Abstract
Although detection of the abnormal isoform of prion protein (PrPsc), the specific feature of transmissable spongiform encephalopathies (TSEs), has been previously demonstrated on formalin-fixed autolytic tissue, no samples with autolysis as severe as tested here (i.e., liquid state) have previously been tested. It is inevitable that a small but significant proportion of brains, especially in summer due to delays in postmortem examination, undergo an extremely severe autolysis that makes samples unsuitable for diagnosis by conventional techniques. In this study, 25 bovine samples were diagnosed by applying immunocytochemistry on the corresponding liquid fraction. Four additional portions of brainstem (positive and negative sheep and cattle) were subjected to one of the autolysis regimens at 56C or environmental conditions for up to 80 days and were analyzed with the same methodology. No abnormal protein could be detected in any of the control animals. PrPsc accumulation was observed by immunocytochemistry in all cases that were positive by either immunohistochemistry on the corresponding filtrates or by Prionics Western blotting, showing an excellent agreement between the methodology assessed and these routine techniques. The results of this study demonstrate immunocytochemistry as a useful tool for diagnosis in liquid-state samples, solving a most relevant problem in BSE and scrapie epidemiology.
T
The detection of the abnormal isoform of prion protein (PrPsc), TSE's specific feature (Bolton et al. 1982), has been widely demonstrated to solve the problem of scrapie and BSE diagnosis in unsuitable samples, detecting PrPsc in the corresponding formalin-fixed tissue (Scott et al. 1992; Miller et al. 1993; Race et al. 1994; Chaplin et al. 2002).
However, the question of whether the disease specific protein is also detectable in fully autolyzed tissues (i.e., liquid state) has not been yet studied. To our knowledge, only a few experiments have been performed on such samples but have always been applied on formalin-fixed filtrates obtained from the autolyzed tissue (Debeer et al. 2001). This extremely severe autolysis is present in a small but significant proportion of cases in Spain, especially during the hot summer months, due to the high temperatures together with a long period from the time of the animal's death on the farm to the extraction of the brainstem. Considering the difficulty of analyzing these liquid-state samples and the fact that they are sometimes the only material available for diagnosis, and therefore most relevant for BSE and scrapie epidemiological studies, there is a need to test the capacity of diagnostic techniques to detect PrPsc in these troublesome samples.
In this study, 25 positive and negative samples were used to assess whether immunocytochemistry using monoclonal antibodies (L42 and 6H4) is able to detect PrPsc in liquid-state neural tissue. Furthermore, the effects of controlled autolysis on the results obtained by this technique were assessed in this study. In this last experiment, scrapie samples were also included because of the relevance of analysis of these animals in the new ovine surveillance program.
Materials and Methods
Samples
Five brainstem samples from suspect cattle aged over 24 months submitted by several regional diagnostic laboratories for BSE confirmation were used. Twenty control negative samples of the same age were provided by the Regional Laboratory for TSEs in Aragón.
All samples presented a very advanced degree of autolysis (mostly liquid state) and were obtained through the Spanish BSE Surveillance Programme, i.e., dead cattle on farm, during the summer of 2001. One subsample of each of them was analyzed using Prionics Western immunoblotting (Schaller et al. 1999), one aliquot was subjected to immunocytochemistry, and the remainder was immediately placed at −70C for further studies. In cases for which any portion of tissue could be recovered, the filtrate was placed in embedding cassettes for formalin fixation (10%), routinely processed, and subjected to immunohistochemistry (i.e., on fixed tissue) as described below to confirm the results on the liquid sample.
Controlled Autolysis
A portion of undamaged brainstem comprising 1 g of tissue was taken caudally to the obex belonging to one positive and one negative BSE cases and one positive and one negative scrapie cases (all of them previously confirmed by immunohistochemistry) and placed in containers. The samples were then subjected to controlled treatments as follows. One sample from each animal was incubated at 56C and another one subjected to the environment (in a screened outdoor enclosure in full sunlight; the ambient temperature exceeded 37C on several days). After incubation or exposure, respectively, an aliquot or swab (in cases when the liquid state was not yet present) was recovered twice a week for 80 days, for PrPsc detection by immunocytochemistry according to the methodology described below.
Immunocytochemistry
A swab from each sample was diluted and vortexed in 1 ml of TBS (0.5 M Tris, 8% NaCl; pH 7.6) and a 40-μl subaliquot from this dilution, together with two subsequent serial decimal dilutions, was dispensed onto Vectabond-pretreated glass slides to optimize the dilution for microscopic assessment. After 24 hr of drying at 56C, samples were immersed in formalin 10% for 1 hr and subjected to the same protocol applied for immunohistochemistry, as described below.
PrPsc detection using a monoclonal antibody was performed following pretreatment as previously described (Hardt et al. 2000). Briefly, sections were pretreated with 98% formic acid and hydrated autoclaving to enhance antigen retrieval. After proteinase K digestion, primary antibodies L42 (R-Biopharm, Darmstadt, Germany; dilution 1:500) and 6H4 (Prionics, Zurich, Switzerland; dilution 1:500) were incubated at RT for 30 minutes. EnVision (DAKO; Glostrup, Denmark) was used as the visualization system and DAB as chromogen. Control sections for each sample with no antibody incubation (antibody diluent instead) were always included.
Results
PrPsc deposits, presenting a granular type in all cases, were detected in five samples with both monoclonal antibodies by immunocytochemistry (Figure 1A). Because of the advanced degree of autolysis in all samples analyzed, neither protein accumulation was identifiable anatomically, nor were tissue components (neurons or neuropil) distinguished. Consequently, it was not possible to assess whether immunolabeling was confined to gray matter or whether it extended to white matter, and most affected nuclei were impossible to distinguish. No positive staining was observed in 20 samples (Figure 1B), even though background immunostaining was observed in some of them (non-specific staining was always distinguished from the characteristic granule-positive deposits). There was excellent agreement between these results and those obtained by immunohistochemistry on fixed tissue filtrates and by Prionics WB on fresh subsamples. A single disagreement arose when the Prionics immunoblotting technique was used. One truly positive sample gave a positive result by immunocytochemistry but a negative result by Prionics WB. Nevertheless, PrPsc was also detected when concentration and no proteinase K digestion was followed.
Concerning the controlled autolysis experiment, PrPsc was still detectable on the positive samples by immunocytochemistry even after 80 days of environmental exposure, although decreasing with time. No PrPsc was detected in the sample from the control animal at day 0 or at any other interval during this treatment. Moreover, there was no difference in the detection of PrPsc in the samples exposed compared with the others heated at 56C.
Discussion
On the basis of the results obtained in this study, immunocytochemistry is a useful tool for diagnosis in very severely autolyzed samples, particularly those in a liquid state, which present such difficulty for analysis. Several experiments have confirmed that neither immunohistochemistry (Debeer et al. 2001) nor other techniques, such as detection of scrapie-associated fibrils (SAF; Scott et al. 1992) or Western immunoblotting (Race et al. 1994; Chaplin et al. 2002), are affected by autolysis. However, in none of them was the degree of autolysis as severe as in the samples we tested. In addition, none of these studies had further or exclusively assessed the liquid phase of the autolytic sample.
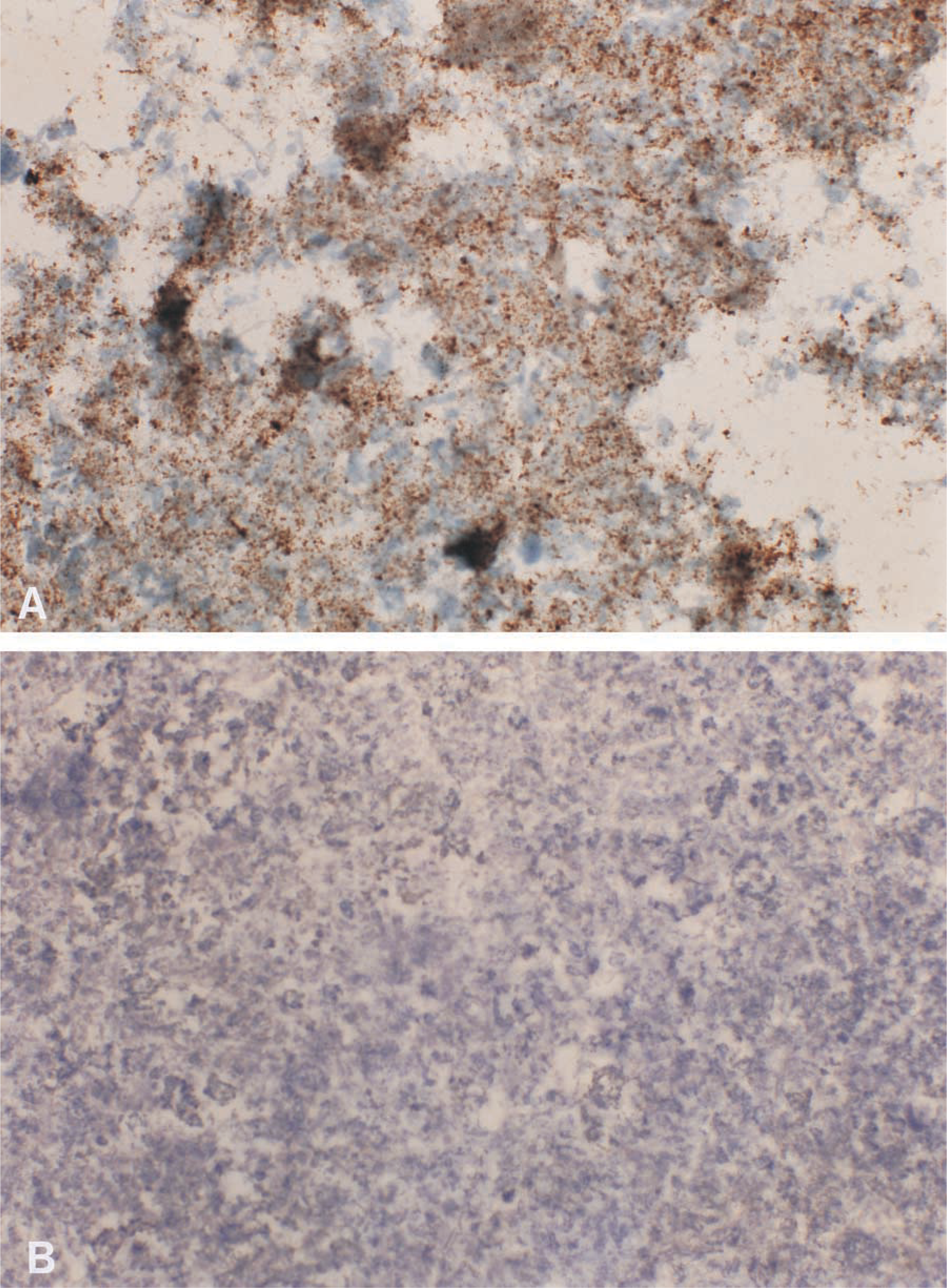
Representative illustrations of PrPsc immunocytochemistry corresponding to (
The specificity of immunolabeling is validated for the absence of any PrPsc deposits in BSE-negative samples. The greater difficulty of interpretation posed by the degree of background staining in the autolyzed material, corresponding to some samples examined, did not compromise the accurate final diagnosis because nonspecific staining could never be confused as a positive deposit because of the characteristic granular type of the latter. In spite of experiments previously performed that had shown no background labeling (Debeer et al. 2001), according to some of them that also showed no specific staining associated with autolysis (Chaplin et al. 2002), it is important to point out that the fraction analyzed in this study consisted of the liquid phase of very advanced autolyzed samples, not the autolyzed tissue or filtrates that are usually examined.
Furthermore, the resistance of the autolyzed material to all pretreatments for antigen unmasking has been confirmed in this experiment. Formic acid incubation, autoclaving, and proteinase K digestion were compatible, as previously described (Debeer et al. 2001), with preservation of the material on the glass slides. Adherence was slightly altered in some cases, but a significant part of the autolyzed material remained adhered on all sections. This exceptional stability of PrPsc despite degradative conditions (Taylor 2000) is the major feature that allows the potential diagnosis of BSE and scrapie in animals that previously could not be evaluated because the tissues were unsuitable for histopathological examination (mainly because of autolysis).
On the other hand, the strong agreement between the routine methods (Prionics WB and IHC) and the technique assessed in this study concerning all the samples analyzed confirms the accuracy of the latter on liquid samples, the main objective of the experiment. The one disagreement found between them (although disappearing as indicated above) might suggest a failure of the Prionics immunoblotting technique in such severely autolyzed material. Nevertheless, further studies will be necessary to overcome this potential problem.
This work should encourage laboratories to address the problem of those cattle and sheep brains that present extreme difficulties for TSE analysis because of a severe degree of autolysis. It is important to note that such difficulty involves a consequent loss of data for epidemiological studies about such a relevant disease. Furthermore, the results recorded here should also provide confirmation of immunocytochemistry as an appropriate diagnostic tool for such samples.
Footnotes
Acknowledgements
Supported by a grant from the European Commission (FAIR 98–7021).
We gratefully thank Dr Groschup for kindly supplying antibody, regional laboratories for TSEs for providing the samples, and the technical staff (National Reference Centre and Regional Laboratory in Aragón) for assistance in processing the samples.
