Abstract
Expression of SMR2, a member of the gene family encoding salivary glutamine/glutamic acid-rich proteins, is regulated by androgens in rat submandibular gland acinar cells. To further characterize SMR2 regulation, we analyzed SMR2 expression during submandibular gland postnatal development and rat puberty at both a global and a single-cell level. Using in situ detection of mature and primary SMR2 transcripts, we show that SMR2 expression is heterogeneous among acinar cells. However, only one cell population with various amounts of mRNAs can be defined. The number of high-expressing cells increases in males during puberty and in females up to 6 weeks of age, suggesting that some factor in addition to acinar differentiation might be important for SMR2 expression in female rats. Involvement of the β-adrenergic system in regulating SMR2 expression was tested in rats exposed daily to isoproterenol for 4 days. Under these conditions we found an increase in SMR2 expression in female rats, associated with an increase in SMR2 mRNA levels in most acinar cells. This suggests that a signaling cascade, elicited by β-adrenergic stimuli, might act in concert with androgens to regulate SMR2 expression.
Keywords
D
Two androgen-responsive genes, VCSA1 and SMR2 (Rosinski–Chupin et al. 1988,1993; Rosinski–Chupin and Rougeon 1990), were previously identified in rat submandibular gland (SMG) acinar cells. Recent results on VCSA1 transcription regulation have revealed that androgens can elicit both a binary and a graded response in the physiological context of rat SMG (Rosinski–Chupin et al. 2001). VCSA1 transcripts, which encode the SMR1 protein, a precursor for sialorphin, are found in the rat SMG at a 1000-fold greater level in males than in females (Rosinski–Chupin et al. 1988). Using in situ hybridization (ISH), we showed that VCSA1 expression is activated in only a small proportion of differentiated acinar cells in the presence of low levels of circulating androgens in prepubescent and in castrated males, as well as in females. During the course of sexual maturation in males, the VCSA1 response to androgens involves both an increase in the proportion of acinar cells expressing VCSA1 and an increase in VCSA1 mRNA content in the positive cell population. VCSA1 response to androgens is strongly amplified by the combination of these two mechanisms (Rosinski–Chupin et al. 2001).
The SMR2 gene is a member of a multigene family encoding salivary glutamine/glutamic acid-rich proteins (GRPs) and proline-rich proteins (PRPs) (Rosinski–Chupin and Rougeon 1990). Whereas PRPs are believed to be involved in tannin neutralization (Bennick 2002), GRPs may bind to tooth mineral and mediate adhesion of specific microorganisms to the tooth surface (Mirels et al. 1987). To date, SMR2 is the only androgen-responsive member of this family. Castration reduces SMR2 mRNA accumulation in the gland, and injection of androgens into females or castrated males induces SMR2 gene expression up to male levels (Rosinski–Chupin and Rougeon 1990). Unlike VCSA1, the steady-state levels of SMR2 mRNA are only 20–30-fold greater in male than in female SMGs.
Acinar cells are, along with duct cells, the major cell type of rat SMG, accounting for about 50% of the total cell number. The presence of an androgen receptor in these cells has been demonstrated (Zhuang et al. 1996). Unlike many target cells for androgens, acinar cells do not appear to depend on androgens for their differentiation. Differentiation of acinar cells occurs during the first 3 weeks of postnatal life (Leeson and Jacoby 1959; Cutler and Chaudhry 1974; Moreira et al. 1991), when only low levels of androgens circulate in the bloodstream. In addition, there is no obvious sex-linked difference in the morphology or the relative number of acinar cells. Therefore, these cells are excellent models to discriminate between the respective contributions of tissue-specific effects and androgen responses on gene expression. In addition to androgens, acinar cells of rat SMGs are responsive to a number of stimuli originating from the nervous system. In particular, isoproterenol, a β-adrenergic agonist, affects the expression of a number of genes expressed in acinar cells, such as the genes encoding PRP (Mehansho and Carlson 1983), GRP (Cooper et al. 1991; Ten Hagen et al. 1997), and cystatin (Shaw and Barka 1989). Isoproterenol also has general effects on acinar cells, stimulating DNA synthesis and cell division (Muir et al. 1975).
To determine whether the “binary plus graded” model of response to androgens can be generalized to other androgen target genes in the rat SMG, we have studied in more detail SMR2 induction during postnatal development at both a global and a single-cell level. We show that SMR2 expression is dependent on acinar cell differentiation and androgen concentration. As previously observed for VCSA1, SMR2 expression is heterogeneous in the acinar cell population in the glands of female or immature rats. However, in contrast to VCSA1, there is no evidence for a bimodal pattern of SMR2 mRNA level distribution among acinar cells. In addition, SMR2 expression was found to be upregulated after chronic administration of the β-adrenergic agonist isoproterenol.
Materials and Methods
Animals
Male, female, and pregnant female Wistar rats were purchased from Iffa Credo (Lyon, France). All rats were housed in a temperature-controlled room with free access to food and water. Newborn (no discrimination was made according to the sex of animal) male and female rats of 5, 12, and 18 days or 4, 6, 9, and 11 weeks of age were sacrificed by cervical dislocation or carbon dioxide. For castration experiments, rats were castrated at the age of 4 weeks and sacrificed at 11 weeks. For isoproterenol injection, 8-week-old male and female rats, housed for 1 week, were injected daily with isoproterenol (20 mg/kg body weight; dissolved in 0.15 M NaCl containing 0.3 mM Na2S2O5) or vehicle alone for 4 consecutive days and sacrificed on the next day. In all experiments, care and euthanasia of study animals were in accordance with the European community standards on the care and use of laboratory animals (Ministère de l'Agriculture, France; authorization no. 005329; date 1/26/93). Submandibular glands were dissected and either immediately frozen in liquid nitrogen and stored at −80C before RNA or DNA isolation or were fixed overnight at 4C with 4% paraformaldehyde buffered in PBS.
Probes
The following plasmids were used to generate probes: (a) SMR2 probes: pcS2–1, a 400-bp BglII fragment (nt 132–531; GenBank accession number J05490) cloned in pcDNAII vector (Invitrogen; Leek, The Netherlands) (Rosinski–Chupin et al. 1993) was used to generate probes for Northern blotting analysis and for in situ detection of mRNAs; pInt1SMR2, a 405-bp sequence of SMR2 first intron (nt 992–1397 of SMR2 gene; GenBank accession no. J05491), generated by PCR and cloned in pcDNAII vector, was used for in situ detection of SMR2 heterogeneous nuclear RNA (hnRNA); (b) GRP Ca probes: pcG-1 and pUC42 were described (Rosinski–Chupin et al. 1993) and used for in situ detection of GRP mRNAs and Northern blotting analysis, respectively. Although both probes correspond to GRP Ca sequences (Cooper and Tabak 1991), they also hybridize to GRP Cb mRNAs; pIntGRPCa, a 337-bp sequence of GRP Ca first intron (nt 2289–2626, GRP Ca gene; GenBank accession no. M76536) was used for in situ detection of GRP heterogeneous RNA; (c) Actin probe: rpcAct corresponds to the 1.8-kb nearly full-length cDNA for rat β-actin (Nudel et al. 1983) cloned in pcDNAII; (d) SMGA probe: pcSMGA, a 450-bp sequence of SMGA cDNA (Mirels and Ball 1992; Mirels et al. 1998), corresponding to nt 115–555 (GenBank accession no. M83210) and cloned in pcDNAII vector, was used for in situ detection of SMGA mRNAs.
RNA Blotting Analysis
RNA was prepared from individual submandibular glands as previously described (Rosinski-Chupin et al. 1988). Total RNA was electrophoresed in an agarose/formaldehyde gel, transferred to a nylon membrane (Hybond N; Amersham, Les Ulys, France) and hybridized with probes labeled by the random priming method (Feinberg and Vogelstein 1983). Hybridization signals were quantified with the Phosphor-Imager system (Molecular Dynamics; Sunnyvale, CA).
To normalize the results obtained from different Northern blotting experiments and correct for differences of exposure times or transfer efficiency, the same three RNA preparations were included in each experiment and were used to calculate a correction factor. To control the mRNA quality, the blots were hybridized with a β-actin cDNA probe. Although a decrease in β-actin mRNA relative quantities in relation to age could be noted during the first weeks after birth (Figure 1A), no significant difference in actin mRNA quantities among individuals at the same age, whatever their sex, were observable, indicating that no sample degradation had occurred.
Deadenylation Experiments
Deadenylation experiments were adapted from Carrazana et al. (1988). Briefly, 10 μg total RNA for control or isoproterenol-injected females was hybridized with 500 pmoles oligo(dT) 12–18 in 20 μl KCl 0.1 M, EDTA 0.1 mM by heating for 2 min at 65C, followed by 30 min at 22C. Hydrolysis of polyA tails was performed by incubating RNA samples with 1 U RNase H in RNase buffer (80 mM KCl, 10 mM MgCl2,1 mM DTT, 50 mM Tris-HCl, pH 7.5) for 30 min at 37C.
In Situ Hybridization
In situ hybridization experiments on SMG sections were performed as previously described, with digoxigenin- or 33P- or 35S-labeled cRNA probes (Rosinski–Chupin et al. 2001). Partial hydrolysis to reduce probe size was performed only for intron probes. For detection of radiolabeled probes, an NTB2 (Kodak) emulsion was used. Digoxigenin-labeled probes were revealed by nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate alkaline phosphatase detection as previously described (Rosinski–Chupin et al. 1993). When simultaneous detection of radiolabeled and Dig probes was performed, the Dig probe was revealed first, and the color development time was chosen to not interfere with the further detection of the radiolabeled probe.
The proportion of SMG cells expressing SMR2 mRNA at a given age was determined on an average of 500–1000 acinar cells by microscopic observation. To count the positive acinar cells on highly labeled sections, we performed the observation under high magnification. An acinar cell was considered as positive when we found a localized higher density of labeling (at least 3 times the background level appreciated on negative cells, e.g., duct cells), associated with a nucleus (for determination of the number of cells, an acinar cell was counted only if its nucleus was detected). When the proportions of labeled cells were very low, the number of acinar cells in a field was estimated and 50–150 fields were observed.
Results
Expression of the SMR2 Gene During Post-Natal Development of Rat SMGs
We have previously shown that SMR2 is expressed in the SMG acinar cells in adult male rats (Rosinski–Chupin et al. 1993). These cells begin to differentiate during the first postnatal days and acinar differentiation is complete by 3–4 weeks after birth (Leeson and Jacoby 1959; Cutler and Chaudhry 1974; Moreira et al. 1991). To determine the kinetics of SMR2 induction in the SMG, we quantified SMR2 mRNAs by Northern blotting analysis in the SMG during the first 11 weeks of postnatal life (Figure 1). This period also includes the period of sexual maturation in male rats. Indeed, rat plasma testosterone is minimal around 10–15 days after birth and then increases, reaching maximal values by 7–9 weeks of age (Resko et al. 1968; Paz et al. 1980). To control for the extent of acinar differentiation, we quantified in parallel the mRNAs encoding GRPs. GRPs (glutamine/glutamic acid-rich proteins) are specifically expressed in rat SMG acinar cells and their expression parallels acinar differentiation (Moreira et al. 1989). Although GRPs belong to the same multigene family as SMR2, their expression does not depend on androgens. Two GRP genes, GRP-Ca and GRP-Cb, have been described (Heinrich and Habener 1987; Mirels et al. 1987; Cooper et al. 1991; Ten Hagen et al. 1997). They correspond to similar proteins except for their carboxy-terminal end. Under our conditions of Northern blotting analysis, the two GRP mRNAs could not be discriminated and were quantified together. GRP mRNA quantifications reveal that the maximal increase in GRP mRNA levels occurs between days 0 and 12. A plateau is reached by 4 weeks of age, in males as in females, correlating with the end of the acinar differentiation period.
SMR2 mRNAs are not detected at birth and are barely detected in 5-day-old rats. SMR2 mRNA levels increase thereafter. In contrast to GRP, the plateau is reached only by 6 weeks of age in female rats, 2 weeks after the end of the acinar differentiation period. Differences in SMR2 expression between males and females are apparent as early as 18 days and become statistically significant (p<0.05, Student' test) by 4 weeks of age. In males the plateau is reached by 9 weeks of age. The values for SMR2 mRNA steady-state levels are on average 20-fold higher in males than in females at 11 weeks of age (oscillating between 10- and 100-fold due to individual variations in females).
Altogether, these results are consistent with the fact that the increase in SMR2 expression depends on both acinar differentiation and plasma testosterone concentration. More surprising is the increase in SMR2 expression in females after the end of the acinar differentiation period. This suggests that SMR2 expression might occur at a late stage of acinar differentiation or that it requires, in addition to cell-specific factors, an inducing signal, whose intensity would be maximal by 6 weeks of age in females.
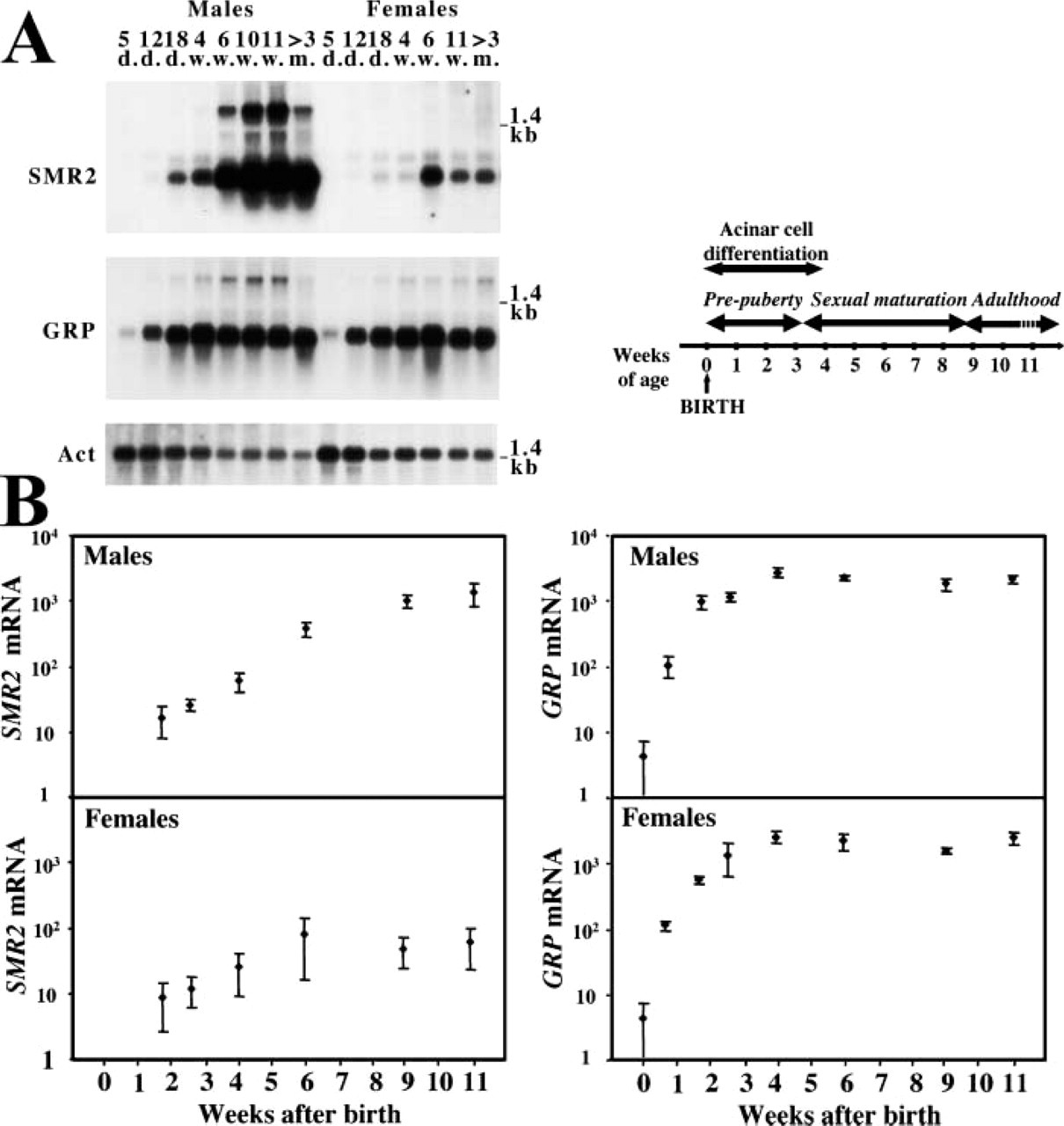
Quantitative analysis of SMR2 mRNA expression during development. (
SMR2 mRNAs Are Expressed in Differentiating Acinar Cells
The perinatal development of SMG acinar cells occurs throughout proliferation and differentiation of proacinar or type III cells, which differentiate into seromucous mature acinar cells (Cutler and Chaudhry 1974; Ball et al. 1988; Moreira et al. 1990,1991; Hand et al. 1996; Denny et al. 1997). Both types of cells are characterized by a different set of secretory proteins (SMGA, SMGB1, and SMGB2 for type III cells, and mucin and GRP for mature cells). Cells in intermediate stages (type IIIP cells) have been reported to express both sets of proteins (Moreira et al. 1991). To more precisely determine the relationships between SMR2 expression and acinar differentiation, we studied SMR2 expression by in situ hybridization, either alone or in combination with detection of GRP mRNA, during the first 2 weeks of life.
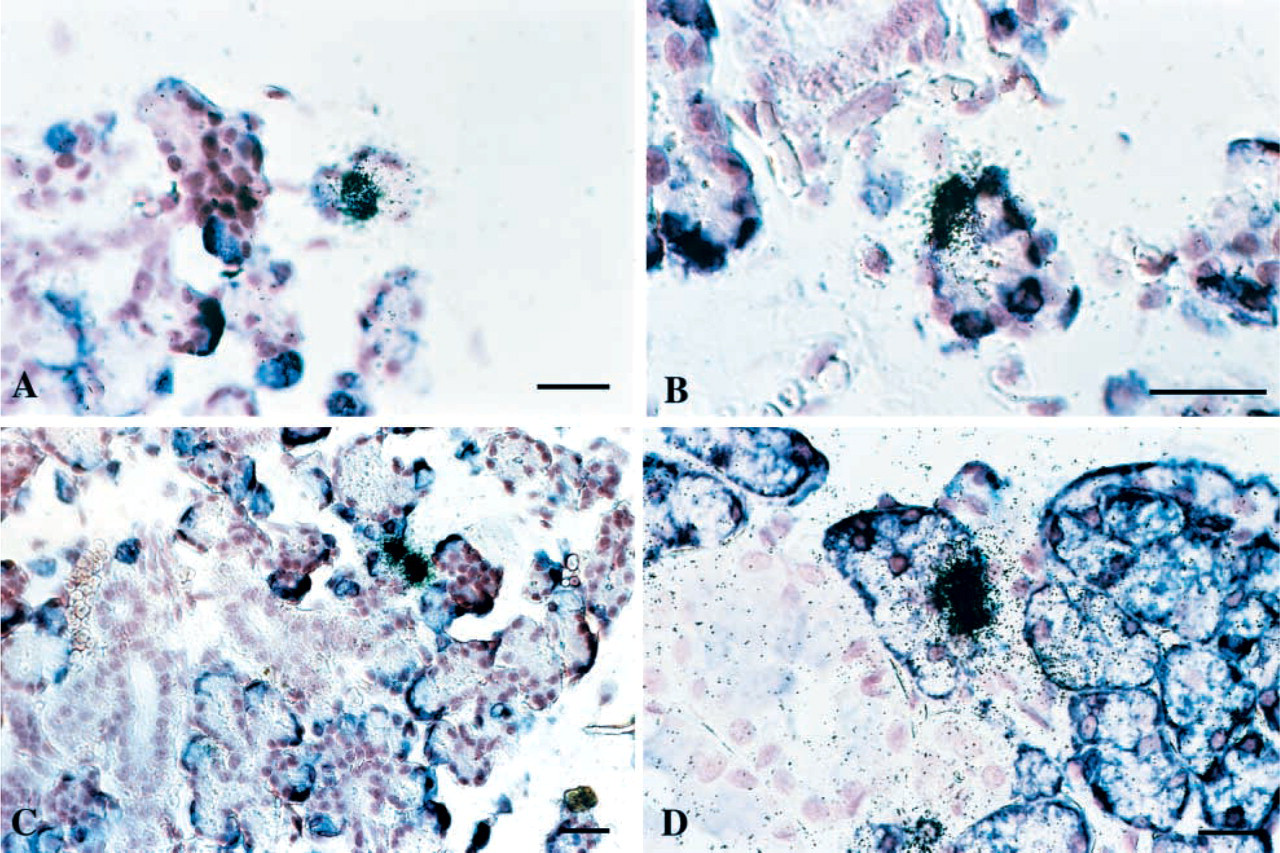
Heterogeneous expression of SMR2 mRNA in acinar cells. Detection of SMR2 transcripts by ISH in the SMG at the beginning of acinar cell differentiation [(
A few GRP mRNA highly positive cells were detected as soon as birth and their number considerably increased by day 5, correlating with the higher level of GRP expression detected by Northern blotting analysis (data not shown). In contrast, only a few SMR2-positive cells were detected at day 5 (Figure 2A). Both the number of SMR2-positive cells and the intensity of labeling were enhanced in 12-day-old rats. However, the proportion of SMR2-positive cells remained low compared with the proportion of GRP-positive cells (Figure 2C). Double hybridization experiments with GRP and SMR2 probes in 5- and 12-day-old rats confirmed that, although most SMR2 highly positive cells also expressed GRP, they represented only a fraction of the GRP-positive cells.
Using longer (21 days compared to 5–7 days) exposure times for SMR2 mRNA detection, a large population of acinar cells with labeling intensity near the background level was revealed in addition to the high-expressing cells in 12-day-old rats (not shown). This suggests that, at 12 days of age, all acinar cells might express SMR2 but at very different levels.
To further investigate the differentiation state of SMR2 high-expressing cells, we also performed double ISH experiments at days 5 and 12 after birth with probes for SMR2 and the proacinar marker SMGA (Figure 2B). A number of cells were found to co-express SMR2 and SMGA mRNAs, suggesting that SMR2 high-expressing cells do not necessarily correspond to a late stage in acinar differentiation.
Altogether, these experiments show that SMR2 is likely expressed as early as the type III P stage. In contrast with GRP, whose expression is maximally induced in all differentiating acinar cells, SMR2 is expressed at very heterogeneous levels in differentiating acinar cells.
SMR2 Expression Gradually Increases in Acinar Cells in Males During Puberty and in Females up to 6 Weeks
We next studied SMR2 expression during the first 9 weeks of life in male and female rats, by in situ detection, using medium exposure times (7 days). The percentage of GRP-expressing cells in the entire cell population was determined in parallel to follow acinar cell differentiation. The proportion of GRP-positive cells in the total cell population was found to reach a plateau by 3–4 weeks of age, suggesting that acinar differentiation might be complete by 4 weeks of age. This is consistent with the fact that, by 4 weeks of age, most acini were found to have gained their typical morphology (Moreira et al. 1991; and data not shown).
As shown in Figure 3E, the percentage of acinar cells expressing SMR2 increased up to about 6 weeks in females. However, heterogeneity of SMR2 expression levels among acinar cells was conserved (Figures 3A, 3C, and 2D). A population of highly positive cells, which increased in number with age, was observed, together with a large population of cells with medium-intensity labeling. Cells with low SMR2 expression levels, near the background level and not taken into account in the determination of the percentage of positive cells, could be detected at longer exposure times. Therefore, SMR2 expression appears to display a continuous range of values in the acinar cell population in female rats, from low- to high-expressing cells. This is compatible with a continuous distribution of SMR2 mRNA levels around a mean value and an increase in this mean value up to 6 weeks of age in females.
In males, the number of cells with medium- and high-intensity labeling increased more rapidly than in females and the percentage of these cells among acinar cells reached 100% by 9 weeks of age (Figure 3E). However, some heterogeneity of labeling among acinar cells was maintained up to adulthood (Figures 3B and 3D; see also Figure 4F) and, as in females, the proportion of cells expressing SMR2 at medium levels was high. These results are compatible with a continuous range of SMR2 mRNA levels in the acinar cell population and a progressive increase in the mean SMR2 expression level during puberty.
A distribution of labeling similar to that in adult females was found in castrated males (not shown).
In conclusion, no evidence for a bimodal pattern of SMR2 expression was obtained under conditions of low or medium concentrations of androgens. Moreover, the continuous increase in the mean SMR2 mRNA levels at the cell level during male puberty strongly suggests a graded induction of SMR2 expression in males. A graded induction of SMR2 expression up to 6 weeks of age is also observed in females.
Detection of SMR2 Primary Transcripts
Increases in SMR2 mRNA steady-state levels, in response to androgens during puberty, might be due to an increase in transcription rate and/or in mRNA half-life. Primary transcripts (hnRNAs), which are temporally associated with the transcription process, are short-lived because of rapid splicing and are therefore considered to be accurate indicators of ongoing or very recent transcription. Therefore, to determine whether the increase in the SMR2 mRNA levels is correlated with a change in transcription rate, we performed in situ detection of SMR2 hnRNAs using an antisense RNA probe corresponding to part of the SMR2 intron 2 sequence. Under these conditions, labeling is observed specifically in the acinar cells, in the SMG of males, before puberty (4-week-old rats), or at adulthood (9-week-old rats), as well as in the SMG of female rats. As expected for primary transcripts, labeling is concentrated in the cell nuclei.
Labeling occurs in a higher percentage of acinar cell nuclei, and is more intense, in adult compared to pre-pubertal males (40% vs 6%) (compare Figures 4D and 4B), and in males compared to females (40% vs 8%) (Figures 4D and 4C). This indicates that androgen effects on SMR2 expression rely, at least in part, on an increase in transcription rate. Surprisingly, only 40–50% of acinar cells contain detectable amounts of hnRNAs in adult 9-week-old male rats. This might be due to the difficulty in detecting low levels of hnRNAs in these experiments. The existence of cells in which SMR2 is transcribed at a lower rate might explain the heterogeneity of SMR2 mRNA levels among acinar cells in male rats and, more generally, in females and immature rats. However, the comparison of SMR2 mRNA and hnRNA labeling patterns on serial sections reveals that some acinar cells contain high amounts of SMR2 mRNA but are negative for hnRNA (Figures 4E and 4F). This suggests that, in these cells, transcription might have recently been turned off, leading to the disappearance of hnRNAs, whereas mRNAs, which are more stable, would still be detected in the cytoplasm. Therefore, SMR2 transcription might be a discontinuous process even in the continuous presence of androgens. The same phenomenon has previously been observed for VCSA1, the second androgen-responsive gene expressed in rat submandibular gland acinar cells (Rosinski–Chupin et al. 2001). Interestingly, cyclic assembly of a transcription complex involving estrogen receptor and co-activators and resulting in waves of transcription has been reported (Shang et al. 2000).
In contrast, a higher percentage of positive nuclei (Figures 4D and 4G) associated with a lower heterogeneity in mRNA levels among acinar cells is observed using a GRP hnRNA probe or a GRP mRNA probe.
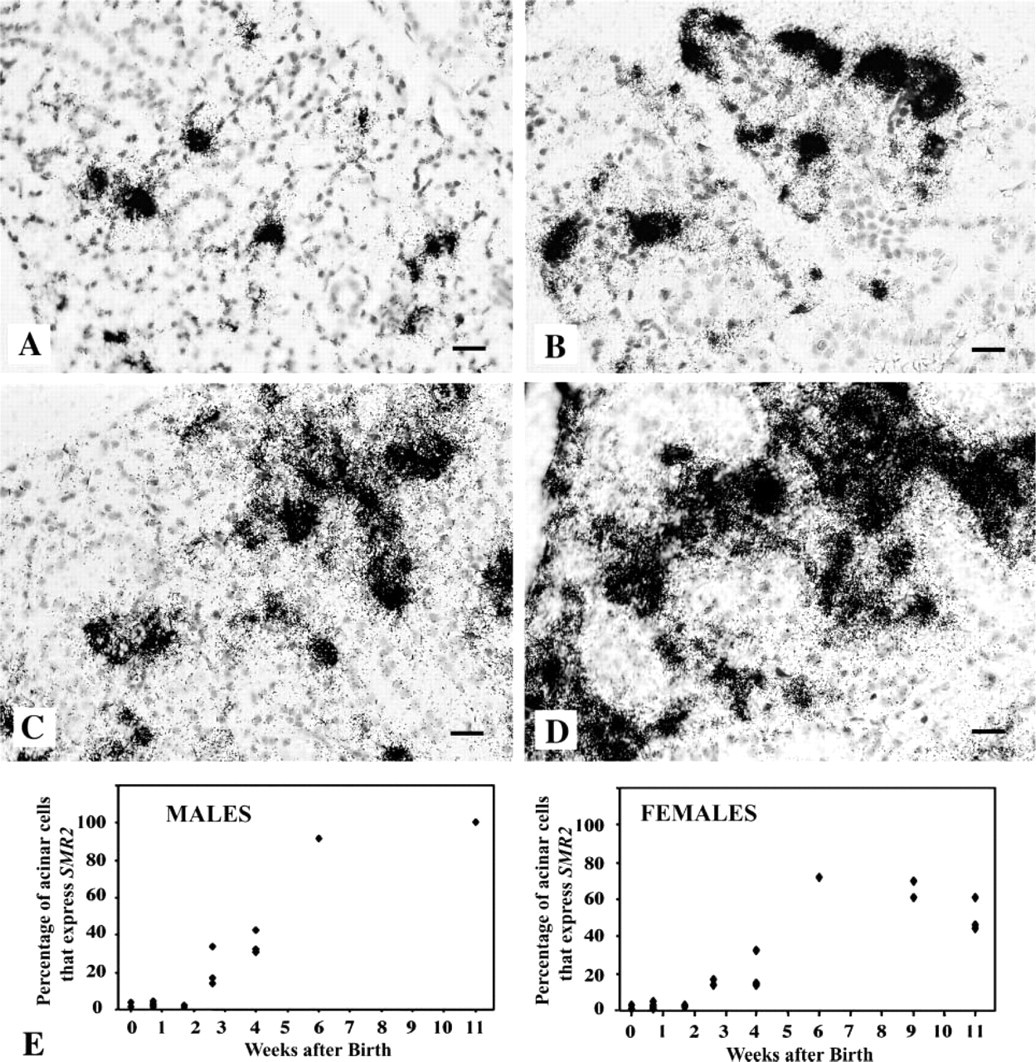
Variations of the number of SMR2 highly-expressing cells during SMG development in male and female rats. (
An increase in the percentage of acinar nuclei containing detectable amounts of SMR2 hnRNA is also observed in females between 4 and 9 weeks of age (0.5 vs 8%) (Figures 4A and 4C), correlating with the increase in mRNA steady-state levels and in the number of acinar cells positive for SMR2 mRNA detection.
Repeated Exposure to Isoproterenol Has a Complex Effect On SMR2 mRNA Accumulation, Length of PolyA Tail, and Cellular Distribution
Chronic daily exposure to isoproterenol is known to induce acinar proliferation and differentiation and to affect the expression of a number of acinar genes. For example, isoproterenol has been shown to decrease GRP Ca but not GRP Cb mRNA levels (Ten Hagen et al. 1997).
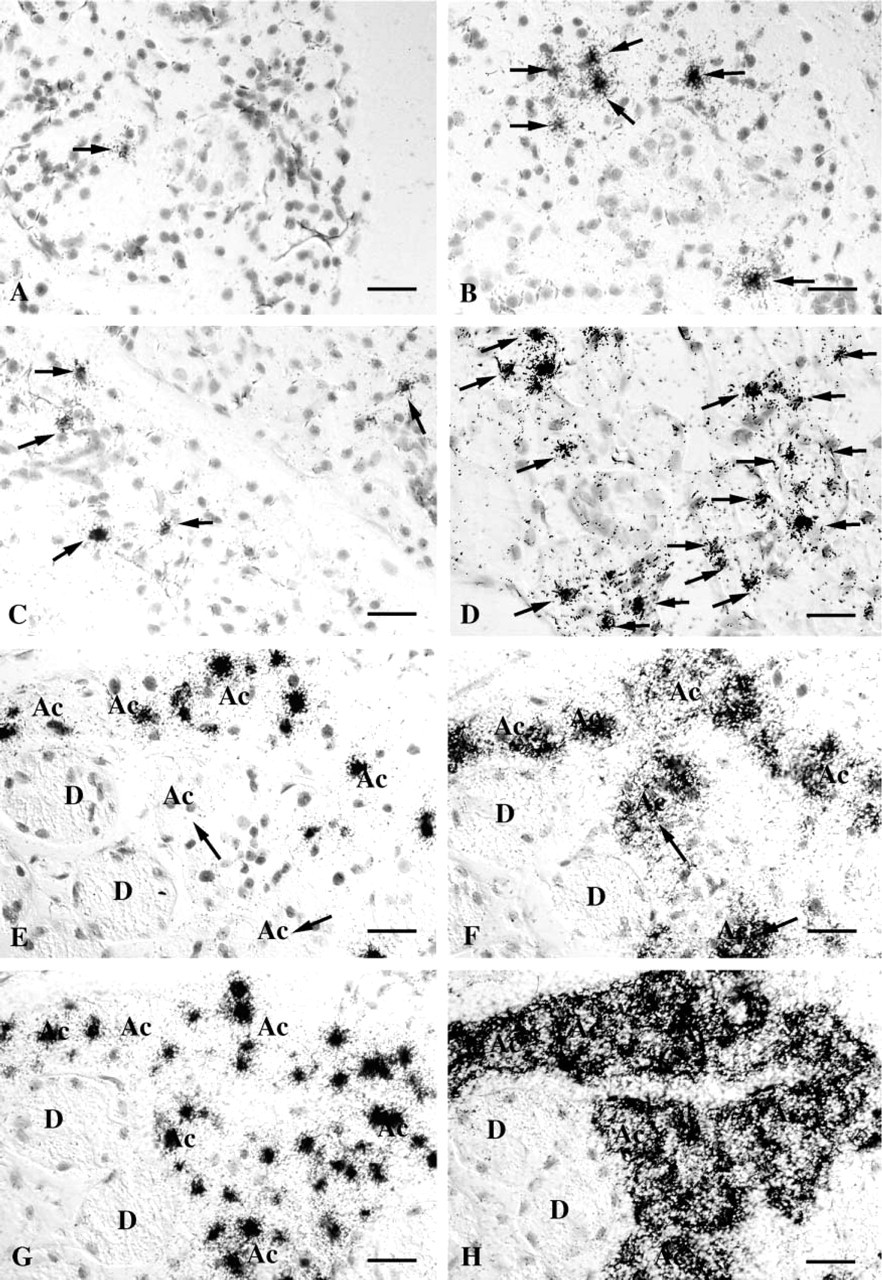
Variations of SMR2 hnRNAs during SMG development. SMR2 primary transcripts were detected by ISH on SMG sections from a 4-week-old female (
To determine if β-adrenergic agonists could also have an effect on SMR2 mRNA accumulation, we studied the effects of daily IP injection of isoproterenol for 4 days in 8-week-old male and female rats. Northern blotting analysis revealed that repeated administration of isoproterenol increases the accumulation of SMR2 mRNA in both males and females (Figures 5A and 5C), with a slightly stronger effect in females than in males.
In addition to the increase in amounts of SMR2 mRNA after exposure to isoproterenol, an increase in the size of the hybridizing mRNA was observed in most samples (Figure 5A). To determine the origin of the increase in mRNA size, an increase in the length of the polyA tail was tested. A selective degradation of the polyA tails was performed and the deadenylated mRNAs were analyzed by Northern blotting. As shown in Figure 5B, after deadenylation the SMR2 mRNAs obtained from isoproterenol-injected or control rats migrate with the same apparent electrophoretic mobility, indicating that RNA size differences were due to different polyA tail lengths.
Northern blotting analysis using a GRP probe showed that GRP expression is globally decreased after chronic exposure to isoproterenol (Figures 5A and 5C). Surprisingly, as previously observed for SMR2, the values are slightly more affected in females than in males. In addition, as for SMR2 mRNA, an increase in the mRNA size (Figure 5A) can be observed. This effect also appears to be linked to an increase in the polyA tail length (Figure 5B).
Finally, the effects on SMR2 expression of chronic daily exposure to isoproterenol were studied at the single-cell level by ISH with a probe for SMR2 mRNA. Under these conditions, isoproterenol induces an increase in the volume of acinar cells. In female rats, most acinar cells are now clearly identified as SMR2-expressing cells, consistent with an increase in SMR2 expression per cell. Interestingly, the heterogeneity of SMR2 mRNA levels among acinar cells appears to be reduced, although expression remains higher in a few cells (Figure 6A and 6B). In particular, inside the same acinus all cells express SMR2 at relatively equivalent levels, which contrasts with intra-acinus heterogeneity of labeling in control females, in which high and low expressing cells are found together (Figure 6C). Therefore, chronic daily exposure to isoproterenol has a complex effect on SMR2 expression, not only modifying the levels of SMR2 expression and the length of the polyA tail of the mRNA but also the expression pattern among acinar cells. Despite the increase in SMR2 mRNA levels per cell in females, detection of hnRNA failed to reveal an increase in SMR2 transcription rate in these experiments (data not shown).
Discussion
When studied at the single-cell level, responses to inducers have usually been described as either graded or binary. Using the VCSA1 gene as a model, we previously obtained evidence that, in the physiological context of rat SMG, androgens can elicit both types of responses. To determine whether these androgen effects can be generalized, we have studied the induction of SMR2 expression during postnatal development and during sexual maturation. We show here that the pattern of SMR2 induction differs notably from that observed with either VCSA1 or GRP, a marker of acinar differentiation. In particular, whereas SMR2 is expressed heterogeneously in acinar cells under conditions of low concentrations of circulating androgens (before puberty and in castrated males), the distribution of SMR2 expression levels appears to be unimodal; the androgen response does not include a binary component.
During SMG development, SMR2 expression occurs during the differentiation of acinar cells from their precursors, the so-called type III cells. Several lines of evidence suggest that SMR2, like GRP, is first expressed at the intermediate IIIP cell stage. The first SMR2-expressing cells are detected during the first days after birth, at a time when type IIIP cells are predominant. Furthermore, we found that some cells co-express SMR2 and SMGA, a marker of types III and IIIP cells. However, whereas GRP genes are rapidly expressed at a relatively uniform and high level in most differentiating acinar cells, SMR2 is expressed at very heterogeneous levels in differentiating acinar cells. This heterogeneity is reminiscent of what has previously been observed for VCSA1 (Rosinski–Chupin et al. 2001) and might be a characteristic property of inducible genes.
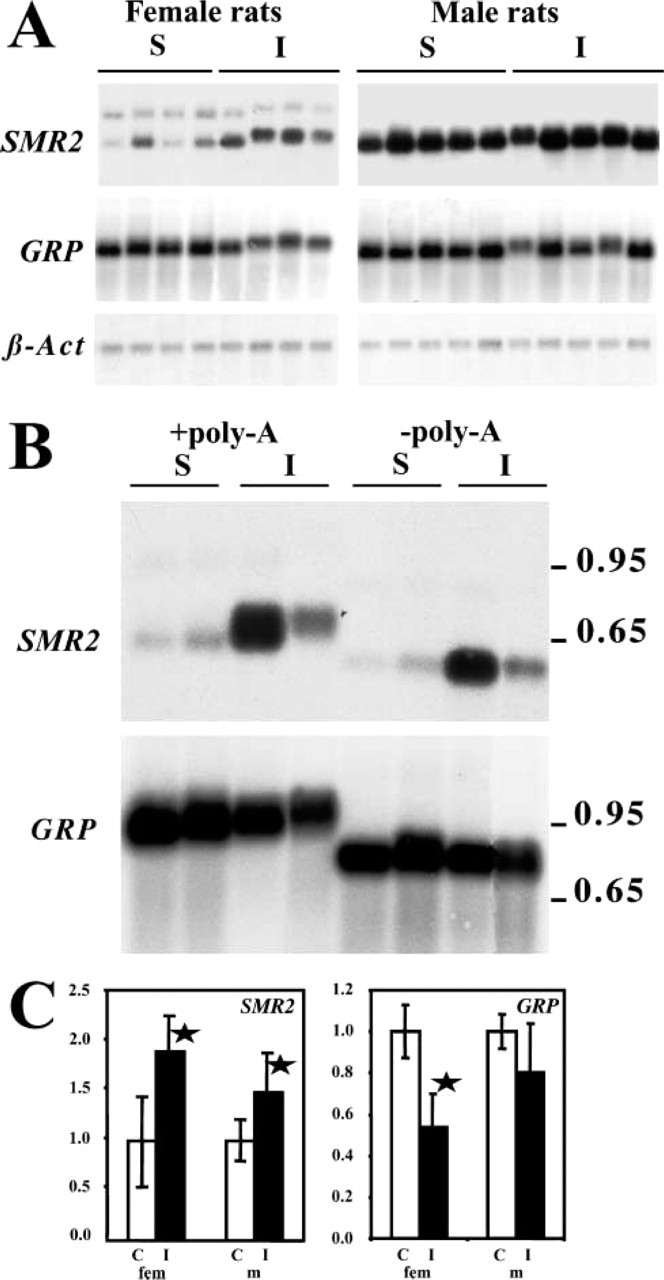
Variation of SMR2 and GRP mRNA levels in rat SMG after isoproterenol treatment. Male and female 8-week-old rats were given daily injections of either saline (S) or isoproterenol (I) during 4 days. SMG were harvested 24 hr after the last injections and RNA were prepared and analyzed. (
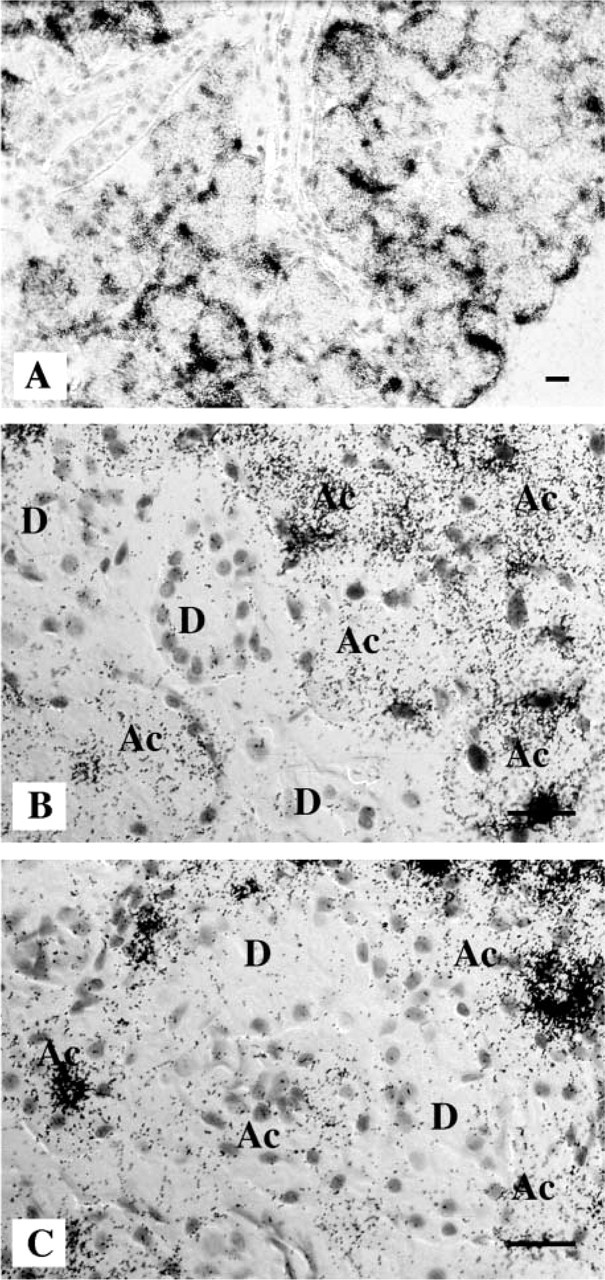
In situ hybridization of SMR2 transcripts on an SMG section from an isoproterenol-treated (
Beyond this resemblance, there are a number of differences between VCSA1 and SMR2 patterns of expression in acinar cells. We previously demonstrated a bimodal distribution of VCSA1 expression in the acinar cell population in rats before puberty. Two sub-populations that either express VCSA1 at high levels or do not express VCSA1 at all were clearly identified. Although some cells with intermediate labeling levels were observed, they did not constitute a large part of the total cell population. In addition, no further detection of new VCSA1-expressing cells was possible using long exposure times (Rosinski–Chupin et al. 2001), indicating that negative cells were really non-expressing cells. The distribution pattern of SMR2 expression is quite different. Despite the wide range in SMR2 mRNA levels among acinar cells, our observations suggest a continuous distribution from low- to high-expressing cells. Using long exposure times, a majority of differentiating acinar cells were found to express SMR2, at least at low levels, in 12-day-old rats. In addition, a larger number of cells with intermediate labeling intensity than of cells with high labeling intensity was observed, suggesting a continuous variation in SMR2 expression levels in the cell population. Under these conditions, the increase in SMR2 expression at puberty appears to involve only a progressive increase in the mean SMR2 expression level in individual cells.
Because SMR2 expression depends on acinar differentiation, a surprising observation was that SMR2 mRNA levels continue to increase after the end of acinar differentiation period in female rats and reached a plateau value later than GRP mRNAs. This appears to be specific for SMR2 because VCSA1 expression was previously shown to be maximal in females by 3–4 weeks of age, like GRP. At the single-cell level, the global increase in SMR2 expression correlated with a gradual enhancement of the SMR2 mRNA content per cell. In addition, a higher number of cells actively transcribing the gene were observed in adult than in 4-week-old females, using in situ detection of primary transcripts, revealing an increase in transcription rate. This suggests that other age-dependent factors, in addition to acinar differentiation, regulate SMR2 expression in females. These factors do not appear to be directly linked to female physiology because both the global levels of SMR2 mRNA and the pattern of SMR2 mRNA distribution were similar in females and in castrated males.
The secretory function of the SMG is regulated, at the level of protein synthesis and secretion, by numerous hormones and by the autonomic nervous system. Both sympathetic and parasympathetic branches of the autonomic nervous system innervate the rat SMG. Whereas parasympathetic innervation of rat SMG is present at birth, sympathetic innervation reaches the glands by postnatal day 5 (De Champlain et al. 1970; Bottaro and Cutler 1984). In adult rats, prolonged electrical stimulation of the autonomic nervous system via the superior cervical ganglion causes enlargement of salivary glands by increasing cell size and cell number. Similarly, isoproterenol was shown to induce hypertrophic and hyperplasic enlargements of rodent salivary glands and to induce the expression of a number of genes in these organs, such as cystatins and proline-rich proteins (Mehansho and Carlson 1983; Ann et al. 1987; Humphreys-Beher 1987; Shaw et al. 1990; Bedi 1991; Cooper et al. 1991; Matsuura and Hand 1991; Nishiura et al. 1991; Ten Hagen et al. 2002). These effects of isoproterenol are mediated, at least in part, by activation of β-adrenergic receptors, which triggers an increase in cAMP cellular content and a cascade of molecular events leading to gene activation.
In a search for inducers of SMR2 expression, we looked for an effect of chronic exposure to isoproterenol on SMR2 expression. Isoproterenol was previously shown to be able to induce expression of PRPs, which are phylogenetically related to GRP and SMR2. We show by Northern blotting analysis that chronic injection of isoproterenol induces, particularly in females, an increase in SMR2 mRNA levels while decreasing global GRP mRNA levels. A decrease in GRP-Ca expression was previously reported (Ten Hagen et al. 1997). Therefore, although they belong to the same gene family, SMR2 and GRP are differentially regulated by isoproterenol. Both effects of isoproteronol on SMR2 and GRP expression were found to be less marked in males than in females, as was also previously reported for cystatin induction by isoproterenol (Shaw et al. 1990). Whether or not isoproterenol stimulates SMR2 transcription is not yet clear. Surprisingly, although an increase in SMR2 mRNA amounts was observed using Northern blotting analysis and in situ detection of mature transcripts, we did not find an increase in the number of cells positive for SMR2 primary transcripts. Because in situ detection of SMR2 hnRNA was performed 24 hr after the last injection of isoproterenol and because primary transcripts are unstable, this result does not rule out the possibility that a transient stimulation of transcription might have occurred rapidly after isoproterenol injection. Alternatively, isoproterenol might have increased SMR2 mRNA steady-state levels through a post-transcriptional mechanism. Interestingly, we observed an increase in SMR2 mRNA polyA tail size during SMR2 response to isoproterenol. Regulation of polyadenylation was reported to modulate translatability and/or stability of some mRNAs (Bernstein and Ross 1989; Nielsen and Shapiro 1990). An increase in polyA tail length was shown to be associated with the post-transcriptional response of the gene coding for EGF to androgens in mouse SMG (Sheflin et al. 1996). However, the same increase in polyA tail length is observed for GRP, whose expression decreases after isoproterenol injection.
In conclusion, although the precise mechanisms remain to be determined, our results indicate that SMR2 expression can be induced through at least two different pathways, one involving androgens and the other β-adrenergic signaling. This is reminiscent of previous reports on certain prostatic genes (Guthrie et al. 1990). This dual regulation by two different pathways could explain why, at low concentration of androgens such as found in females and in immature or castrated males, SMR2 expression, in contrast to VCSA1 expression, is not completely turned off in a majority of acinar cells. In such a context, androgens may not be needed to turn on SMR2 transcription and may affect only expression levels. This might also explain why a binary response to androgens is not observed. Therefore, the type of response elicited by androgens in a physiological environment may be strongly dependent on whether other factors co-regulate gene expression.
Footnotes
Acknowledgements
Supported by grants from the Institut Pasteur and from the Centre National de la Recherche Scientifique.
We would especially like to thank Dr M. Goodhardt for helpful discussions and critical reading of the manuscript.
