Abstract
It has been suggested that brush cells (BCs), a distinct type of cell occurring in various epithelia of the respiratory and gastrointestinal tracts, may function as receptor cells. The major characteristics of BCs are a prominent brush border and an unusually highly ordered arrangement of cytoskeletal elements (F-actin, microtubules, and intermediate filaments). In this study we aimed to characterize the nature of the intermediate filaments in BCs by light and electron microscopic immunostaining. Gallbladder and stomach specimens from mice and rats, respectively, were fixed in various solutions, embedded either in paraffin or epoxy resin, and processed for immunodetection. Commercially available, well-characterized antibodies against neurofilaments, peripherin, and cytokeratin peptide 18 were used. The polyclonal antiserum cocktail to neurofilaments was applied as a supplement in a double-labeling procedure with anti-actin and anti-cytokeratin 18 antibodies. The results demonstrate that the BCs of both organs express two types of intermediate filaments, i.e., neurofilaments and cytokeratin 18 filaments, and that these have a compartmentalized distribution in the cytoplasm. BCs do not express peripherin. The immunodetection of intermediate filaments distinctive for mature neurons in BCs supports their putative receptor function. The co-expression of neurofilaments and cytokeratins is shown for the first time in healthy tissues.
Keywords
B
In this study we demonstrate by light (LM) and electron microscopy (EM) immunostaining that, in addition to CK 18-containing filaments, the BCs express IF type IV: the neurofilaments (NFs) which, as generally acknowledged, characterize the mature neurons. A combination of optimal morphological preservation and efficent immunolabeling of all cytoskeletal elements enabled us to recognize that NF and CK 18 IFs each have a distinct compartmentalized distribution in the cytoplasm.
The presence of neuron-specific IFs in BCs is a new characteristic that adds support to their putative function as receptor cells.
Materials and Methods
Animals and Tissue Preparation
The gallbladders of six adult mice (male, NMRI breed; Hannover) and the stomachs of six male adult Wistar rats (200–250 g bw) were used. The animals were decapitated and the organs were quickly removed in toto and immersed in the fixative. Tissue strips were then excised from both organs in the regions known to be rich in BCs (Luciano and Reale 1990; Luciano et al. 1993). They were transferred into fresh fixative solution, cut into small blocks, and further fixed at room temperature (RT) as follows: (a) for 4 hr in 4% formaldehyde freshly prepared from paraformaldehyde according to Karnovsky (1965) diluted in PBS, pH 7.4; (b) for 2 hr in 3% glutaraldehyde in 0.1 M sodium cacodylate-HCl buffer, pH 7.3, followed by 45-min treatment in the dark at RT with 1% tannic acid (Mallinckrodt; St Louis, MO) dissolved in cacodylate buffer, washed in the same buffer to which 1% sodium sulfate (Merck; Darmstadt, Germany) was added and further treated with 1% aqueous uranyl acetate (Merck) for 30 min (schedule modified from Phend et al. 1995); (c) for 2 hr in 3% glutaraldehyde in cacodylate buffer as above. The specimens fixed in a and b were then processed directly for embedding, while the specimens fixed in c were postfixed for 90 min either in cacodylate-buffered 2% osmium tetroxide or in 1% ferrocyanide-reduced osmium tetroxide (Karnovsky 1971).
The formaldehyde-fixed specimens (a) were washed in PBS, dehydrated in ascending concentrations of ethanol, and embedded either in the paraffin equivalent Histocomp (Vogel; Giessen, Germany) or in epoxy resin (Serva; Heidelberg, Germany). The specimens fixed in glutaraldehyde (b) and (c) were washed in cacodylate buffer, dehydrated in ethanol, and embedded in epoxy resin.
For LM, immunostaining was applied on both paraffin and epoxy (semithin) sections. Once the presence of BCs and their successful immunolabeling had been ascertained on semithin sections, parallel thin sections were prepared from the same tissue blocks and processed for immunogold EM.
Primary Antibodies
The rabbit polyclonal antiserum cocktail to neurofilaments (cat. no. NA 1297; batch Z02787; Biotrend Chemikalien, Köln, Germany), the mouse monoclonal anti-peripherin (clone PJM50; Novocastra Laboratories, Newcastle, UK), anti-cytokeratin peptide 18 (clone Ks 18.04; Progen, Heidelberg, Germany and clone CY-90; Sigma, Deisenhofen, Germany), anti-actin (clone C4; Chemicon International, Hofheim, Germany) were used. The working dilutions of primary antibodies had been assessed in preliminary tests and are indicated in Table 1.
Immunostaining Procedures
Paraffin sections (about 4 μm thick) from gallbladder and stomach specimens were collected on glass slides pretreated with 3-(triethoxysilyl)propylamide (silane; Merck), dewaxed in xylene, and rehydrated in descending concentrations of ethanol. Endogenous peroxidase activity was blocked by treating the sections with 0.6% H2O2 in 96% ethanol for 20 min at RT. Later these sections were processed for antigen retrieval and immunostaining in the same way as the epoxy semithin sections (see below).
Epoxy semithin sections (about 1 μm thick) were collected on silane-treated slides, etched for 15 min in sodium ethoxide diluted to 50% with absolute ethanol, and rehydrated.
For both paraffin and epoxy sections, antigen retrieval was performed in a microwave oven (Sharp R-2S67; Sharp, Hamburg, Germany) by heating them in 0.01 M citrate buffer, pH 6.0 (Cattoretti et al. 1992) at 700 W three times for 5 min and then allowing to cool for 30 min. After washing with PBS, nonspecific protein binding was blocked using 5% bovine serum albumin (BSA) in PBS for 30 min at RT. The sections were then incubated in a humid chamber overnight at 4C with the primary antibodies diluted in PBS containing 1% BSA, as indicated in Table 1. Biotinylated (goat anti-rabbit and goat anti-mouse) secondary antibodies (Jackson Immunoresearch Laboratories; West Grove, PA) diluted 1:100 in PBS containing 1% BSA were applied for 60 min at RT, followed by 30-min treatment with streptavidin conjugated with peroxidase (Jackson Immunoresearch Laboratories) diluted 1:100 in PBS. Peroxidase labeling was visualized with 3,3′diaminobenzidine (Sigma) as chromogen. After washing in PBS and dehydration in ethanols, the sections were mounted in Entellan (Merck) and examined with a Leitz Orthoplan microscope.
Thin sections (about 70 nm thick) were collected on nickel grids. Etching and antigen retrieval were performed as described elsewhere (Groos et al. 2001). In brief, after 10-sec corrosion of the section surface (etching) in saturated sodium ethoxide diluted to 50% with absolute ethanol, the sections were rehydrated and processed for antigen retrieval by heating them at 95C for 15 min and then cooling to 21C at a rate of 0.04C/sec in a thermocycler UNO-Thermoblock (Biometra; Göttingen, Germany). Incubation with the primary antibodies was carried out in a humid chamber overnight at 4C at the dilutions indicated in Table 1.
Dilutions of primary antibodies a
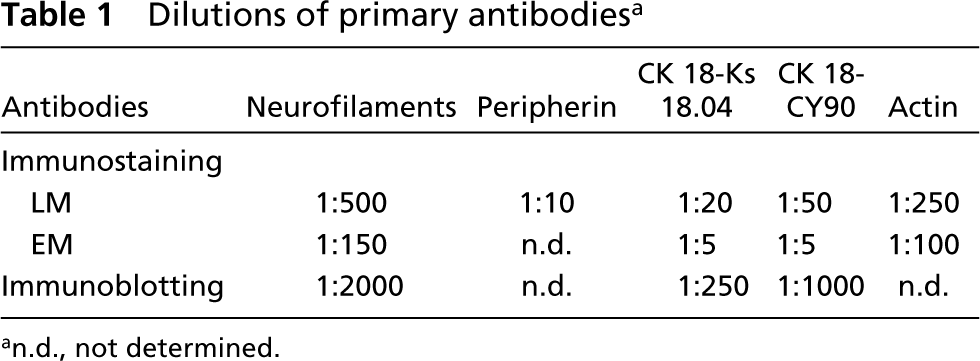
an.d., not determined.
For all sections, after washing in PBS the appropriate secondary antibodies, goat anti-mouse and goat anti-rabbit, both conjugated with 10-nm gold particles (British BioCell; Cardiff, UK), were applied for 60 min at RT.
The immunoreaction was stabilized with 2.5% glutaraldehyde in PBS for 10 min (Merighi 1992). The sections were stained with uranyl acetate and lead citrate. The electron microscope used was a Siemens Elmiskop 101.
Double labeling was performed on thin sections to detect NFs and F-actin as well as NFs and CK 18. For this purpose, the sections on nickel grids were processed in the same way as described previously except that both primary and both secondary antibodies were present simultaneously in the incubation solutions and that the binding of anti-actin and anti-CK 18 antibodies was detected with a goat anti-mouse IgG conjugated with 5-nm gold particles (British BioCell).
Controls
Negative Controls. The primary antibodies were either omitted from the incubation solution or substituted by normal mouse or preimmune rabbit sera (Jackson Immunoresearch Laboratories) at the same concentrations as the primary antibodies.
Positive Controls. The specificity of the NF immunoreaction was tested on thin sections of the rat cerebellum from tissue blocks fixed according to procedure c and processed in parallel with thin sections from gallbladder and stomach specimens.
Immunoblotting
The antibodies detecting IFs in BCs by immunohistochemistry were also tested for specificity and species crossreactivity in Western blotting analysis. HeLa cells, which are known to contain a high amount of CK 18 and from which the immunogen for antibody Ks 18.04 had been derived, served as positive control.
Procedure. HeLa cells, whole gallbladders, continuous strips of the stomach severed circumferentially along the stomach ridge (at the transition zone between pre- and glandular stomach; see Luciano et al. 1993), and samples of mouse and rat cerebellum were homogenized in 25 mM Tris-HCl buffer, pH 8.0, containing 2.5% sodium dodecyl sulfate (SDS), 12.5% glycerin, 0.5 mM EDTA, and 2.5% mercaptoethanol. Proteins were separated in 9–19% gradient SDS-PAGE and electrophoretically transferred onto Protran nitrocellulose membranes (Schleicher & Schuell; Dassel, Germany). Nonspecific protein binding was blocked for 1 hr at RT with 5% nonfat dry milk dissolved in PBS to which 0.1% Tween-20 was added (PBS-Tween). The blots were incubated for 1 hr at RT with the primary antibodies, diluted as indicated in Table 1, in PBS-Tween containing 3% BSA. They were then treated with horseradish peroxidase-labeled goat anti-mouse and anti-rabbit immunoglobulins (Cappel, ICN; Eschwege, Germany) diluted 1:1000 in PBS-Tween and reacted with ECL Western blotting detection reagent (Amersham Pharmacia; Little Chalfont, UK). The chemiluminescent signals were detected on Hyperfilm (Amersham).
Results
Immunodetection of NFs
In both paraffin sections and epoxy resin semithin sections, the BCs of the mouse gallbladder (Figures 1a–1d) and rat stomach (Figures 1e–1g) showed distinctive specific staining after incubation with antibody against NFs. In the gallbladder, the staining was prominent in the supranuclear region of the BCs where, in longitudinal sections, it appeared in the form of dark, parallel rows directed apicobasally (Figures 1a–1c). This localization clearly indicates that the NFs were intermingled and ordered between the F-actin and microtubule bundles that characterize this cytoplasmic region of BCs. Serial semithin sections revealed that the immunostaining further delineated the periphery of the cell (Figures 1d and 1d 1), but this was only just visible in its basal cytoplasmatic processes and, moreover, only when BCs had lost contact with the lumen (Figure 1d 1). In contrast, in the stomach of the rat, the many BCs present in the epithelium lining the distal slope of the ridge (Luciano and Reale 1992) were immunolabeled in their whole length, independent of the section thickness and embedding medium (paraffin or epoxy resin; Figures 1e–1g). However, in semithin sections the row-like localization of NF in the apical cytoplasm was very distinct (Figures 1f and 1g). The immunolabeling in the subnuclear region, and especially in the BC basal processes, was exceptionally clear (Figure 1g).
In EM, the highly specific immunogold localization of F-actin was a major criterion that helped to identify, at least initially, the IF bundles intermixed with the actin and microtubule assemblies in the apical cytoplasm (Figure 2a). In fact, F-actin immunolocalization, by leaving the IF bundles unlabeled, made the differences in diameter between the two filament types immediately evident (Figure 2b) and even allowed their detection, otherwise difficult, in transverse sections of BC apical cytoplasm (Figure 2c). When thin sections, parallel to those used to demonstrate immunostaining of F-actin, were treated with the rabbit polyclonal antiserum cocktail to NFs, the IF bundles became positively labeled whereas the F-actin bundles remained negative (compare Figure 2b with Figure 2d).
These results were confirmed in double-labeling experiments (Figure 2e), which further revealed the complex interlacing between F-actin and NFs with microtubules in some areas of the BC apical cytoplasm (Figure 2e). At the cell margins, around the nucleus, and below it, a large number of IFs organized in bundles of different thickness and orientation were present. However, in these regions they remained mostly negative after application of the antibody against NFs (see below).
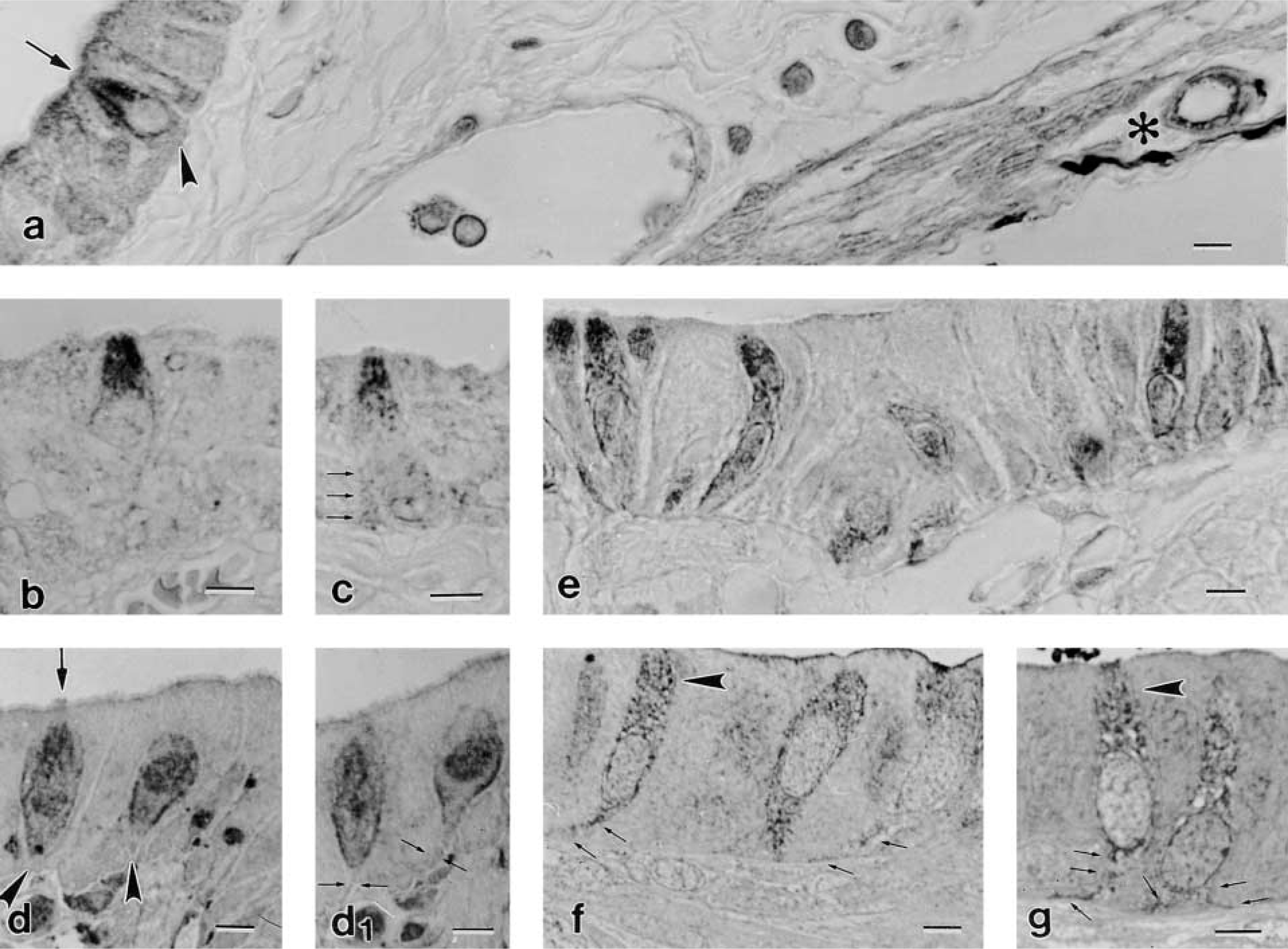
LM immunolocalization of NFs in BCs in paraffin sections (specimen fixation a: (
Negative Control. After incubation in the absence of the primary antibody, all LM sections (paraffin and epoxy semithin) as well as thin sections (Figure 3a) remained unlabeled.
Positive Control. The first indication of the specificity of the reaction was already given by LM sections showing a positive stain of both BCs and small nerve fibers in the gallbladder wall (Figure 1a) or BCs and small assemblies of ganglionic cells in the submucosa of the rat stomach (not shown). Nevertheless, final proof was provided by EM immunolabeling in sections of rat cerebellum, showing highly specific staining of axon NFs (Figure 3b).
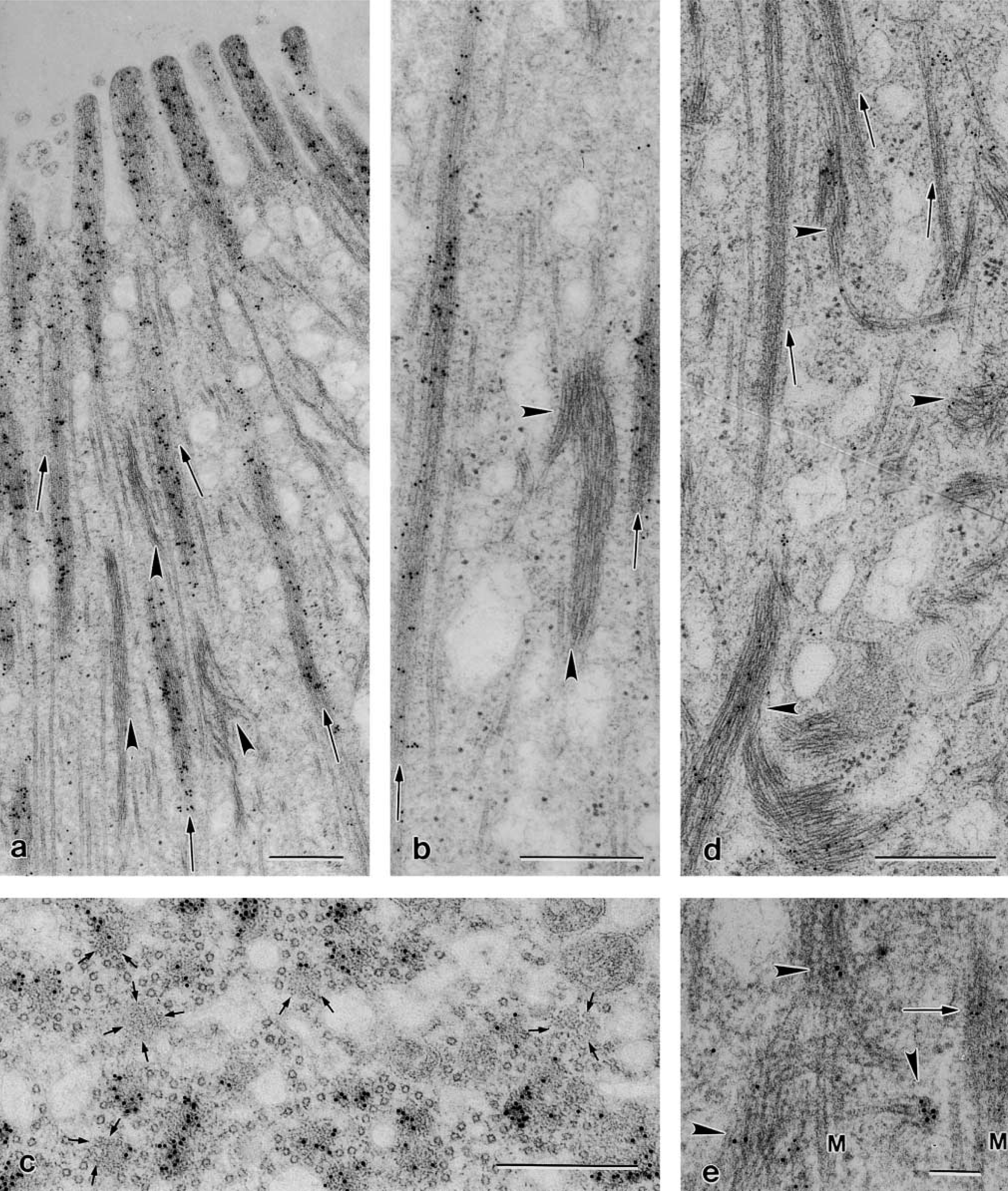
Thin sections showing immunogold labeling in apical cytoplasm of BCs. Gallbladder specimens fixed in c. (
(
Immunodetection of Peripherin
The use of the mouse monoclonal antibody against peripherin protein failed to detect BCs (Figures 4a and 4b). The epithelium covering the stomach-limiting ridge in which many BCs occur was always negative, and thus were all epithelial cells lining the stomach glands (Figure 4b). In contrast, peripherin antibody labeled small assemblies of ganglion cells present in the stomach submucosa in the same section (Figures 4a and 4c) as well as the ganglia lining the muscle sheets in both the gallbladder and stomach (not shown).
Immunodetection of CK 18-containing Filaments
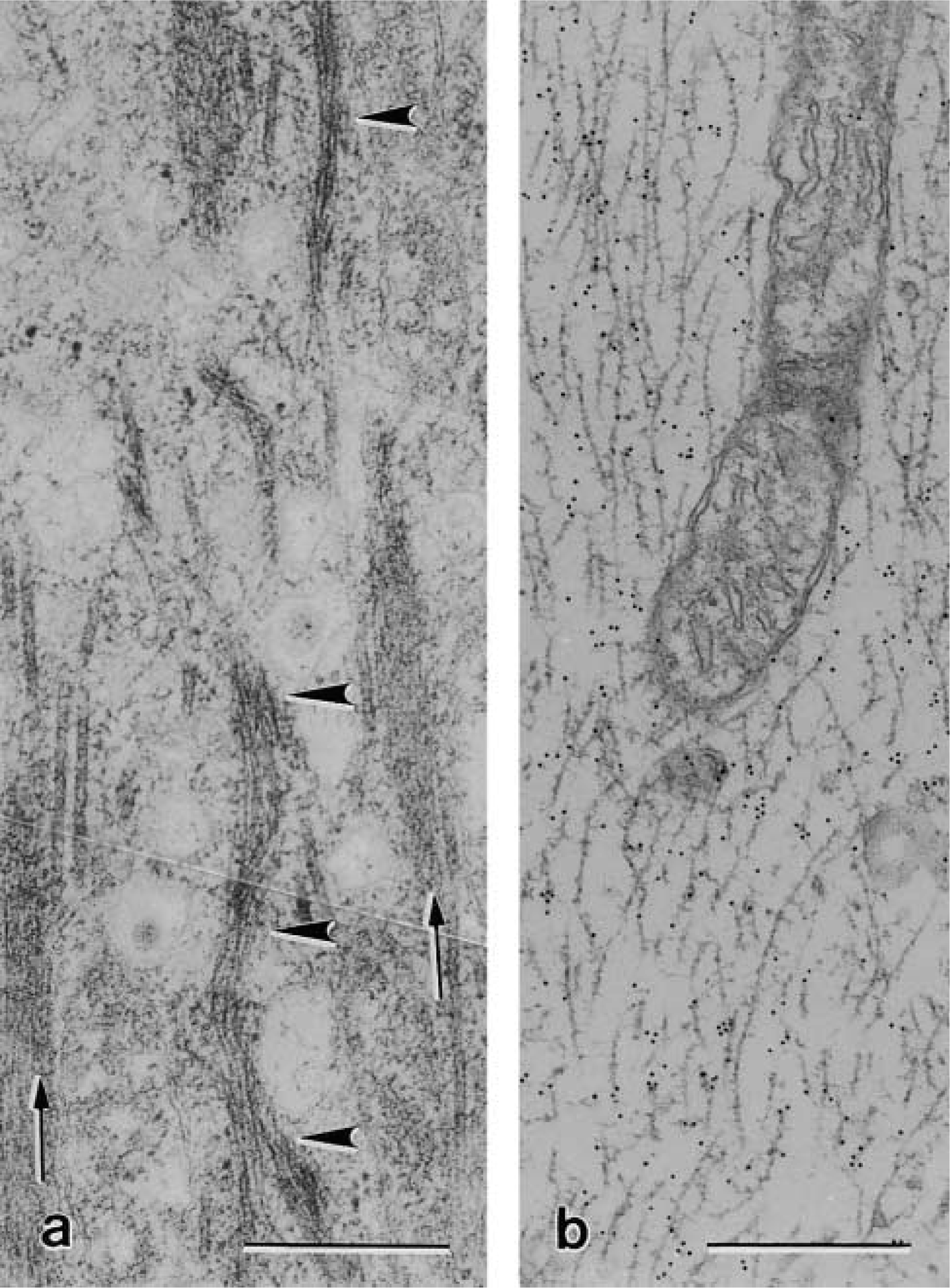
The two antibodies against CK peptide 18 applied in this study (Table 1) showed differences in species crossreactivity. The mouse monoclonal anti-CK 18 (clone CY 90) stained BCs only in the rat stomach and not in the mouse gallbladder (results not shown). In contrast, independent of the procedure with which the specimens were fixed and embedded, the mouse monoclonal anti-CK 18 clone Ks 18.04 labeled BCs in both mouse gallbladder and rat stomach (Figures 5a and 5b). The immunoreaction in these organs was highly specific so that the BC shape was brought up among neighboring epithelial cells (Figure 5a and 5b). The staining especially delineated the cell borders and the basal processes (Figure 5b). The application of this same antibody on thin sections revealed that CK 18-positive bundles had a distinctive distribution in the cytoplasm. They were excluded in the region where F-actin, microtubules, and NFs dominate. In contrast, favorable section planes grazing BCs along a median plane enabled recognition that CK 18-positive bundles almost exclusively formed the prominent network that surrounds the nucleus (Figure 6a). However, and in agreement with LM observations, the double-labeling procedure demonstrated that near the lateral cell borders CK 18-positive bundles co-localized with NFs (Figures 6c and 6d). The efficiency of the double labeling was highly dependent on the thin section plane, transverse or oblique to the filament bundles, there being more epitopes exposed in an oblique plane (Kellenberger et al. 1987). Bundles of CK 18-positive filaments were frequently observed to converge toward the unlabeled desmosomes (Figure 6b).
Immunoblotting
In Western blotting analysis, the rabbit polyclonal antiserum cocktail to NFs detected bands at about 68–70, 150, and 200–210 kD in both mouse and rat brain extracts (Figure 7a). These bands corresponded to the low, medium, and heavy molecular weight of NF triplet proteins, thus demonstrating the species crossreactivity of the antibody for all three NF protein isoforms. The specificity and species crossreactivity of the two antibodies directed against CK 18 peptide were confirmed in Western blotting experiments. The monoclonal anti-CK 18 clone Ks 18.04 labeled a band of molecular weight slightly over 45 kD in HeLa cells, in mouse gallbladder, and in rat stomach (Figure 7b). In contrast, the anti-CK 18 clone CY 90 detected a corresponding band only in HeLa cells and in rat stomach but not in mouse gallbladder (Figure 7c), indicating, in parallel with the immunohistochemical results, differences in species crossreactivity.
Discussion
The Cytoskeletal Network of BCs
It is rare for all three basic components of the cytoskeleton of eukaryotic cells—microtubules, F-actin, and IF—to be so abundantly present and with a so highly ordered distribution in cytoplasm as in BCs. Many F-actin assemblies extend from the apex of the microvilli deep into the supranuclear region, where they become flanked by microtubule assemblies. This alternating arrangement of F-actin and microtubules, particularly evident in longitudinal sections of the cell, entails all organelles in between assuming the same apicobasal orientation and bestows the BC with its typical stiff aspect (Luciano and Reale 1997). Concerning the IFs, they have been regarded in earlier studies (Luciano and Reale 1969) as the most dominant structures in the regions around and below the nucleus.
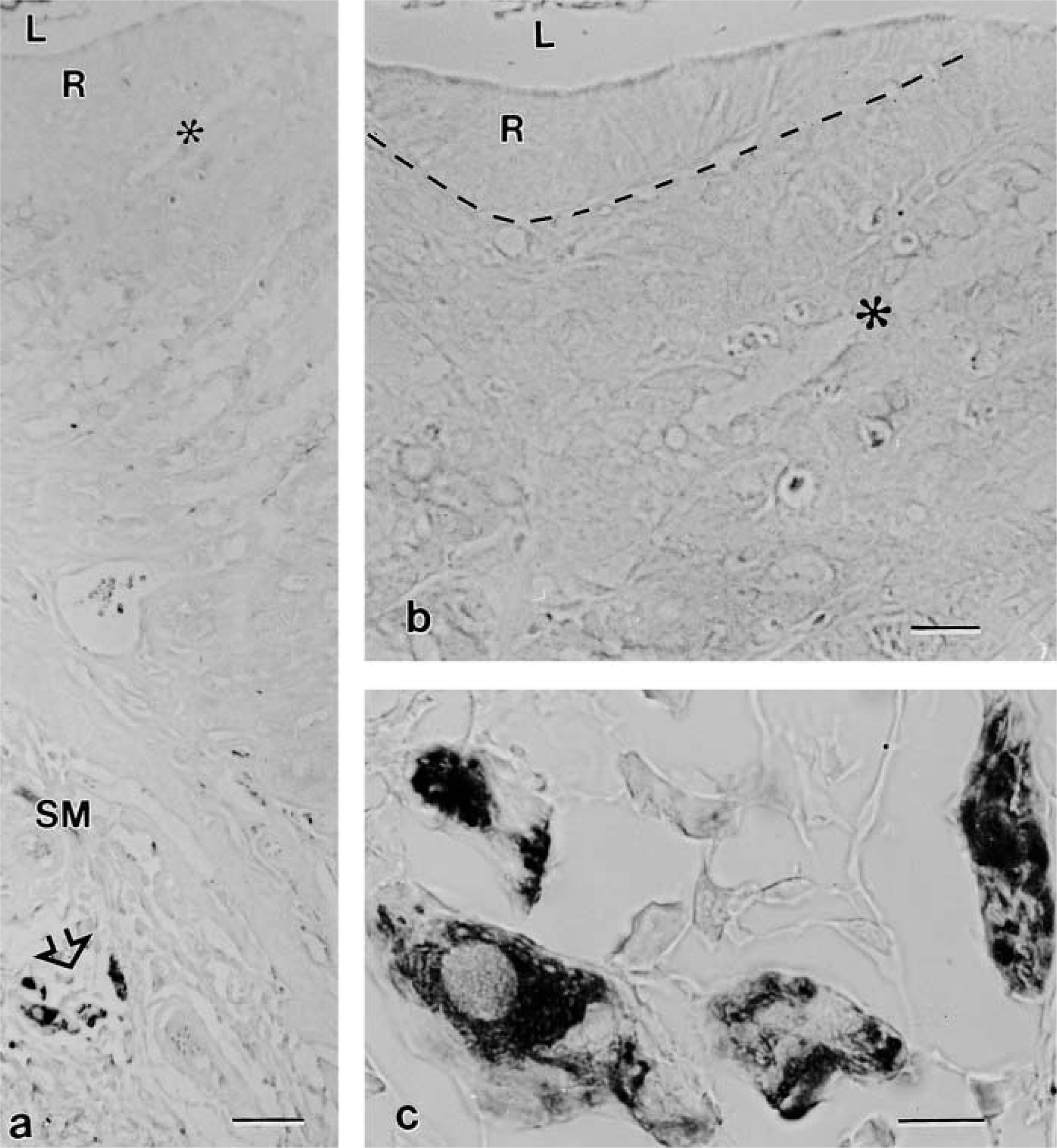
LM immunolocalization of peripherin in paraffin sections of stomach specimens fixed in a. The section crosses the stomach wall in the region of the limiting ridge (R) from the lumen (L) of the organ to the submucosa (SM). A positive reaction is visible only in some ganglion cells (open arrow), which are shown at higher magnification in
The cytoskeletal elements are known to be, in general, very delicate structures, subjected both in vivo and in vitro to alterations such as depolymerization, disassembly, and/or unraveling, being greatly influenced by chemical, thermal, and mechanical stress (reviewed by Herrmann and Aebi 1999). Our previous studies have shown that the cytoskeleton of BCs is strongly influenced by fixatives, buffers, and the osmolarity of the solution used (Luciano et al. 1981). Therefore, the structural preservation of cytoskeletal elements with their specific distribution in the cytoplasm of BCs was an essential prerequisite of this study. However, the treatment of the tissue specimens with suitable fixatives and embedding media can generate serious drawbacks if an immunotechnique is to follow. Therefore, we applied the LM and EM immunodetection procedure recently proposed for epoxy resin sections (Groos et al. 2001) that fulfills the requirements of good morphology and efficient immunolabeling. Even in conventionally glutaraldehyde prefixed and postosmicated specimens, it was possible to detect the occurrence of two types of IF in the cytoplasm of the BCs in two different organs and animal species.
Compartmentalized Distribution of IFs in the Cytoplasm of BCs
Immunodetection of NFs. This study clearly demonstrates the presence of NFs in BCs. Western blotting analysis and immunostaining of thin sections of rat cerebellum (positive control) confirm the specificity of the rabbit antiserum cocktail to NFs used.
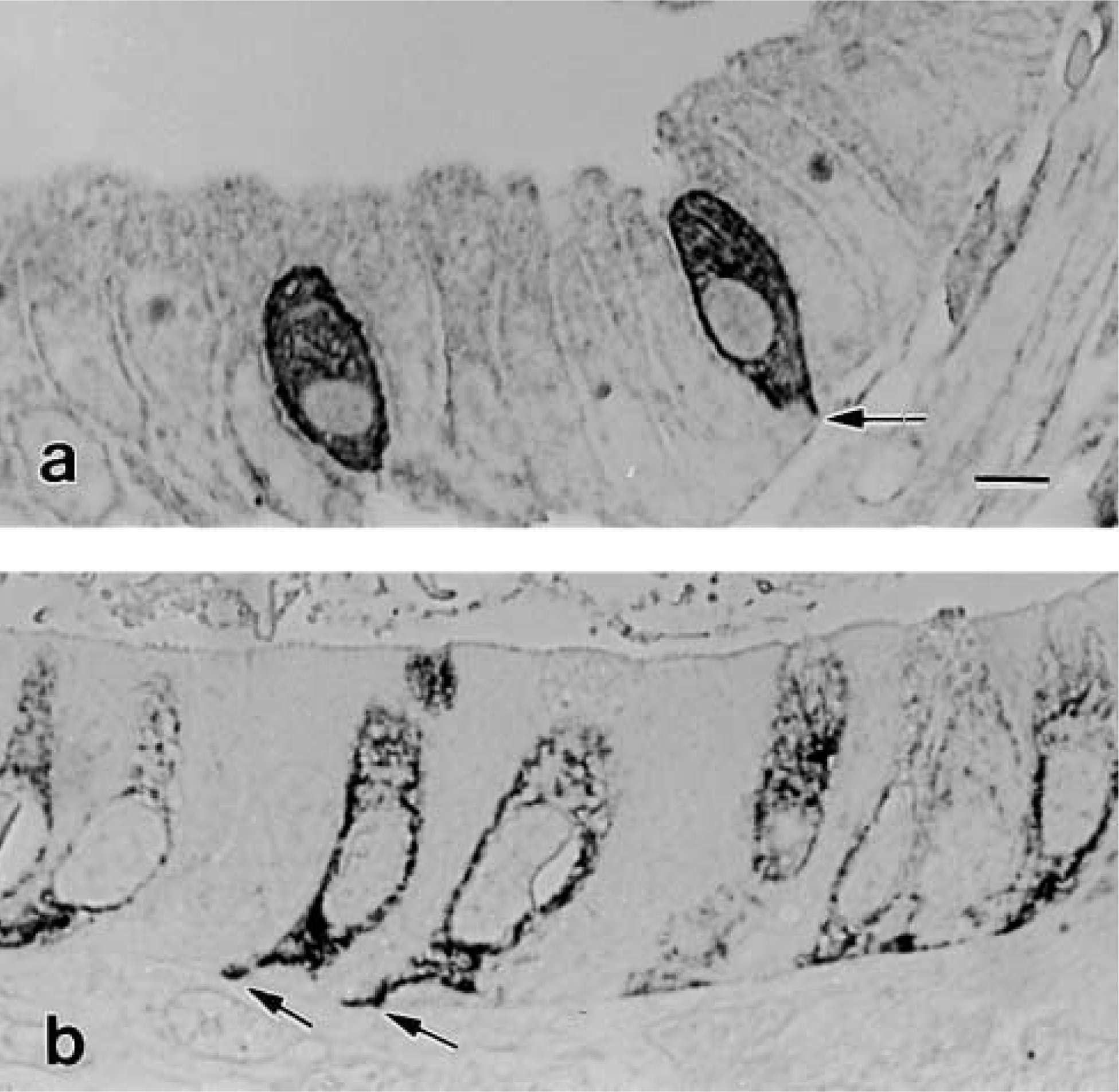
LM immunolocalization of cytokeratins in epoxy semithin section from specimens fixed in b: mouse gallbladder (
Immunogold labeling reveals that bundles of NFs are mainly intermingled with actin and microtubules in the apical cytoplasm, where they assume the same orientation. This location and arrangement, already predictable from the row-like labeling observed in LM, was at first surprising. In fact, the presence of IFs at this location had escaped notice in previous morphological studies. Conceivably, attention was distracted by the prominent number of F-actin and microtubule assemblies occurring here.
The presence of NFs in the BC cytoskeleton is of biological significance. It implies that the cell is able to synthesize this type of IF, a property generally considered to be restricted to mature neurons (Hirokawa 1993). In fact, neuronal IFs are developmental stage-specific or -dependent and cell type-specific in differentiated neurons (Ching et al. 1993; Coulombe et al. 2001). This concept is not consistent with the presence of NFs in BCs unless it can be assumed that BCs had acquired the ability to synthesize NFs during development and that, once differentiated, they can work as sensory receptive cells despite being located in common epithelia. However, BCs do have several morphological characteristics that can be paradigmatically related to those of well-established sensory cells, such as Merkel cells and Pacinian corpuscles (Luciano and Reale 1990). In addition, like the hair cells of the mammalian inner ear and photoreceptors, BCs maintain structural polarity after isolation (Luciano et al. 1993). Similar to some (sensory) neurons, BCs express nitric oxide (NO) because they react with antibodies directed against neuronal NO synthase (Kugler et al. 1994).
With the detection of NFs the BCs acquire a characteristic property of neurons (Hirokawa and Takeda 1998) and, although irregularly, of some types of hair cells in the sensory epithelia of the inner ear (Dechesne et al. 1994, and literature therein).
In neurons the NFs are synthesized in the cell body and transported to the axon, but the mechanism of transport is controversial (Brady 2000; reviewed by Chou and Goldman 2000). Our present knowledge of the function of NFs comes largely from investigations of the axon cytoskeleton. Studies in genetically engineered animals have demonstrated that NFs in the axon act to maintain the structural integrity of the cytoskeleton, are responsible for radial growth, and thus determine axon diameter (reviewed by Hirokawa and Takeda 1998). Therefore, in neurons, in addition to structural scaffolding, IFs fulfill cell type-specific functions (Coulombe et al. 2000). With the mere presence of NFs as a component of the BC cytoskeleton, it is too early to speculate on their function. However, it is interesting that, on the basis of resemblance analogy, the cytoskeletal elements, and in particular their arrangement in the apical cytoplasm of BCs, have close structural similarities to the organization of the axon cytoskeleton with regard to straight arrangement of microtubules and NFs (Hirokawa 1993), whereas the assemblies of F-actin strongly recall those in the hair cells of the inner ear (Tilney et al. 1992). NFs are also present at the BC margins directly below the lateral plasma membrane as well as in the BC basal branches. An analogue localization of NFs at the cell border can be observed in type I vestibular hair cells (see Figure 3 in Dechesne et al. 1994).
Further studies are needed to determine whether there are functional relatives beyond the structural analogies in cytoskeletal organization between BCs, axons, and hair cells.
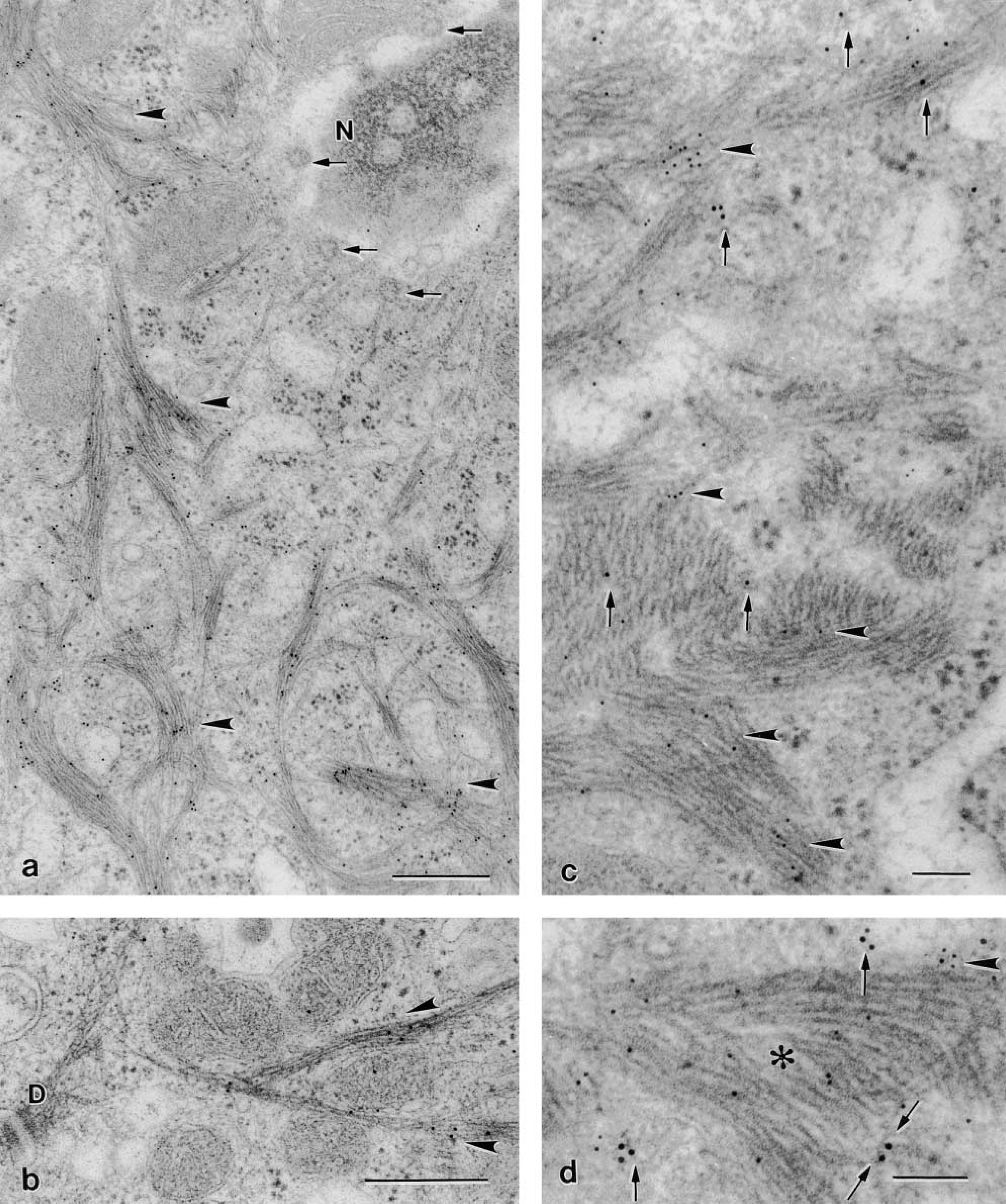
Thin section immunogold labeling in BCs of mouse gallbladder specimens (fixation c). (
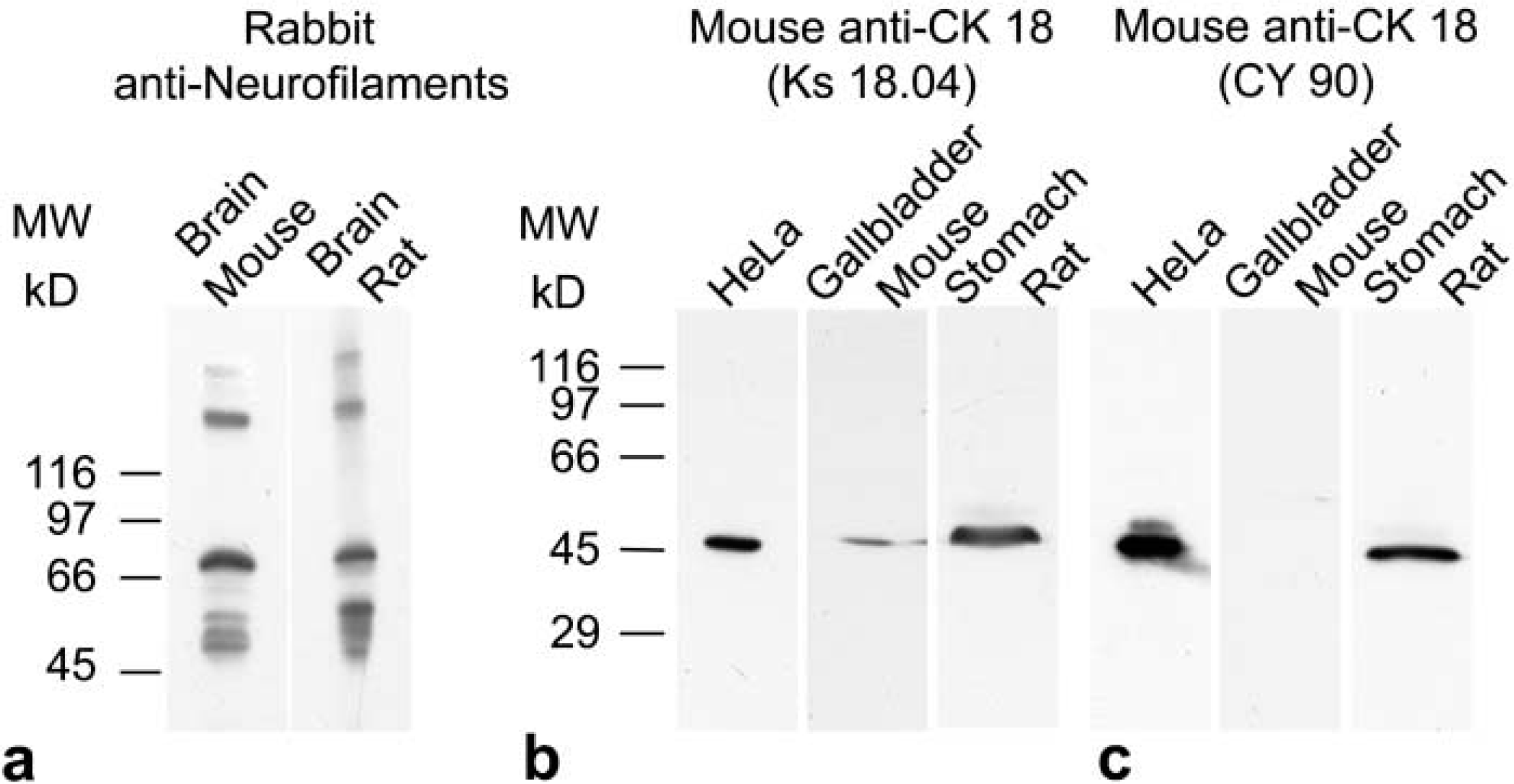
Western blotting analysis showing specificity and species cross-reactivity of antibodies against NFs and CK 18. (
Immunodetection of Peripherin. Peripherin is an IF protein first described in neuroblastoma cells and subsequently found to be expressed mainly in neurons of the peripheral nervous system (Portier et al. 1983–1984) including myenteric ganglion cells during pre-and postnatal life of the rat (Matini et al. 1997). Our results demonstrate that BCs did not express peripherin because the epithelium covering the stomach-limiting ridge, regarded as a warrant for the presence of BCs, was always negative, whereas the ganglion cells in the stomach wall were peripherin-immunoreactive. Antibodies against peripherin were used during development and in adult rats for the immunohistochemical characterization of spiral and vestibular ganglion cell bodies and their projections directed to the membranous labyrinth (Hafidi et al. 1993; Hafidi 1998; Leonard and Kevetter 2002). The results of these studies showed strong peripherin immunostaining of the fiber bundles directly innervating the sensory epithelia. However, the hair cells type I and II of the vestibular labyrinth and the inner and outer hair cells of the organ of Corti remained unstained. The lack of peripherin expression in BC is apparently an additional finding that associates BCs to the sensory cells.
Immunodetection of CK 18. CK 18 belongs to the group of acidic keratins type I IF (Herrmann and Aebi 2000) that has been shown to be expressed in BCs of epithelia covering different organs of the GI tract (Kasper et al. 1994). Because of the strong immunoreaction that BCs display after application of antibodies directed against CK peptide 18, these have been proposed and used as specific LM markers to identify BCs in various epithelia (Höfer and Drenckhahn 1996; Gebert et al. 2000), although there are differences in species crossreactivity between antibody clones (Gebert et al. 2000; and this study).
EM immunogold labeling reveals that CK 18-positive IFs were absent in the apical cytoplasm where NFs, microtubules, and F-actin assemblies occur, although, from LM sections one would be induced to think so, the stain being mainly distributed over the whole cell. Assemblies of CK 18-positive filaments form, in contrast, the bulk of cytoskeletal elements in the perinuclear region, and in places they show close juxtaposition and even linkage with the nuclear envelope. CK 18-containing filaments and NFs are regularly co-expressed at the lateral cell margins and in the BC basal processes.
In recent years, growing evidence from studies using the green fluorescent protein-tagged IF fusion proteins, had led to the interpretation that these polymers are highly dynamic structures, able to assemble and disassemble, and to convert into protofilamentous aggregates. Their constituent proteins can move rapidly throughout the cytoplasm during various cell activities (Chou et al. 2001; Coulombe et al. 2001). This property may give new insight into how the basal branches of BCs, which are highly variable structures mostly detectable only using serial sections (Luciano and Reale 1990), different from BC to BC, may change their thickness and length and vary their course within the epithelium.
On the other hand, among IF types, cytokeratins are considered as major promoters of a specific epithelial cytoarchitecture and protectors against mechanical stress (Herrmann and Aebi 2000; Coulombe et al. 2000). According to this role, the network of cytokeratin bundles extending from the cell periphery toward the nucleus could provide the BCs with a stable scaffold that protects against mechanical stress and maintains intact cell shape. Our previous studies have demonstrated that BCs retain their morphological polarity in the dissociated state, i.e., after abolishment of intercellular contacts and up to 2 hr after permanent mechanical stress, such as shaking and repeated centrifugation, which had been necessary for their isolation (Luciano et al. 1993).
Co-expression of NF- and CK 18-containing Filaments in BCs
IFs are subdivided into six distinct types, which differ in the number of the genes that regulate their function, in sequence type, assembly group, molecular weight, and distribution in various tissues (Coulombe et al. 2001). IFs are believed to be cell type-specific. For example, cytokeratins are characteristic of the epithelia as NFs are of the mature neurons (Coulombe et al. 2001). A review of the literature reveals that, unlike during tissue development, the co-expression of different IF types in already differentiated cells is mostly regarded as an unusual event, especially when the co-expression involves the NFs.
Under normal physiological conditions and in adult animals, co-expression of NFs and IF type III vimentin has thus far been immunohistochemically demonstrated in the horizontal cells of the mouse retina (Dräger 1983; Shaw and Weber 1984), and in the type I vestibular hair cells of the guinea pig inner ear (Dechesne et al. 1994).
Under pathological conditions however, a co-expression of NFs and cytokeratins has been repeatedly reported. It occurs, for example, in tissue malignancies such as endocrine carcinomas of the respiratory and gastrointestinal tracts (Van Muijen et al. 1984; Lehto et al. 1985; Miettinen et al. 1985), in Merkel cell tumors (Gould et al. 1985; Van Muijen et al. 1985; Merot et al. 1986), and in a cultured cell line, PC12, derived from rat adrenal pheochromocytoma and frequently used as an in vitro model system for neuronlike cell differentiation (Franke et al. 1986).
In the present study, in contrast, we demonstrated the co-existence of NF- and CK 18-containing filaments in normal and fully differentiated cells residing in common epithelia. This unexpected co-existence in BCs could reflect a functional requirement for tasks that are divergent enough to be fulfilled by only one type of IF. This possibility may, in turn, imply that some cells, including BCs, can retain the ability to synthesize IFs in different and more convenient molecular configurations, according to their functional needs. Conceivably, the co-expression of NF- and CK 18-containing filaments goes beyond the context of the BC function, assuming much more general biological interest.
Conclusions
The results of the present study seem to provide a clear indication that the architecture of the BC cytoskeleton holds the secret to the way in which the cell functions. The presence of NFs in the BC cytoskeleton may add a piece to the puzzle.
New knowledge about the dynamics of cytoskeletal elements has steadily grown in recent years, and the numbers of associated proteins involved as crosslinkers and/or as motor proteins acting in transport mechanisms have progressively expanded (Baas and Ahmad 2001; Chou et al. 2001). The peculiar arrangement of the cytoskeletal elements (microtubules, F-actin, and NFs), as well as that of the cell organelles in between that characterize the apical cytoplasm of BCs, appear to suggest that these structures are engaged in a transport process. Future studies aiming to determine whether the cytoskeleton of BCs also possesses the repertoire of associated proteins needed elsewhere for the functional dynamics of cytoskeletal elements (Baas and Ahmad 2001) could drastically change our present notion of an apparently “rigid” BC cytoskeleton to that of a highly dynamic compartment.
Footnotes
Acknowledgements
We wish to thank Prof Dr E. Ungewickell for critical reading of the manuscript, Mr F. Hurkuck for excellent technical assistance, Ms A. Hundt for photographic work, and Ms S. Fryk for linguistic corrections. The generous gift of anti-peripherin antibody by Ms A. Michalski (Loxo; Dossenheim, Germany) is greatly appreciated.
