; Media Cybernetics, Silver Spring, MD). Four pancreatic tissues were analyzed in each experimental group and five sections at 500-μm intervals were chosen from each pancreatic tissue.
Western and Northern Blotting Analyses
Western blots for clusterin and Northern blotting analyses for clusterin and insulin were performed as previously described (Park et al. 1999; Kim et al. 2001). Expression of β-actin and α-tubulin was also assessed as control for Northern and Western blotting analysis, respectively. Pancreatic tissues obtained at 1, 3, or 7 days and 4 weeks after Px were used for these blotting analyses.
Overexpression of Clusterin in Pancreatic Duct Cells
Clusterin cDNA from nucleotide + 63 to nucleotide +1406 (GenBank accession number M64723) was obtained from rat brain total RNA by RT-PCR and cloned into the expression vector pcDNA3 (Invitrogen; Groningen, Netherlands). Ducts were isolated by modifying the method developed for isolation of pancreatic islets (Bouwens et al. 1995). In brief, whole pancreatic tissue was injected with 5 ml of collage-nase (1.5 mg/ml; Roche) and digested for 30 min at 37C. The digest was washed two or three times with HBSS and then transferred to the isolation medium. After rapid sedimentation, the ducts were collected and sliced into small pieces. The duct tissues were transferred onto Matrigel (Becton Dickinson; Bedford, MA)-coated coverslips and cultured with RPMI medium for 3 days until the cell explants developed from the primary duct tissue. After discarding the primary duct tissues by aspiration, the cell explants were transfected with clusterin cDNA (pcDNA3-rCLU) using the Effectene reagent (Qiagen; Valencia, CA) according to the procedure recommended by the manufacturer. Briefly, 0.4 μg of pcDNA3-rCLU was mixed with enhancer in 100 μl of DNA condensation buffer and 10 μl of Effectene was added to the DNA mixture for the formation of a transfection complex. The duct cells were incubated with the culture medium containing the Effectene–DNA complex for 6 hr and the medium was changed to normal growth medium. For control, the vector plasmid (pcDNA3) was used in the same manner. Overexpression of clusterin in the transfected duct cells was confirmed by immunostaining and Western blotting analysis for clusterin, while replication and differentiation were determined by immunostaining for PCNA and cytokeratin 20 using the appropriate antibodies (Dako). For determination of the transfection efficiency, we carried out co-transfection of pGFP vector (Clontech; Palo Alto, CA) with pcDNA3-rCLU into the duct cells under the same experimental condition and assessed the percentage of the GFP-producing cells to the whole duct cells 24 hr after transfection. The transfection efficiency was found to be 53.35 ± 3.06% (mean ± SEM; n = 12 from three independent experiments).
Statistical Analysis
Results were statistically analyzed by ANOVA. We considered p values of less than 0.01 as significant. All results are expressed as mean values ± SEM and were analyzed using SAS software (version 6.12; SAS Institute, Cary, NC).
Results
Changes in Blood Glucose Concentrations
The Px rats displayed a wide spectrum of fasting blood glucose concentrations. Some Px rats showed no or slight increases in blood glucose (<8.3 mmol/liter), whereas others displayed very high levels (>11.1 mmol/liter) early after Px. The rats showing prolonged hyperglycemia died within 2 weeks and were therefore excluded from the experiments. Most of the rats used remained euglycemic during the experimental period, although they did show higher blood glucose concentrations compared with sham-operated animals (Figure 1).
Characterization of Regenerating Pancreatic Tissue After Partial Px
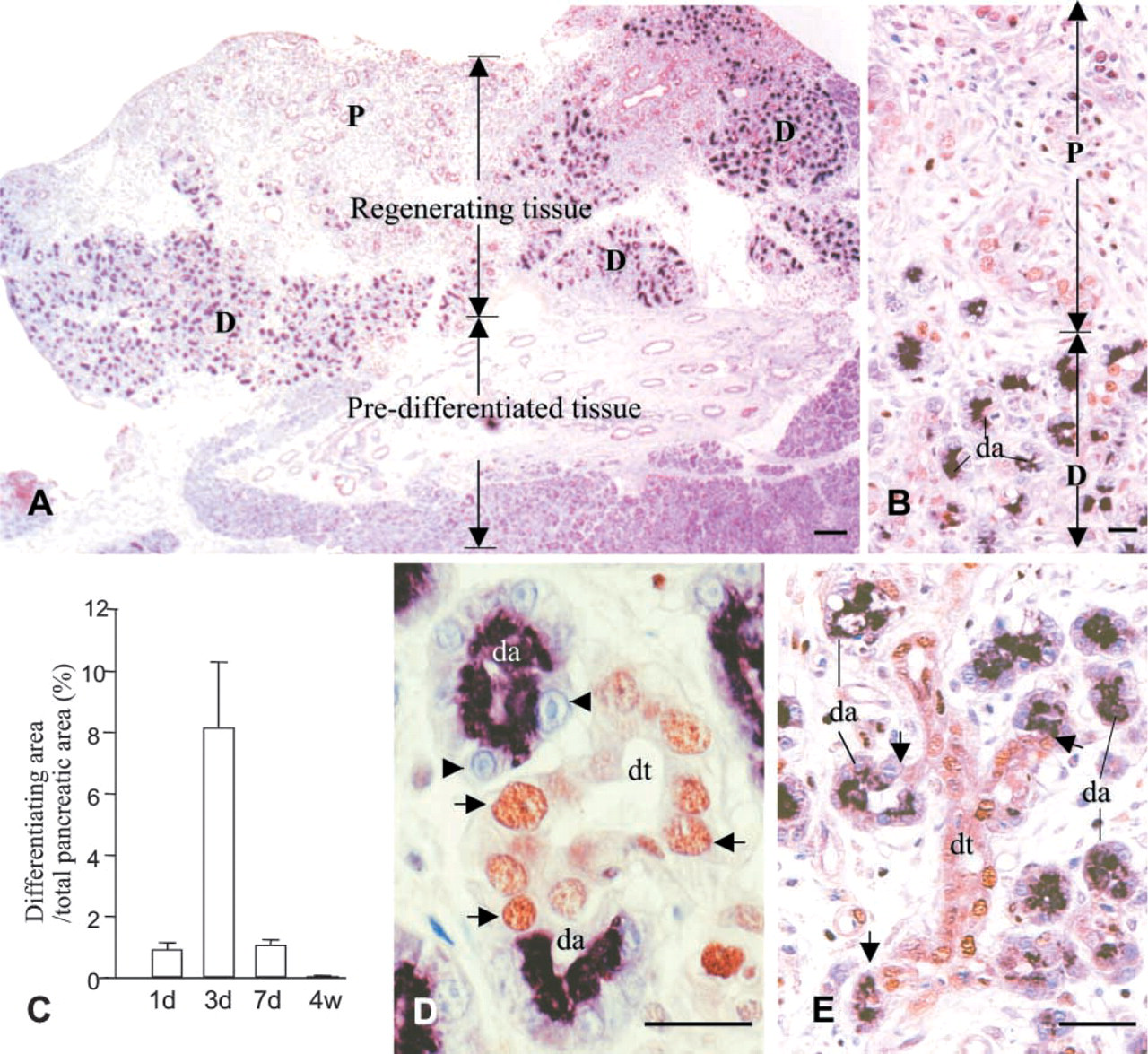
Regeneration can be depicted in two phases, the proliferation-dominant phase (1–3 days after Px) followed by a differentiation phase (1–4 weeks). The early phase was characterized by development of neogenic tissue showing extensive proliferation. Although sporadically seen on the first day after Px, considerable regeneration of the tissue was found during the second and third days. Most of the regenerating tissue developed near the outer limit of the tissue or was surrounded by injured tissue induced by the Px. On observation of several series of serial tissue sections, we found no direct histological connection between those neogenic foci and preexisting ducts. The area of regenerating tissue reached up to 22% at this time point but decreased rapidly at day 7.
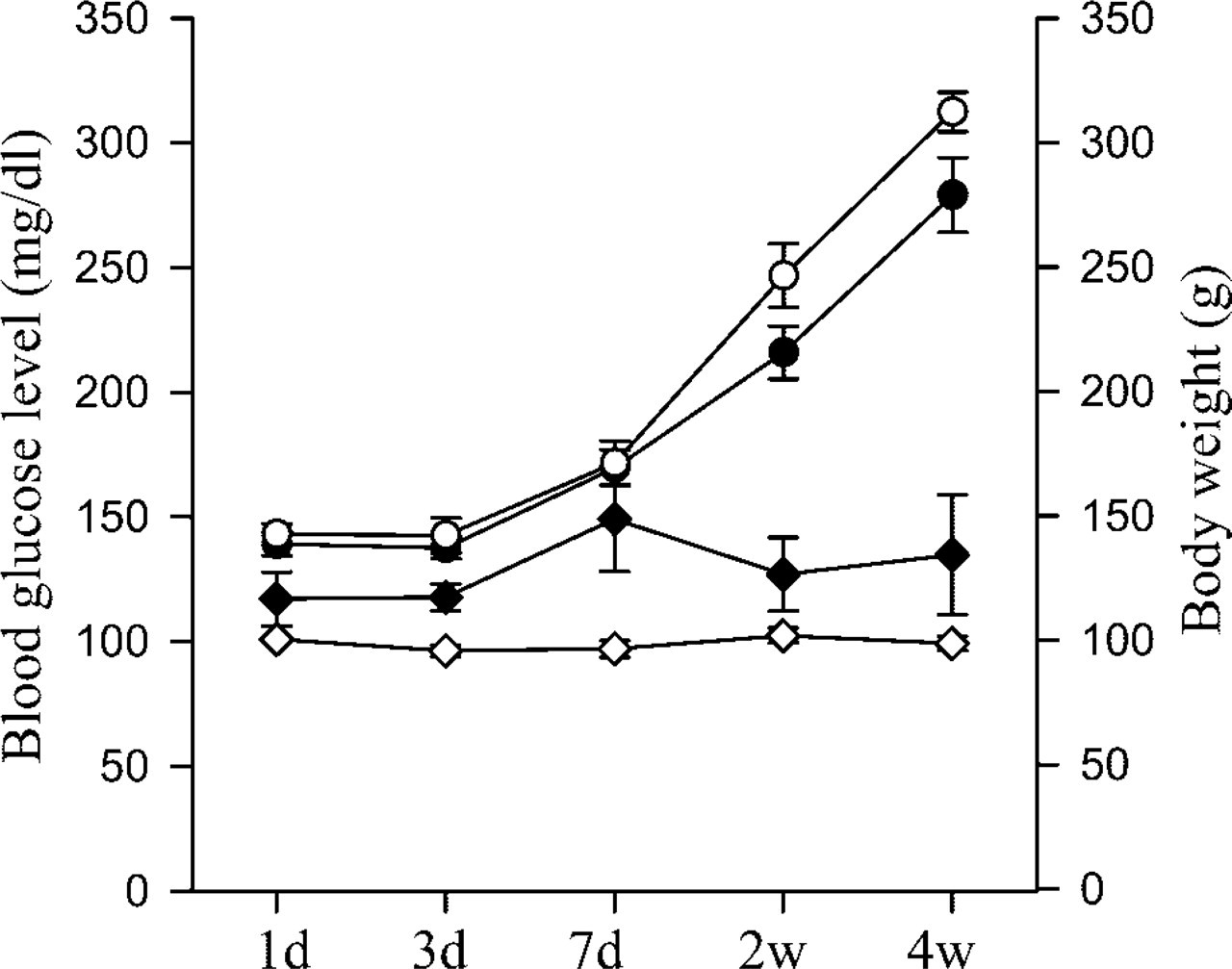
Fasting blood glucose concentration and body weight after Px and sham operation. Solid diamonds, blood glucose of Px rats; open diamond, blood glucose of sham-operated controls; solid circles, body weight of Px rats; open circles, body weight of sham-operated controls. The numbers of animals in the Px group are 39, 38, 26, 27, and 13 for days 1, 3, 1 week, 2 weeks, and 4 weeks, respectively, while 18, 20, 20, 16, and 20 animals were used at the same time points for the sham-operated controls. Mean values ± SEM.
Regeneration persisted for 4 weeks (Figure 2). The regenerating tissue was heterogeneous in composition (Figures 3A–3C). Some areas were composed of tubules and ductules in surrounding loose connective tissue, whereas others were denser with differentiating acini derived from tubules or ductules (Figures 3A–3C). Small tubular structures formed by elongated epithelial cells with irregular lumens in the neogenic tissue were considered to be tubules, while similar structures, displaying larger lumens lined by low epithelial cells, were considered to be ductules (Figures 3A and 3C). Differentiating acini showed direct connections with the epithelial lining of the tubules or ductules (Figures 3B, 5D, and 5E). This may indicate sequential processes of neogenic renewal of pancreatic tissue with formation of tubules evolving into ductules and differentiating acini. Most epithelial cells in tubules and ductules were PCNA-positive, reflecting an active process of replication (Figures 3C, 5D, and 5E). The rate of replication assessed by PCNA labeling increased significantly in regenerating tissue compared to sham-operated pancreaatic tissue or to intact pancreatic tissue away from regenerating areas (Figure 3E). In contrast, no sign of proliferation was detected in the differentiating acinar cells (Figures 3B, 5D and 5E). During the differentiating phase (1–4 weeks), the areas of regeneration gradually decreased with reduced proliferating activity, whereas areas of fully differentiated tissue gradually increased (Figure 2). This suggests that cells in regenerating tissues evolved gradually into well-differentiated pancreatic cells.
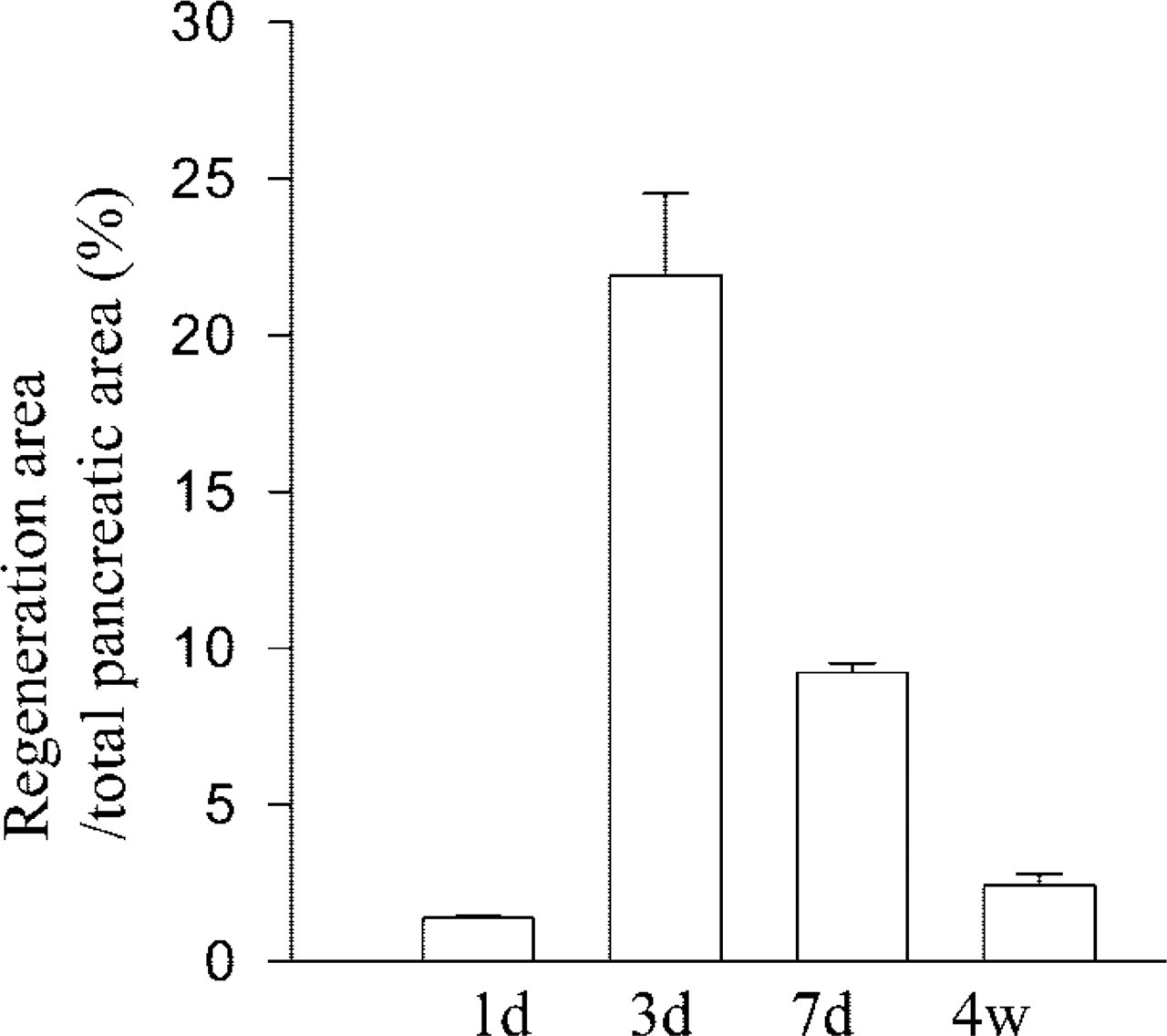
Percent area of regenerating tissue to whole pancreatic tissue at 1, 3, 7 days, and 4 weeks after Px (n=4 rats in each group). Mean values ± SEM.
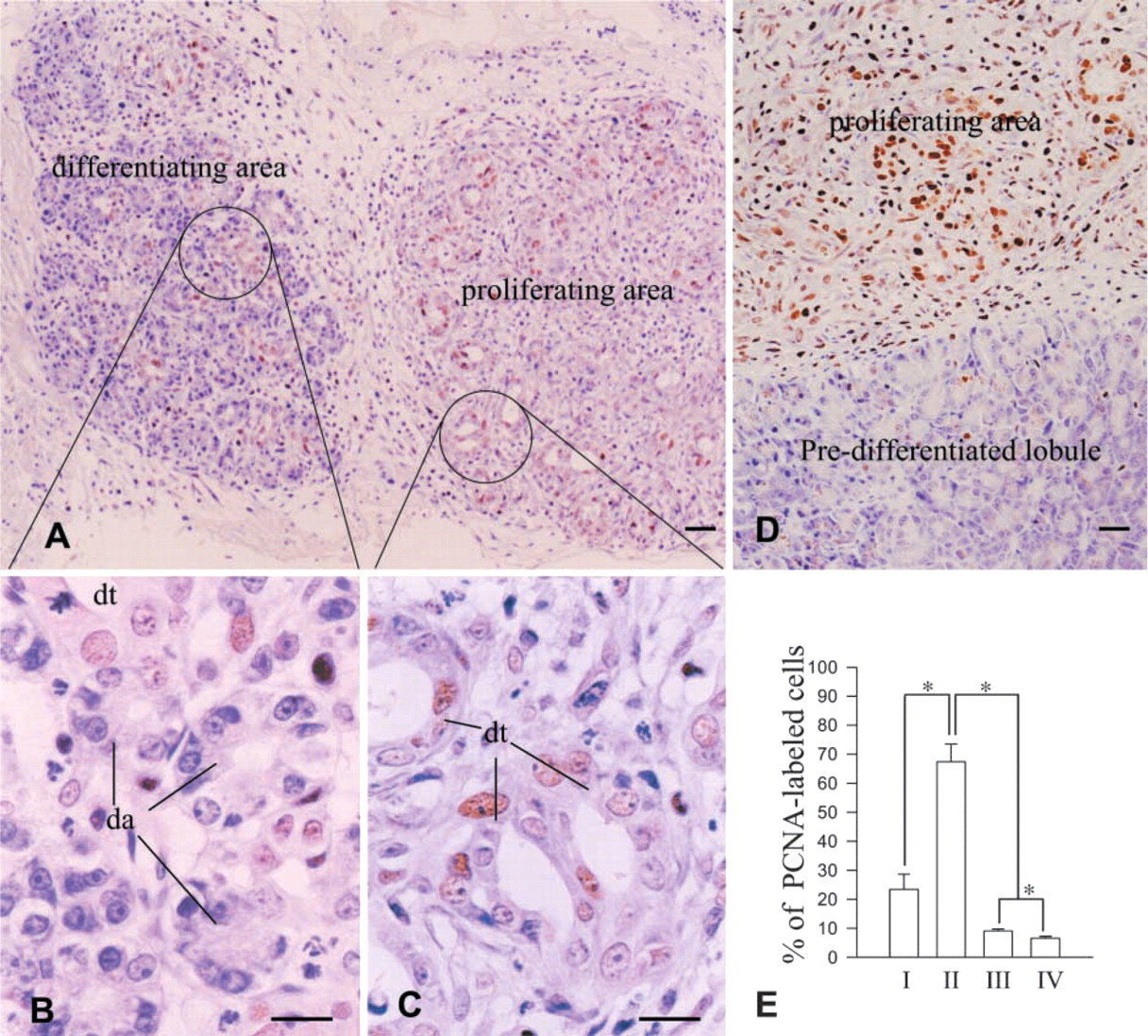
PCNA immunolabeling for morphological features and proliferating activity of the regenerating tissues. (
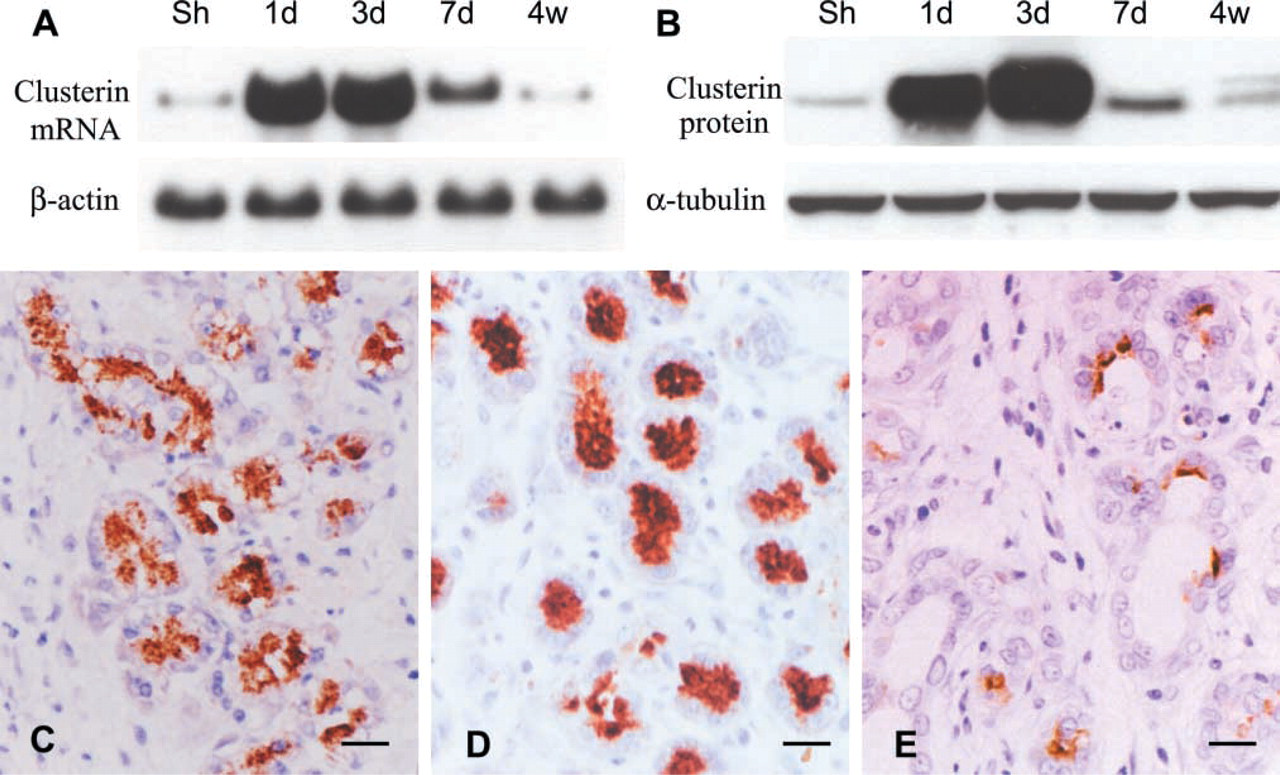
Changes in clusterin expression after Px demonstrated by Northern (
Changes in Clusterin Expression
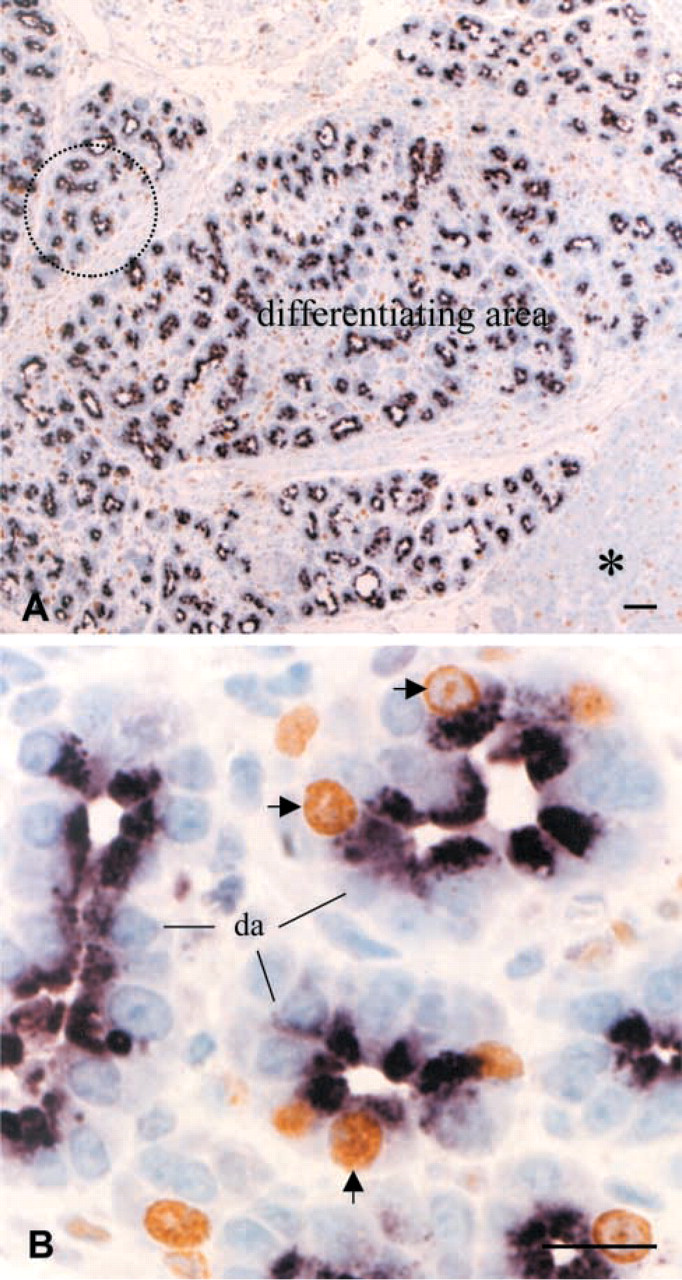
Clusterin expression was assessed by immunocytochemistry and Western blotting, and its mRNA expression was evaluated by Northern blotting. As reported previously (Park et al. 1999), only weak immunoreactivity was detected in some glucagon cells of the islets in pancreatic tissue of sham-operated animals. However, clusterin expression was remarkably increased after Px in correlation with pancreatic regeneration. Clusterin mRNA expression was upregulated during the proliferation-dominant phase, particularly at 1–3 days after Px, and decreased thereafter in the differentiation-dominant phase toward the levels of the sham-operated animals (Figure 4A). This was confirmed by Western blotting, clusterin being remarkably enhanced at 1 and 3 days after Px and declining thereafter (Figure 4B). Immunocytochemistry provided more detailed information on clusterin expression at the cellular level. During the proliferation-dominant phase, intense clusterin expression was detected in the regenerating tissues, particularly in the differentiating acinar cells at day 3 after operation (Figure 4D). Interestingly, most of the clusterin cells in the differentiating acini were devoid of nuclear PCNA labeling, and PCNA-labeled cells in the tubules or ductules showed no clusterin immunoreactivity (Figures 5B and 5D). Such dissimilarity in immunoreactions was distinctively represented at the junctions between tubules and differentiating acini (Figures 5B and 5D). However, a subpopulation of clusterin-positive cells in developing acini were labeled with BrdU at the nuclear level in the pancreatic tissues obtained 3 days after BrdU injection and Px (Figure 6). This indicates that the BrdU-labeled post-mitotic cells were derived from duct cells and expressed clusterin. It also indicates that clusterin-positive cells of the regenerating tissue are in a more differentiated state and that clusterin may be involved in the differentiation process. Therefore, we referred to the clusterin cells lining the differentiating acini as being on the “regeneration front.” The clusterin cells might be initiating their differentiation into exocrine or endocrine cells. We therefore divided the regenerating tissues into the proliferating area and the area of clusterin expression, which is identical to the differentiating area (Figures 5A and 5B). The ratio of clusterin expression area over total pancreatic tissue displayed peak at day 3 after Px and then was reduced (Figure 5C).
Double immunolabeling for clusterin and PCNA. (
Insulin Content and Biosynthesis in β-cells During Pancreatic Regeneration
Insulin and glucagon cells were detected as single cells or as small cell clusters in the regenerating tissue 2–3 days after Px. They evolved into primitive islets by forming larger and irregular masses during the second phase of regeneration (Figure 7). Clusterin expression was detected not only in the developing islets but also in the duct epithelium in the regenerating tissue. Clusterin was co-localized with glucagon in the α-cells but not with insulin-secreting β-cells (Figures 7A–7D). In the regenerating tissue the β-cells displayed intense insulin immunoreactivity as well as insulin mRNA in situ hybridization signal, implying a dynamic biosynthetic activity (Figure 7E). This was supported by Northern blotting analysis for insulin mRNA (Figure 7F).
Proliferation and Differentiation by Overexpression of Clusterin in Duct Cells During Primary Culture
To elucidate its action, clusterin was overexpressed in duct cells by transfecting clusterin cDNA. Transfection of clusterin cDNA (pcDNA3-rCLU) induced transformation of non-differentiated duct cells into differentiated cells. Clusterin cDNA-transfected cells displayed immunoreactivity for cytokeratin-20, whereas no such immunolabeling was found in the pcDNA3-transfected cells (Figures 8G and 8H). Proliferation activity was assessed by PCNA immunolabeling (Figures 8D and 8E). Overexpression of clusterin gave rise to a 2.5-fold-increase in duct cell proliferation (Figure 8F).
Clusterin expression in post-mitotic cells labeled with BrdU in regenerating pancreas of the Px rats. The tissue was removed 3 days after BrdU injection and double immunostained for BrdU (brown reaction of DAB) and clusterin (blue-black reaction of nickel-DAB). The BrdU-incorporated cells distributed in the differentiating area as well as in the predifferentiated lobule (asterisk in
Discussion
Slow and limited renewal of human pancreatic tissue leads to poor recovery of β-cells in diabetic patients. However, regeneration of β-cells with improvement of diabetic symptoms can be induced in animal models (Brockenbrough et al. 1988; Bonner–Weir et al. 1993; Watanabe et al. 1994; Xu et al. 1999). Subtotal Px provokes neogenic regeneration of pancreatic tissues, mimicking embryonic development (Bonner–Weir et al. 1993). Because of the rapid reappearance of islets of Langerhans with a normal distribution of β-cells, subtotal Px is considered to be an experimental model for diabetes research (Bonner–Weir et al. 1983; Watanabe et al. 1994; Okamoto 1999; Xu et al. 1999). However, molecular mechanisms and cellular processes underlying the neogenic tissue renewal are in need of intensive investigation. Two pathways of pancreatic regeneration have been suggested in the model of subtotal Px: (a) replication of preexisting tissue, and (b) proliferation and differentiation of the duct epithelium (Bonner–Weir et al. 1993; Sharma et al. 1999). In this study we have demonstrated that substantial replication is confined to the focal area of regeneration. More extensive proliferation was found in the tissue area of growing tubules and ductules than in the differentiating area showing clusterin expression. These results indicate that pancreatic regeneration takes place by the pathway of proliferation and differentiation of the duct epithelium, as suggested by Bonner–Weir et al. (1993). However, we cannot concur with the suggestion that ducts and duct-like structures in the regeneration areas originate from preexisting larger ducts (Bonner–Weir et al. 1993). We observed the appearance of primitive tubular structures displaying high replication activity in swirls of loose connective tissue located at a distance from the larger ducts (Figures 3A–3D). These might represent starting events in pancreatic regeneration. In addition, the epithelial cells in larger pancreatic ducts were quiescent after a transient manifestation of proliferation as described previously (Bonner–Weir et al. 1993). The proliferating tubules of the regenerating area demonstrated active replication 3 days after Px. We therefore could not relate the formation of these small tubules in the areas of regeneration to the small ducts budding from the larger ones. We suggest instead that regeneration in this model may be initiated from persistent stem cells located in the mesenchyme-like loose connective tissue with the help of epithelio–mesenchymal interactions, as already proposed for pancreatic development (Gittes et al. 1996; Miralles et al. 1999; Edlund 2001). Cells in the tubular structures transform not only into duct epithelial cells and differentiating acini but also into endocrine cells.
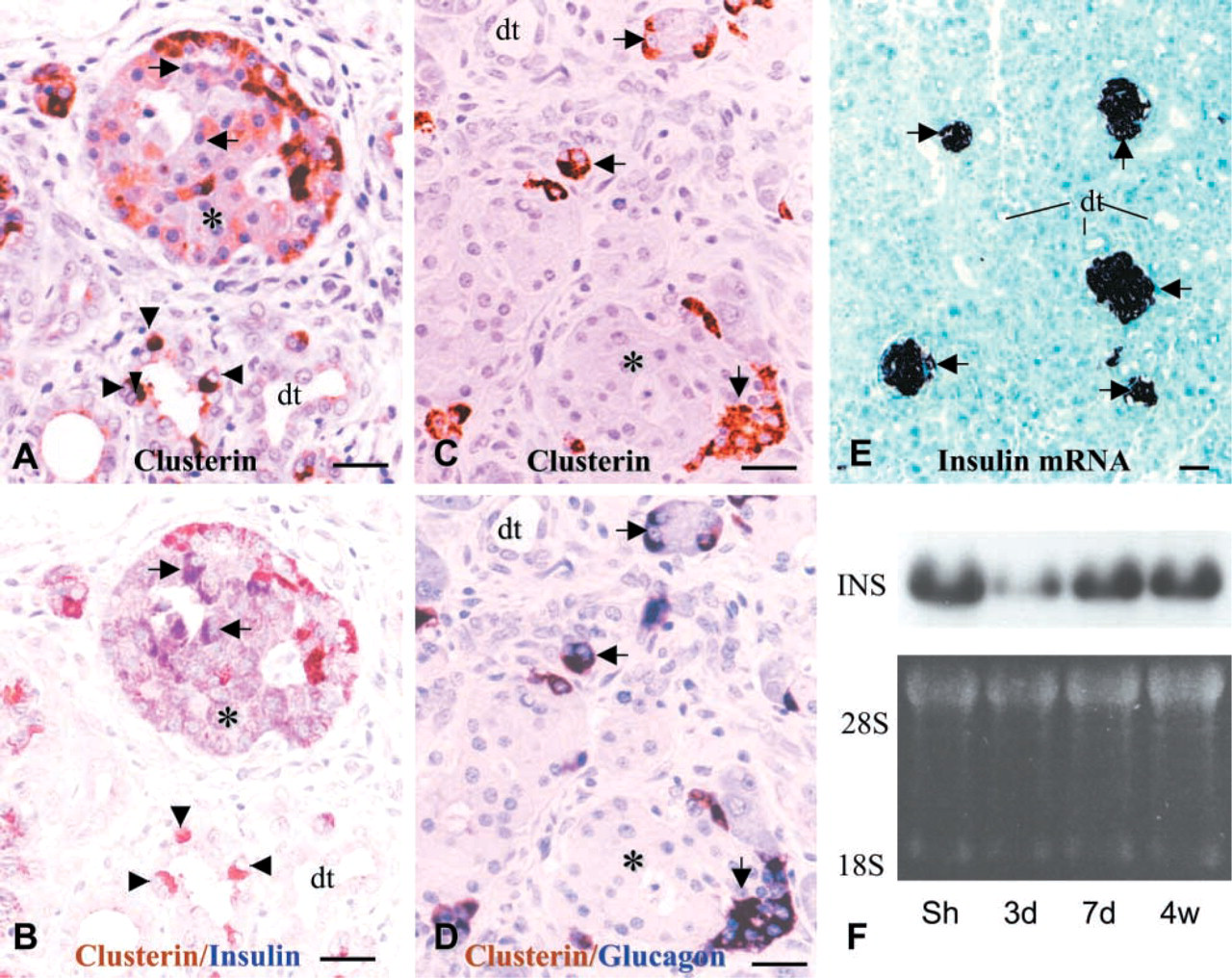
Clusterin expression in developing islets of regenerating tissue and changes in insulin mRNA expression after Px. During formation of the primitive islets at day 7 after Px, clusterin expression (brown reaction in
The mechanisms of neogenic regeneration after Px, which may provide an important clue for β-cell replication in diabetes, remain largely unknown, particularly at the molecular level. Several factors, including IDX-1 (Sharma et al. 1999) and IGF-1 (Smith et al. 1991), have been proposed to stimulate the formation of new pancreatic tissue induced by subtotal Px. This process of neogenic regeneration of pancreatic tissue is probably regulated by the combined action of multiple genes and gene products. Certain endogenous molecules must play very specific roles and cooperate with IDX-1 and IGF-1 in regulating pancreatic neogenesis. We found an upregulation of clusterin associated with neogenic renewal of pancreatic tissues after Px. As reported for both IDX-1 and IGF-1 (Smith et al. 1991; Calvo et al. 1997; Sharma et al. 1999), clusterin was transiently upregulated as early as 1–3 days after Px, the peak time of regeneration, and then declined towards basal levels. Clusterin-expressing cells differ from those expressing IDX-1 and IGF-1. Expression of IDX-1 (Sharma et al. 1999) and IGF-1 (Smith et al. 1991; Calvo et al. 1997) was reported to occur in duct cells and islet cells not only in the neogenic tissue but also in the preexisting tissue. In contrast, clusterin was predominantly expressed in the developing acini and only in very few duct cells and endocrine cells present in the focal area of regeneration. This may indicate that clusterin' actions differ from those of IDX-1 and IGF-1, playing more critical and specific roles in neogenic growth of pancreatic tissue.
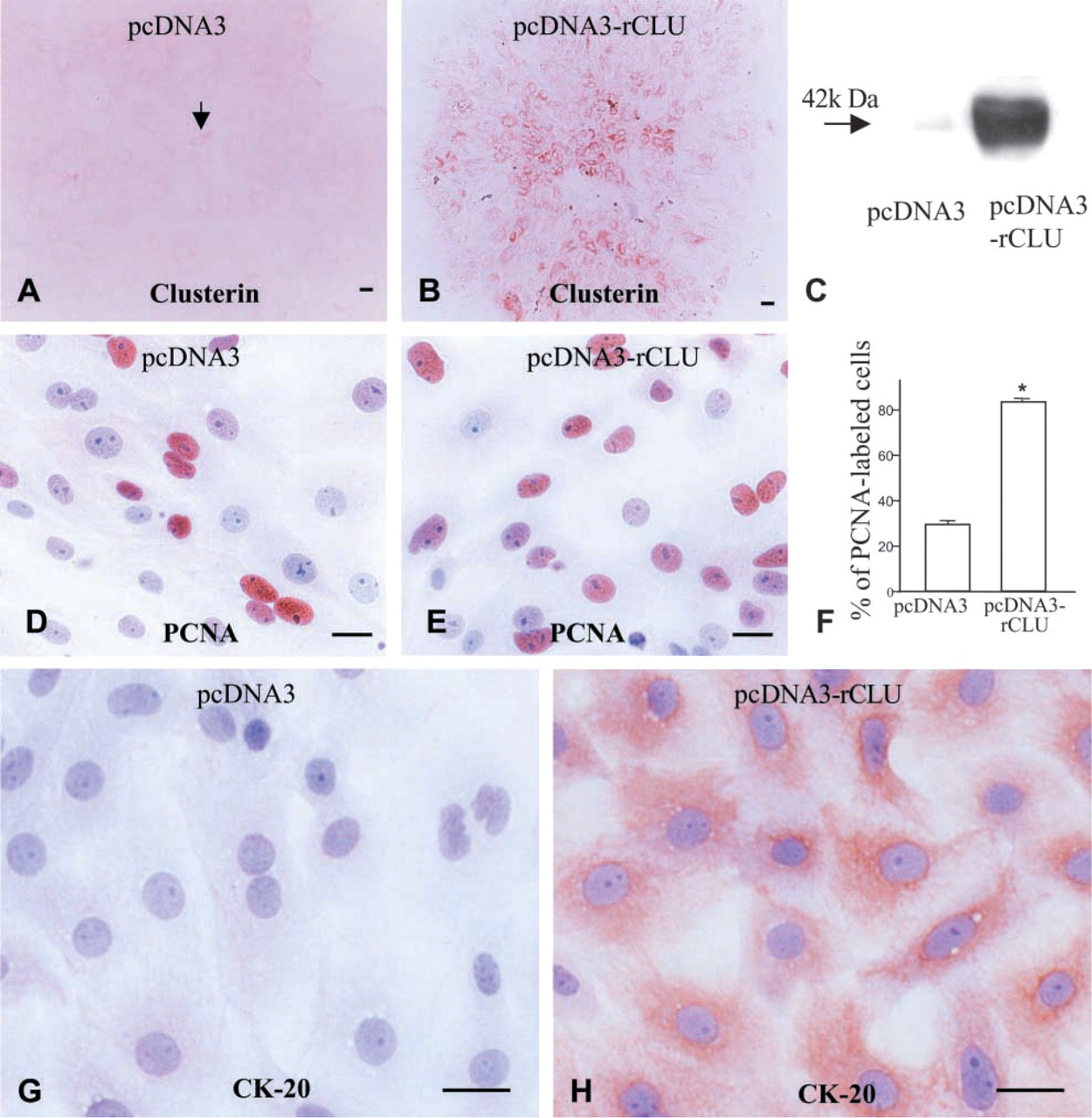
Clusterin overexpression in duct cells and modification of cell character. Overexpression of clusterin was confirmed by clusterin immunostaining (
Clusterin is an amphipathic glycoprotein originally identified in the luminal fluid of the rete testis as an aggregating molecule (Fritz et al. 1983). In pancreas, clusterin is not a regular secretory product but instead is expressed transiently during fetal development or in response to certain cytotoxic injuries and subsequent proliferation (Scaglia et al. 1995; Calvo et al. 1998; Min et al. 1998; Park et al. 1999). Our recent experimental data also demonstrated that this protein is involved in proliferation of islet cells after β-cell death induced by streptozotocin (Kim et al. 2001). Although mechanisms of clusterin action have not been defined, clusterin is known to be regulated by growth factors (Reddy et al. 1996a,b; Jin and Howe 1997,1999; Gutacker et al. 1999; Wegrowski et al. 1999). TGF-β induces expression of clusterin gene through the activator protein-1 (AP-1) binding site (Jin and Howe 1997,1999; Wegrowski et al. 1999). Clusterin directly interacts with the intracellular segment of TGF-β receptors, inducing their phosphorylation (Reddy et al. 1996a, b). This implies that clusterin may instead function as a signaling molecule for inducing proliferation and differentiation with the help of growth factors. We therefore propose a dual action of clusterin in neogenic regeneration of pancreatic tissue. By an autocrine action, clusterin may be involved in the differentiation of acinar cells. On the other hand, on secretion clusterin appears to exert a paracrine action regulating replication and differentiation of duct elements. In the present study we found that overexpression of clusterin stimulates duct cell proliferation and differentiation. Transfection of clusterin cDNA into duct cells resulted in a 2.5-fold increase of proliferation. It also coincided with a similar effect on MIN6 cells, whose proliferation increased 31% with clusterin overexpression (Kim et al. 2001). We further found that overexpression of clusterin induces transformation of undifferentiated duct cells into more differentiated cells expressing cytokeratin (Figures 8G and 8H). This line of evidence may indicate that clusterin acts as a regulatory factor involved not only in replication but also in differentiation during the neogenic renewal of pancreatic tissue. Clusterin-expressing cells lining the differentiating acini should be considered as the “regeneration front” during pancreatic neogenic renewal.
Footnotes
Acknowledgements
Supported by a grant from the Korea Health 21 R&D Project, Ministry of Health & Welfare, Republic of Korea, to I-S Park (HMP-00-B-20700–0027).
