Abstract
Introduction
Protein kinase A (PKA) and protein kinase G (PKG) are the main downstream effectors of second messengers cAMP and cGMP, which play important roles in physiological and pathological processes. Recently, there are two findings: one is PKA catalytic subunits α (PKACα) and PKG II can be secreted, the other is that the two secretory protein kinases are associated with the progression of tumors. Previous data also demonstrate that the two kinases, as signal cascades, involved in inflammation-associated disorders. However, it remains unclear whether the secreted PKACα or PKG II could serve as diagnostic biomarkers for inflammation-associated disorders.
Methods
The serum from suffered coronary disease, diabetes, rheumatoid arthritis, and schizophrenia were collected. The serum PKACα and PKG II were detected by ELISA. All the patients were consent informed.
Results
Our results showed that the serum PKACα and PKG II had obvious changes in coronary disease, rheumatoid arthritis, schizophrenia patients. However, the trends was opposite, especially in rheumatoid arthritis.
Conclusions
Serum PKACα and PKG II could serve as potentially diagnostic biomarkers for some inflammation-associated disorders, such as coronary disease, rheumatoid arthritis, and schizophrenia.
Keywords
Introduction
Inflammation-associated disorders are characterized by dysregulation of immune response, including a group of common, chronic, and complex disorders, such as type-1 diabetes, hypertension, coronary heart disease, rheumatoid arthritis, inflammatory bowel disease, autoimmunee diseases. Together, inflammation-associated disorders have an estimated incidence of 80 per 100 000 person-years and affect 3–5% of the population.1,2 Chronic diseases result in significant health care and high diseases burden. Protein kinases, especially, cAMP-dependent protein kinase (PKA) and cGMP-dependent protein kinase (PKG), 3 as signal cascades, play key roles in inflammation-associated disorders.3,4 Recently, more and more data indicated the secretion phenomenon of PKACα and used a biomarker for tumor diagnosis.5–7 Our group also noticed that PKACα and PKG II could be detected in the serum of human and mice, as well as in the cell culture supernatant.8,9 Since PKACα and PKG II play an important role in inflammation-associated disorders, it remains unclear whether the secreted PKACα and PKG II can serve as potential biomarkers for these diseases. Therefore, in the present study, we assess the diagnostic effects of PKACα and PKG II for some of inflammation-associated disorders, such as, coronary disease, diabetes, rheumatoid arthritis, and schizophrenia. Our results showed that the PKACα and PKG II in the serum could be used for screening, diagnosis of coronary disease, rheumatoid arthritis, and schizophrenia.
Materials and Methods
Serum samples
This is a basic research to explore the potential diagnostic biomarkers for some inflammation-associated disorders. The patients suffered from diabetes (n = 42, with recent-onset diabetes diagnosed during 2 years), coronary (n = 31, diagnosis based on previous coronary angiograms), rheumatoid arthritis (n = 18, diagnosed according to 2010 ACR/EULAR classification criteria), schizophrenia (n = 30) and depression disorders (n = 30) (diagnosis based on Diagnostic and Statistical Manual of Mental Disorders fourth version (DSM-IV), never treated with antipsychotics and normal body weight) were included. Twenty volunteers were included as control. All the selected patients were from the Affiliated Hospital of Jiangsu University, Taizhou People’s Hospital of China, and Zhenjiang Fifth People’s Hospital (Zhenjiang Mental Health Center), did not combine other diseases nor supplemented drugs that affect the test, and aged from 40 to 75 years old. All the patients were consent informed. The number of patients determines the cases’ number from 2018 to 2020. The study involving human subjects was approved by the Medical Ethics Committee of Jiangsu University and Taizhou People’s Hospital (KY202001601), respectively. The serum was obtained from the above patients or volunteers, centrifugated at 2000 r/min for 20 min at 4°C and stored at −80°C until use.
ELISA assay
The serum samples were detected by ELISA according to ELISA kit manufacturers’ instructions obtained from Cloud-Clone-Corp (FANKEW) (Katy, TX, USA) and read at 450 nm immediately. The data were from three independent experiments and were expressed as mean ± SD.
Statistical Analysis
All data analyses were conducted with SPSS 19.0 software (IBM, Chicago, IL, USA) and GraphPad Prism 8.0, and p < .05 was considered statistically significant. The relationship between PKACα, PKG II and various diseases was analyzed by Spearman rank correlation, respectively. Two groups were compared by t-test. Three or more groups were analyzed by one-way ANOVA, followed by the Bonferroni post hoc test. ROC curve analyses for PKACα and PKG II were used to determine the optimal cutoff values of diabetes, coronary disease, rheumatoid arthritis, schizophrenia, and depression disorder, and the AUC values were used to compare the predictive potential of the prognosticators.
Results
The levels of PKACα and PKG II were significantly changed in patients
To clarify the differences in patients suffering from coronary disease, rheumatoid arthritis, schizophrenia and depression, and volunteers, the serum PKACα and PKG II levels were analyzed. The results showed that PKACα was significantly decreased in the serum of coronary disease, rheumatoid arthritis, and schizophrenia, compared to volunteers (*p < .05, *p < .05, ** p < .01, respectively). Conversely, PKG II was increased in the serum of rheumatoid arthritis, schizophrenia, and depression disorders compared to their volunteers (**p < .01, ***p < .001, *p < .05, respectively). There was no difference in PKACα in patients with diabetes and depression disorders compared to corresponding volunteers (p > .05). Besides, for PKG II, there were no obvious changes between diabetes and volunteers, as well as coronary disease and volunteers (p > .05) (Figure 1(a) and (b)). The levels of PKACα and PKG II in the serum of different patients with inflammation-associated disorders. (a) The level of PKACα in human sera from Control healthy subjects (n = 20, 131.4 ± 37.49), patients with Diabetes (n = 42, 118.9 ± 23.45) or Coronary disease (n = 31, 111.9 ± 21.34), Rheumatoid arthritis (n = 18, 107.4 ± 8.466), Schizophrenia (n = 30, 104.6 ± 15.81), and Depression disorder (n = 30, 130.1 ± 37.21) were analyzed by the ELISA arrays. (b) The level of PKG II in human sera from Control healthy subjects (n = 20, 121.3 ± 55.79), patients with Diabetes (n = 42, 100.5 ± 21.24) or Coronary disease (n = 31, 124.2 ± 42.76), Rheumatoid arthritis (n = 18, 173.1 ± 22.19), Schizophrenia (n = 30, 171.0 ± 67.48), and Depression disorder (n = 30, 154.8 ± 32.63) were analyzed by the ELISA arrays. The levels represent the mean ± Std. Deviation (M ± SD) of PKACα or PKG II level. *p < .05, ** p < .01, ***p < .001, versus Control; ns, not significant. NC group, normal saline-treated group.
The correlation analysis of serum PKACα and PKG II with various biomarkers in some inflammation-associated disorders
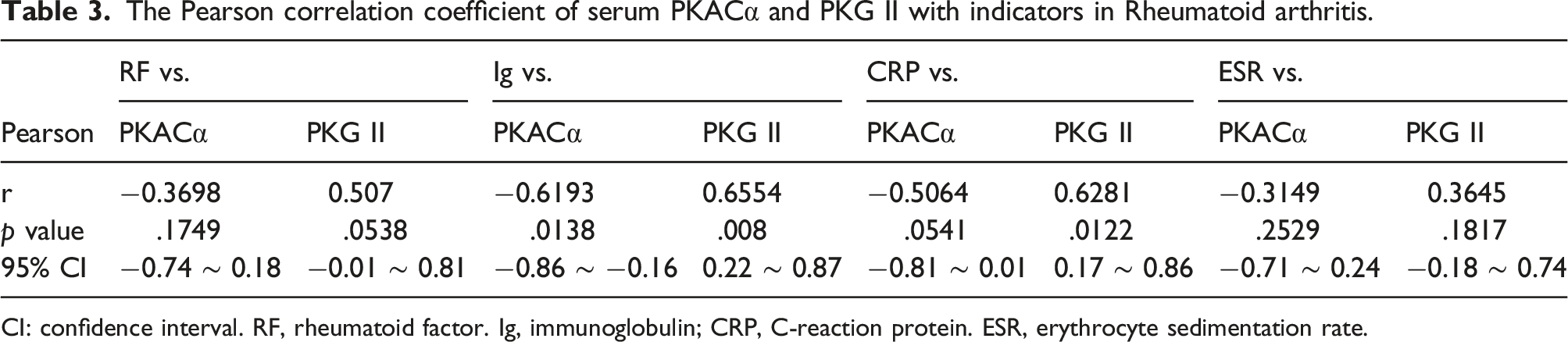
The Pearson correlation analysis was used to evaluate the relationship between serum PKACα/PKG II with varous biochemical biomarkers in different inflammation-associated disorders. In diabetes, there was a significant positive correlation between creatinine and PKACα (r = 0.3996, p = .0478), creatinine and PKG II (r = 0.4417, p = .0271), while no obvious relationship between PKACα or PKG II with glucose, glycosylated hemoglobin (HBA1C) %, and urea nitrogen. For coronary disease, there was a significant negative correlation between cholesterol (CHOL) and PKACα (r = −0.3907, p = .0328), triglyceride (TG), and PKACα (r = −0.3951, p = .0307). In rheumatoid arthritis, PKACα and Ig, RF, CRP, ESR were negatively correlation (r = −0.6193, p = .008; r = −0.3698, p > .05; r = −0.5064, p > .05; r = −0.3149, p > .05), while PKG II and Ig, RF, CRP, ESR were positively correlation (r = 0.6554, p = .0328; r = 0.507, p > .05; r = 0.6281, p > .05; r = 0.3645, p > .05). However, there is an opposite trends of PKACα and PKG II in rheumatoid arthritis (Figure 2 and Tables 1, 2, and 3) The Pearson correlation analysis of serum PKACα and PKG II with indicators in different inflammation-associated disorders. (a) Correlation plot between PKACα or PKG II with Glucose, HBA1C%, Creatinine, and Urea nitrogen in diabetes. (b) Correlation plot between PKACα or PKG II with CK-MB, CHOL, and triglyceride in Coronary Disease. (c) Correlation plot between PKACα or PKG II with CRP, RF, Ig, and ESR in Rheumatoid arthritis. HBA1C, glycosylated hemoglobin; CK-MB, creatinine kinase, MB isoenzyme; CHOL, cholesterin; TG, triglyceride; CRP, C reactive protein; RF, rheumatoid factors; Ig, immunoglobulin; ESR, erythrocyte sedimentation rate. The Pearson correlation coefficient of serum PKACα and PKG II with indicators in Diabetes. CI: confidence interval. HBA1C, glycosylated hemoglobin. The Pearson correlation coefficient of serum PKACα and PKG II with indicators in Coronary disease. CI: confidence interval. CK-MB, creatinine kinase, MB isoenzyme; CHOL, cholesterol; TG, triglyceride. The Pearson correlation coefficient of serum PKACα and PKG II with indicators in Rheumatoid arthritis. CI: confidence interval. RF, rheumatoid factor. Ig, immunoglobulin; CRP, C-reaction protein. ESR, erythrocyte sedimentation rate.


The diagnostic potential of serum PKACα and PKG II in different inflammation-associated disorders
The optimal cutoff value of PKACα and PKG II for the different inflammation-associated disorders was selected using ROC curve analysis. As the results showed that, for coronary disease, the sensitivity and specificity of PKACα were 96.55% and 50%, respectively, the cutoff value was 134.1 pg/mL. For rheumatoid arthritis, the sensitivity and specificity of PKACα were 100% and 55% with a cutoff value of 126 pg/mL, and for PKG II was 100% and 65% with a cutoff value of 131 pg/mL. For schizophrenia, the sensitivity and specificity of PKACα were 93.1% and 60% with a cutoff value of 123 pg/mL, and for PKG II was 76.67% and 75% with a cutoff value of 137.4 pg/mL. Besides, the sensitivity and specificity of PKG II were 86.21% and 65% for depression with a cutoff value of 130.5 pg/mL. All the data indicated that PKACα and PKG II have the potential diagnostic value for the above diseases (Figure 3 and Table 4) The ROC analysis curve of PKACα and PKG II of serum in various diseases. The ROC plots were from Control healthy subjects (n = 20), patients with Coronary disease (n = 31; a), Rheumatoid arthritis (n = 18, b), Schizophrenia (n = 30, c), and Depression disorder (n = 30, d). ROC, Receiver operating characteristic. The ROC analysis of serum PKACα and PKG II in various diseases. CI: confidence interval; AUC: Area under the ROC curve; SE: Std. Error.

Discussion
Inflammation-associated disorders include rheumatoid arthritis, inflammatory bowel disease, asthma, multiple sclerosis, connective tissue disorders, etc. Furthermore, inflammation-associated disorders are often accompanied by different co-morbidities, such as cardiovascular disease, metabolic and bone disorders, neuropsychiatric disease, and cognitive deficit, which further influences the quality of life. There are no effective measures to cure these diseases.10–12 Therefore, it is crucial to detect and control the progression of the disease timely. However, because of the pathogenic complexity of inflammation-associated disorders, there are no broad-spectrum diagnostic markers.
PKACα and PKG II are typically considered as classical intracellular molecules and play important roles in different pathological and physiological processes. PKA contains catalytic subunits and regulatory subunits; there are three isoforms of the catalytic subunits (PKA-C: Cα, Cβ, Cγ) and four isoforms of the regulatory subunits (PKA-R: RIα, RIβ, RIIα, RIIβ). In the absence of cAMP, PKA is an inactive heterotetramer with two identical catalytic and regulatory subunits. Whereas, in the presence of cAMP, R subunits binds to cAMP, and the C subunits are released from holoenzyme, phosphorylating target substrates, 13 including ion channels, enzymes, and transcription factors, and regulates their activity. PKA plays multiple roles in heart functions, including contraction, metabolism, ion fluxes, gene transcription,13,14 neuronal development, synaptic plasticity, brain homeostasis, as well as in the regulation of feeding, energy expenditure, and glucose homeostasis.15,16 Furthermore, dysregulation of PKA signaling contributes to the progression of several neuropsychiatric and neurodegenerative diseases and inflammation-associated disorders. 17 Conversely, PKG is encoded by two genes, prkgI and prkgII, which are transcribed into three enzymes PKGIα, PKGIβ, and PKG II. The PKG I isozymes are prominent in all types of smooth muscle, platelets, and specific neuronal areas. 18 As a membrane-bound protein, PKG II is mainly located in the secretory epithelium of the small intestine, brain, kidney, chondrocytes, prostate, gallbladder, pancreas, and lung.19–22 PKG II is composed of three functionally distinct domains 23 : an N-terminal domain, a regulatory domain, and a catalytic domain, and the physiologically most relevant activator of PKG II is cGMP. 21 PKG II regulates the activity of processes in cells by phosphorylating specific target proteins on serine or threonine residues. 21 PKG II is known to be linked to fluid and salt balance in epithelial tissues, 24 intestinal fluid secretion, 25 chondrogenesis, renin secretion, and memory formation, cytoskeleton regulation, proliferation, and apoptosis in tumor cells.26,27 However, an extracellular form of PKA and PKG II has been found in the conditioned medium and serum from cancer patients, including prostate, renal, bladder, breast, and lung carcinomas, and can be considered as a diagnosis biomarker.6,7,28–30
The present study demonstrated that the serum PKACα and PKG II had obvious changes in coronary disease, rheumatoid arthritis, schizophrenia patients. However, serum PKACα and PKG II have opposite changes tendency, especially in rheumatoid arthritis. It seems that PKACα and PKG II can serve as potentially diagnostic markers for inflammatory associated disorders. However, our research still has some limitations, such as, the sample number not enough, the defect of comparison between the classical clinical diagnostic biomarkers and PKACα and PKG II, lack of the relationship between serum PKACα and PKG II levels and disease progression. However, our results at least indicated that the secreted PKA and PKG are potential diagnostic biomarkers for inflammation-associated disorders. Of course, to further confirm the diagnostic value of PKACα and PKG II, more clinical studies are needed.
Conclusions
Our results showed that the serum PKACα and PKG II could serve as diagnostic biomarkers for coronary disease, rheumatoid arthritis, and schizophrenia. Of course, further researches are also needed to approve the conclusions (Figure 4). The schematic image of PKACα and PKG II in different diseases. PKACα and PKG II could be secreted to the blood from the cells and regulate several immune-mediated inflammatory diseases, such as diabetes, coronary disease, rheumatoid arthritis, schizophrenia, and depression disorder. And whether extracellular PKACα and PKG II is friend or foe is also need to be investigated.
Footnotes
Acknowledgements
We thanks Dr Javaid Akhter Bhat for editing the revision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health and Health Commission of Jiangsu Province (LGY2018025); 333 Project Funding Plan of Jiangsu Province (BRA2019172).
Ethical statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Informed consent
Written informed consent was obtained from all subjects before the study.
