Abstract
Our previous studies on an inhibitor of O-glycosylation of glycoproteins, Gal-NAcα-O-bn, in the model of enterocytic HT-29 cells, have shown at the cellular level an alteration of the normal localization of apical glycoproteins, and at the biochemical level an in situ synthesis and storage of sialylated GalNAcα-O-bn oligosaccharides. The purpose of this study was to examine if a relation existed between these two events, using different cell lines. Intracellular storage of GalNAcα-O-bn metabolites occurred in HT-29 and CAPAN-1 cells but not in Caco-2 cells. On the other hand, an accumulation of endosomal/lysosomal compartments was observed in HT-29 and CAPAN-1 cells but not in Caco-2 cells. These data focused on a GalNAcα-O-bn-derived storage phenotype in HT-29 and CAPAN-1 cells. The apical membrane glycoproteins MUC1 and CEA showed an abnormal localization inside intracytoplasmic vesicles in HT-29 cells, whereas they kept their normal localization in Caco-2 and CAPAN-1 cells. Studies on the glycosylation of these apical glycoproteins showed that GalNAcα-O-bn inhibited the glycosylation in a cell-specific manner. The alteration in the apical targeting of glycoproteins, and the appearance of a GalNAcα-O-bn-derived storage phenotype are two independent and cell type-specific events. The former depends on the inhibition pattern of the glycosylation of endogenous glycoproteins, whereas the latter is connected to the intracellular accumulation of GalNAcα-O-bn metabolites.
T
During recent years, several studies have shown that treatment of cell lines with GalNAcα-O-bn induced different types of alterations at the cellular level. First of all, our work on HT-29 cells showed that long-term GalNAcα-O-bn treatment of differentiated HT-29 cells, of goblet or enterocytic phenotype, induced alterations in intracellular trafficking of apically secreted and/or apical membrane glycoproteins. We reported an inhibition of mucin secretion and a defect in the apical localization of membrane brush border glycoproteins such as dipeptidylpeptidase IV, carcinoembryonic antigen (CEA), and the mucin-like glycoprotein MUC1 (Hennebicq-Reig et al. 1998; Huet et al. 1998). Furthermore, we also observed that the cells displayed a heterogeneous pattern of different intracytoplasmic vesicles, some of which contained the mistargeted glycoproteins. All these data suggested a putative role of O-linked oligosaccharides in the regulation of intracellular trafficking in polarized intestinal cells. Such a hypothesis was also supported by several other studies on other cell types (Monlauzeur et al. 1998; Alfalah et al. 1999; Benting et al. 1999; Ait-Slimane et al. 2000; Jacob et al. 2000).
However, long-term GalNAcα-O-bn treatment of Caco-2 cells did not induce such a cytoplasmic accumulation of brush border glycoproteins as in HT-29 cells, suggesting a relative cell type-specific effect of GalNAcα-O-bn (Gouyer et al. 2001). No mechanism could yet be given for the origin of this cell-specific behavior. However our previous work demonstrated that the intracellular fate of GalNAcα-O-bn could be widely different according to the cell type. Enterocyte-like HT-29 cells, but not Caco-2 cells, are able to generate considerable amounts of a series of sialylated GalNAcα-O-bn-derived oligosaccharides (Zanetta et al. 2000). In this line, Ulloa et al. (2000) then reported that long-term GalNAcα-O-bn treatment of mucinsecreting cells induced alterations in the maturation of the endosomal/lysosomal system that were reminiscent of sialic acid storage disease (SASD).
Altogether, these data raised the question of a potential connection between the intracellular metabolization of GalNAcα-O-bn by the cell-specific glycosyltransferases and the morphological and/or functional alterations observed in the cell. To address this question, we have undertaken a study of these metabolic and cellular aspects in a series of cell lines that display polarity when grown in culture.
Materials and Methods
Materials
GalNAcα-O-bn was from Sigma Chemical (St Quentin Fallavier, France). Mouse MAbs AC17 against LAMP1 and 517 against CEA, 4B10 against ST3 Gal I (Vallejo-Ruiz et al. 2001), and 214D4 against MUC1 were a gift from Dr. A. Le Bivic (IBDM; Marseille, France), Pr. H. Clausen (School of Dentistry, Copenhagen, Denmark), and Dr. J. Hilkens (Département de Biologie Tumorale, Amsterdam, Netherlands), respectively. Digoxigenin- or alkaline phosphatase-labeled Maackia amurensis agglutinin (MAA) (Wang and Cummings 1988), peanut (Arachis hypogaea) agglutinin (PNA) (Lotan et al. 1975), and Erythrina cristagalli (ECA) (Iglesias et al 1982), which recognize the oligosaccharide species Neu5Acα2–3Gal-R, Galβ1–3GalNAc-R, and Galβ14GlcNAc, respectively, were from Roche Diagnostics; Meylan, France.
Cell Culture
Cell maintenance and all experiments were done in 25-cm2 T flasks (Corning Glass Works; Corning, NY). Cells were seeded at 2 × 104 cells/cm2. For maintenance purposes, cells were passaged weekly, using 0.025% trypsin, 0.53 mM EDTA in Ca2+/Mg2+-free PBS (PBS-). Enterocyte-like HT-29 cells (enterocyte-like HT-29 G− subpopulation derived from the parental cell line by culture in a glucose-free medium (Zweibaum et al. 1985) and an HT-29 5M12 clone, isolated from a HT-29 MTX subpopulation selected by adaptation to 10−5 M methotrexate (Lesuffleur et al. 1998), were grown in standard Dulbecco's modified Eagle's minimal essential medium (Eurobio; Paris, France), supplemented with 10% (v/v) heat-inactivated fetal calf serum (FCS). Cells were cultured at 37C in a 10% CO2/90% air atmosphere. Cells of the pancreatic cancer cell line CAPAN-1 (American Type Culture Collection; Manassas, VA) were grown in RPMI 1640 with 2 mM glutamine supplemented with 15% heat-inactivated FCS. These cells were propagated in a humidified atmosphere of 5% CO2/95% air at 37C. Caco-2 cells were cultured as previously reported (Pinto et al. 1983). GalNAcα-O-bn was added at different concentrations ranging from 2–10 mM in the culture medium. For long-term treatment, cells were treated from day 2 after seeding up to day 14, and for short-term treatment (kinetic study), cells were cultured in standard conditions up to day 5 and then in the presence of GalNAcα-O-bn from 3 to 44 hr. The medium was changed daily.
Analysis of GalNAcα-O-bn Derivatives
Control and GalNAcα-O-bn-treated cells were lyophilized and submitted to a Folch extraction and partition as previously described (Zanetta et al. 2000). Extracts were then submitted to a methanolysis and the monosaccharide composition was determined by gas chromatography/mass spectrometry (GC/MS). Monosaccharides were analyzed in a single step using the electron impact detection mode and quantified as the heptafluorobutyrate derivatives of the O-methyl glycosides obtained after anhydrous acid methanolysis in the presence of lysine as an internal standard (Zanetta et al. 1999). Evaluation of monosaccharides linked to GalNAcα-O-bn was obtained by subtraction of the monosaccharide amounts in GalNAcα-O-bn-treated cells and control cells. Results were expressed in nmol of monosaccharide per mg of cell protein.
Immunoprecipitation and Western Blotting with Lectins
For immunoprecipitation of MUC1 or of CEA, cells were lysed in hot buffered 5 mM Tris-HCl, pH 8.2, containing 0.5% SDS, 5 mM EDTA, 150 mM NaCl, 100 μM AEBSF, then heated at 100C for 5 min, sonicated, and centrifuged at 12,000 × g for 10 min. The supernatants were adjusted to 2.5% Triton X-100 and incubated with the antibody (MAbs 214D4 and 517, respectively) overnight at 4C. Immune complexes were collected on protein G-Sepharose 4B (Sigma), eluted in the SDS sample buffer (0.2 M Tris-HCl buffer, pH 6.8, containing 2% SDS and 30% glycerol) at 60C for 5 min, and then analyzed on 5–30% polyacrylamide gels (Laemmli 1970). For detection of MUC1 and CEA by Western blotting, the proteins were transferred to a hybond C extra membrane (Amersham; Aylesbury, UK), the membrane was then treated with the anti-MUC1 (MAb 214D4) or anti-CEA (MAb 517) antibody and then with peroxidaseconjugated anti-mouse antibody (Sigma) diluted 4000-fold. Detection was carried out by luminescence using the ECL Western blotting system (Amersham). For lectin blotting, glycoproteins were transferred to a nitrocellulose membrane (BioTrace NT; Gelman Sciences, Ann Arbor, MI) as described in Vaessen et al. (1981). The membranes were treated for 2 hr with 2% polyvinylpyrrolidone K 30 in 10 mM Tris-HCl, pH 7.4, containing 0.15 M NaCl (TBS). Membranes were incubated with digoxigenin-labeled (MAA) or phosphatase-labeled lectins (ECA, PNA) at a concentration of 5 μg/ml in TBS. After a previous incubation with alkaline phosphatase-labeled anti-digoxigenin Fab fragment (1 μg/ml in TBS) for MAA blotting, the labeled glycoproteins were revealed by 4-nitroblue tetrazodium chloride/5-bromo-4-chloro-3-indolyl phosphate staining.
Confocal Microscopy
For immunofluorescence and confocal microscopy, cells were grown on 8-chamber coverslips. Cells were fixed with 4% paraformaldehyde for 20 min, quenched with 50 mM NH4Cl for 30 min, then permeabilized with 0.2% saponin or 0.05% saponin (for HT-29 and CAPAN-1 cells, respectively) for 20 min, and a saturation step was done for 30 min in PBS/0.2% saponin/1% BSA or PBS/0.05% saponin/1% BSA, respectively. To detect CEA, LAMP1, MUC1, and ST3Gal I, respectively, 517 (1:500), AC17 (1:400), 214D4 (1:3), or 4B10 (undiluted) in PBS containing 1% BSA/0.2% saponin for HT-29 or 1% BSA/0.05% saponin for CAPAN-1, was added overnight. Secondary FITC-conjugated goat antimouse antibody (1:150) was incubated for 1 hr and 30 min.
Confocal microscopy was carried out using a Leica instrument (Model TCS-NT). Serial sections of 0.5 μm were performed in z projection. Three xy sections at the top, middle, and bottom of the cell layers and one xz section are shown for each experiment.
Transmission Electron Microscopy and Ultrastructural Immunochemistry
For classical transmission electron microscopy, the cells were fixed in 25-cm2 T flasks for 30 min at RT with 2% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4). The monolayers were then washed with phosphate buffer and postfixed with 1% osmium tetroxide for 1 hr before dehydration with a graded series of ethanol and hydropropyl methacrylate. The monolayers were recovered with Epon and heated at 37C for 12 hr and at 60C for 3 days. The 25-cm2 T flasks were cut and the monolayers re-embedded in Epon to make sections perpendicular to the bottom of the flask. Ultrathin sections were stained with uranyl acetate and lead citrate and examined under an electron microscope (Léo 906).
For ultrastructural immunochemistry using anti-MUC1 (undiluted) and anti-CEA (1:50) MAbs, the cells were fixed in phosphate buffer containing 4% paraformaldehyde, 0.2% glutaraldehyde for 10 min. The cell layers were scraped with a rubber policeman, the cell pellets were infiltrated with phosphate buffer containing 2.3 M sucrose and 20% polyvinylpyrolidone for 20 hr, and then frozen in liquid nitrogen. Ultrathin cryosections were successively incubated with PBS containing 10% fetal bovine serum (30 min), primary antibodies (1 hr), rabbit anti-mouse antibody (1:10, 1 h) followed by gold-conjugated protein A. All antibodies were diluted in PBS containing 10% fetal bovine serum. The grids were finally counterstained with methylcellulose uranyl acetate and observed with an electron microscope (Léo 906).
Results
After a first examination of several cell types, we selected for this study enterocyte-like cells derived from two cell lines [i.e., HT-29 G− subpopulation and HT-29 5M12 clone (Zweibaum et al. 1985; Lesuffleur et al. 1998), and the colon carcinoma cell line Caco-2 which spontaneously differentiates into enterocyte-like cells (Pinto et al. 1983)], and duct-like pancreatic cells [i.e., the CAPAN-1 cell line (Fanjul and Hollande 1993)].
Evaluation of GalNAcα-O-bn Derivatives
We first evaluated the amount of stored GalNAcα-O-bn derivatives after treatment of the three cell lines by GalNAcα-O-bn at the concentration used in previous work, i.e., 2 mM. We have isolated the GalNAcα-O-bn derivatives from the three different cell lines by Folch extraction and determined the monosaccharide composition in the presence of an internal standard (Table 1).
An important amount of different monosaccharides, i.e., galactose (Gal), N-acetylglucosamine (GlcNAc), N-acetylgalactosamine (GalNAc), fucose (Fuc), and sialic acid (Neu5Ac), was found in HT-29 cells. No complex GalNAcα-O-bn derived oligosaccharides could be detected in Caco-2 cells. GalNAcα-O-bn derivatives were present in CAPAN-1 cells, but in a much lower amount than in HT-29 cells, and Neu5Ac was detected in trace amounts.
For the two cell lines able to store GalNAcα-O-bn derivatives, we analyzed their dose-response to GalNAcα-O-bn. According to the previous results at the concentration of 2 mM, GalNAcα-O-bn was used at the concentration of 5 mM for HT-29 cells and at the concentrations of 5 mM and 10 mM for CAPAN-1 cells, respectively. The results especially showed that the higher concentrations of GalNAcα-O-bn resulted in a large increase in GalNAcα-O-bn derivatives stored in CAPAN-1 cells. At the concentration of 10 mM, CAPAN-1 cells accumulated GalNAcα-O-bn derivatives at a similar level as that obtained in HT-29 cells at the concentration of 2 mM. At the higher concentrations, Neu5Ac became clearly detected in GalNAcα-O-bn derivatives, but in a lower relative amount than in HT-29 cells.
Monosaccharide composition of GalNAcα-O-bn derivatives in the three treated cell lines a
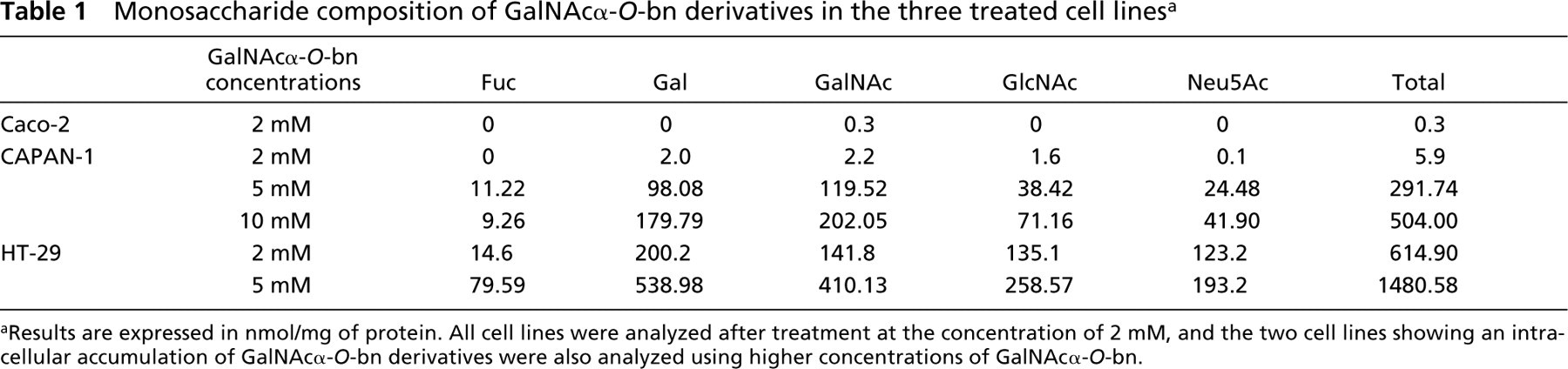
aResults are expressed in nmol/mg of protein. All cell lines were analyzed after treatment at the concentration of 2 mM, and the two cell lines showing an intracellular accumulation of GalNAcα-O-bn derivatives were also analyzed using higher concentrations of GalNAcα-O-bn.
Cell Morphology
We first analyzed the morphology of the three cell types in control conditions and after treatment by GalNAcα-O-bn at the concentration of 2 mM, using transmission electron microscopy (Figure 1). At postconfluence (day 14), HT-29 and Caco-2 control cells are polarized cells of enterocytic phenotype, with the brush border of microvilli clearly apparent. CAPAN-1 control cells had scanty cytoplasm and displayed some villi at the cell surface. The ultrastructural morphology of GalNAcα-O-bn-treated Caco-2 cells was almost similar to the control cells, with only a few cytoplasmic vesicles appearing under the cell surface. In contrast, the treated HT-29 and CAPAN-1 cells differed from the control cells by having a larger amount of cytoplasm filled with intracytoplasmic vesicles and late endosomes/lysosomes of important size. The morphological abnormalities were found at a higher level in HT-29 cells than in CAPAN-1 cells. We then analyzed the ultrastructural morphology of CAPAN-1 and HT-29 cells at the higher concentrations of GalNAcα-O-bn (data illustrated in Figure 1 for CAPAN-1 cells at the concentrations of 5 and 10 mM). CAPAN-1 cells treated by higher concentrations of GalNAcα-O-bn showed a similar phenotype as that observed in HT-29 cells treated with the lowest concentration of 2 mM GalNAcα-O-bn, with a cytoplasm filled with large endosomes/lysosomes.
Expression of Markers for TGN and of Endosomes/Lysosomes
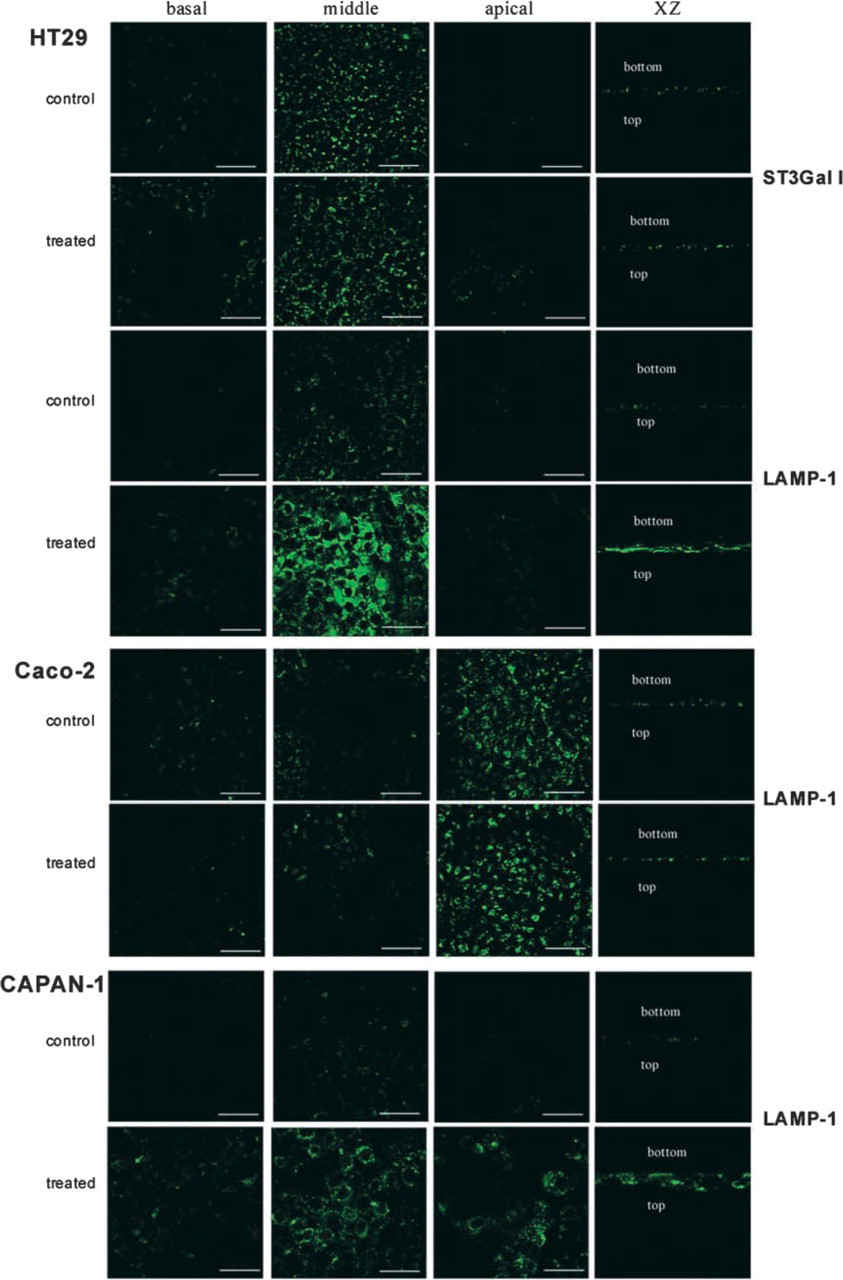
We analyzed the cellular distribution of the glycosyl-transferase ST3Gal I, a marker for TGN, and of the lysosomal protein LAMP-1 (Figure 2).
ST3Gal I labeling was localized in large cisternae corresponding to TGN in HT-29 cells and was unchanged under GalNAcα-O-bn treatment. This result showed that the integrity of the Golgi apparatus was preserved, thus indicating that the intracytoplasmic vesicles observed in these cells did not result from a dislocation of the Golgi apparatus. ST3Gal I was not detected in the two other cell lines (data not shown).
Using the antibody against LAMP-1, we observed a perinuclear labeling in HT-29 and CAPAN-1 cells. Under GalNAcα-O-bn treatment, a strong increase in LAMP-1 labeling was visualized in these two cell lines, with a broader distribution all over the cytoplasm. In control Caco-2 cells, the expression of LAMP-1 was strong and was also localized at the apical side. No effect of GalNAcα-O-bn treatment was observed on the cellular distribution of this marker in these cells.
Glycosylation of Apical Glycoproteins
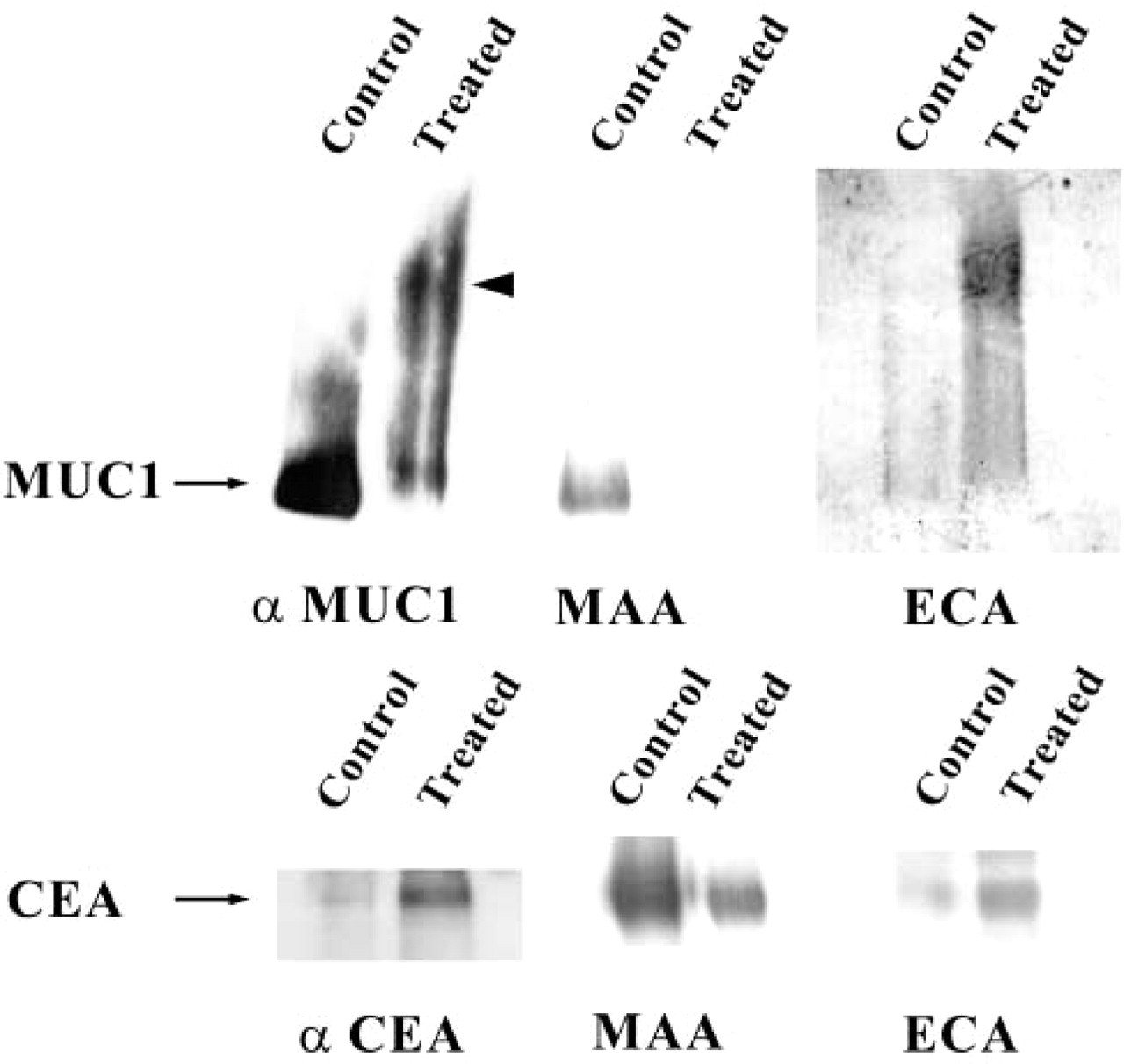
In HT-29 cells, we have previously shown that GalNAcα-O-bn treatment inhibited the α2–3 sialylation by ST3Gal I and ST3Gal IV on apically targeted glycoproteins (Huet et al. 1998; Gouyer et al. 2001). ST3Gal I is highly expressed in HT-29 cells and substitutes T-antigen (Galβ1–3GalNAc) into the sialyl T motif Neu5Acα2–3Galβ1–3GalNAcα. The inhibition of ST3Gal I thus induced the abnormal expression of T-antigen. In Caco-2 cells, in contrast, the sialylation of glycoproteins that occurred mainly in the α2–6 linkage was not affected by GalNAcα-O-bn treatment. We have thus analyzed in CAPAN-1 cells the effect of GalNAcα-O-bn on the glycosylation of two brush border glycoproteins, MUC1 and CEA (Figure 3). Both glycoproteins were labeled by MAA lectin in control CAPAN-1 cells, showing their substitution by α2–3-linked sialic acid. The MAA labeling was decreased in GalNAcα-O-bn-treated cells and the inhibition in the sialylation of MUC1 resulted in a slower migration on SDS-PAGE, as previously observed for secreted mucins of HT-29 MTX cells (Hennebicq-Reig et al. 1998). Nevertheless, in contrast to HT-29 cells, no PNA labeling occurred, indicating the absence of expression of T-antigen (Galβ1–3GalNAc) (data not shown). Instead, we observed the appearance of strong labeling with ECA, a lectin that binds the lactosamine sequence Galβ1–4GlcNAc. Two sialyltransferases involved inα2–3 sialylation were shown to be expressed in CAPAN-1 cells by RT-PCR analysis, i.e., ST3Gal III and ST3Gal IV (data not shown), whereas ST3Gal I, the enzyme involved in the α2–3 sialylation of the T-antigen, is weakly expressed at the RNA level in this cell line. Thus, in CAPAN-1 cells, GalNAcα-O-bn treatment affected ST3Gal III and/or ST3Gal IV, and the lack of sialylation unmasked the subsequent disaccharide unit (Galβ1–4GlcNAc) present in core 2 structures, but also in the terminal position of elongated core 1 (Galβ1–4GlcNAcβ1–3Galβ1–3GalNAc).
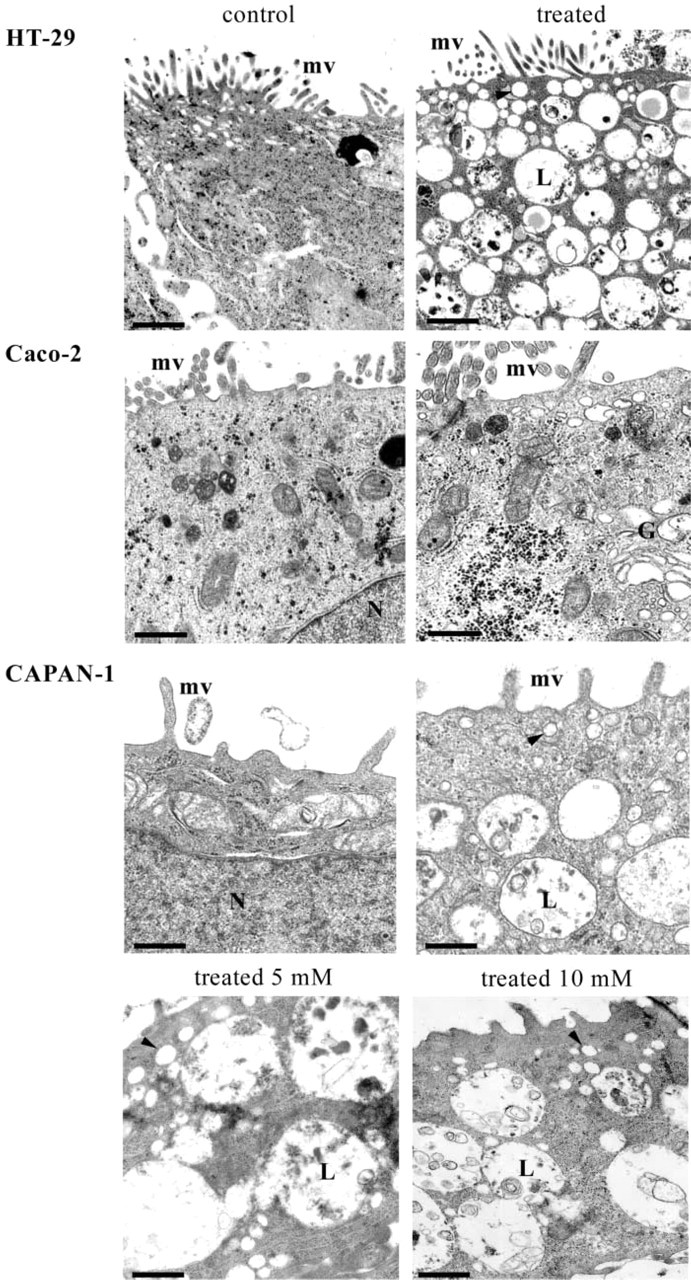
Ultrastructural morphology of control and permanently GalNAcα-O-bn-treated HT-29, Caco-2 and CAPAN-1 cells. Cells were analyzed after 14 days of culture in the absence or in the presence of 2mM GalNAcα-O-bn. CAPAN-1 cells treated with 5 and 10 mM of GalNAcα-O-bn are also shown. Transmission EM of sections perpendicular to the bottom of the flask shows the apical microvilli (mv) in both control and treated cells, and the very numerous vesicles (arrowhead) and late endosomes/lysosomes (L) in treated HT-29 and CAPAN-1 cells. Bars: HT-29 control = 1.1 μm; HT-29 treated = 1.4 μm; Caco-2 control = 0.84 μm; Caco-2 treated = 0.65 μm; CAPAN-1 control and treated 2 mM = 0.5 μm; CAPAN-1 treated 5 and 10 mM = 0.84 μm.
Western blot with lectin staining on CEA and MUC1 immunoprecipitates. CEA and MUC1, synthesized in control and GalNAcα-O-bn-treated CAPAN-1 cells, were immunoprecipitated with mouse MAbs 517 and 214D4. The glycoproteins were checked by Western blotting with their corresponding antibody and then studied by lectin blotting with different lectins. CEA and MUC1 reacted with MAA in control CAPAN-1 cells. After GalNAcα-O-bn treatment, the MAA labeling decreased and ECA labeling appeared on both these glycoproteins.
Distribution of Apical Glycoproteins
We analyzed and compared the cellular distribution of MUC1 and CEA in the three cell lines using antibodies that recognize peptide epitopes of these glycoproteins (Figure 4). As expected, MUC1 and CEA displayed an apical expression in the three polarized cell lines. In Caco-2 cells, the expression of MUC1 was very low and was therefore not illustrated. In accordance with our previously described results, MUC1 and CEA lost their apical expression and were found accumulated in the cytoplasm in HT-29 cells treated with 2 mM GalNAcα-O-bn. The intracellular accumulation of CEA and MUC1 was localized inside intracytoplasmic vesicles, as observed using ultrastructural immunohistochemistry (Figure 5). In contrast, no alteration in the cellular distribution of these two glycoproteins was observed after treatment of CAPAN-1 and Caco-2 cells with 2 mM GalNAcα-O-bn. For CAPAN-1 cells, the use of higher concentrations of GalNAcα-O-bn (5 and 10 mM) did not alter the apical expression of MUC1 and CEA (data illustrated in Figure 4 for MUC1 at the concentration of 10 mM). The absence of an abnormal cytoplasmic localization of other glycoproteins was also checked by lectin labeling of control and GalNAcα-O-bn-treated CAPAN-1 cells and analysis by confocal microscopy (data not shown). In particular, the labeling with MAA and ECA was restricted to the cell surface and particularly at the apical membrane in both control and treated CAPAN-1 cells.
Kinetic Study of Morphological Changes in HT-29 Cells
The abnormal localization of apical glycoproteins inside GalNAcα-O-bn-treated HT-29 suggested that their intracellular transport via apical carrier vesicles could be inhibited. We therefore analyzed in depth the evolution of the morphology of HT-29 cells as they advanced along the GalNAcα-O-bn exposure starting from confluent cells at day 5 of culture, with special focus on fusion events of carrier vesicles with the plasma membrane (Figure 6).
Control cells already expressed a well-identified brush border with many microvilli and extremely rare cytoplasmic small vesicles under the apical membrane. In these control cells, many instances of fusion of vesicles with the basolateral or apical plasma membranes were observed. After 3 hr of GalNAcα-O-bn exposure, many instances of fusion of carrier vesicles with the plasma membrane were still observed. After 6 hr of exposure, vesicles began to accumulate close to the Golgi apparatus, which was slightly dilated, and fusion of carrier vesicles with the plasma membrane was seen at a lesser extent. After 18 hr of exposure, the number of intracytoplasmic vesicles was increased and some rare clustered late endosomes/lysosomes and/or multivesicular bodies began to be detected. After 44 hr of exposure, the accumulation of these degradative structures was clearly seen.
Cellular distribution of ST3Gal I and LAMP1 in control (upper row) and GalNAcα-O-bn-treated (14 days) cells (lower row). The three cell types were successively analyzed with two antibodies directed against ST3Gal I and LAMP1. ST3Gal I was expressed in HT-29 cells but not in Caco-2 and CAPAN-1 cells. No change was induced by the permanent GalNAcα-O-bn treatment. With the anti-LAMP1 antibody, a perinuclear labeling was observed in the three cell-types, with also a localization at the apical side in Caco-2 cells. GalNAcα-O-bn treatment induced a marked increase in the labeling in both HT-29 and CAPAN-1 cells, but not in Caco-2 cells. Bars =
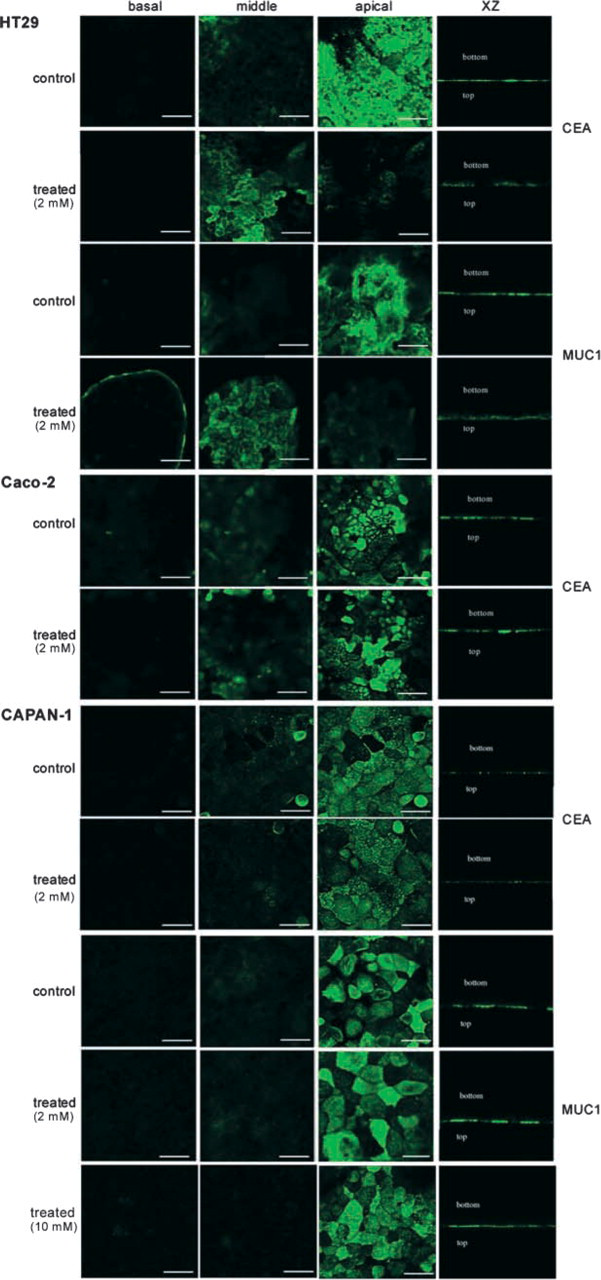
Analysis of the cellular distribution of CEA and MUC1 in control and permanently GalNAcα-O-bn-treated (14 days) cells by confocal microscopy. In the three types of control cells, CEA and MUC1 showed typical expression at the apical membrane. After treatment by 2 mM GalNAcα-O-bn, both glycoproteins became localized in HT-29 cells, whereas no change occurred in Caco-2 and CAPAN-1 cells. Similar results were obtained after treatment of CAPAN-1 cells with 10 mM GalNAcα-O-bn. Bars =
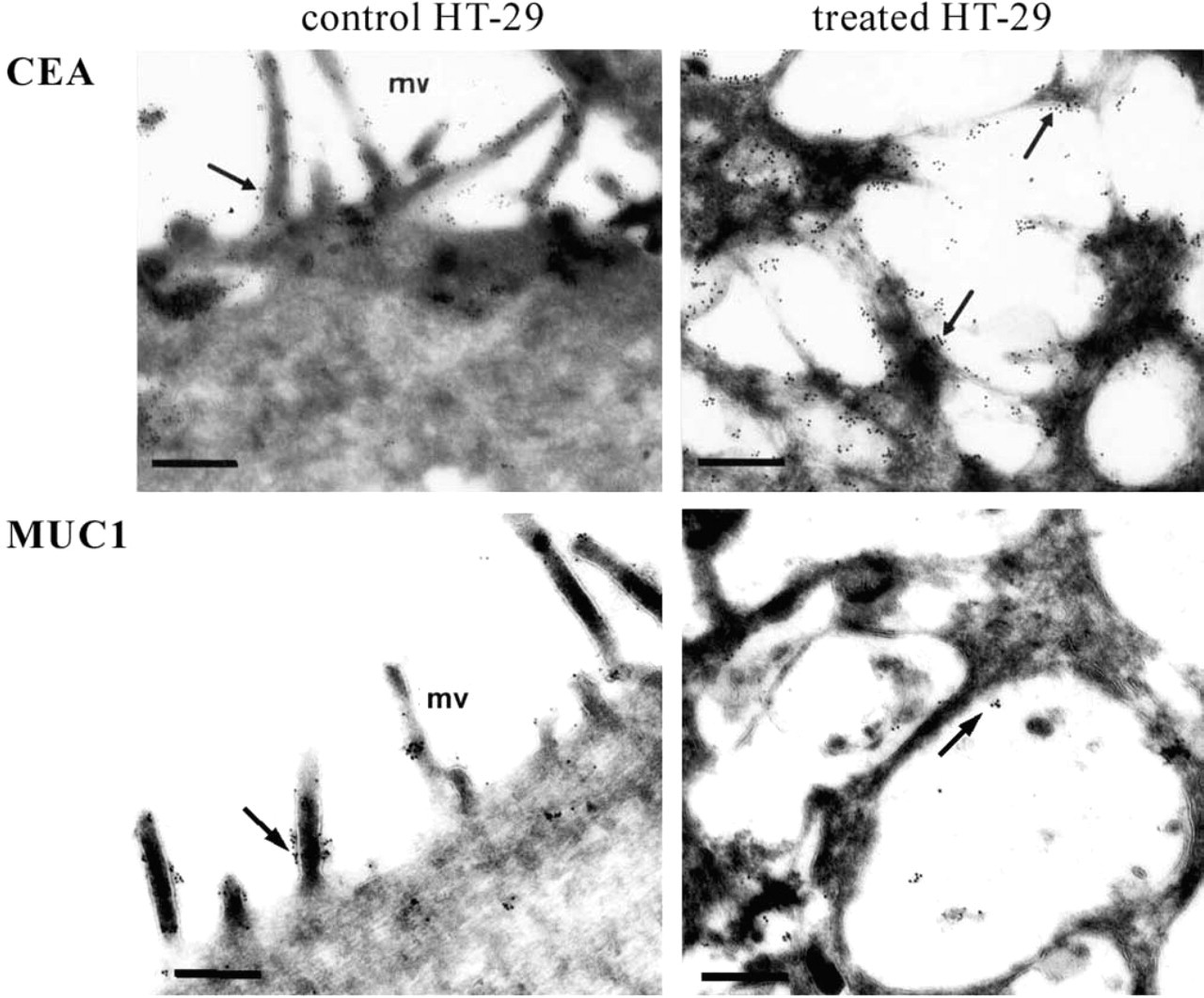
Immunogold labeling of CEA and MUC1 in control and permanently GalNAcα-O-bn-treated (14 days) HT-29 cells. In control cells, the gold particles are present at the brush border (arrows). In treated cells, the gold particles are present in intracellular vesicles (arrows). Bars: CEA control = 0.46 μm; CEA treated = 0.36 μm; MUC1 = 0.3 μm.
The same kinetic study carried out in CAPAN-1 cells clearly showed that fusion of carrier vesicles with the plasma membrane continued to be seen throughout the GalNAcα-O-bn exposure (data not shown). Therefore, the accumulation of intracellular vesicles had no connection with an inhibition in intracellular transport via carrier vesicles.
These results showed inhibition of fusion of carrier vesicles with the plasma membrane in GalNAcα-O-bn HT-29 cells, with the latter phenomenon preceding the alterations at the level of the endosomal/lysosomal system. This observation was in line with the alteration in the apical localization of glycoproteins specifically observed in HT-29 cells.
Discussion
The present study shows that permanent treatment by GalNAcα-O-bn, a competitive inhibitor of O-glycosylation, can induce two types of cell response that correspond to different mechanisms induced by the penetration of this exogenous substance into the cells.
The first type is localized at the level of endosomal/lysosomal compartments. Previous work on differentiated mucin-secreting or enterocyte-like HT-29 cells has suggested that prolonged treatment with GalNAcα-O-bn led to the appearance of characteristics of a storage phenotype. We found that GalNAcα-O-bn was extensively substituted in both HT-29 cell types, generating a series of complex sialylated GalNAcα-O-bn derivatives that accumulated inside the cells (Zanetta et al. 2000; and data not shown). On the other hand, Ulloa et al. (2000) showed an inhibition in the processing of α-glucosidase and cathepsin D in GalNAcα-O-bn-treated HT-29 cells of mucin-secreting phenotype, and regarding the fact that the GalNAcα-O-bn derived oligosaccharides were mainly sialylated (Zanetta et al. 2000). This observation has been connected to the inhibition in the processing of lysosomal hydrolases described in the immature lysosomes of sialic acid storage disease (SASD) fibroblasts (Schmid et al. 1999; Ulloa et al. 2000).
In the present work, we first sought to specify the storage phenotype induced by GalNAcα-O-bn treatment of enterocyte-like HT-29 cells and of other cell lines. We have quantified the amount of GalNAcα-O-bn-derived oligosaccharides after treatment with 2 mM GalNAcα-O-bn. In HT-29 cells, we found a high content of monosaccharides linked to GalNAcα-O-bn, including Gal, GlcNAc, Fuc, and Neu5Ac, confirming the high rate of metabolization of GalNAcα-O-bn by a series of glycosyltransferases. In CAPAN-1 cells, GalNAcα-O-bn-derived oligosaccharides were also found but to a lesser extent than in HT-29 cells. In contrast, no substitution of GalNAcα-O-bn was detected in Caco-2 cells. At the morphological level, a spectacular accumulation of late endosomes/lysosomes of important size was shown in HT-29 cells, with many multivesicular bodies. The accumulation of such compartments was also confirmed by an increased expression of the lysosomal protein marker LAMP-1. CAPAN-1 cells displayed similar alterations as in HT-29 cells, although at a lower level, whereas no changes appeared in enterocyte-like Caco-2 cells. To see if the morphological alterations were connected to intracellular storage of GalNAcα-O-bn derivatives, we studied the effect of higher concentrations of GalNAcα-O-bn. The morphological analysis of CAPAN-1 cells treated with 5 or 10 mM GalNAcα-O-bn showed a storage phenotype similar to that observed in HT-29 cells treated with 2 mM GalNAcα-O-bn. The results showed that the effect of GalNAcα-O-bn treatment on the lysosomal system was associated with the cells' capacity to accumulate GalNAcα-O-bn derivatives. The differences in the intracellular fate of GalNAcα-O-bn according to cell type may be related to the expression pattern of glycosyltransferases able to substitute this exogenous acceptor and/or the transport of GalNAcα-O-bn to Golgi vesicles.
GalNAcα-O-bn-treated HT-29 and CAPAN-1 cells showed some morphological differences in comparison to the SASD phenotype. The lysosomes became larger, than normal lysosomes, whereas they are smaller in SASD fibroblasts, and the accumulation of vesicles seen in GalNAcα-O-bn-treated cells is not a typical feature of SASD fibroblasts (Schmid et al. 1999). Rather, the GalNAcα-O-bn-derived storage phenotype can be connected to the phenotype induced by exposure of fibroblasts to sucrose, this phenotype being characterized by similar vesicles called “sucrosomes.” The latter have been shown to correspond to swollen late endosomes, and the underlying mechanism was that the high concentration in sugar inhibited the fusion between pre-existing lysosomes and late endosomes and for re-formation of dense core lysosomes (Cohn and Ehrenreich 1969; De Courcy and Storrie 1991; Montgomery et al. 1991; Bright et al. 1997). Therefore, the alterations seen at the level of endosomes/lysosomes in cells treated with GalNAcα-O-bn mimic the storage phenotype induced by the uptake of a sugar into cells that cannot digest it. This suggests that, in addition to glycosyltransferases, the occurrence of a GalNAcα-O-bn storage phenotype can be also widely modulated by the expression of glycosidases.
In addition to the possible induction of a storage phenotype, GalNAcα-O-bn treatment was reported to impair the normal localization of apical glycoproteins in differentiated HT-29 cells, such glycoproteins becoming localized inside the cells (Huet et al. 1998). In contrast, in Caco-2 cells, such intracellular accumulation of apical glycoproteins was not observed (Gouyer et al. 2001). Similarly, the storage abnormalities were found in HT-29 cells but not in Caco-2 cells. An important question emerged from the finding that both types of cell responses were observed in HT-29 cells, whereas none occurred in Caco-2 cells, i.e., the existence or not of a connection between these two events. A substantial response was obtained by analyzing the CAPAN-1 cell line. This cell line, which displayed a typical storage phenotype, kept on targeting the apical glycoproteins CEA and MUC1, even at the highest concentration of GalNAcα-O-bn, and no intracellular localization of these or other glycoproteins was detected. This observation enabled us to determine that the abnormal intracellular localization of apical membrane glycoproteins induced by GalNAcα-O-bn treatment of HT-29 cells was a phenomenon independent of that of the induction of a storage phenotype. Furthermore, morphological studies showed an inhibition in the fusion of carrier vesicles to the plasma membrane in HT-29 cells, but not in CAPAN-1 cells, and the latter phenomenon even occurred well before the appearance of the storage phenotype. All these data indicated that the apical targeting of glycoproteins was specifically inhibited in HT-29 cells and that this response did not result from the intracellular accumulation of GalNAcα-O-bn derivatives but rather from another mechanism induced by the intracellular penetration of this competitive inhibitor of glycosylation.
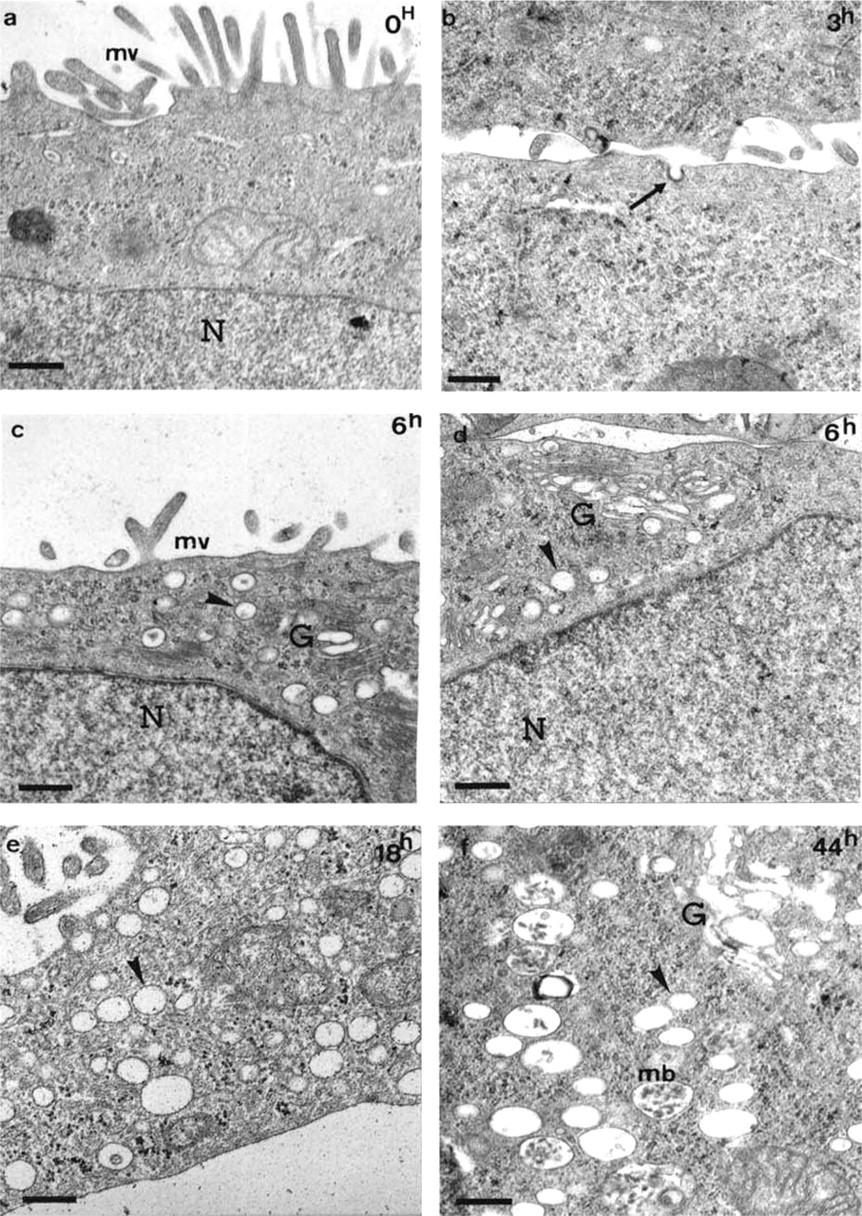
Ultrastructural morphology of cells in a kinetic experiment after short exposure times to GalNAcα-O-bn. Cells were analyzed after 5 days of culture in standard conditions (
This other mechanism likely relies on the competitive effect of GalNAcα-O-bn and/or its derivatives against the cellular glycosylation machinery. In that context, we have previously shown in HT-29 cells that GalNAcα-O-bn treatment primarily inhibited the terminal sialylation of O-glycosylproteins by the sialyltransferase ST3Gal I (Hennebicq-Reig et al. 1998; Gouyer et al. 2001) and of O- or N-glycosylproteins by the sialyltransferase ST3Gal IV. The inhibition of ST3Gal I resulted in the expression of the immature T-antigen. Here we have shown that ST3Gal I was not significantly expressed in CAPAN-1 cells. In these cells, GalNAcα-O-bn treatment inhibited the α2–3 sialylation of glycoproteins by the sialyltransferases ST3Gal III and/or ST3Gal IV, and resulted in the expression of the mature N-acetyl lactosamine sequence. These results show that the effect of GalNAcα-O-bn treatment on the glycosylation of endogenous glycoproteins can vary considerably according to the cell type. It can be suggested that the expression of an immature glycan epitope in GalNAcα-O-bn-treated HT-29 cells is responsible for the abnormalities of apical targeting in these cells. Indeed, the hypothesis for a putative lectin involved in the apical targeting pathway has been proposed (Fiedler et al. 1994; Alfalah et al. 1999; Gouyer et al. 2001). Such a lectin would be able to recognize, with broad specificity, different mature glycan epitopes. The existence of a variable pattern of critical signals for apical targeting would be compatible with the heterogeneity of terminal glycan epitopes according to individual variations.
In conclusion, treatment of polarized cells with the inhibitor of O-glycosylation, GalNAcα-O-bn, can induce two types of cellular response, which depend on different mechanisms. One is the GalNAcα-O-bn-derived storage phenotype, induced by the accumulation of metabolites of this sugar analogue. The other is the inhibition of the apical targeting of glycoproteins, and several arguments suggest that the latter response in HT-29 cells results from abnormal expression of the immature T-antigen induced by the competitive inhibition of the sialyltransferase ST3Gal I.
