Abstract
One of the pathological changes characteristic of the transmissible spongiform encephalopathies (TSEs) is the accumulation of disease-specific PrP (PrPsc). Immunola-beling of PrPsc was compared using a panel of monoclonal and polyclonal antibodies. To determine the effects of tissue fixation on immunostaining, we performed a supplementary investigation reviewing the fixatives formol saline and periodate-lysine-paraformaldehyde (PLP). The main target sites of the antibodies were similar. However the monoclonal antibodies (MAbs) 6H4, 7A12 and 8H4 revealed targeted PrPsc labeling with no background labeling. Although 7A12 and 8H4 did not detect early PrP deposition, we propose that during the later stages of disease 7A12 and 8H4 can be used with equal effectiveness in place of 6H4. Tissues taken during the early stages of disease that had been fixed in PLP displayed more PrP immunolabeling than tissues that had undergone formol fixation. PLP fixation on 6H4-immunostained tissue revealed interweaving granular linear PrP deposits in the hippocampus. This labeling was not observed in tissue that had undergone formol fixation, suggesting that PLP fixation might enhance the sensitivity of the immuno-histochemical (IHC) detection of PrP. In the two scrapie mouse models studied here, PLP fixation and immunolabeling with the anti-PrP antibody 6H4 gave superior results.
T
To further clarify the role of PrP during disease pathogenesis, the deposition of disease-specific PrP has been studied in experimental murine scrapie models using IHC techniques. In normal mice, BSE-infected mice, and mice infected with scrapie, PrP is associated with the follicular dendritic cells of the spleen, lymph nodes, Peyers patches, and the islets of Langerhans in the pancreas (McBride et al. 1992). In the central nervous system (CNS) of TSE-infected mice, accumulation of PrPsc is observed in different forms: amyloid plaques, fine granular or synaptic deposits, and coarser depositions (Bruce et al. 1989). Ultrastructural studies have shown that it is localized to amyloid fibrils and accumulates on the plasmalemma of neurite membranes (Jeffrey et al. 1994). Previous studies using scrapie mouse models revealed that the pattern of PrP pathology in the brain depends upon the agent used, route of injection and mouse genotype (Bruce et al. 1989).
Immunolabeling of PrP using the antibodies 1A8 and 1B3 (Farquhar et al. 1989) in the 87V/VM and ME7/CV scrapie mouse models has been shown in previous investigations (Bruce et al. 1989; Jeffrey et al. 1994). 1A8 is a high-titer anti-mouse PrP serum that recognizes many different epitopes on the PrP molecule, particularly amino acid residues 89–114, 114–131, 140–182, and 187–234. Similarly, 1B3 targets the amino acid residues 14–36, 83–102, 119–139, and 188–212 on the PrP molecule (Langeveld et al. 1993).
Recent investigations have also used the mouse MAb 6H4 (Prionics; Zurich, Switzerland) to reveal PrP deposition in the CA2 region of the hippocampus in the 87V/VM model of scrapie (Jamieson et al. 2001). In contrast to the rabbit polyclonal antibodies, 6H4 specifically recognizes the epitope DWEDRYYRE (amino acids 143–151) in the murine PrP sequence. Immunolabeling PrP with the comprehensively characterized MAbs 7A12 and 8H4 has been achieved in wild-type (PrP+/+) mice (Liu et al. 2001). However, immunolabeling PrP with these antibodies in scrapie mouse models has yet to be investigated. The mouse MAbs 7A12 and 8H4 react with epitopes located on the central region or the C-terminal domain of the PrP molecule. 7A12 reacts with PrP23-145 and PrP90-231, both of which are located on the central region of the PrP protein, in contrast to 8H4, which binds to epitopes located in the C-terminal region between amino acid residues 144 and 231 (Li et al. 2000). The monovalent nature of 6H4, 7A12 and 8H4 could result in weaker immunodetection due to their high specificity for only a few epitopes or the inability to recognize the differential expression of certain PrP glycoforms in the mouse brain during development (Liu et al. 2001).
The two scrapie mouse models used here differ in their targeting of PrPsc deposition in the brain. The ME7 scrapie strain produces widespread diffuse vacuolation and PrP accumulation of varying intensities throughout the brain (Bruce et al. 1989). In the ME7/CV scrapie mouse model, PrP deposition is first detected by IHC as granular accumulations at 60 days post infection (dpi) in the hippocampus and thalamus, gradually appearing diffusely throughout the brain (Jeffrey et al. 2001). However, the 87V strain of scrapie consistently produces targeted vacuolation, PrP deposition, and amyloid plaques in specific neuroanatomical areas in the brain (Bruce et al. 1989).
The principal aim of our investigation was to determine if there were variations in PrPsc immunolabeling using five PrP antibodies and if any differences were evident at various stages of disease. We compared the immunolabeling of anti-PrP monoclonal and polyclonal antibodies in the ME7/CV experimental murine scrapie model over several time points. We also compared the immunolabeling of anti-PrP monoclonal and polyclonal antibodies in the 87V/VM murine scrapie model at the 80-day point to reproduce the early PrP labeling observed in previous investigations (McBride et al. 1998).
It has been previously reported that PLP-fixed brains of mice can reveal extensive PrPsc immunoreactivity in comparison to brains fixed in formol saline (Bruce et al. 1989). Therefore, we also compared the effects of two fixatives, PLP and formol saline, on the amount of PrPsc labeling revealed.
Materials and Methods
The ME7 murine scrapie model was produced by intracerebrally injecting the F1 cross between the C57BL/DK and VM/DK mouse strains (known as CV) with 20 μl of a 1% (w/v) brain homogenate from a C57BL mouse terminally infected with the ME7 strain of scrapie. This model develops consistently severe hippocampal pathology (Scott and Fraser 1984). As negative controls, age- and strain-matched mice were intracerebrally inoculated with uninfected brain tissue. Four scrapie-infected and two age-matched normal brain-injected mice were sacrificed by cervical dislocation at specific time points throughout the incubation period and the brains were removed for further analysis. The time points studied were 50, 70, 96, 126, 152, 184, and terminal point of disease (230 dpi).
The 87V murine scrapie model was produced by intracerebrally inoculating VM/Dk mice with 20 μl of a 1% (w/v) brain homogenate from a mouse terminally infected with the mouse passaged 87V scrapie strain. The 87V scrapieinfected and normal brain-injected control mice were sacrificed by cervical dislocation and their brains removed at 80 dpi and terminal point of disease (320 dpi).
Once removed, the brains were fixed in 10% formol saline or PLP and trimmed to give four coronal levels [approximating Figures 25, 46, 64, and 85 in Franklin and Paxinos (1997)] and 6-μm paraffin sections prepared on Superfrost slides. All experimental procedures were carried out in strict accordance with UK Home Office Project Licence regulations.
The pretreatments applied are based on the standard immunolabeling protocols for 1A8, 1B3 and 6H4 used at the Neuropathogenesis Unit (NPU) to optimize labeling and minimize nonspecific background staining during immunolabeling in mouse brain, and has been referred to in previous publications (Brown et al. 2000; Baxter et al. 2002).
With combined pretreatments of formic acid immersion (Kitamoto et al. 1987) and hydrated autoclaving (Haritani et al. 1994), PrP detection is considerably enhanced (Brown et al. 2000). Therefore, to unmask the antigen the sections were pretreated by hydrated autoclaving at 121C for 15 min, followed by immersion in 98% formic acid for 10 min to enhance PrP visualization.
To reduce the risk of unwanted background labeling, endogenous peroxidase activity was inhibited with hydrogen peroxidase in methanol. For the polyclonal antibodies, nonspecific binding sites were blocked by preincubation in normal goat serum at 1:20 (Diagnostics Scotland; Edinburgh, UK). For the MAbs, the nonspecific binding sites were blocked by preincubation in normal rabbit serum at 1:20 (Diagnostics Scotland).
Primary antibodies 1A8, 1B3 (NPU) and 6H4 (Prionics) were applied optimally titrated and diluted to 1:2000; 7A12 and 8H4 (kindly provided by Dr. Man-Sun Sy of Case Western Reserve University; Cleveland, Ohio) were diluted to 1:800. The sections were exposed to the primary antibodies overnight at room temperature. For the polyclonal antibodies, negative control sections were incubated with normal rabbit serum (Diagnostics Scotland). The secondary antibody was biotinylated goat anti-rabbit (Jackson ImmunoResearch, Stratech Science; Luton, UK) used at 1:400 for 30 min. For the MAbs, negative control sections were incubated with normal mouse serum (Diagnostics Scotland). The secondary antibody was biotinylated rabbit anti-mouse (Jackson ImmunoResearch) used at 1:400 for 30 min. All sections were incubated in avidin-biotin complex (ABC) Elite kit PK-6100 (Vector Laboratories; Peterborough, UK) and the reaction product was visualized with 0.025% diaminobenzidine (Sigma; Dorset, UK).
Results
The five anti-PrP antibodies were compared at different stages of disease and the results are summarized in Table 1 and 2. Differences between the antibodies were more noticeable during the earlier stages of disease. In the ME7/CV scrapie model at 70 dpi, 1A8 (Figure 1A), 1B3 (Figure 1B), 6H4 (Figure 1C), 7A12 (Figure 1D), and 8H4 (Figure 1E) detected the deposition of PrP in the pyramidal cell body layer of the CA1 region of the hippocampus. However, the intensity of staining of the sections stained with 1A8 (Figure 1A) and 1B3 (Figure 1B) was compromised in the neuropil by the nonspecific binding partly concealing PrP labeling.
During the progression of disease, visualization of the increasingly widespread diffuse PrP pathology in the ME7 model was minimally affected by nonspecific labeling. This was initially noticeable at 96 dpi in the hippocampus of sections stained with 1A8 (Figure 1F), 1B3 (Figure 1G), 6H4 (Figure 1H), 7A12 (Figure 1I), and 8H4 (Figure 1J). Although the background labeling masked the early diffuse PrP staining in the CA1 region of the hippocampus (Figures 1A-E), the deposition of PrP by 96 dpi was sufficiently established to counteract this effect.
Immunohistochemical detection of PrP at various time points with five anti-PrP antibodies in the ME7/CV scrapie-infected mouse brain fixed in both PLP and formol saline a
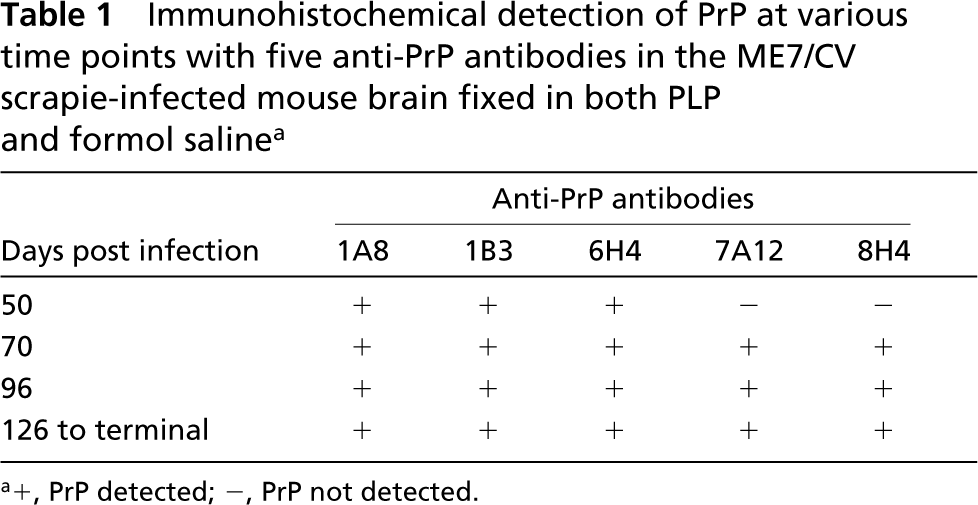
+, PrP detected; −, PrP not detected.
Immunohistochemical detection of PrP at 80 dpi with five anti-PrP antibodies in the 87V/VM scrapie-infected mouse braina

+, PrP detected; −, PrP not detected.
In the 87V/VM murine scrapie model which has targeted PrP pathology, during the terminal stages of disease 1A8 (Figure 1K) and 1B3 (Figure 1L) labeled sections showed nonspecific background staining that partially masked the PrPsc labeling. However, 6H4 (Figure 1M), 7A12 (Figure 1N), and 8H4 (Figure 1O) revealed intense and specific PrPsc labeling of the amyloid plaques, fine granular depositions, and diffuse accumulations, with no evident nonspecific background staining. During the early stages of disease (80 dpi), 6H4 (Figure 2A), 1B3 (Figure 2B), and 1A8 (not shown) all detected early punctate PrP deposition in the dorsal raphe, median raphe nucleus, and vestibular nucleus, with no apparent difference in targeting or intensity. In contrast, 7A12 and 8H4 did not detect this early PrP deposition (not shown).
PLP-fixed 87V/VM murine scrapie brains demonstrated early PrP staining at 80 dpi in the vestibular nucleus, dorsal raphe, and median raphe nucleus with 6H4 (Figure 2A), 1B3 (Figure 2B), and 1A8 (not shown). However, PrP deposition was not detected with 7A12 or 8H4 (not shown). PrPsc immunolabeling was not revealed at 80 dpi in 87V/VM brains fixed in formol saline with any of the antibodies tested (Figure 2C). This fixative effect is not repeated with the ME7/CV murine scrapie model. Sections stained with 6H4, 1B3 and 1A8 showed no such difference at 51 dpi (polyclonal results not shown), where PrP was observed in the dorsal raphe of brains fixed in PLP (Figure 2D) and formol saline (Figure 2E). As in the early stages of disease in the 87V/VM scrapie mouse model, the early deposition of PrP in the ME7/CV model was not detected by 7A12 and 8H4 when tissues were fixed in either formol saline or PLP (not shown).
We also observed that only PLP-fixed tissues immunostained with 6H4, 1B3, and 1A8 revealed delicate tracking of PrP along processes in the stratum radiatum of the hippocampus (Figure 2F) (polyclonal results not shown).
Discussion
Few studies have concentrated on determining if there are variations in PrPsc immunolabeling using different PrP antibodies at various stages of disease. We show that the main targeting sites of 1A8, 1B3, 6H4, 7A12, and 8H4 in the brains of ME7 scrapie-infected mice were similar, with PrPsc labeling observed in the same neuroanatomic areas in the later stages of disease. Similarly, in 87V scrapie-infected mice, the main target sites of the five antibodies studied were identical in the later stages of disease.
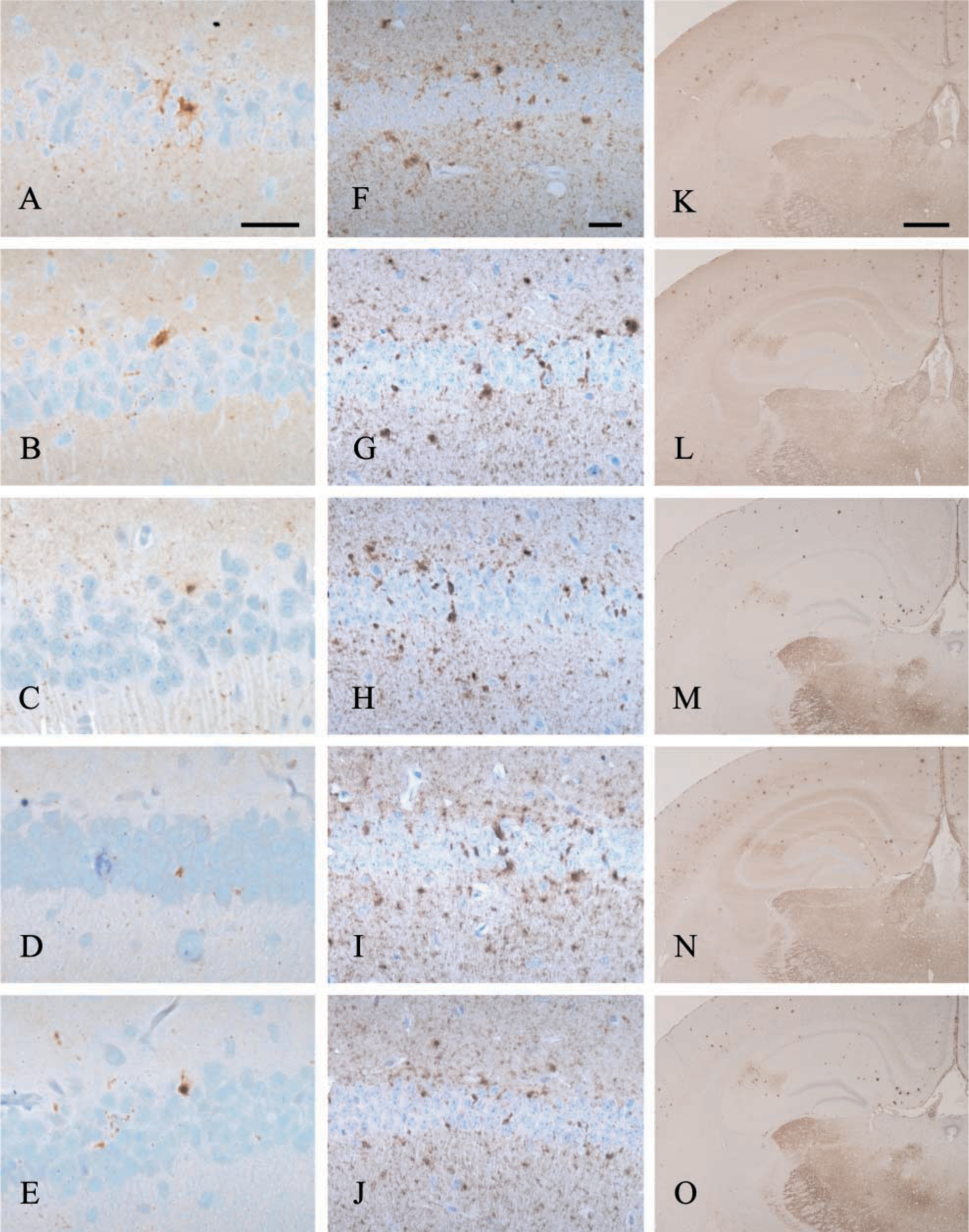
Immunolabeling of anti-PrP rabbit polyclonal antibodies 1B3 and 1A8 and mouse MAbs 6H4, 7A12, and 8H4 on scrapie-infected (87V and ME7) mouse brains at various time points. In the ME7/CV murine scrapie model at 70 dpi, 1A8 (
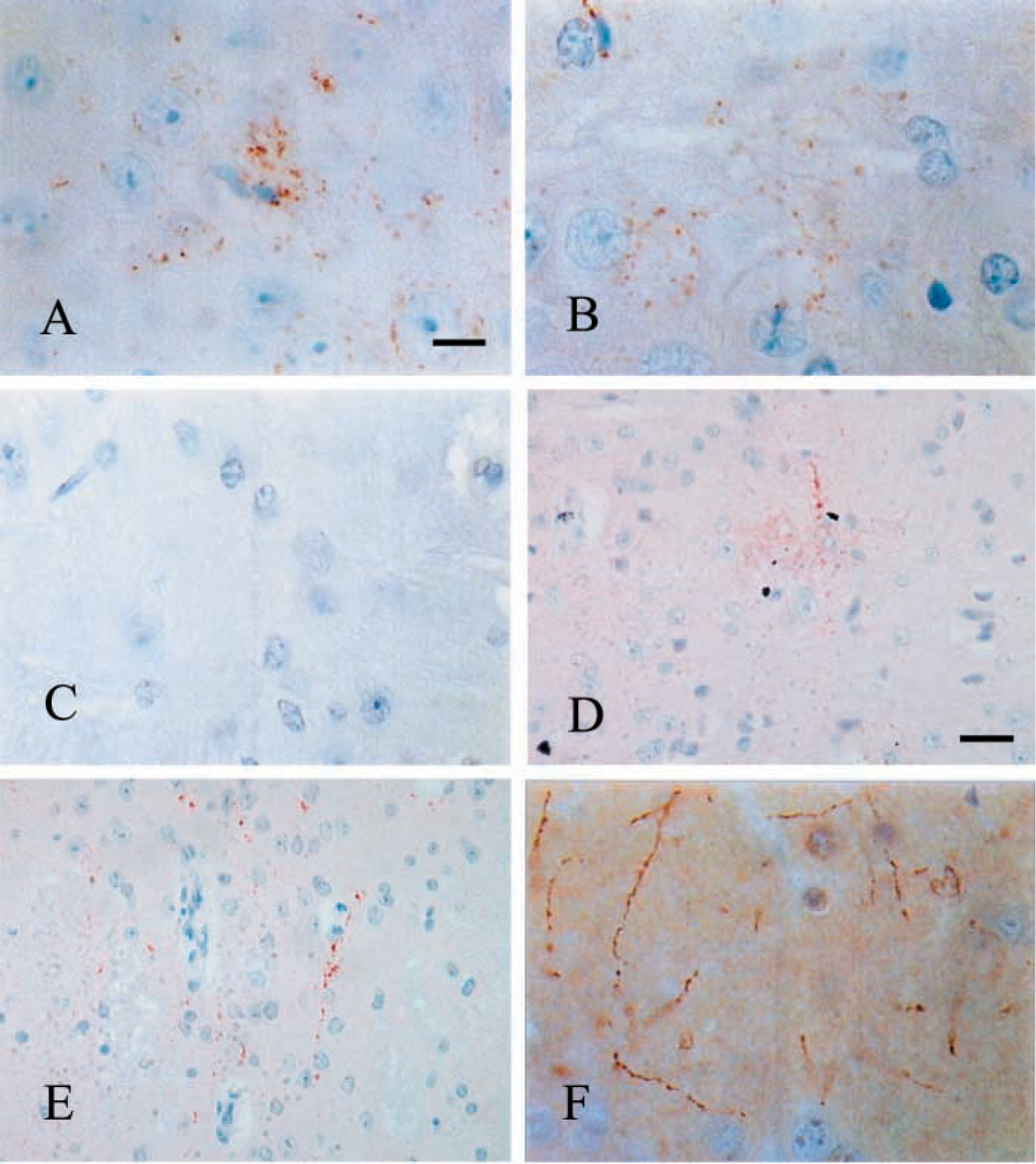
In the PLP-fixed 87V/VM model at 80 dpi, 6H4 (
However, differences emerged during the early stages of disease in both murine scrapie models studied. Only 1A8, 1B3, and 6H4 detected early punctate PrP deposition in the 87V scrapie model at 80 dpi and the ME7 scrapie model at 51 dpi. These results are consistent with reports that 87V scrapie-associated PrP is first detected at 70 dpi (McBride et al. 1998). The results also show that only 1A8, 1B3, and 6H4 detected early PrP deposition in the ME7/CV scrapie model at 51 dpi, which is in agreement with previous reports of the detection of abnormal PrP at 60 dpi (Jeffrey et al. 2001). However, of these three antibodies 6H4 alone revealed more targeted PrPsc labeling with minimal nonspecific reaction.
That the monoclonal antibodies 7A12 and 8H4 did not label early PrP deposition is not an indication of their lack of efficacy but does lend support to the complex nature of PrP expression as various glycoforms in different brain regions (Somerville 1999; Liu et al. 2001) and possibly during the development of the disease. It is conceivable that the targeted nature of 7A12 and 8H4 is directed towards specific PrP glycoforms that may not be expressed during the onset of disease or found in the neuroanatomic areas in the CNS that express early PrP deposition. This finding is in itself highly intriguing and needs additional in-depth analysis to fully appreciate the complexities of PrP expression.
Previous reports that PLP-fixed brains of mice reveal extensive PrPsc immunoreactivity have been partially supported by this investigation. Tissues that had been fixed in PLP displayed more PrP immunolabeling than tissues that had undergone formol fixation. Although during the early stages of disease in the ME7/CV model there is no significant difference in PrP immunolabeling with fixation in either PLP or formol saline, a subtle difference between the effects of the two fixatives on immunolabeling was apparent on closer inspection.
A delicate tracking of PrP was seen in the stratum radiatum of the hippocampus, similar in appearance to the interweaving granular linear PrP deposits observed and described in more detail by McLean et al. (1998). This labeling was not observed in tissue that had undergone formol fixation, suggesting that PLP fixation might enhance the sensitivity of the IHC detection of PrP. It is the absence of early PrP labeling in the 87V/VM model fixed in formol saline and its presence in PLP fixed brains that further supports this finding and is consistent with previous reports that PLP fixation reveals extensive PrPsc immunoreactivity (Bruce et al. 1989). Furthermore, PLP is known to react with sugars and to stabilize proteins (McLean and Nakane 1974), which may be crucial in enhancing the detection of PrP, a sialoglycoprotein, in the early stages of disease in the 87V/VM model. Therefore, we report that immunolabeling with PLP-fixed tissues produces superior immunolabeling in the CNS. However, this discrepancy in fixative effect between the 87V/VM and ME7/CV models of scrapie is as yet unexplained and warrants further investigation.
Conclusion
The principal aim of our investigation was to determine if there are differences in PrPsc immunolabeling using different PrP antibodies. We report that there is no discernible difference between the targeting of the monoclonal antibody 6H4 and the polyclonal antibodies 1A8 and 1B3 in the scrapie-infected mouse brain. Furthermore, we report that the MAbs 7A12 and 8H4 do not detect the early deposition of PrP. However, it is conceivable that the targeted nature of 7A12 and 8H4 is directed towards specific PrP glycoforms that may not be expressed during the onset of disease and early PrP deposition.
Comparing the five anti-PrP antibodies studied, we suggest that 6H4 is a more appropriate antiserum for PrP immunodetection in the mouse CNS throughout the course of disease, revealing targeted PrPsc labeling with no nonspecific reaction. Given the panel of antibodies currently available for PrP immunodetection, we also propose that 7A12 and 8H4 can be used with equal effectiveness in place of 6H4 for PrP immunolabeling in the later stages of disease in the mouse CNS. That 6H4 targets a single epitope in the PrP sequence and demonstrates comparable targeting to 1B3 and 1A8 that targets multiple epitopes suggests that monovalency does not result in weaker immunodetection. It appears that it is the location of the epitope on the PrP molecule to which the antibody is targeted that ultimately determines the accuracy of immunodetection.
Footnotes
Acknowledgements
We thank Patricia McBride (Neuropathogenesis Unit) for helpful discussion. We also thank Drs Man-Sun Sy and Rona Barron for kindly providing the antibodies 7A12 and 8H4.
