Abstract
Glycoprotein 2 (GP2), initially identified as a primary membrane protein of zymogen granules in pancreatic acinar cells, is a marker of intestinal microfold cells (M cells) and involved in bacterial transcytosis in M cells. Recent studies have reported GP2 expression in the pancreas and intestine among various other organs. We aimed to elucidate the expression of GP2 and its glycosylation modifications in human Cowper’s glands. We generated an anti-human GP2 monoclonal antibody suitable for formalin-fixed, paraffin-embedded tissue sections. This antibody, designated GP2-71, successfully stained both the plasma membrane and cytoplasm of normal pancreatic acinar cells. Subsequently, we performed GP2-71 immunostaining on normal human Cowper’s gland tissues. Our findings revealed that the luminal cell membrane and contents of the Cowper’s glands reacted strongly with GP2-71. Moreover, the regions stained with GP2-71 were partially immunostained with the anti-sialyl Lewis x (sLex) monoclonal antibody HECA-452. Furthermore, western blot analysis of protein A-purified GP2-immunoglobulin G fusion proteins demonstrated that GP2 was decorated with HECA-452-positive glycans. Collectively, these findings indicate that GP2 is expressed in human Cowper’s glands and is potentially decorated with sLex-related glycans.
Introduction
Glycoprotein 2 (GP2), a glycosylphosphatidylinositol (GPI)-anchored membrane protein with a molecular weight of 74 kDa, was first identified as a major membrane protein in zymogen granules of pancreatic acinar cells. 1 Produced by pancreatic acinar cells, GP2 is secreted into the intestinal tract, where it binds to enterobacterial FimH, a component of the type I pili on the bacterial outer membrane. 2 This binding prevents the entry of intestinal bacteria into the intestinal mucosal epithelium and regulates bacterial infections.3,4 Furthermore, GP2 serves as a marker of microfold cells (M cells) located within Peyer’s patches of the intestine, where it facilitates bacterial recognition and transcytosis from the intestinal lumen by binding to enterobacterial FimH. 2 Bartlitz et al. 4 demonstrated that mannose residues comprising high-mannose-, hybrid-, and complex-type N-glycans present on GP2 are critical for this binding; however, the structures of GP2-modifying glycans remain unclear.
Cowper’s glands, also known as bulbourethral glands, are a pair of exocrine glands approximately 5 mm in length, located within the urogenital diaphragm near the prostatic apex.5,6 In response to sexual stimulation, the Cowper’s glands secrete glycoprotein-containing mucus, which acts as a lubricant to reduce friction between the penis and vaginal mucosa during sexual intercourse. In addition, the alkaline component of the mucus helps neutralize the urethra and vagina, thereby aiding sperm viability. 5 Piludu et al. 7 reported that Cowper’s gland secretions contain MG1 mucin whose major protein component is encoded by the MUC5B gene. 8 In mice, GP2 secretion has been observed in multiple secretory glands outside the pancreas, including Cowper’s glands; 9 however, no studies have examined GP2 expression in human Cowper’s glands.
In this study, we developed an anti-GP2 monoclonal antibody that can be used for formalin-fixed paraffin-embedded (FFPE) tissue sections. Using this antibody, we conducted immunohistochemical analysis to determine whether GP2 is expressed in human Cowper’s glands. In addition, we performed western blot analysis on GP2-immunoglobulin G (IgG) fusion proteins secreted from sialyl Lewis x (sLex)-expressing Chinese hamster ovary (CHO) cells to determine whether GP2 is decorated with sLex-related glycans.
Materials and Methods
Construction of Expression Vectors Encoding Human GP2
DNA fragments encoding human GP2 (amino acid residues 1–387) 10 were polymerase chain reaction (PCR)-amplified with the oligonucleotides 5′-TCAAgCTTAgTgACCTgCATgCCTCACCTTAT-3′ and 5′-TgCTCgAgTCAgAACAgCCAAgCCAggAggAC-3′ using a human small intestine cDNA library as a template. The fragments were inserted into HindIII/XhoI sites of pcDNA3.1/Hygro (Invitrogen, Carlsbad, CA), resulting in pcDNA3.1/Hygro-GP2. Similarly, DNA fragments encoding amino acid residues 45–119 of GP2 were PCR-amplified with 5′-CgggATCCCgCCCCgAggAggAgTgCCTTg-3′ and 5′-TAgAATTCTACTCCTCTgTCTgCAAgATgC-3′. The fragments were inserted into BamHI/EcoRI sites of pCold-GST (Takara Bio, Kusatsu, Japan), which carries a glutathione S-transferase (GST) tag, resulting in pCold-GST-GP2. DNA fragments encoding the extracellular domain (amino acid residues 1–363) of GP2 were PCR-amplified with 5′-ACCCAAgCTggCTAgCAgTgACCTgCATgCCTCA-3′ and 5′-CTCACCCTCgggATCCCCATTCATgACACCgggA-3′, and the fragments were subcloned into NheI/BamHI sites of pcDNA3.1/Hygro-IgG, 11 resulting in pcDNA3.1/Hygro-GP2-IgG.
Production of Anti-GP2 Monoclonal Antibodies
GP2-GST fusion protein was expressed and purified as previously described. 12 Wistar Kyoto rats were immunized with GP2-GST fusion protein as previously described. 11 Lymphocytes collected from inguinal lymph nodes were mixed with SP2/0 mouse myeloma cells and electrofused as previously described. 13 Cells were suspended in a selection medium containing hypoxanthine, aminopterin, and thymidine, dispensed into 96-well plates, and cultured until the surviving cells formed colonies. Culture media were initially screened by enzyme-linked immunosorbent assay using the immunogen, and positive ones were further screened by immunostaining of FFPE human terminal ileum tissue sections. Hybridoma cells in the culture media that selectively stained intestinal M cells were cloned by limiting dilution. Among these hybridoma lines, the line designated GP2-71 was selected for this study. The experimental protocol was approved by the Animal Experimentation Committee of Kansai Medical University. In addition to GP2-71, the monoclonal antibody, HECA-452 (BD Pharmingen, San Diego, CA), which recognizes sLex and its enantiomer sLea,14,15 was used in this study.
Expression and Purification of GP2-IgG Fusion Proteins
To obtain full length of human GP2, human embryonic kidney (HEK) 293T cells were transfected with pcDNA3.1/Hygro-GP2 using Lipofectamine 3000 (Thermo Fisher Scientific, Waltham, MA). After 36 hr, cells were harvested in phosphate-buffered saline supplemented with a protease inhibitor cocktail (Roche, Basel, Switzerland), and the membrane fraction was obtained as previously described.16,17 To obtain GP2-IgG fusion proteins potentially glycosylated with sLex glycans, CHO cells stably expressing sLex glycans (CHO/CD34/F7/C2) 18 were transfected with pcDNA3.1/Hygro-GP2-IgG. After 36 hr, conditioned medium was recovered and purified using Protein A-Sepharose 4B Fast Flow from Staphylococcus aureus (Sigma-Aldrich, St. Louis, MO). These protein samples were subjected to western blot analysis as described previously.12,19
Human Tissue Samples
Normal Cowper’s gland tissue samples (n=3) were obtained at autopsy from patients without Cowper’s gland disease at University of Fukui Hospital. The tissues were fixed in 10% neutral-buffered formalin and embedded in paraffin. Normal pancreatic and prostate tissues were used as controls. The human tissue analysis was approved by the Research Ethics Committee of University of Fukui.
Immunohistochemistry
Immunohistochemical staining was performed using an indirect method, 20 and double immunofluorescence staining for GP2-71 and HECA-452 was performed as previously described. 21
Results
GP2-71 Monoclonal Antibody Recognizes GP2
To determine whether the GP2-71 monoclonal antibody specifically recognizes GP2, we first conducted western blot analysis on membrane fractions of HEK 293T cell lysates transfected with pcDNA3.1/Hygro-GP2. As presented in Fig. 1A, a band corresponding to GP2 (~70 kDa), was detected in the lane of HEK 293T cells transfected with GP2 cDNA. In contrast, no band was detected in HEK 293T cells transfected with the empty vector, indicating that the GP2-71 monoclonal antibody specifically recognizes GP2.
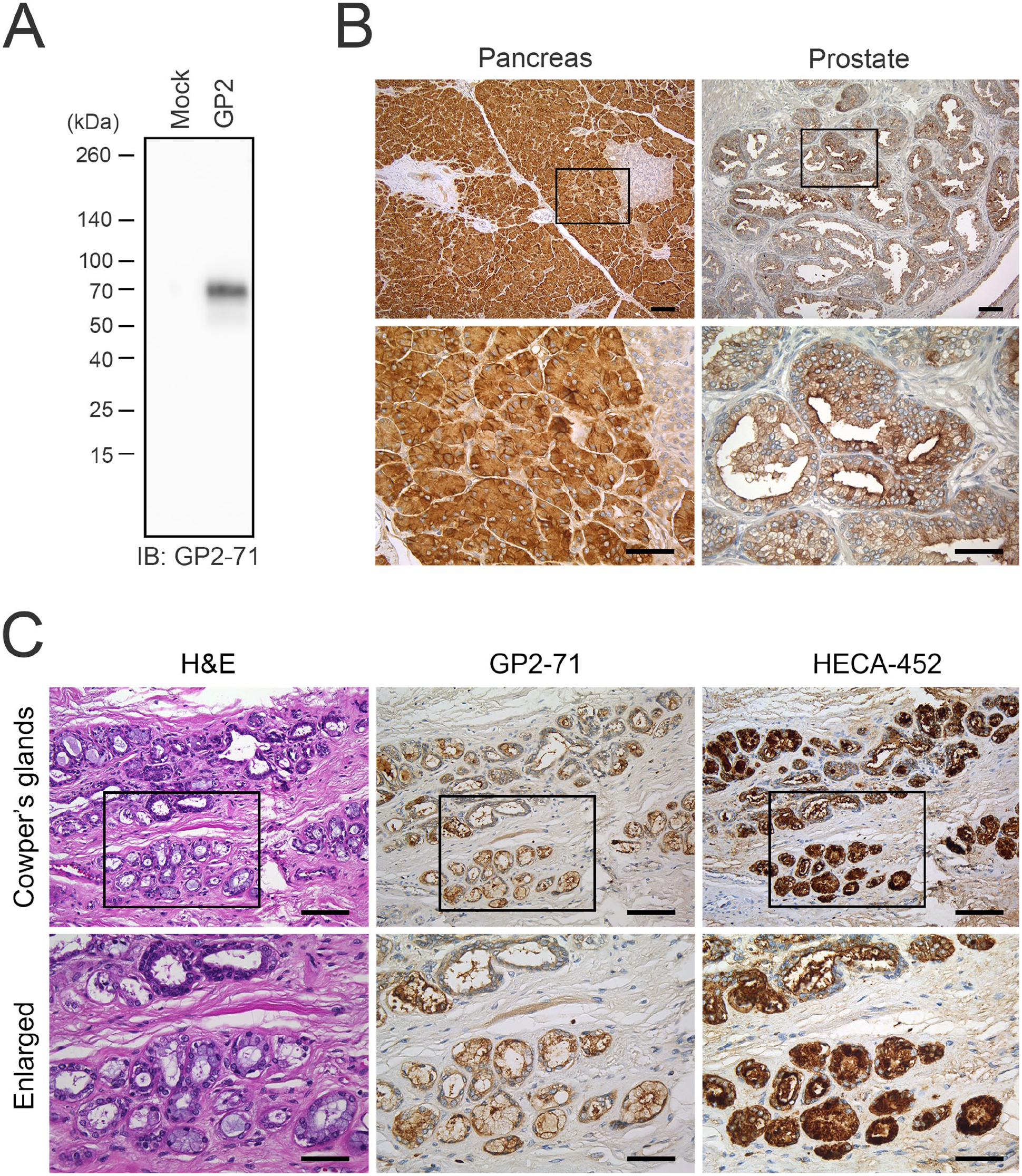
(A) Western blot analysis of the membrane fraction of HEK 293T cell lysates transfected with either an empty vector (mock; left lane) or with vector harboring cDNA encoding GP2 (GP2; right lane). Membranes were immunoblotted with GP2-71 antibody. The numbers on the left indicate the molecular mass (kDa). IB: immunoblot. (B) GP2 expression in human pancreas and prostate. The left and right panels show GP2-71 immunostaining of normal human pancreatic and prostate tissue, respectively. (C) Immunohistochemical profiles of human Cowper’s glands. Serial tissue sections were stained with hematoxylin and eosin (H&E) or immunostained with GP2-71 or HECA-452. In (B) and (C), signals were visualized with 3,3′-diaminobenzidine (brown), and the tissues were counterstained with hematoxylin. The lower panels show enlarged images of the boxed area of the upper panels. Scale bar = 100 µm for the upper panels and 50 µm for the lower panels.
GP2-71 Stains GP2 on FFPE Tissue Sections
Subsequently, we investigated whether GP2-71 could recognize GP2 in FFPE tissue sections. During hybridoma screening, GP2-71 was found to preferentially recognize Spi-B-positive M cells within the intestinal epithelium. 12 Moreover, as shown in Fig. 1B (left panels), the cytoplasm and plasma membrane of pancreatic acinar cells were strongly immunolabeled with GP2-71, whereas pancreatic ducts and islets of Langerhans cells were not stained. This staining pattern is consistent with a previous report, 22 confirming that the GP2-71 antibody selectively recognizes GP2 in FFPE tissue sections. In contrast, GP2-71 immunostaining of five normal prostate tissues was inconsistent, with some areas stained and others not. Glands stained with GP2-71 represented ~40% of the prostate glands, and in these glands the apical membrane and cytoplasm of prostate acinar cells were immunolabeled (Fig. 1B, right panels).
Cowper’s Glands Are Stained with GP2-71 and HECA-452
To assess GP2 expression in human Cowper’s glands, we immunostained three Cowper’s gland tissue samples with GP2-71. As shown in Fig. 1C (middle panels), in all specimens examined, the apical and lateral membranes of the glandular cells of the Cowper’s gland and luminal contents were strongly stained, whereas the cytoplasm of the glandular cells was weakly stained. These findings are consistent with those of Kimura et al., 9 who reported that in mouse Cowper’s glands, the lateral membranes of glandular cells and luminal contents were immunoreactive for GP2. Furthermore, immunostaining of human Cowper’s glands with HECA-452 showed positive immunolabeling of glandular cell cytoplasm and luminal contents; this expression pattern was similar to that of GP2. Moreover, double immunofluorescence staining for GP2-71 and HECA-452 demonstrated that these two epitopes were partially colocalized in human Cowper’s glands (Fig. 2A). These findings strongly suggest that GP2 expressed in human Cowper’s glands is decorated with glycans that are recognized by HECA-452.
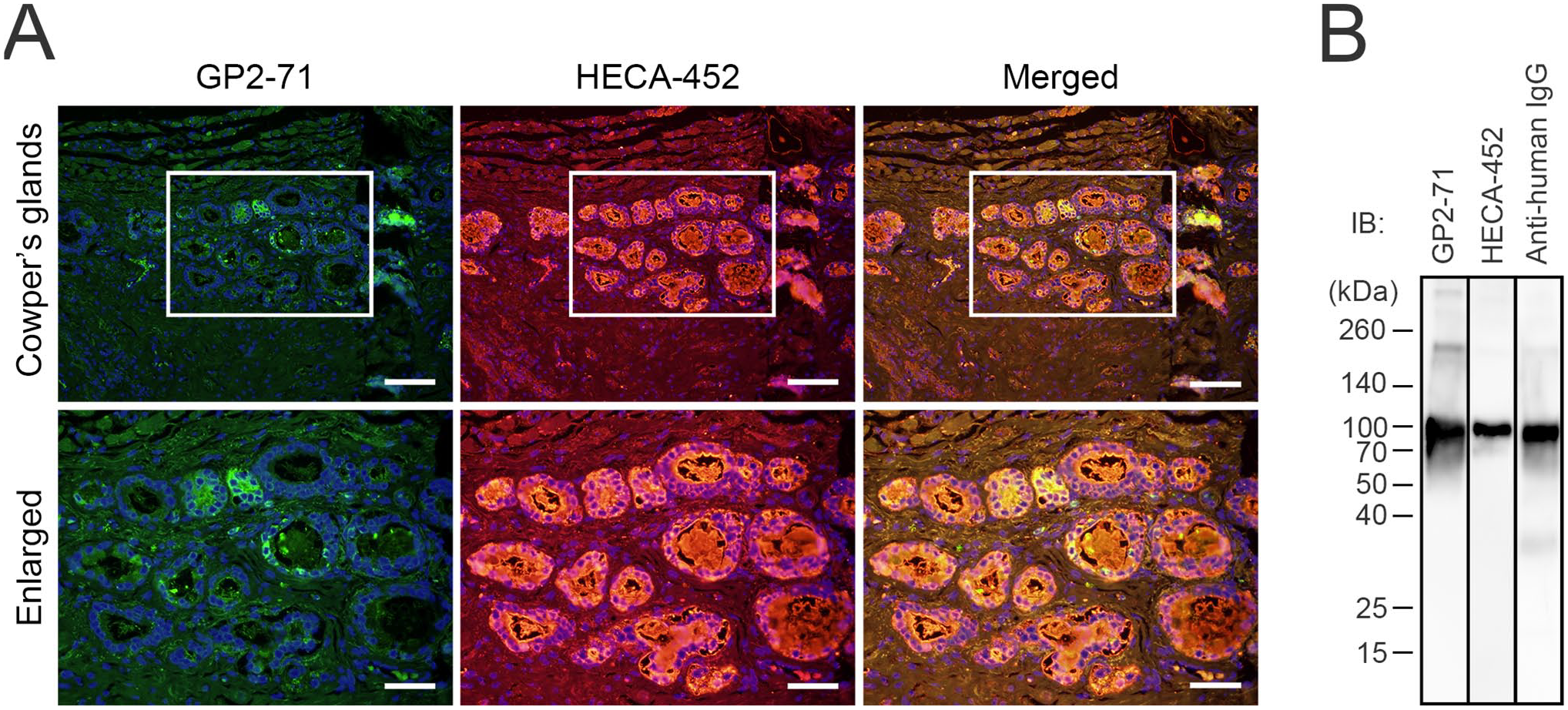
(A) Double immunofluorescence of Cowper’s glands for GP2-71 (green) and HECA-452 (red). Yellow signals in merged images indicate antigen colocalization. Sections were counterstained with 4′,6-diamidino-2-phenylindole (blue). The lower panels show enlarged images of the boxed area of the upper panels. Scale bar = 100 µm for the upper panels and 50 µm for the lower panels. (B) Western blot analysis of GP2-IgG fusion proteins immunoblotted (IB’d) with GP2-71 (first lane), HECA-452 (second lane), and anti-human IgG antibody (third lane). The numbers on the left indicate molecular mass (kDa).
GP2 Functions as a Scaffold Protein for HECA-452-Positive Glycans
To determine whether GP2 proteins were decorated with glycans recognized by HECA-452, we performed a western blot analysis of a fusion protein consisting of the extracellular domain of GP2 and the Fc portion of IgG. As shown in Fig. 2B, a band at ~110 kDa was detected for both GP2-71 and anti-human IgG antibodies, indicating that this band corresponded to the GP2-IgG fusion protein. Moreover, a band of the same size was detected by HECA-452. These findings collectively indicate that GP2 proteins are decorated with HECA-452-reactive glycans and suggest that GP2 secreted in human Cowper’s glands is likely to be glycosylated with sLex-type glycans.
Discussion
In this study, we successfully generated a monoclonal antibody that specifically binds to the human GP2 protein. Using this antibody, we demonstrated that GP2 is expressed in human Cowper’s glands. Moreover, double immunofluorescence staining and western blot analysis of GP2-IgG fusion proteins indicated that GP2 proteins secreted in human Cowper’s glands were likely decorated with glycans recognized by the HECA-452.
Our findings reveal that GP2-71 stains the apical and lateral membranes of Cowper’s glandular cells and luminal contents, with only faint staining in the cytoplasm of Cowper’s glands. Kimura et al. 9 previously demonstrated that GP2 is present in the lateral membranes of glandular cells and in the luminal contents of mouse Cowper’s glands; their immunoelectron microscopy findings indicated that GP2 is present in both the apical and basolateral membranes of glandular cells, with minimal levels in the cytoplasm. These observations are consistent with those in human Cowper’s glands; however, the staining in the apical membrane of human tissue was notably stronger.
Although rare, Cowper’s glands may be included in prostate needle biopsy specimens, in which case they must be differentiated from prostate cancer. 23 Can GP2-71 be useful in this differentiation? In the present study, we showed that ~40% of normal prostate glands express GP2, not just Cowper’s glands. Moreover, Uhlig et al. 22 reported that 6.9% of 14,463 prostate cancer samples expressed GP2. Thus, we conclude that GP2-71 is not a suitable antibody for differentiating Cowper’s glands from prostate cancer.
Western blot analysis revealed bands migrating at ~110 kDa when detected using both GP2-71 and anti-human IgG antibodies. This finding indicates that these bands correspond to the fusion proteins. GP2 is a GPI-anchored membrane protein, and the molecular weight of its extracellular domain is ~70 kDa. The molecular weight of the Fc portion of IgG is ~50 kDa as a dimer, 24 but is estimated to be ~25 kDa as a monomer under reducing conditions. Thus, the expected molecular weight of the fusion protein would be ~95 kDa. Given that the molecular weight of the band detected by HECA-452 immunoblotting was also ~110 kDa, it is reasonable to conclude that the fusion protein was glycosylated with HECA-452-reactive glycans, leading to an increase in its molecular weight.
Few studies have investigated the presence of glycans on GP2,4,25 and the glycosylation of GP2 remains poorly understood. Our findings indicate that GP2 is glycosylated with glycans recognized by HECA-452, an antibody that binds to both sLex and its enantiomer sLea,14,15 although the physiological roles of these glycans remain unclear. Easton et al. 26 previously reported that uromodulin is decorated with sLex glycans during pregnancy, which increases its immunosuppressive effects. Thus, it can be speculated that sLex-related glycans on GP2 are also associated with immunomodulation. Further studies are needed to support this hypothesis.
Footnotes
Acknowledgements
We thank Hisataka Kato and Maiko Yamanaka for technical assistance.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to this article as follows: YF designed and performed the research, analyzed the data, and wrote the manuscript; SL designed and conducted the research; AM, YA, and HH performed the research; TN and TOA designed and conducted the research; MK conceived and designed the research, analyzed the data, and wrote the manuscript. All the authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Grant-in-Aid for Scientific Research (B) 21H02702 from the Japan Society for the Promotion of Science (to MK).
