Abstract
Previous works have shown that glycoconjugates with terminal fucose (Fuc) are located in the primordial germ cells (PGCs) of some mammals and might play a role in the migration and adhesion processes during development. The aim of this work was to identify the terminal Fuc moieties of Xenopus PGCs by means of three Fuc-binding lectins: from asparagus pea (LTA), gorse seed (UEA-I), and orange peel fungus (AAA). The histochemical procedures were also carried out after deglycosylation pretreatments: β-elimination with NaOH to remove O-linked oligosaccharides; incubation with PNGase F to remove N-linked carbohydrate chains; and incubation with α(1,2)- and α(1,6)-fucosidase. The PGCs were always negative for LTA and UEA-I, two lectins that have the highest affinity for Fuc α(1,2)-linked. However, the PGCs were strongly labeled with AAA, which preferentially binds to Fuc with α(1,3) or α(1,4) linkages and to Fuc α(1,6)-linked to the proximal N-acetylglucosamine. There was fainter labeling with AAA when the sections were preincubated with α(1,6)-fucosidase, but the labeling remained strong when the sections were pretreated with α(1,2)fucosidase. When the β-elimination procedure was carried out, the PGC labeling with AAA was slight. If the PNGase F incubation was performed, the PGCs remained moderately positive for AAA. These data suggest that the Xenopus PGCs have Fuc moieties in O- and N-linked oligosaccharides, including Fuc α(1,6) linked to the innermost GlcNAc, and that the Fuc was not in α(1,2)-linkage.
I
On the other hand, one of the major difficulties in study of the PGCs in most animals is the identification and isolation of these cells, owing to the absence of good markers. This is specially true for Xenopus embryos (Gomperts et al. 1994). Recently, some lectins have been shown to be good markers for identification of the PGCs in the embryos of some species (Fazel et al. 1990; Takagi et al. 1997).
The aim of the present work was to characterize, by use of Fuc-binding lectins, fucosylated glycans as a first approach to determine the possible role of glycoconjugates in Xenopus PGC migration and adhesion and to investigate if some of these lectins might be used as PGC markers.
Materials and Methods
Sample Preparation
Xenopus eggs obtained after injection of 1000 U hCG (Profasi HP; Serono, Madrid, Spain) were fertilized in vitro by gently rubbing with testis fragments. The embryos were reared in darkness in 1:10 normal amphibian medium (NAM; Slack 1984) at 18C in a Selecta Prebatem refrigerated incubator (Izasa; Barcelona, Spain), until 43, 46–47, and 48 stages were reached (Nieuwkoop and Faber 1956). Then the embryos were anesthetized with 0.1% (w/v) 3-aminobenzoic acid ethyl ester (MS-222; Sigma Química, Alcobendas, Spain), and fixed with 2.5% (v/v) glutaraldehyde in 0.1 M Sorensen phosphate buffer at pH 7.4 for 4 hr. To block free aldehyde groups of the glutaraldehyde, the samples were washed in the buffer and then immersed for 1 hr in 0.5 M ClNH4. Then the embryos were dehydrated with ethanol solutions, embedded in Epon, and polymerized into gelatin capsules. Finally, 1-μm-thick sections were obtained with an LKB Ultratome-III.
Lectin Histochemistry
In this study, horseradish peroxidase (HRP)- and digoxigenin (DIG)-conjugated lectins were employed to stain semithin sections of epoxy-embedded embryos by the methodology previously described (Alonso et al. 2001). Their specificities, suppliers, and working solutions are shown in Table 1. The lectin-binding patterns were established by methods previously reported (Sáez et al. 1999). First, the Epon resin was removed with a saturated solution of KOH in ethanol. It has been reported that this method neither degrades glycans nor removes O-linked residues (Stoddart and Jones 1998). Then the endogenous peroxidase was blocked by immersing the sections for 30 min in H2O2, after which every section was incubated with a lectin in a moist chamber for 1.5 hr. After washing in the TBS buffer, the sections processed with the DIG-conjugated lectin were incubated with an anti-DIG/HRP-labeled goat antibody (Roche; Barcelona, Spain) for 1 hr. Finally, the HRP-conjugated lectins and the HRP-labeled antibody were developed with 3,3′-diaminobenzidine and H2O2. For the stage 46–47 embryos, the histochemical procedure for each lectin was realized in three ways: (a) without previous treatment; (b) after β-elimination, a chemical deglycosylation procedure that removes the O-linked oligosaccharides (Ono et al. 1983); and (c) after incubation with endoglycosidase F/peptide N glycosidase F (PNGase F; Roche), an enzymatic deglycosylation method to remove the N-linked oligosaccharides (Lucocq et al. 1987). Moreover, AAA histochemistry was carried out after incubation with α(1,2)-fucosidase and α(1,6)-fucosidase (Sigma Química).
The intensity of the staining was quantified by two independent observers that classified the PGC labeling into four arbitrary categories: no labeling (0), weak (1), moderate (2), and strong (3). This quantification is useful because it enabled us to compare the PGC labeling without the pretreatments and combined with them.
Deglycosylation Pretreatments
The enzymatic pretreatment using PNGase F to remove the N-linked oligosaccharides was performed as described previously (Lucocq et al. 1987; Sáez et al. 1999). Briefly, the method employed consisted of incubating the sections with the enzyme at 6 U/ml for 2 days at pH 9. At this pH, PNGase activity predominated over Endo F activity, so that the cleavage of the β-aspartyl glycosyl linkage [GlcNAcβ(1-N)Asn] was preferred. To remove the O-linked oligosaccharides, chemical β-elimination with 0.5 N NaOH in 70% (v/v) ethanol, at 4C for 10 days, was performed as previously reported (Ono et al. 1983; Sáez et al. 1999).
To remove Fuc, α(1,2)- and α(1,6)-fucosidases were employed. The sections were incubated with the enzyme in a 1:200 dilution in 250 mM sodium phosphate buffer, pH 5.0, for 2 days at 37C in a moist chamber.
Controls
The following controls were used: (a) substitution of the lectins, anti-DIG antibody, and enzymes by the buffer alone; (b) preincubation of the lectins with 0.2 M Fuc (Sigma Química) (Spicer and Schulte 1992); (c) preabsorption of the anti-DIG antibody with the corresponding antigen; and d) staining of sections of other tissues of known altered binding pattern for each of the chemical and enzymatic pretreatments (Madrid et al. 1994,2000; Sáez et al. 1999,2000a,b; 2001a,b).
Results
The control sections were always negative.
The binding patterns of each lectin observed in stages 43, 46–47, and 48 were very similar. Then deglycosylative pretreatments were performed only with the stage 46–47 embryos. These results are shown in Table 2. No lectin can be employed as a marker of the PGCs because the PGCs and the surrounding mesentery cells showed similar lectin binding patterns.
The PGCs were negative for LTA (Figure 1) and UEA-I (Figure 2) with all the pretreatments employed. AAA labeled the plasma membrane and the cytoplasm of the PGCs and the surrounding mesentery cells (Figure 3a). This labeling was notably decreased by the β-elimination pretreatment (Figure 3b). However, after incubation with PNGase F, labeling with AAA was slightly decreased and remained moderate (Figure 3c).
Lectins employed, with indication of their working dilution in μg/ml and major carbohydrate binding specificity

aHRP, horseradish peroxidase; DIG, digoxigenin.
bAlso named OFA.
Fuc-binding lectin labeling pattern of the PGCs of stage 46–47 Xenopus embryos a

aStaining intensity: 0, negative; 1, weak; 2, moderate; 3, strong.
When the sections were previously incubated with α(1,2)fucosidase, AAA labeled the PGCs (Figure 3d). However, when the incubation was carried out with α(1,6)fucosidase pretreatment, they were scarcely positive.
Discussion
One of the major difficulties in research on Xenopus PGC migration is the absence of a specific marker for these cells, a problem that led some groups to study germ-cell migration in other animals (Gomperts et al. 1994). Unfortunately, data from the present work did not enable us to use any of the Fuc-binding lectins employed as PGC markers because the binding pattern of PGCs was similar to that of the surrounding tissues.
In this study, LTA and UEA-I showed a different binding pattern from that of AAA, in spite of the fact that all of them bind Fuc. It has been previously reported that several lectins with affinity for the same sugar could show different labeling patterns (Spicer and Schulte 1992). In this sense, it has been observed that Fuc-binding lectins show different reactivity with fucoconjugates, in accordance with the link between Fuc and its neighbor (Laden et al. 1984; Hennigar et al. 1985; Tsukada and Spicer 1988; Spicer and Schulte 1992). It is generally assumed that AAA bind preferentially to Fuc α(1,6)-linked to the proximal GlcNAc of the N-linked glycans, and to terminal Fuc with an α(1,3) or α(1,4) linkage (Kochibe and Furukawa 1980; Yamashita et al. 1985; Osawa and Tsuji 1987; Spicer and Schulte 1992), whereas LTA and UEA-I are considered to have high affinity for Fuc with an α(1,2) linkage (Pereira and Kabat 1974; Allen et al. 1977; Pereira et al. 1978; Susz and Dawson 1979; Debray et al. 1981; Sugii et al. 1982).
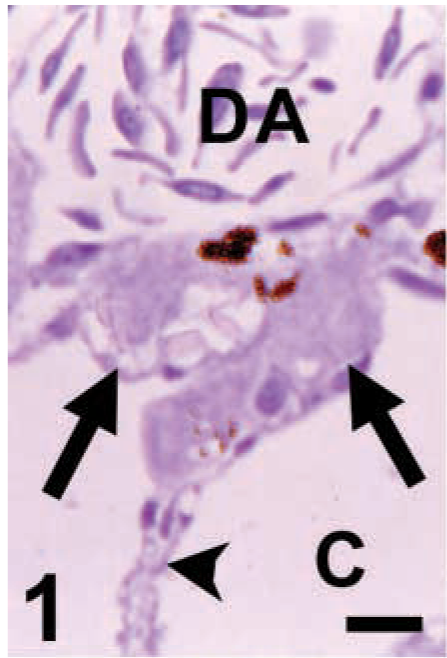
LTA. The PGCs (arrows) are in the dorsal mesentery (arrowhead), under the dorsal aorta (DA). There is no labeling in PGCs. The mesentery cells and other tissues are negative. C, coelom. Bar = 10 μm.
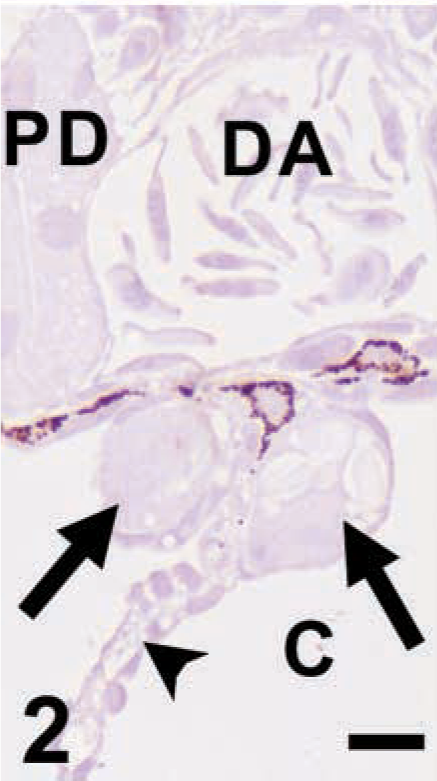
UEA-I. The PGCs (arrows) and most of the surrounding tissues are negative for UEA-I. Arrowhead; dorsal mesentery; C, coelom; PD, pronephric duct. Bar = 10 μm.
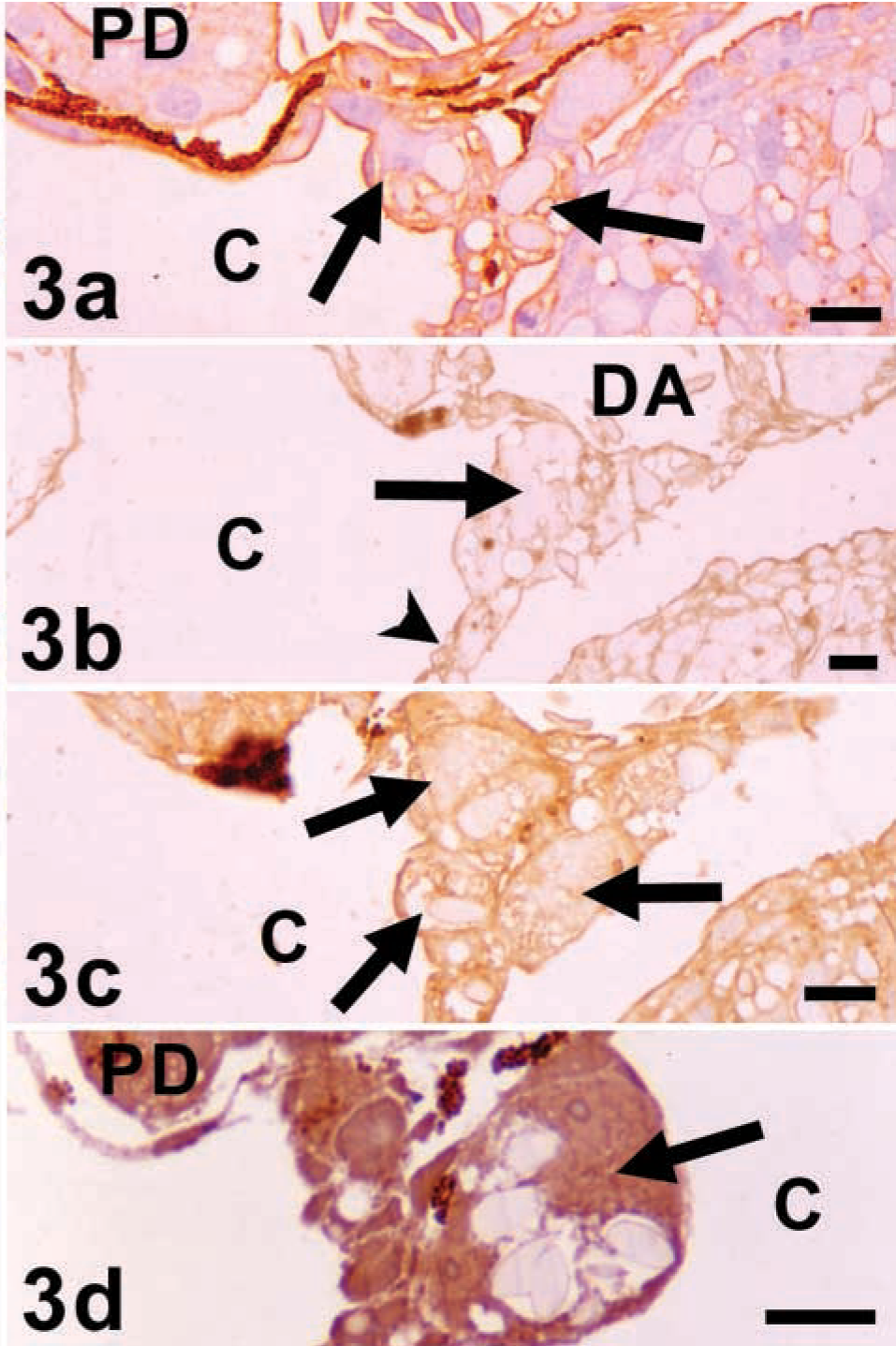
AAA. Without pretreatments (
The analysis of the results obtained with LTA and UEA-I in the present work in the Xenopus PGCs allows us to suggest the absence of α(1,2)-linked Fuc in these cells. This was previously suggested by using UEA-I by Delbos and colleagues (1984), and could be supported by one of our previous works using Gal-NAc-binding lectins (Alonso et al. 2002). In that work, the PGCs were negative for LBA and SBA, two lectins that bind to N-acetylgalactosamine (GalNAc) linked to galactose (Gal) substituted by an α(1,2)-linked Fuc (Roberts and Goldstein 1984; Sikder et al. 1986; Piller et al. 1990; Wu and Sugii 1991), suggesting the absence of this sequence in Xenopus PGCs. In the same work, DBA labeled the PGCs of Xenopus. Interestingly, it has been shown that LTA lectin has the highest reactivity for some α(1,2)-linked Fuc-containing oligosaccharides but has no reactivity for the oligosaccharides with terminal GalNAc α(1,3)[Fuc α(1,2)]Gal (Pereira and Kabat 1974). This means that LTA can recognize α(1,2)-fucosylated glycans different from those recognized by LBA and SBA. Therefore, from these data it could be inferred that no fucosylated glycans with α(1,2) linkage are in the PGCs of Xenopus. The results with AAA suggest that α(1,3), α(1,4), or α(1,6)-linked Fuc is in the glycoconjugates of the PGCs. To verify this, lectin histochemistry was performed again after incubation with α(1,2)fucosidase, and the labeling pattern was not modified, supporting the proposition that AAA did not label α(1,2)-linked Fuc. Moreover, the staining was notably decreased when the O-linked oligosaccharides were removed (β-elimination pretreatment) and remained almost invariable if N-linked oligosaccharides were removed (PNGase F incubation). These data suggest that some Fuc moieties were in O-linked oligosaccharides, although others could be in N-linked oligosaccharides. Perhaps Fuc with an α(1,6) linkage to GlcNAc-Asn, which is present only in N-linked chains, could be responsible for the faint staining after β-elimination, whereas Fuc with α(1,3) or α(1,4) could be present in O-linked chains and could remain after PNGase F pretreatment. Preincubation of the samples with α(1,6)fucosidase resulted in fainter labeling of the PGC compared with cells without pretreatment, supporting the concept that some Fuc residues have an α(1,6) linkage. If we consider that α(1,6) linked Fuc can be mostly linked to the GlcNAc-Asn core of the N-linked chains, it would be considered contradictory that the staining with AAA after α(1,6)fucosidase incubation was slighter than after PNGase F digestion, because it would be expected that the change in lectin staining should be very similar after both pretreatments. However, the moderate staining with AAA lectin after PNGase F incubation could be explained if we assume that removal of N-linked glycans could result in unmasking of other O-linked sugar chains with Fuc moieties that were not accessible to the lectin without pretreatment. The unmasking of sugar residues after oligosaccharide removal has been previously described in other tissues (Leis et al. 1997; Madrid et al. 2000). A similar unmasking of sugar chains could be unexpected after α(1,6)fucosidase incubation because this enzyme removes one sugar moiety from the glycan, but not all the N-linked oligosaccharide chain.
Most of the vertebrate species studied have shown scarce or null reactivity in their PGCs for the Fuc-binding lectins (Delbos et al. 1984; Fazel et al. 1987; Didier et al. 1990; Yoshinaga et al. 1992; Wrobel and Süss 1998). However, fucosylated oligosaccharides have been identified in chick, pig, and mouse PGCs by cytochemistry with lectins (LTA) and antibodies (EMA-1 and anti-SSEA-1) (Hahnel and Eddy 1986; Urven et al. 1988; Fazel et al. 1990; Takagi et al. 1997). The positivity of the mouse PGCs for LTA might suggest the presence of α(1,2) Fuc. However, a previous work with human salivary glands from secretors and nonsecretors provided evidence of affinity for LTA but not UEA-I for α(1,4)-linked Fuc (Laden et al. 1984; Spicer and Schulte 1992), and therefore binding of LTA but not UEA-I to mouse PGCs has been attributed to α(1,4)-linked Fuc (Fazel et al. 1990).
In summary, in this work we have shown by lectin histochemistry that the Xenopus PGCs have Fuc moieties, mainly in O-linked oligosaccharides, and probably with α(1,3) or α(1,4) linkages. Moreover, the Fuc localized in N-linked chains could be attributed to α(1,6) linkages to the innermost GlcNAc. The absence of α(1,2)-linked Fuc can be inferred.
Footnotes
Acknowledgements
Supported by grants from the University of the Basque Country (EA137/97 and G10/99). EA was supported by fellowships from the University of the Basque Country and the Spanish Government (Ministerio de Educación, Cultura y Deporte). Ms M. Portuondo and Ms C. Otamendi contributed to sample preparation.
