Abstract
In this study we compared the immunohistochemically quantified fiber type area with the myosin heavy chain (MyHC) contents of a bundle of fibers from a human masticatory muscle. The total cross-sectional areas were determined immunohistochemically for the three major fiber types (I, IIA, and IIX) in bundles of fibers (n = 42) taken from the anterior and posterior belly of the human digastric muscle (n = 7). The relative MyHC contents of the same fiber bundles were determined electrophoretically (MyHC-I, -IIA, and -IIX; anterior, 32%, 35%, and 33%; posterior, 39%, 42%, and 19%) and compared with the immunohistochemical data (MyHC-I, -IIA, and -IIX; anterior, 32%, 31%, and 37%; posterior, 39%, 45%, and 15%). No significant differences were seen in the mean fiber type distribution between the two techniques; the correlation coefficient ranged from 0.71 to 0.96. The correlation coefficient was higher for MyHC type I and MyHC type IIX than for MyHC type IIA. The MyHC contents of single fibers taken from the posterior belly indicated that many fibers in this belly co-express MyHC-IIA and MyHC-IIX. Despite the presence of these hybrid fibers, the correspondence between both methods was relatively large.
H
To describe the fiber type composition of various muscles, investigators calculated the relative number or the cross-sectional area that a particular fiber type occupies, as determined by either ATPase histochemistry (Johnson et al. 1973; Eriksson et al. 1982; Travnik et al. 1995; Monemi et al. 2000) or immunohistochemistry (Schiaffino and Reggiani 1994; Korfage et al. 2000), or by a combination of the two methods (Sciote et al. 1994; Zhou et al. 1995). Another technique is to separate the MyHC isoforms by SDS-PAGE and then to calculate the integrated density of the bands (Adams et al. 1993). This technique allows the determination of the MyHC contents of a muscle even in muscles that are difficult to cut, such as diseased muscles.
Some studies have made quantitative comparisons between the relative area, or the relative fiber type distribution, vs the MyHC contents determined by gel electrophoresis in human muscle biopsy specimens (Perrie and Bumford 1986; Adams et al. 1993; Fry et al. 1994) or in single fibers (Staron 1991; Staron and Hikida 1992; Sant'Ana Pereira et al. 1995). In general, a good correlation between these two methods was found. These studies, however, investigated limb muscles (in particular the vastus lateralis muscle) and used ATPase histochemistry to determine the fiber types. ATPase histochemistry can lead to a mismatch of fiber types, especially among the fast fiber types (Andersen et al. 1994; Serrano et al. 2001). Furthermore, in most studies the fiber type composition was estimated on a small portion of the investigated biopsy, whereas the gel electrophoresis was performed on the total cross-section of the biopsy. A recent study included all fibers of a fiber bundle (human vastus lateralis muscle) and related the methods of immunohistochemistry with gel electrophoresis (Serrano et al. 2001).
To our knowledge, a comparison of the fiber type area of all the fibers, as determined by immunohistochemistry, and the MyHC contents, as determined by gel electrophoresis, has not been performed on whole bundles of muscle fibers in human masticatory muscles. The masticatory muscles are different from limb and trunk muscles. Their fibers are four to five times smaller than those in limb and trunk muscles (Staron and Hikida 1992), and often the type II fibers are smaller than the type I fibers, whereas in limb and trunk muscles the opposite is true. Furthermore, the masticatory muscles contain many hybrid fibers (Korfage and Van Eijden 1999, 2000; Korfage et al. 2001).
The primary focus of the present study was to compare the cross-sectional area of a certain fiber type in a bundle of masticatory muscle fibers, as determined by immunohistochemistry, with its MyHC contents, as determined by gel electrophoresis. We used the digastric muscle. Both bellies of this muscle were investigated separately because they differ in fiber type composition (Eriksson et al. 1982; Korfage et al. 2000).
Materials and Methods
In this study the anterior and posterior bellies of the digastric muscle of seven white cadavers were used, four males and three females, mean age 70.3 ± 15.6 years. The individuals were without known systemic disorders. None of the muscle specimens contained signs of muscle disease on the basis of histological examination. Of these seven cadavers, five had upper and lower dental prostheses and two were partially dentate. The muscles were obtained within 12–36 hr post mortem, which was found not to influence the results greatly (Eriksson et al. 1980). After the muscles were exposed they were cut from their attachment sites. The muscles were rapidly frozen in liquid nitrogen-cooled isopentane. From each frozen belly, three bundles of fibers (approximately 5 mm long) were cut out and stored at −80C until required for further processing. In total, 42 bundles (= 7 muscles, 2 bellies, 3 bundles) were investigated. The cross-sectional area of the bundles was 2.5 ± 0.8 mm2 (mean ± SD). The number of fibers per bundle area ranged from approximately 700 to 3100. The data were grouped regardless of gender.
Immunohistochemistry
Serial transverse sections of 10 μm were cut in a cryomicrotome (Model HM 500 M; Adamas Instruments BV, Leersum, The Netherlands). The first and last 10 sections were collected for gel electrophoresis analysis. The 15 sections in between were mounted on coverslips and three of these sections were used for immunohistochemical analysis. After overnight fixation at −20C in a mixture of methanol:acetone:acetic acid:water (35:35:5:25) (Wessels et al. 1988), the sections were incubated with monoclonal antibodies raised against purified myosin (Bredman et al. 1991), i.e., antibody 219–1D1, which recognized MyHC-I, antibody 333–7H1, which recognized MyHC-IIA, and antibody 332–3D4, which recognized MyHC-IIA and MyHC-IIX. Because the human digastric muscle contains only a few fibers that express MyHC fetal or MyHC cardiac (Korfage et al. 2000), these two antibodies were not included. The indirect unconjugated immunoperoxidase technique (PAP technique) was applied to detect the specific binding of the different antibodies. Nickel-DAB was used to visualize the staining (Hancock 1982).
Sample Method and Fiber Cross-sectional Area Measurements
The three sections were photographed with a digital camera attached to a microscope and the fiber area outlines were drawn on a sheet. The fiber areas were classified by means of the three sections. We calculated the cross-sectional area of the fibers by scanning the drawn sheets, together with a grade mark for correction of enlargement into a personal computer via a flatbed scanner (Hewlett-Packard, Scanjet 4c). A custom-made program (Korfage et al. 2000) that converts the number of pixels into μm2 was then used to determine the total cross-sectional area of each muscle fiber type in μm2. The percentage cross-sectional area (csa) of MyHC type I fibers was calculated as csa type I/(csa type I + csa type IIA/IIX) 100%; of MyHC type IIA fibers as csa type IIA/(csa type I + csa type IIA/IIX) 100%; and of MyHC type IIX fibers as (csa type IIA/IIX – csa type IIA)/(csa type I + csa type IIA/IIX) 100%.
SDS-PAGE
The collected frozen sections were washed and homogenized as described by Bamman and co-workers (1999). The washed myofibrils were diluted in sample mix [15% (w/v) glycerol, 2% (w/v) DTT, 0.01% (w/v) bromophenol blue, and 1% (w/v) SDS in 62.5 mM Tris-HCl buffer, pH 6.8]. The samples were lysed for 2 min at 100C and then stored frozen (-80C) until processed for protein separation.
Gel electrophoresis was performed in high glycerol containing (30%) sodium dodecyl-sulfate polyacrylamide gels (0.75-mm thickness) using an acrylamide to bis-acrylamide ratio of 67:1 in the separating gel (9% total acrylamide, pH 8.8) and of 50:1 in the stacking gel (4% total acrylamide, pH 6.8) (modified from Talmadge and Roy 1993). The samples were run at constant current (13.5 mA) for 29 hr using a custom-built electronic timer device (pulse unit) connected to the power supply that switched the running current on and off (Sant'Ana Pereira et al. 2001). After this process the gels were silver-stained. For better quantification of the MyHC isoform bands, the gels were blue-toned according to Berson (1983). The gels were scanned with an LKB 2202 Ultrascan laser densitometer (LKB; Bromma, The Netherlands). The MyHC isoforms were identified on the basis of migration as MyHC types I, IIA, and IIX. The total integrated area of the three MyHC peaks was set to 100 and each individual area was expressed as a percentage of the total MyHC. The densitometric signal was linear over the range of MyHC concentrations loaded on the gels.
To estimate the quantification error of this scanning method of the MyHC isoform bands, 36 lanes were scanned for a second time on a different day. For each individual MyHC isoform, the mean and SD of the differences (n = 36) was determined and was, respectively, MyHC-I 0.36 ± 4.50%, MyHC-IIA −1.89 ± 9.00%, MyHC-IIX 1.52 ± 9.94%.
Single Fibers
Our antibody panel cannot distinguish hybrid fibers that co-express MyHC-IIA and MyHC-IIX from fibers that express only MyHC-IIA. To get an indication whether the digastric muscle used in this study contains hybrid fibers, we analyzed the MyHC contents of a sample of single fibers by gel electrophoresis. Because the posterior belly contains more fibers denoted by immunohistochemistry as MyHC type IIA fibers, which could co-express more MyHC-IIX than the anterior belly (Korfage et al. 2000), a part of the posterior belly of three individuals was freeze-dried. The three individuals were chosen at random. Before dissection, the muscle parts were brought, under vacuum, to room temperature (RT) over 2 hr. From these bellies, single fibers were microdissected (Essén et al. 1975). After dissection, each fiber (approximately 5 mm long) was split into two parts. One part was embedded in 15% gelatin and processed for immunohistochemistry to ascertain if the dissected fiber was indeed single. The other part was dissolved in sample mix and processed for gel electrophoresis using the same method as described above. The embedded single fibers were frozen in liquid nitrogen and cut in a cryomicrotome (10 μm). The slides were incubated with the same three antibodies as used for the determination of the fiber areas but with a modified method (Sant'Ana Pereira et al. 1995). In total, 61 single fibers were investigated.
Statistical Analyses
For each individual the mean was calculated for each MyHC fiber type area and MyHC contents over the three bundles of each belly. Then the grand mean and SD values were calculated over the means of the individuals (n = 7). The Wilcoxon ranking test for paired data was used to analyze differences between the mean fiber type area and its corresponding MyHC isoform contents within a belly, and between the anterior and posterior belly. The level of significance was set at p < 0.05. Spearman's rank correlation was used to examine the relation between MyHC contents, as determined by gel electrophoresis, vs fiber type area. The level of significance was set at p < 0.05.
Results
The fiber type areas of MyHC type I, MyHC type IIA, and MyHC type IIX differed significantly between the anterior and posterior belly (Figure 1). In the anterior belly, a larger MyHC type IIX fiber area and a smaller MyHC type IIA and MyHC type I fiber area were found than in the posterior belly.
In Figure 2 the percentage fiber type area in the anterior and posterior belly of each individual is linked to its percentage MyHC isoform contents. There was a large interindividual variation of the proportion of each fiber type area and MyHC isoform contents. For all MyHCs, no significant differences were found between the proportion of a MyHC fiber type area and its MyHC isoform contents (Table 2).
Of the 61 investigated single muscle fibers, 37 were pure fibers, expressing only one MyHC isoform, and the other 24 fibers were hybrid fibers. The most common hybrid fiber type found in these three muscles was fibers that co-expressed MyHC-IIA and MyHC-IIX (Table 1).
Discussion
Two methods can be used to quantify the fiber type composition of a muscle, i.e., to calculate either the percentage or the area that is occupied by a particular fiber type classified by ATPase histochemistry or immunohistochemistry, or to determine the MyHC contents by gel electrophoresis. An advantage of gel electrophoresis is that even in biopsies of muscles that are difficult to cut in a cryotome, e.g., in diseased material, an MyHC profile can be established.
To justify the use of one of the two methods, the correlation between these methods has to be established. A comparison between fiber types as determined by ATPase histochemistry and the MyHC contents (Perrie and Bumford 1986; Adams et al. 1993; Fry et al. 1994), and as determined by immunohistochemistry and the MyHC contents (Serrano et al. 2001), was made in human limb and trunk muscle biopsies and also in single fibers (Staron 1991; Staron and Hikida 1992; Sant'Ana Pereira et al. 1995). A comparison of fiber type composition determined by immunohistochemistry and gel electrophoresis in human masticatory muscles has, to our knowledge, not been previously performed. In most of the aforementioned studies the vastus lateralis muscle was investigated, in which the distribution of the fiber types in a biopsy was estimated by calculating the cross-sectional area for the different fiber types on approximately 50 fibers. The gel electrophoresis, however, was done on all fibers of the biopsy specimen. The study by Serrano and co-workers (2001) and the present study are, in this respect, more accurate because the total area of all fibers of a particular fiber type was measured.
The correlation coefficients for ATPase histochemistry vs gel electrophoresis in the vastus lateralis muscle found in the literature ranged from 0.71 to 0.87 (Adams et al. 1993; Fry et al. 1994) and for immunohistochemistry vs gel electrophoresis from 0.66 to 0.88 (Staron et al. 2000; Serrano et al. 2001). The correlation coefficients in the present study (range 0.71–0.93) are comparable to those found in these studies.
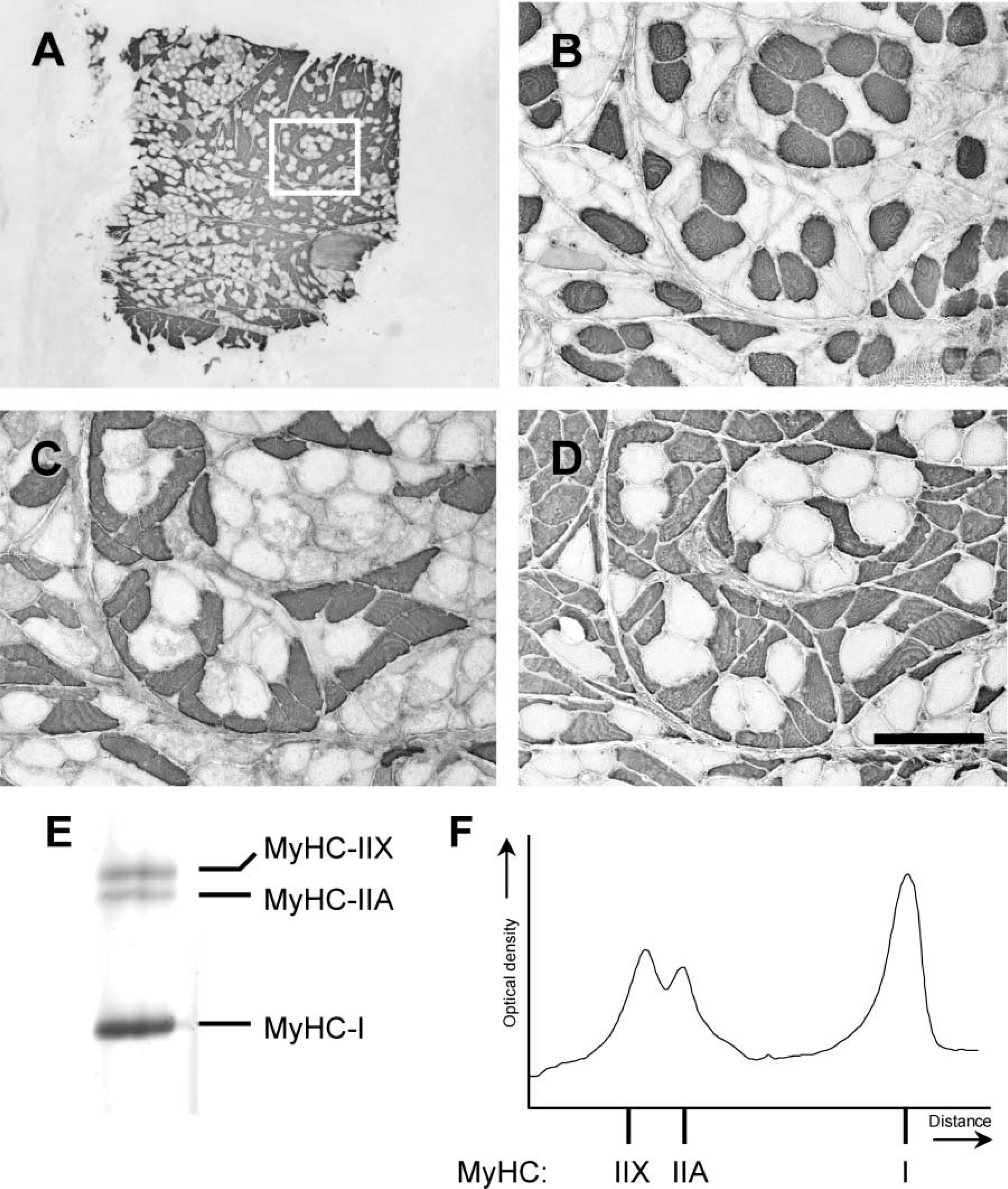
Example of a cross-section of a fiber bundle from the anterior belly of the digastric muscle (
The observed differences between the two methods might be explained as follows. First, the concentration of myosin in a fiber differs between type I and type II fibers. In rabbit, it was found that type I fibers from the soleus contained approximately 22% less myosin than type IIX fibers from the psoas (Tikunov et al. 2001), and in chicken the red portion of the latissimus dorsi muscle contained 25% less myosin than its white portion (Everett et al. 1983). Greater volume of a slow muscle fiber was occupied by mitochondria and sarcoplasmic reticulum than in a fast fiber (Rome and Lindstedt 1998). Because there is no significant difference between the fiber cross-sectional area of type I and type II fibers in the digastric muscle (Korfage et al. 2000), we expected the MyHC contents of type I fibers to be lower than its type I area proportion. However, this was not seen in the present study.
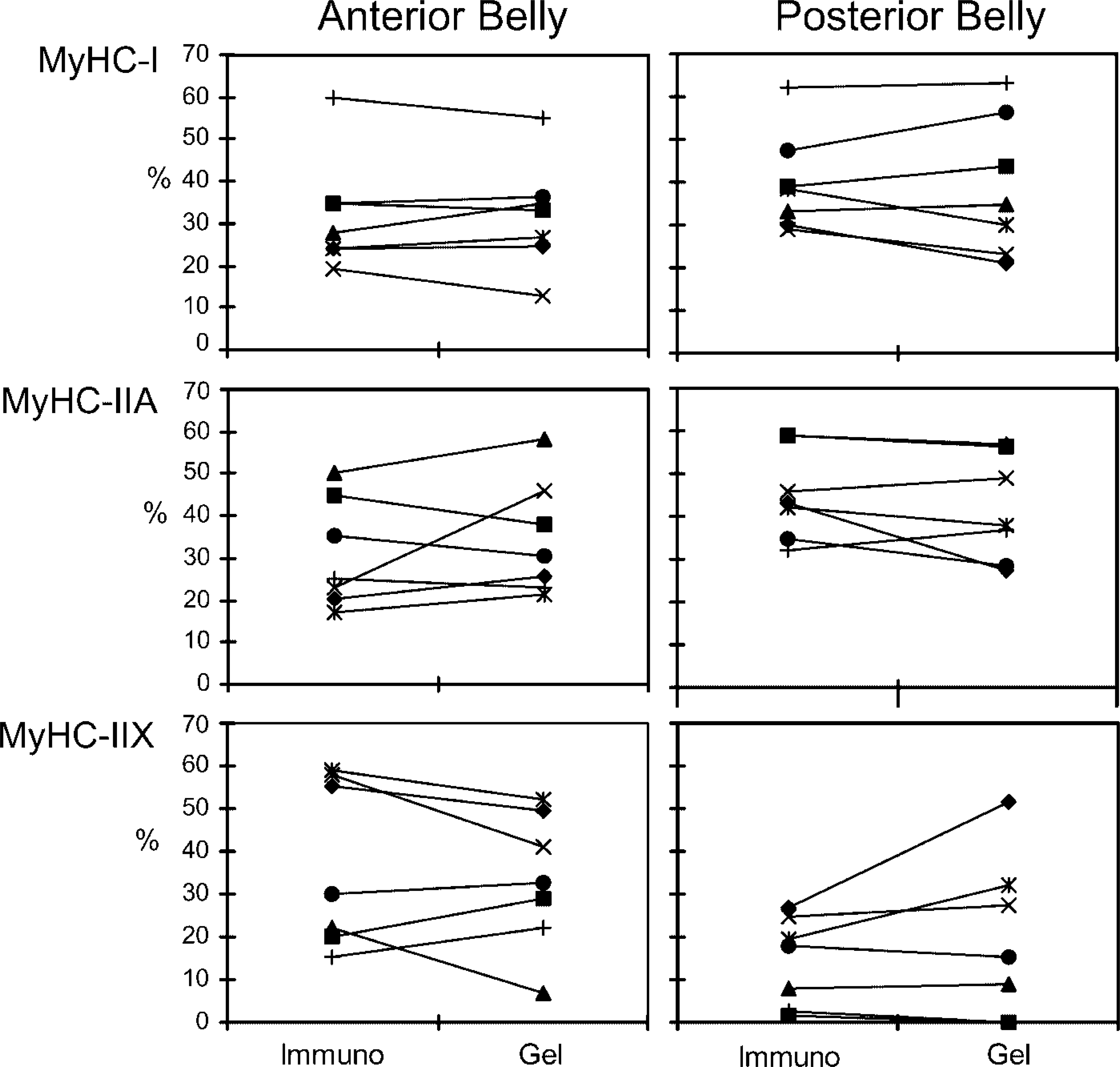
Percentage of MyHC fiber type area as determined by immunohistochemistry linked with their MyHC contents in the anterior and posterior belly (n = 7). Each marker denotes one individual.
Second, hybrid fibers that co-express more than one MyHC isoform could contribute to the difference between the two methods. The single-fiber analysis showed that a significant portion of the investigated fibers of the posterior belly were hybrid fibers. This was also noted in a study (Monemi et al. 2000) in which many fibers co-expressed both fast MyHC isoforms. With the antibody panel used in the present study it was not possible to detect hybrid fibers that co-express MyHC-IIA and MyHC-IIX. Many of these fibers were therefore misclassified as MyHC type IIA fibers by immunohistochemistry when they were actually hybrid fibers that co-express MyHC-IIA and MyHC-IIX. Antibodies that can distinguish this hybrid fiber type exist (Schiaffino et al. 1989; Lucas et al. 2000; Serrano et al. 2001) but are not easily available. Moreover, with immunohistochemistry the exact contents of the various MyHC isoforms in hybrid fibers cannot be determined. Whether hybrid fibers were the reason that the correlation coefficient for MyHC-IIA in the present study was lower than for MyHC-I and MyHC-IIX fibers remains to be determined.
MyHC fiber type composition of single fibers (n = 61) of the posterior belly of the digastric muscle from three individuals, as determined by SDS-PAGE

Mean myosin heavy chain isoform percentages (n = 7)
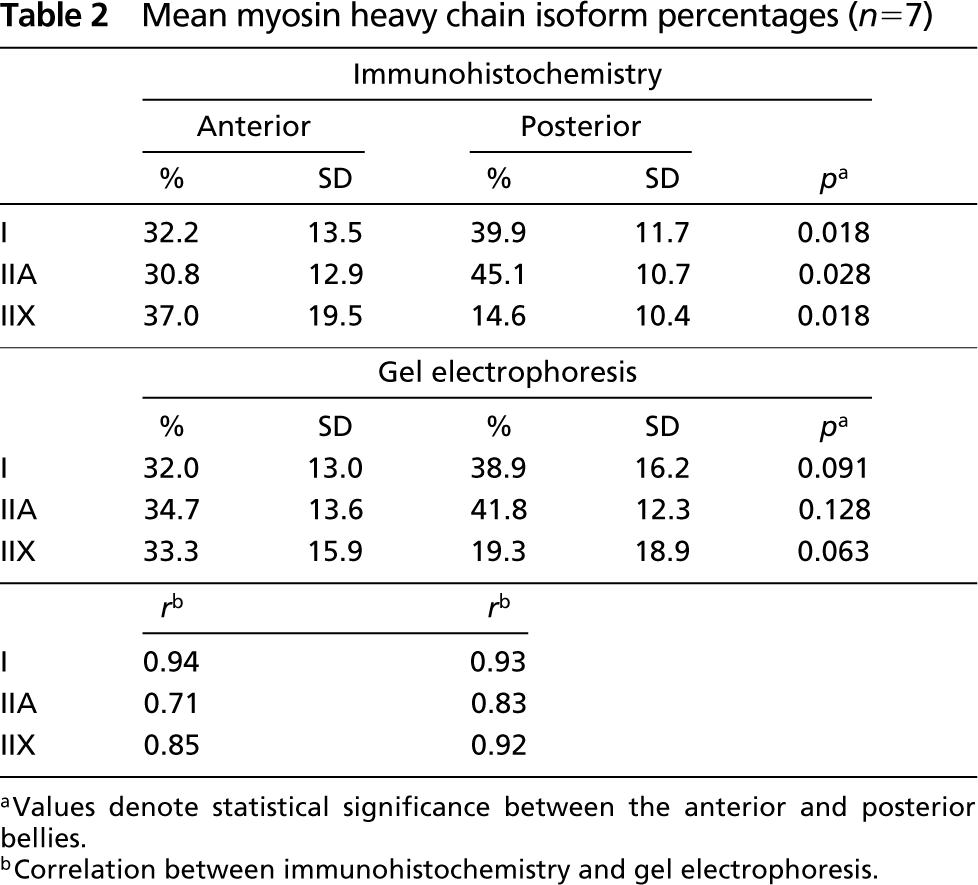
aValues denote statistical significance between the anterior and posterior bellies.
bCorrelation between immunohistochemistry and gel electrophoresis.
The present study confirms in single fibers the previous immunohistochemical results (Monemi et al. 2000) showing that the human digastric muscle contains hybrid fibers that co-express MyHC-IIA and MyHC-IIX. This particular hybrid fiber type was commonly found in limb and trunk muscles of untrained people (Andersen et al. 1994; Williamson et al. 2001) and can be distinguished by standard ATPase histochemistry (Staron 1997), although ATPase histochemistry underestimates this particular hybrid fiber type (Serrano et al. 2001). In an ATPase histochemical study of the digastric muscle in young individuals (Eriksson et al. 1982), these hybrid fibers were not found. This may be explained either by the mismatch between ATPase histochemical fiber type estimation and its MyHC isoform contents or by the high age of the investigated subjects in the present study, because this particular hybrid fiber type increases with age (Klitgaard et al. 1990; Andersen et al. 1999).
Aging can influence the fiber type composition. In a study that compared the fiber type composition of the human digastric muscle in young and elderly individuals (Monemi et al. 2000), no significant differences were found in MyHC content of muscle fibers between young adults and elderlypersons. The masseter of elderly individuals, however, contained smaller amounts of slow and larger amounts of fast MyHCs than the masseter of young individuals (Monemi et al. 1999), and in limb and trunk muscles of elderly individuals it was noted that the proportion of slow MyHC was larger and of fast MyHCs smaller than in young individuals (Lexell 1993; Monemi et al. 1999).
In conclusion, both immunohistochemistry and gel electrophoresis give comparable results. In the human digastric muscle, hybrid fibers co-expressing MyHC-IIA and MyHC-IIX appear to be a common hybrid fiber type. Further studies of single fibers with gel electrophoresis could show the ratio of MyHC-IIA and MyHC-IIX within these hybrid fibers.
Footnotes
Acknowledgements
Supported by the Interuniversitary Research School of Dentistry through the Academic Centre of Dentistry, Amsterdam.
We would like to express our gratitude to Dr J.H. Koolstra, Dr G.E.J. Langenbach, and L.J. van Ruijven for reviewing the manuscript, and to Dr J van der Velden for expert advice on the gel electrophoresis.
