Abstract
Nerve growth factor (NGF) and semaphorin3A (Sema3A) are guidance cues found in pathways and targets of developing dorsal root ganglia (DRG) neurons. DRG growth cone motility is regulated by cytoplasmic signaling triggered by these molecules. We investigated interactions of NGF and Sema3A in modulating growth cone behaviors of axons extended from E7 chick embryo DRGs. Axons extending in collagen matrices were repelled by Sema3A released from transfected HEK293 cells. However, if an NGF-coated bead was placed adjacent to Sema3A-producing cells, axons converged at the NGF bead. Growth cones of DRGs raised in 10-9 M NGF were more resistant to Sema3A-induced collapse than when DRGs were raised in 10-11 M NGF. After overnight culture in 10-11 M NGF, 1-hr treatment with 10-9 M NGF also increased growth cone resistance to Sema3A. Pharmacological studies indicated that the activities of ROCK and PKG participate in the cytoskeletal alterations that lead to Sema3A-induced growth cone collapse, whereas PKA activity is required for NGF-mediated reduction of Sema3A-induced growth cone collapse. These results support the idea that growth cone responses to a guidance cue can be modulated by interactions involving coincident signaling by other guidance cues.
D
Sensory neurons of dorsal root ganglia (DRG) extend peripheral processes to skin, muscle, and other organs, and DRG central processes make synapses in the spinal cord. The neurotrophin NGF and the semaphorin Sema3A regulate the in vitro motility of DRG growth cones and regulate in vivo axon morphogenesis, as shown by experimentation and by analyses of mice with mutations for NGF, Sema3A, and the neuropilin-1 Sema3A receptor (Martin et al. 1989; Taniguchi et al. 1997; Patel et al. 2000; Tucker et al. 2001). NGF and Sema3A are present in peripheral pathways and in targets of DRG axons (Elkabes et al. 1994; Messersmith et al. 1995; Wright et al. 1995; Giger et al. 1996; Puschel et al. 1996; Shepherd et al. 1996,1997; White et al. 1996; Fu et al. 2000; Cahoon-Metzger et al. 2001). This co-distribution prompted us to examine whether NGF and Sema3A interact in regulating DRG growth cones.
NGF promotes differentiation, survival and morphogenesis of trkA-expressing sensory neurons (Snider 1994). Local application of NGF in vitro stimulates growth cone migration and axon branching (Gallo et al. 1997; Gallo and Letourneau 1998). NGF binding to trkA and p75 receptors activates a number of signaling pathways (Lee et al. 2001; Patapoutian and Reichardt 2001), and NGF regulation of growth cone motility involves PLC, PI3kinase, and cAMP pathways (Gallo and Letourneau 1998; Song et al. 1998; Cai et al. 1999; Ming et al. 1999; Song and Poo 1999; Zhang et al. 1999). Sema3A is concentrated in regions avoided by NGF-responsive DRG axons, and soluble Sema3A collapses NGF-responsive DRG growth cones (Luo et al. 1993; Shepherd et al. 1997; Tuttle and O'Leary 1998). Signaling by Sema3A through the neuropilin 1–plexin complex is not well understood, although reports implicate cGMP levels and Rac1 and RhoA GTPases in Sema3A signaling (Jin and Strittmatter 1997; Kuhn et al. 1999; Vastrik et al. 1999; Nakamura et al. 2000; Rhom et al. 2000).
We investigated interactions of NGF and Sema3A signaling in regulating chick DRG growth cones. Elevated levels of NGF reduced the collapse of DRG growth cones by Sema3A. Pharmacological studies indicated opposite roles for protein kinases PKA and PKG in mediating signaling by these molecules. Inhibition of the RhoA effector ROCK also reduced Sema3A-induced growth cone collapse. Our results support the idea that the response of growth cones to a single guidance cue is not invariant but depends on interactions with signaling triggered from other cues and on the activities of other second messenger pathways (Hopker et al. 1999; Song and Poo 1999; Zou et al. 2000; Stein and Tessier-Lavigne 2001).
Materials and Methods
NGF was obtained from R & D Systems (Minneapolis MN). 8-Bromo-cyclic AMP, 8-bromo-cyclic GMP, ODQ, KT5720, KT5823, Sp-cAMP, Y27632, and HA1077 were purchased from Biomol Research Laboratories (Plymouth Meeting, PA). PKI and YC-1 were purchased from Calbiochem (La Jolla, CA). Purified Sema3A and HEK293 cells transfected to produce and secrete Sema3A were generously provided by Drs. Yuling Luo and Sheldon Ng of Exelixis (South San Francisco, CA). Drugs were prepared in water or in DMSO and were aliquotted.
DRG Cultures
Culture dishes were treated overnight with 10 μg/ml laminin. Explants of E7 chick DRGs were cultured overnight in a warmed, humidified incubator in 2 ml F12 medium (Gibco/BRL; Gaithersburg, MD) buffered with 10 mM HEPES and with supplements (5 μg/ml transferrin, 40 μg/ml sodium pyruvate, 5 μg/ml phosphocreatine, 5 μg/ml progesterone, 5 μg/ml Na selenite) and NGF. DRG explants were experimentally treated with neurotrophins and drugs in several ways.
Explants were cultured overnight in 10-11, 10-10, or 10-9 M NGF before the addition of Sema3A or control medium for 30 min. Or
Explants were cultured overnight in 10-11 M NGF. On the next day, 10-9 M NGF was added to some dishes for 1 hr, followed by the addition of Sema3A for 30 min. Or
Explants were cultured overnight in 10-11 M NGF. On the next day, 10-9 M NGF was added to some dishes for 1 hr, followed by addition of pharmacological inhibitors to some dishes for another hour, followed by Sema3A for 30 min. Or
Explants were cultured overnight in 10-11 M NGF. On the next day, a drug was added to some dishes for 1 hr, followed by addition of 10-9 M NGF for 1 hr, followed by Sema3A for 30 min.
All collapse assays were performed similarly. Purified Sema3A or conditioned medium from Sema3A-trasfected 293 cells was added for 30 min, followed by fixation with 0.5% glutaraldehyde in PBS for 30 min. Fixed DRGs were viewed by phase-contrast optics with a X20 objective, and the morphology of randomly selected axon endings was scored as either a normal growth cone with lamellipodia and filopodia or a collapsed growth cone (a tapered axon terminal without lamellipodia or less than 3 filopodia; Luo et al. 1993).
Conditioned Media from Sema3A-transfected 293 Cells
HEK 293 cells stably transfected to express human Sema3A were prepared as described in Luo et al. (1993). The cells were maintained in MEM medium supplemented with 10% fetal bovine serum, 1 ml/100 ml penicillin/streptomycin/fungizone, 300 μl/100 ml of 100 mg/ml Geneticin, 1 ml/100 ml
Immunocytochemistry
DRG explants from E7 chick embryos were cultured on laminin-coated coverslips for 24 hr as described above. After 24 hr of culture, the cultures were fixed with 4% paraformaldehyde (PF) in PBS by adding warm fixative directly to the culture medium for 15 min, followed by immunocytochemistry. After rinsing off the fix, cultures were quenched with 0.1 M glycine in PBS for 15 min, and the cells were blocked and permeabilized with 0.1% Triton X-100 in PBS with 1% fish gelatin for 30 min. The fixed cells were incubated at 1:100 dilutions of polyclonal antibodies against the catalytic subunit of the α-isoform of PKA or PKGI α (both from Stressgen Biotechnologies, San Diego, CA) for 1 hr at room temperature (RT). Staining for tubulin was done with a 1:100 dilution of a monoclonal antibody against β-tubulin (βIII; Covance, Princeton, NJ). RhoA was localized with a monoclonal antibody (Santa Cruz Biothechnology) One percent fish gelatin was incubated together with the primary antibody. After rinsing in PBS, the samples were incubated with secondary rhodamine-conjugated goat anti-rabbit and fluorescein-conjugated goat anti-mouse antibodies (Jackson Laboratories; West Grove, PA), each diluted 1:400 in PBS with 1% fish gelatin for 1 hr at RT. F-actin was labeled with rhodamine-conjugated phalloidin (Molecular Probes; Eugene, OR).
Results
To first examine how elongating axons integrate simultaneous signaling from multiple guidance cues, we cultured explants of E7 chick DRGs for 24–48 hr in collagen gels adjacent either to cell aggregates of HEK293 cells transfected to produce and release Sema3A or to glass beads that release NGF from their surfaces. Figure 1 shows that many more DRG axons extended from the side of a DRG explant that was away from Sema3A-producing HEK cells (Figure 1, upper panels). In the presence of non-transfected HEK cell aggregates, axon outgrowth was equal from all sides of DRG explants (not shown). When DRG explants were adjacent to an NGF-coated glass bead, axon growth toward the bead was denser than elsewhere around the explant, although many axons extended beyond the NGF beads (Figure 1, middle panels). When DRG explants were cultured in collagen gels with both an NGF bead and Sema3A-transfected HEK cell aggregate on the same side of the explant, a different pattern of axon outgrowth was observed. Axons extended from the side of the explant facing the bead and the cell aggregate but, in a unique pattern, the axons converged on the NGF bead, very different from the profuse axon growth beyond the NGF bead in the absence of the Sema3A-secreting HEK cells (Figure 1, lower panels). These in vitro results show that both Sema3A and NGF influence axon growth in a manner reminiscent of the axon outgrowth from DRGs through peripheral tissues towards their targets.
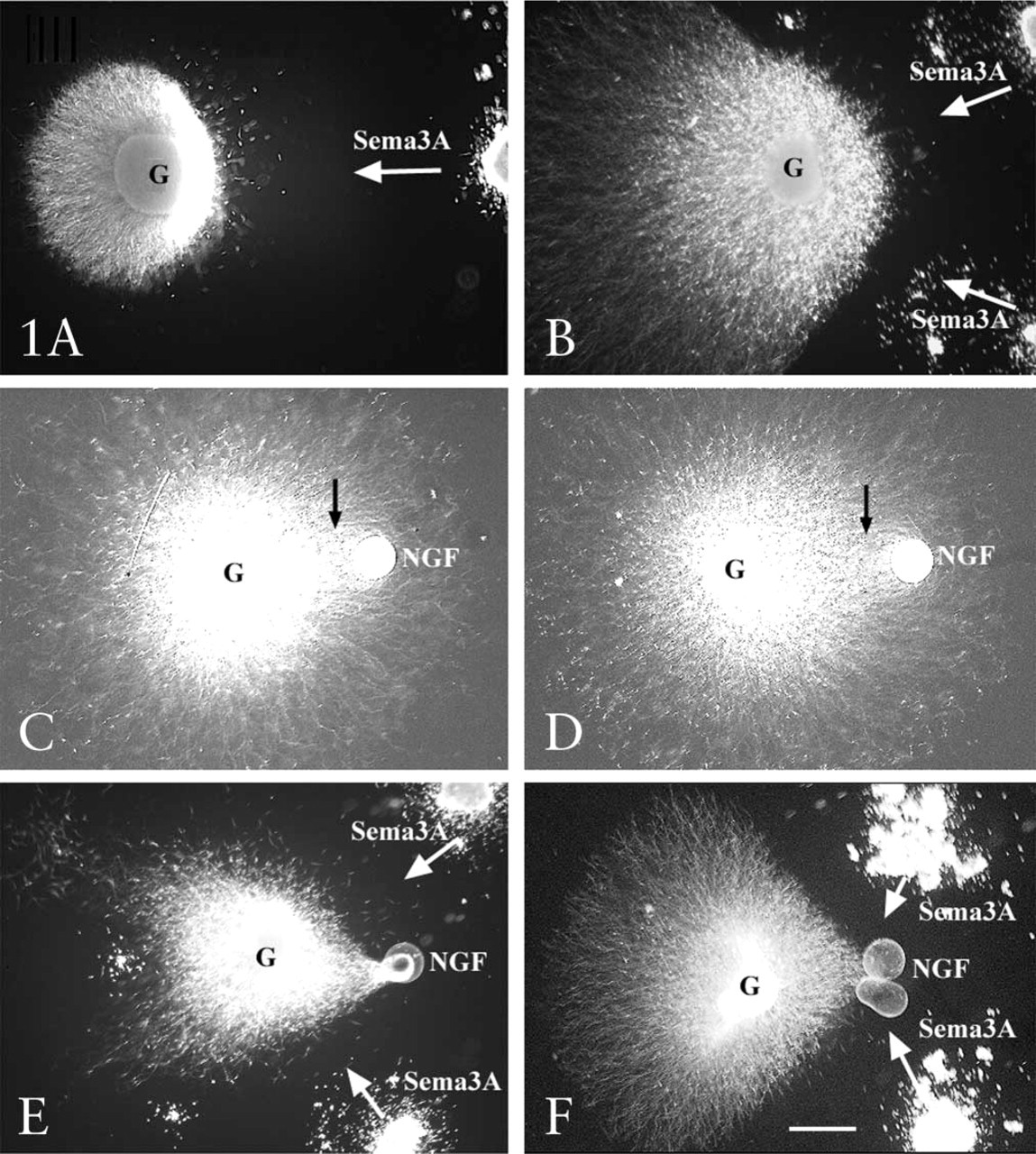
Axon outgrowth from E7 DRGs in collagen gels containing Sema3A-secreting cell aggregates and NGF beads. (
We continued investigating growth cone integration of signaling by Sema3A and NGF with more rapid and efficient in vitro assays of growth cone collapse. Exposure of DRG growth cones to global application of Sema3A causes rapid withdrawal of filopodial and lamellipodial protrusions and collapse of growth cones and distal axons (Luo et al. 1993; Figure 2). The extent of growth cone collapse in response to global application of Sema3A is dose-dependent, and we established a concentration of Sema3A-containing medium that induced approximately 50–60% collapse of growth cones extended from DRG explants cultured overnight in 10-11 M NGF. This concentration was used in the subsequent studies.

This sequence shows the collapse of a growth cone and terminal axon of a DRG neuron raised in 10-11 M NGF and exposed to Sema3A for 30 min. After 30 min the growth cones have collapsed and short axonal branches are retracted. Bar = 10 μm. From Dontchev and Letourneau (2002), with permission. Copyright 2002 by the Society for Neuroscience.
Elevated NGF Concentrations Can Reduce Sema3A-induced Growth Cone Collapse
Explants of E7 chick DRGs were cultured overnight in media containing 10-11 M, 10-10 M, or 10-9 M NGF. When DRG explants were cultured overnight with 10-9 M or 10-10 M NGF, growth cone collapse in response to a standard amount of Sema3A was significantly less than when DRGs were cultured in 10-11 M NGF (Figure 3). We next investigated whether a briefer exposure to high NGF concentrations would reduce the collapse response to Sema3A. Explants were cultured for 24 hr with 10-11 M NGF, and then the neurotrophin concentration of the medium was elevated to 10-9 M NGF for 1 hr before adding Sema3A for 30 min. One hour of exposure to 10-9 M NGF was sufficient to reduce the collapse response of DRG growth cones to Sema3A (Figure 4). Therefore, elevated concentrations of NGF can act within 1 hr to reduce the Sema3A-induced collapse of growth cones of DRG neurons raised in 10-11 M NGF.
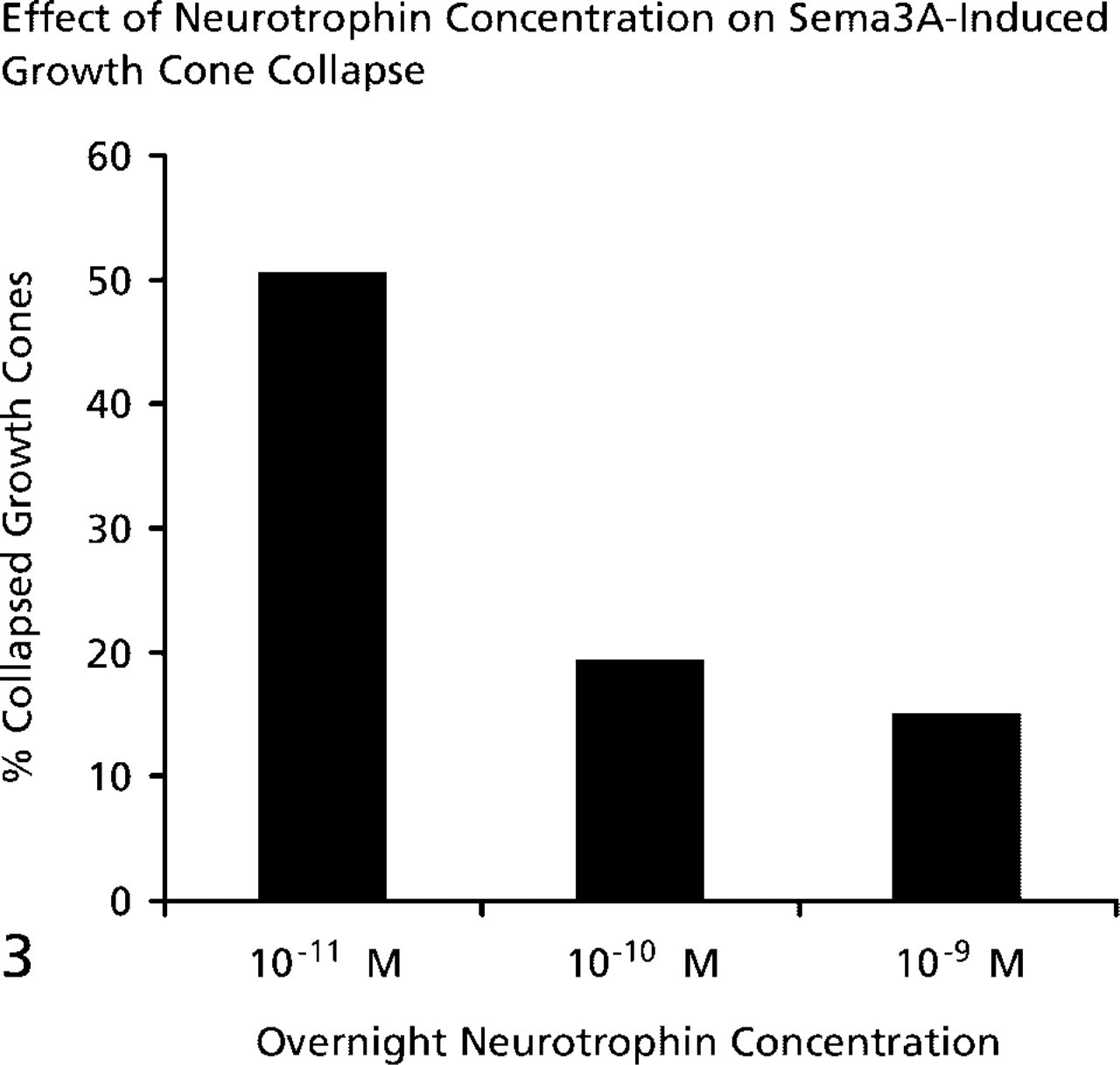
The collapse response to Sema3A is reduced when DRGs are cultured in higher NGF concentrations. E7 DRGs were cultured overnight in media containing the NGF concentrations listed on the X-axis. After 24 hr a standard amount of Sema3A was added to the dishes for 30 min, followed by fixation. The percent collapsed growth cones was determined for each condition.
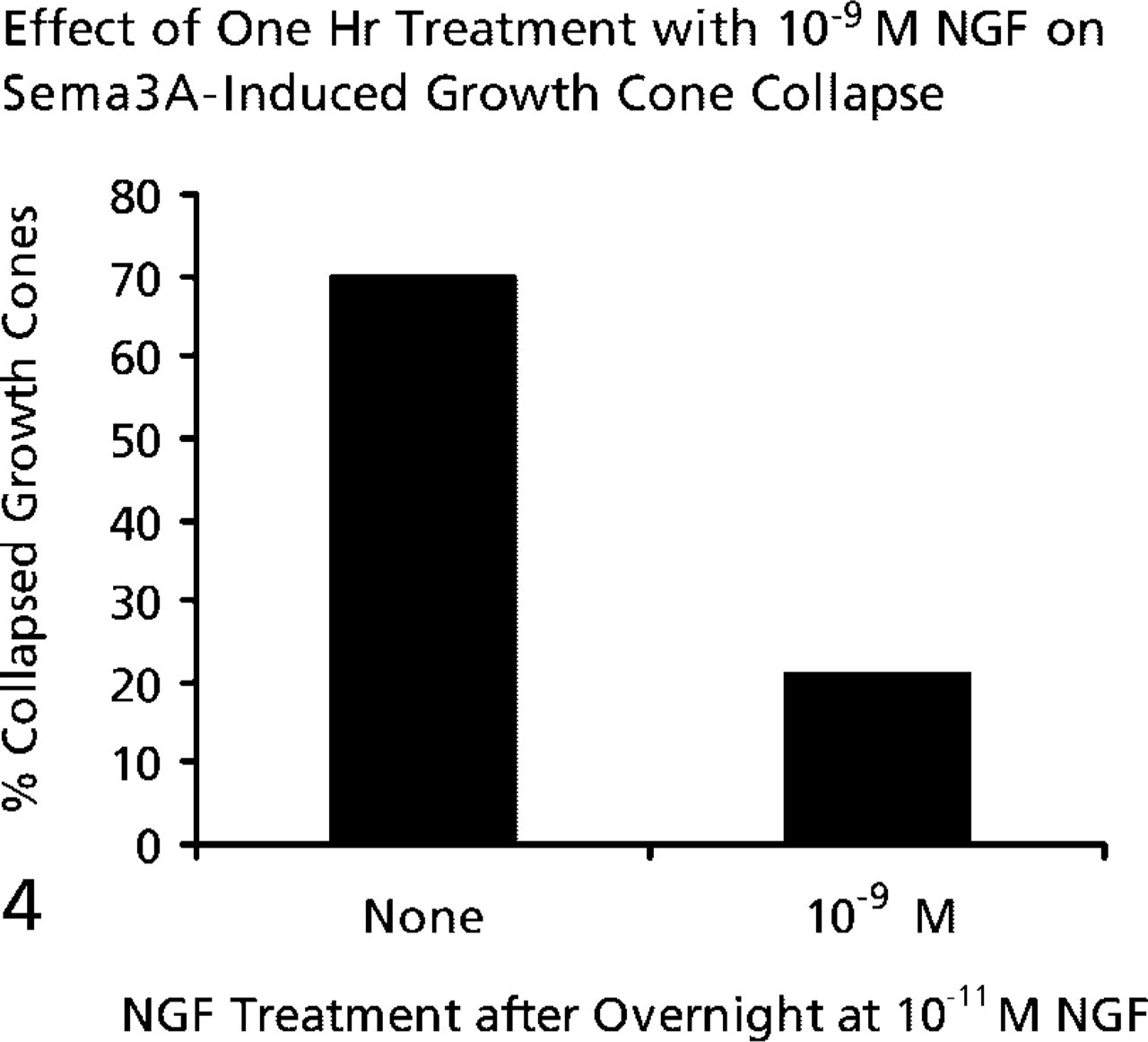
One-hour treatment with 10-9 M NGF is sufficient to decrease the collapse response to Sema3A. E7 DRGs were cultured overnight in medium containing 10-11 M NGF. The NGF concentration was elevated to 10-9 M for one hr before addition of Sema3A for 30 min, followed by fixation and determination of percent collapsed growth cones.
PKA Activity Is Involved in NGF Modulation of Sema3A-induced Growth Cone Collapse
It has been proposed that the cAMP-regulated protein kinase A (PKA) and the cGMP-regulated protein kinase G (PKG) modulate growth cone responses to many extrinsic guidance molecules (Song and Poo 1999). Therefore, we determined the effects of activators and inhibitors of these two kinases on NGF modulation of growth cone responses to Sema3A. We performed immunocytochemistry using commercially available polyclonal antibodies to determine whether PKA and PKG-I isoform are present in E7 DRG growth cones. Western blots of chick embryo brain proteins separated by SDS-PAGE showed that these antibodies specifically recognized chick proteins of the expected approximate molecular weights of 53 kD and 75 kD (not shown). Figure 5 shows images of robust staining of DRG growth cones for both PKA and PKG.
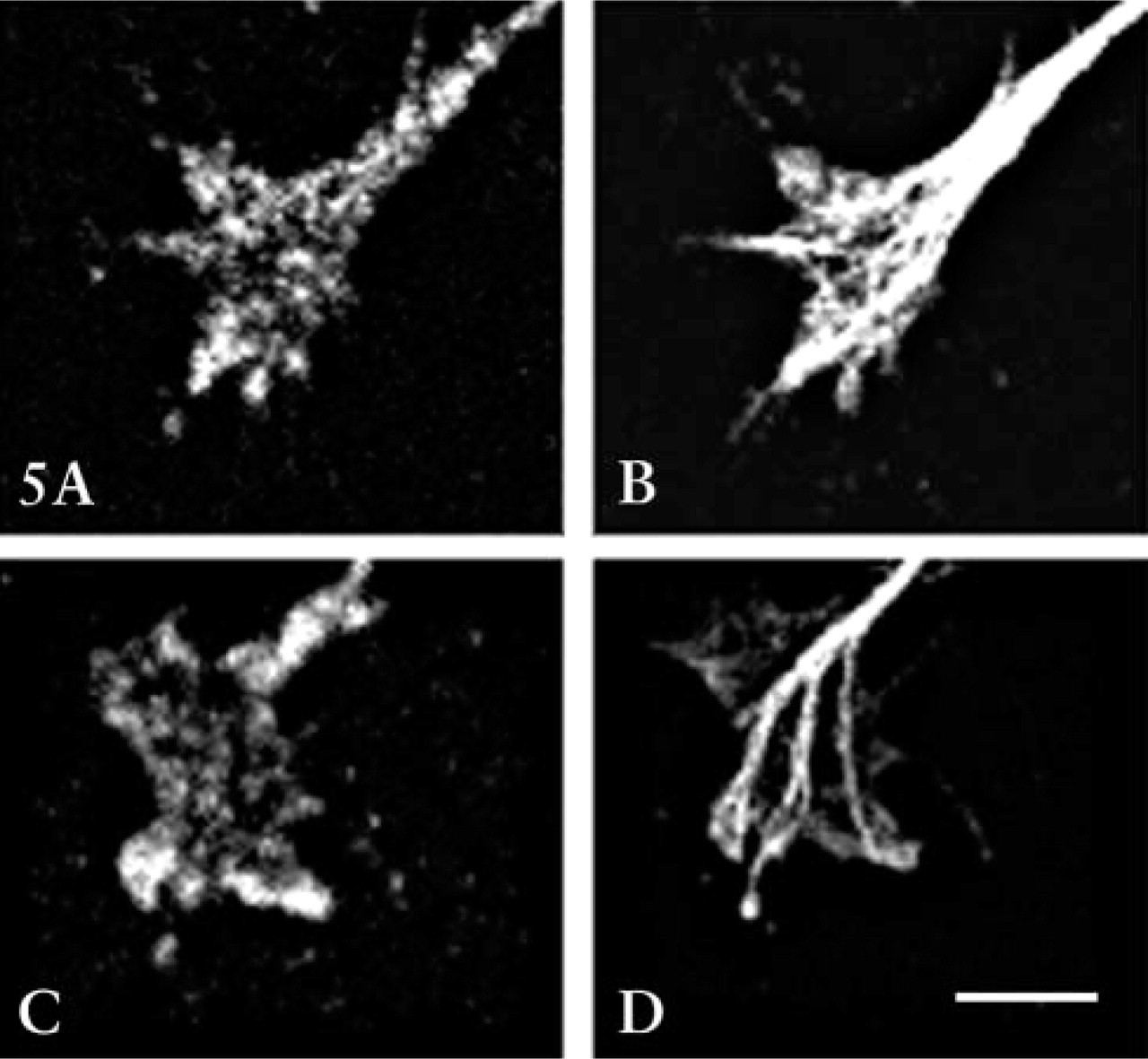
Immunocytochemical labeling of DRG growth cones with antibodies against the catalytic unit of PKA (
We tested the effects of activation of PKA by adding the cAMP analogues 8-bromo cAMP or Sp-cAMP (Figure 6A). Either PKA activator alone significantly reduced the growth cone response to Sema3A. Furthermore, when both PKA activation and elevation of NGF preceded the addition of Sema3A, the reduction in collapse response to Sema3A was even greater. Next we investigated the effect of inhibition of PKA activity on NGF modulation of growth cone responses to Sema3A. Two inhibitors of PKA activity were used (Figure 6B). When PKA was inhibited with KT5720 before addition of Sema3A (Davies et al. 2000), the collapse response to Sema3A was not significantly changed. However, inhibition of PKA by KT5720 significantly diminished the ability of elevated NGF to reduce Sema3A-induced growth cone collapse. We also used a specific PKA inhibitor, a myristolated form of PKI (Walsh et al. 1990). Prior addition of PKI enhanced the collapse of NGF-cultured DRG growth cones in response to Sema3A. We also found that addition of PKI greatly decreased the effect of 10-9 M NGF in reducing Sema3A-induced growth cone collapse. These experiments indicate that activity of PKA is required for NGF signaling to modify growth cone collapse responses to Sema3A. When PKA was inhibited, NGF-mediated reduction of the collapse response was less effective, and under conditions that activate PKA, the effects of elevated NGF were enhanced.
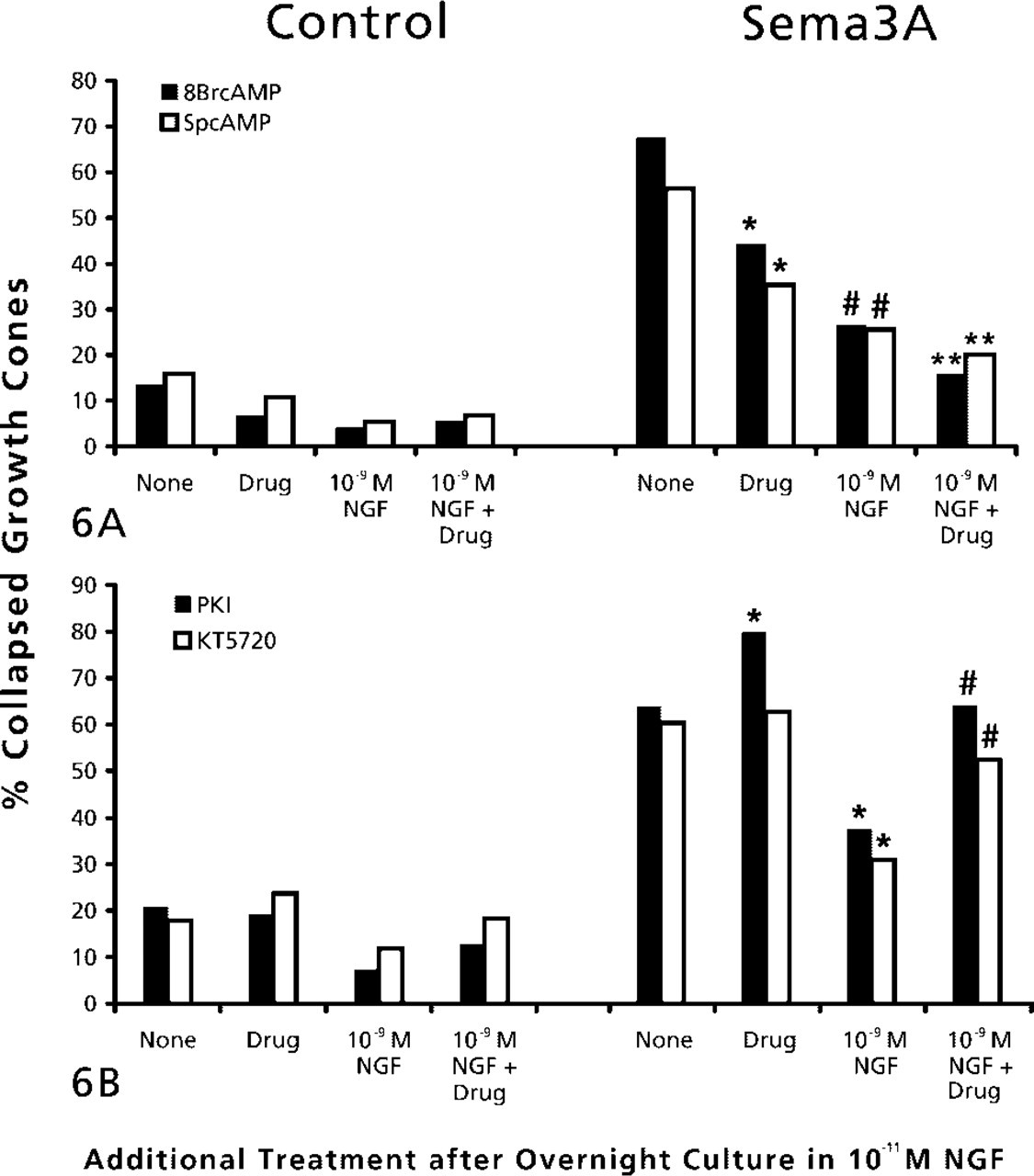
(A) Effects of PKA activation and elevated NGF on growth cone response to Sema3A. DRG explants were cultured overnight in medium containing 10-11 M NGF. At 24 hr, NGF in some dishes was elevated to 10-9 M for 60 min, then 2 mM 8-bromo cAMP or 2 μM Sp-cAMP was added to some dishes for another 60 min, then Sema3A or control medium was added for 30 min, followed by fixation. Percent collapsed growth cones for each sample population is presented. The experiments with 8-br-cAMP and sp-cAMP were conducted at different times, with slightly different levels of collapse in response to Sema3A alone. This accounts for the different height of bars labeled “None.” ∗
p<0.01, significantly different from Sema3A. #p<0.01, significantly different from drug and Sema3A. ∗∗
p<0.01, significantly different from 10-9 M NGF and Sema3A. (
In the above experiments using blockers of PKA activity, NGF was elevated to 10-9 M for 60 min before adding the drugs. This sequence of treatment would allow 60 min of signaling by elevated NGF before addition of a PKA inhibitor. In the next experiments we added the PKA inhibitor PKI either before elevating NGF to block all signaling that might involve PKA activity or after elevation of NGF. We found that the ability of 10-9 M NGF to reduce the 69% Sema3A collapse response to 40% collapse was diminished to the same extent whether PKI was added before (58% collapsed) or after (62% collapsed) elevating NGF to 10-9 M.
PKG Activity Is Involved in Sema3A-induced Growth Cone Collapse
The cGMP-regulated kinase PKG has been implicated in mediating growth cone responses to Sema3A. Therefore, we investigated the effects of manipulations that may affect PKG activity on chick DRG growth cone responses to Sema3A. First, we examined the effects of a PKG inhibitor, the drug KT5823 (Hidaka and Kobayashi 1992; Firestein and Bredt 1998). Prior addition of KT5823 significantly reduced the percent of DRG growth cones that collapsed in response to Sema3A (Figure 7A). When KT5823 pretreatment was combined with prior elevation of NGF to 10-9 M, the percentage of collapsed growth cones after Sema3A addition was not different from that for growth cones not exposed to Sema3A. Another manipulation that might reduce PKG activity is to inhibit soluble guanylyl cyclase, which would lead to reduced cytoplasmic cGMP. We added the selective guanylyl cyclase inhibitor ODQ (Garthwaite et al. 1995) before Sema3A. Treatment with ODQ alone significantly reduced the collapse response to Sema3A and, similar to the effects of the PKG inhibitor KT5823, the combined pretreatment with ODQ and 10-9 M NGF almost eliminated any response to Sema3A.
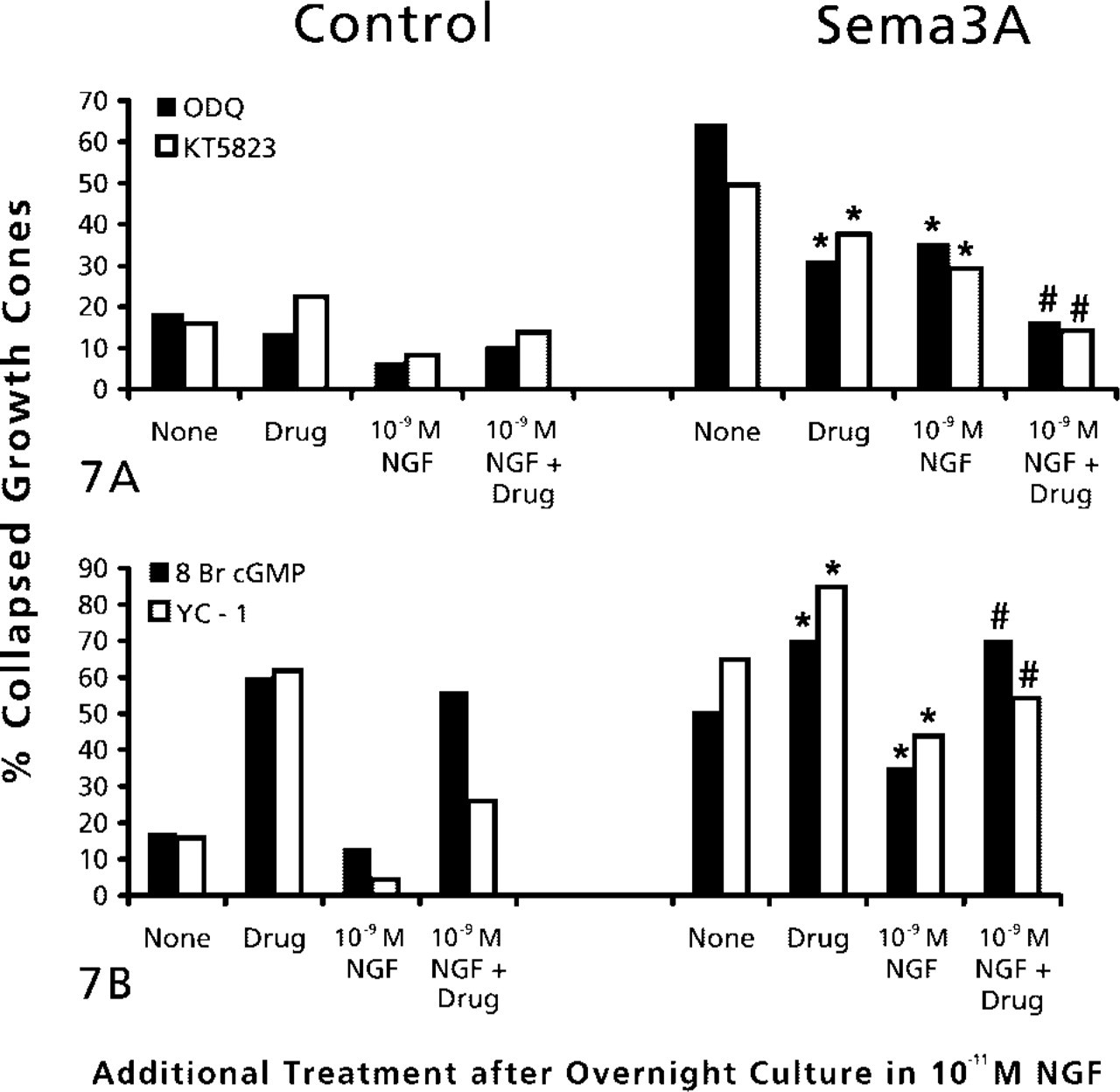
(A) Effects of inhibition of protein kinase G and elevated NGF on growth cone response to Sema3A. DRG explants were cultured overnight in medium containing 10-11 M NGF. At 24 hr, NGF in some dishes was elevated to 10-9 M for 60 min. Then 1 μM KT5823 or 100 nM ODG was added to some dishes for another 60 min, then Sema3A or control medium was added for 30 min, followed by fixation. The percent collapsed growth cones in each sample population is presented. The experiments with KT5823 and ODG were conducted at different times, with slightly different levels of collapse in response to Sema3A alone. This accounts for the different height of bars labeled “None.” ∗
p<0.01, significantly different from Sema3A. #p<0.01, significantly different from 10-9 M NGF and Sema3A. (
To examine the effects of activating PKG, we added the cGMP analogue 8-bromo cGMP before addition of Sema3A (Figure 7B). We found that 8-bromo cGMP alone induced a significant amount of growth cone collapse. This collapse was not attenuated by elevation of NGF to 10-9 M. When DRG explants were exposed to 8-bromo cGMP and then to Sema3A, growth collapse was significantly higher than when explants were treated with Sema3A alone. Elevation of NGF to 10-9 M did not reduce this extensive growth cone collapse. Therefore, PKG activity does appear to be involved in growth cone collapse. Another means to activate PKG is to stimulate quanylate cyclase activity with the drug YC-1 (Ko et al. 1994). Similar to the effect of adding 8-bromo cGMP, treatment with YC-1 alone produced a significant growth cone collapse, and when combined with addition of Sema3A, growth cone collapse was significantly elevated above the effect of Sema3A alone. In addition, YC-1 significantly diminished the reduction of the collapse response, when (NGF) was elevated before adding Sema3A. Therefore, the effects of both these drugs indicate that PKG activity promotes collapse of chick DRG growth cones.
Inhibition of ROCK Reduces Sema3A-induced Growth Cone Collapse
RhoA GTPase and its effector, ROCK, have been implicated in cellular contractility (Katoh et al. 2001) and in growth cone collapse and retraction in response to several guidance factors (Kozma et al. 1997; Kranenburg et al. 1999; Nakamura et al. 2000; Wahl et al. 2000). Figure 8 shows immunocytochemical labeling for RhoA in DRG growth cones, including central regions, lamellipodia, and filopodia. We used two inhibitors of ROCK to investigate collapse mechanisms in DRG growth cones, Y27632 and HA1077 (Davies et al. 2000). When added before addition of Sema3A, either ROCK inhibitor significantly reduced the collapse response to Sema3A, and when combined with elevation of NGF, growth cone collapse in response to Sema3A was no different than for growth cones treated with control medium (Figure 9).
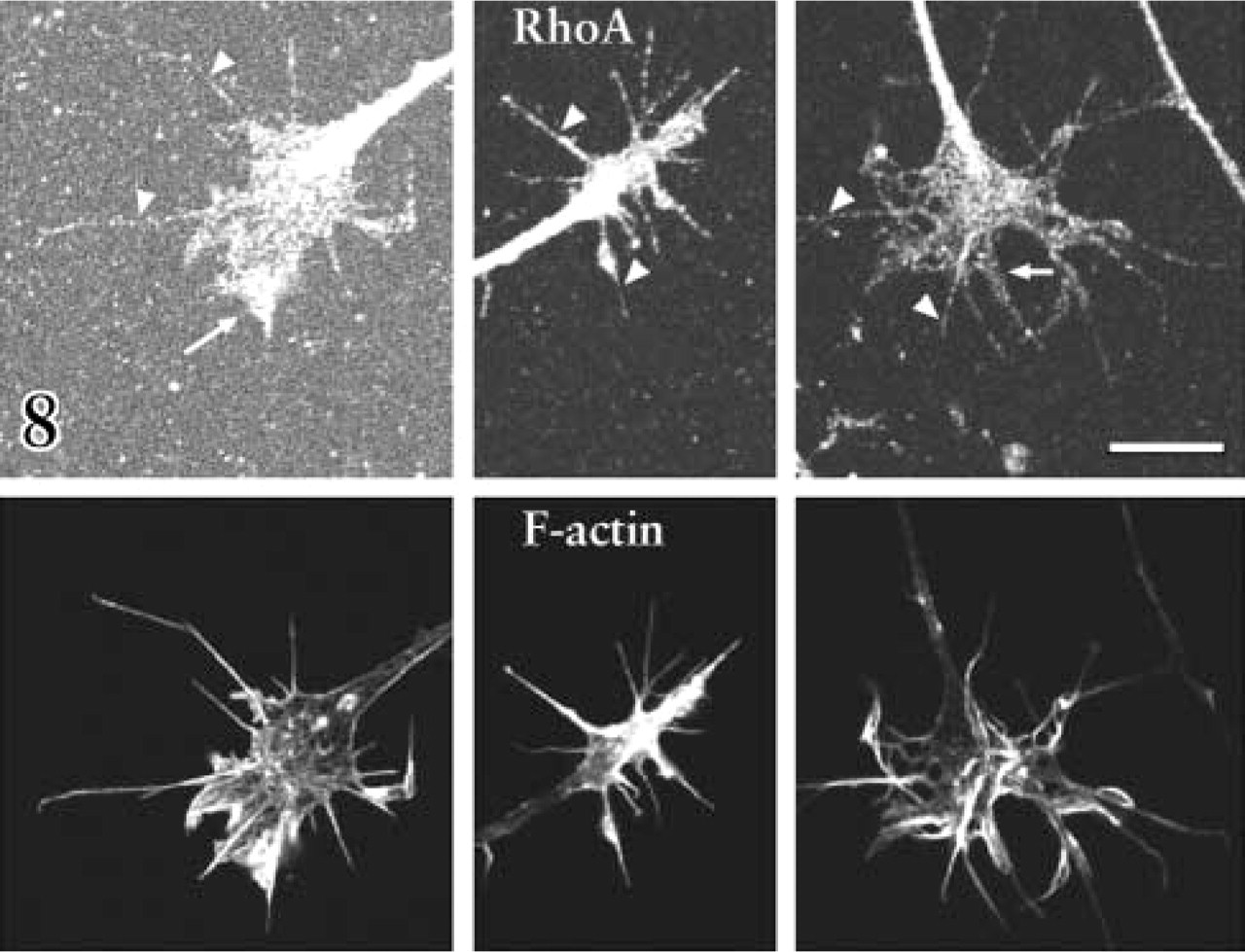
Immunocytochemical double labeling of three DRG growth cones with antibodies against RhoA (upper panels) and with fluorescent phalloidin to label f-actin (lower panels). RhoA is present throughout the growth cones, including filopodia (arrowheads) and lamellipodia (arrows), which are rich in filamentous f-action. Bar = 10 μm.
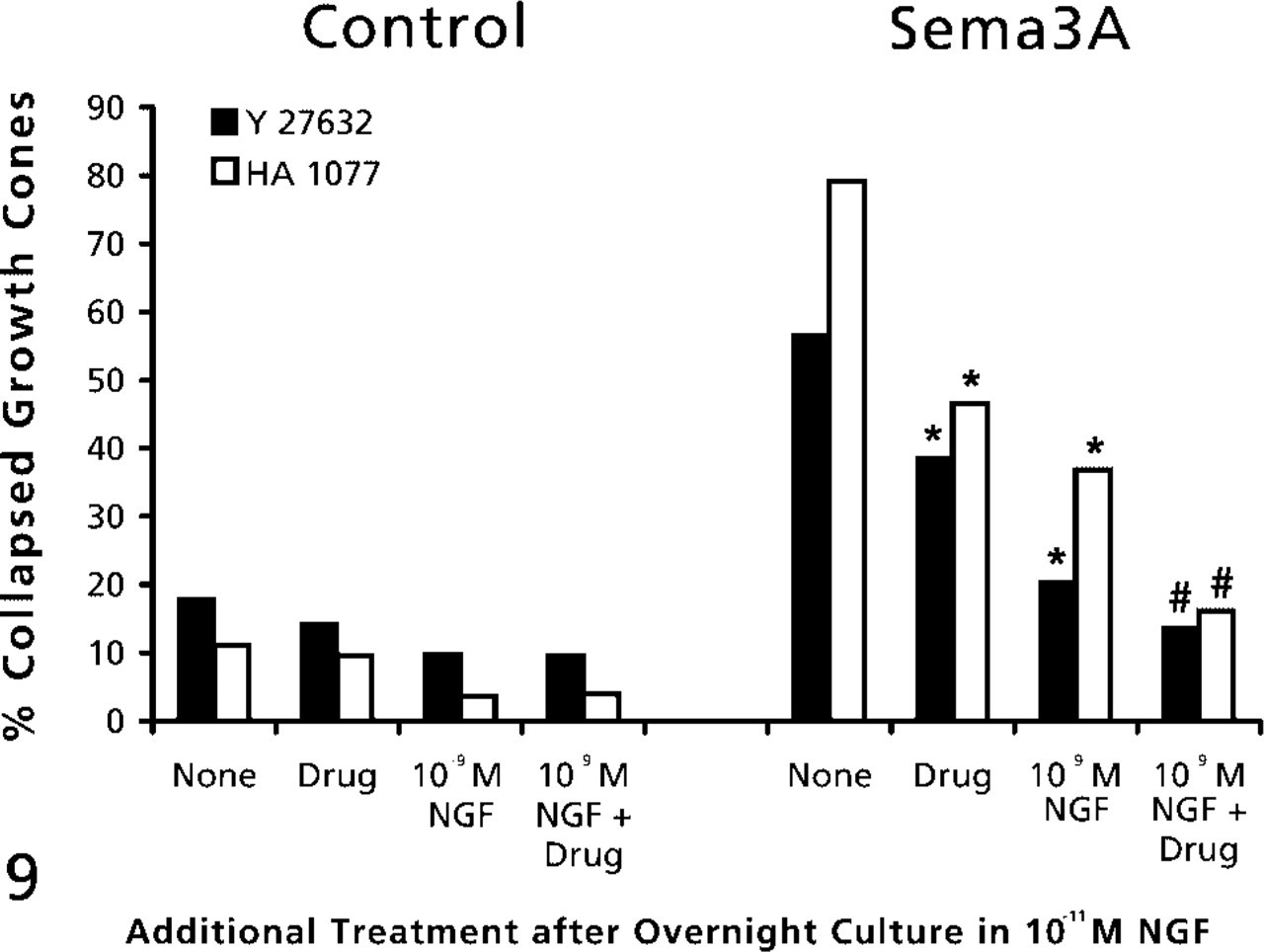
The effects of inhibition of RhoA-activated ROCK and elevated NGF on growth cone response to Sema3A. DRG explants were cultured overnight in medium containing 10-11 M NGF. At 24 hr, NGF in some dishes was elevated to 10-9 M for 60 min. Then 10 μM Y-27632 or 15 μM HA1077 was added to some dishes for another 60 min, then Sema3A or control medium was added for 30 min, followed by fixation. The percent collapsed growth cones in each sample population is presented. The experiments with Y-27632 and HA1077 were conducted at different times, with slightly different levels of collapse in response to Sema3A alone. This accounts for the different height of bars labeled “None.” ∗ p<0.01, significantly different from Sema3A. #p<0.01, significantly different from 10-9 M NGF and Sema3A. From Dontchev and Letourneau (2002), with permission. Copyright 2002 by the Society for Neuroscience.
Discussion
Axon pathfinding is controlled by extracellular proteins that act through ligand-receptor signaling to regulate growth cone behaviors (Muller 1999). Among such proteins, neurotrophins and semaphorins regulate axon navigation and innervation by sensory neurons. Both groups of molecules are present along pathways and in targets of sensory axons (Elkabes et al. 1994; Messersmith et al. 1995; Wright et al. 1995; Giger et al. 1996; Puschel et al. 1996; Shepherd et al. 1996; White et al. 1996; Cahoon-Metzger et al. 2001), suggesting that growth cone behaviors reflect integration of coincident signaling by neurotrophins and semaphorins. This idea provided the rationale for these experiments.
Our studies showed that the collapse response of sensory growth cones to Sema3A was reduced by elevated NGF. This is like our previous finding that BDNF protects retinal growth cones from nitric oxide-induced collapse (Ernst et al. 2000). Growth cones of DRGs raised in 10-10 or 10-9 M NGF collapsed less in response to Sema3A than DRGs raised in 11-11 M NGF. Furthermore, 1-hr exposure to 11-9 M NGF was sufficient to reduce collapse in response to Sema3A. Occupation of trk and p75 neurotrophin receptors would be significantly greater in 11-9 M than in 11-11 M neurotrophin (Lee et al. 2001; Patapoutian and Reichardt 2001), suggesting that increased neurotrophin signaling mediates effects on the response to Sema3A.
Our evidence suggests that NGF triggers signaling that reduces the effectiveness of Sema3A signaling to induce growth cone collapse. Several pathways activated by neurotrophins may modulate Sema3A-induced collapse (Patapoutian and Reichardt 2001), and several studies report that neurotrophins activate cAMP-dependent kinase PKA (Knipper et al. 1993; Cai et al. 1999; Zhang et al. 1999). We also found that PKA activation mediates BDNF protection of retinal growth cones from nitric oxide (Gallo et al. 2002). Our pharmacological results support involvement of PKA activity in NGF-mediated reduction of the collapse response to Sema3A. In view of the protection provided by neurotrophins against growth cone collapse induced by nitric oxide and Sema3A, a common effect of neurotrophins may be to stabilize actin filaments.
Our results indicated that cGMP-activated protein kinase G (PKG) is involved in Sema3A-induced growth cone collapse. Inhibiting PKG or guanylate cyclase reduced the response to Sema3A, whereas activation of PKG or guanylyl cyclase promoted collapse. That elevated NGF did not reduce growth cone collapse induced by PKG activation to the same extent as Sema3A-induced collapse suggests that PKG acts downstream or independently of the influence of NGF signaling. Although cGMP is implicated in growth cone guidance (Song et al. 1998; Song and Poo 1999), it is unclear where cGMP acts. Ion channels, Ca++ release from stores, or myosin contractility may be regulated by cGMP-dependent kinases and affect growth cone motility (Silveira et al. 1998; Vo et al. 1998; Hoffmann 2000). Our results do not strictly fit the model of Song and Poo (1999) for modulation of growth cone guidance by PKA and PKG. However, our findings are consistent with the idea that cyclic nucleotide signaling is important in growth cone responses to guidance cues (Ming et al. 1997; Song et al. 1998).
Rho family GTPases regulate axon morphogenesis and guidance (Gallo and Letourneau 1998; Dickson 2001). In PC12 cells, NGF activates Rac1, localizes Rac1 to the plasma membrane, and inhibits RhoA (Yamaguchi et al. 2001). RhoA is implicated in growth cone collapse stimulated by Sema3A, ephrin-A5, and lysophosphatidic acid (Kozma et al. 1997; Kranenburg et al. 1999; Nakamura et al. 2000; Wahl et al. 2000). Thus, downregulation of RhoA activity by NGF could contribute to reducing the effects of Sema3A. This is supported by our evidence that growth cone collapse in response to Sema3A is reduced by inhibitors of ROCK kinase, a downstream effector of RhoA. Phosphorylation of RhoA by PKA reduces RhoA activity (Dong et al. 1998; Essler et al. 2000; Lang et al. 1996). This modulation of RhoA by PKA may be another site where signals activated by NGF block signaling by Sema3A.
As stated above, these studies investigated the idea that coincident signaling by Sema3A and neurotrophins occurs as DRG growth cones traverse tissues and within their targets. Figure 10 summarizes how interactions between signaling pathways may regulate growth cone behaviors. Several regions of chick and mouse embryos contain high Sema3A levels, the ventral spinal cord and epidermis (chicks) of their targets and the dermamyotome in the peripheral pathway. These regions are not entered by NGF-dependent axons. However, Sema3A is also broadly expressed at low levels in mesenchyme (Giger et al. 1996; Shepherd et al. 1996). It may be that the coincident expression of NGF offsets Sema3A-mediated activation of collapse and allows axons to extend through mesenchyme. In this situation, NGF does not direct growth but maintains growth cone motility. This is supported by evidence that axon extension in peripheral pathways is halted or deficient when neurotrophins are absent or blocked (Patel et al. 2000; Tucker et al. 2001). Thus, a balance between signaling by Sema3A and NGF may contribute to axon growth. When both factors are present at low levels, axons grow in fascicles through tissues. When regions of high Sema3A expression are encountered, sensitive axons do not enter these regions, and when regions of high neurotrophin expression are encountered, such as in a target, other behaviors, such as axon branching, are stimulated. Our experiments emphasize the significance of concentration in determining outcomes of coincident signaling by Sema3A and NGF, but the in vivo concentrations of these molecules are unknown. Many in vitro experiments use these molecules in soluble form. Sema3A and NGF are basic proteins, which may bind negatively charged extracellular matrix components and remain near their release sites. We must learn more about in vivo distributions and actions of Sema3A and neurotrophins.
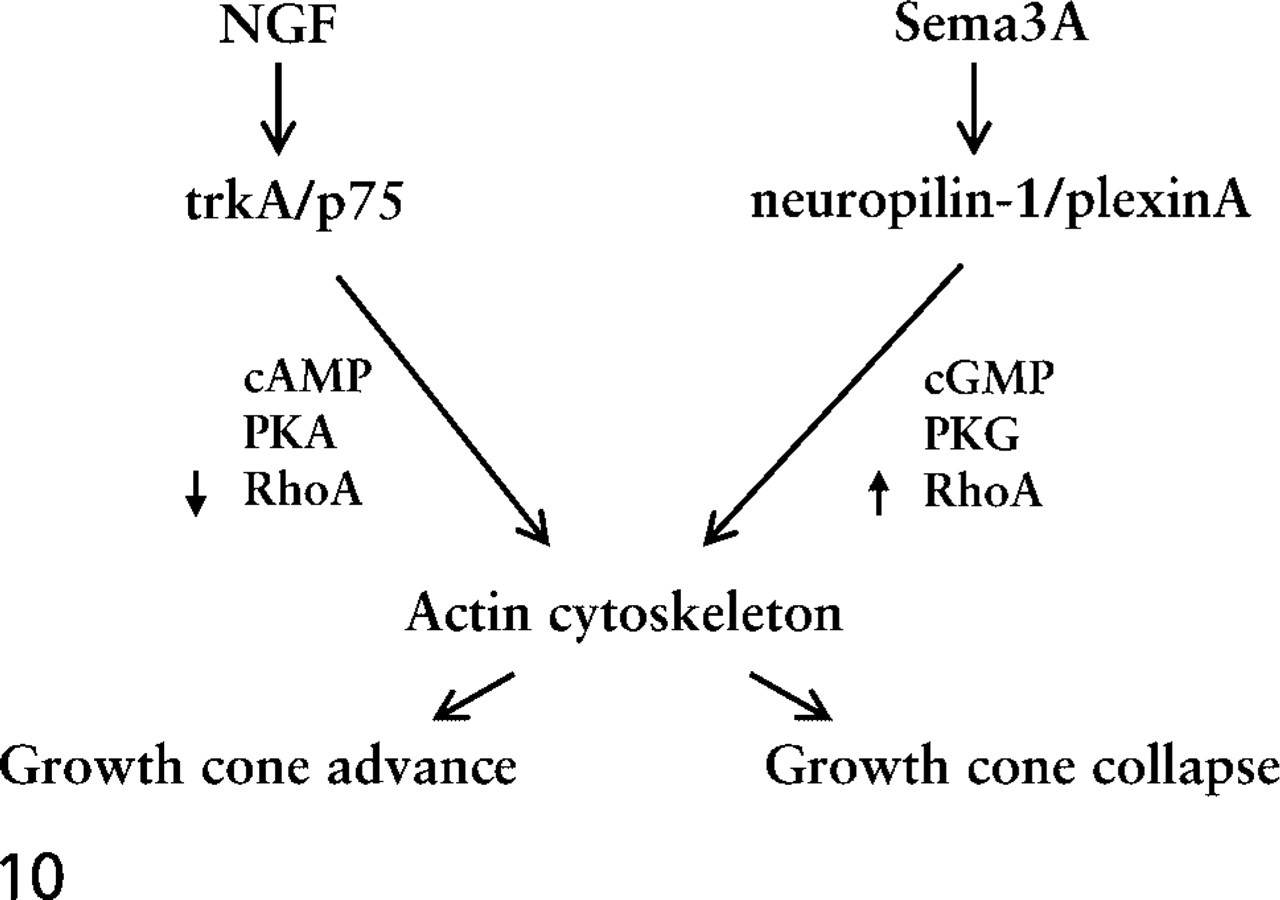
Diagram of signaling from NGF and Sema3A binding to their respective receptors. These signaling interactions modulate the dynamics and organization of the growth cone actin cytoskeleton. The integration of these signaling interactions determines growth cone behaviors.
Footnotes
Acknowledgements
Supported by National Institutes of Health grant HD19950 (PCL), National Science Foundation grant IBN-0080932 (PCL), a grant from the NIH/Fogarty International Center to V.D. Dontchev while in the Letourneau laboratory, and a Minnesota Medical Foundation grant.
Florence Roche and Eric Veien provided valuable technical assistance, and Dr Gianluca Gallo provided valuable comments on the manuscript.
