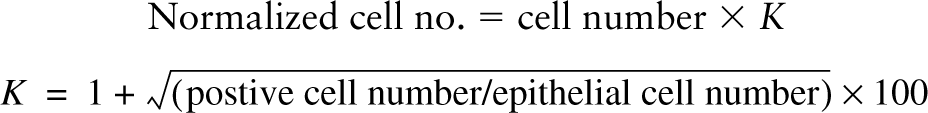Abstract
The FORSE-1 (forebrain-surface-embryonic) monoclonal antibody (MAb) recognizes a carbohydrate cell surface epitope related to the Lewis-X (LeX) and stage-specific embryonic antigens (SSEAs). In the developing CNS, the FORSE-1 epitope is believed to serve as a marker of progenitor cells. We studied the expression of the FORSE-1 epitope in pulmonary neuroendocrine cells (PNECs) and related neuroepithelial bodies (NEBs), cell types implicated in paracrine regulation of lung development. We used dual immunolabeling to identify PNECs/NEBs in tissue sections from developing rabbit fetal lungs and corresponding primary lung cell cultures. During the early stage (E16), the FORSE-1 MAb labeled primitive airway epithelium, whereas serotonin (5HT) immunoreactivity, a marker of PNEC/NEB differentiation, was negative. After E18, FORSE-1 labeling became restricted to PNECs and NEBs, identified by co-expression with 5HT, then decreased coincident with an increase in 5HT. Expression of the FORSE-1 epitope correlated inversely with 5HT expression in PNEC/NEB cells. FORSE-1 immunoreactivity correlated with cell proliferation assessed by BrdU labeling. Downregulation of the FORSE-1 epitope correlated with maturation of PNECs/NEBs. The presence of few FORSE-1/5HT-positive cells in postnatal lung suggests retention of progenitors. The FORSE-1 epitope was associated with a high molecular weight (286 kD) glycoprotein that decreased with increasing gestational age, as demonstrated by immunoblotting. Overall expression of SSEA-1, −3, and −4 antigens was similar to FORSE-1/5HT, although the former was preferentially localized to neurite-like processes. Because the role of the FORSE-1 epitope in the CNS probably involves cell adhesion and differentiation, we propose a similar function in developing lung. The demonstration of LeX/SSEA antigen expression in the PNEC/NEB cell lineage underscores the importance of these cells in developing lung. Furthermore, the FORSE-1 antigen may identify committed progenitors of the PNEC/NEB cell system.
Keywords
P
The FORSE-1 (forebrain-surface-embryonic) MAb raised against a surface antigen expressed by retinoic acid-induced human embryocarcinoma (NT2D1) was shown to specifically recognize a cell surface antigen related to the Lewis-X (Le-X) carbohydrate epitope expressed during neuronal development (Allendoerfer et al. 1999). The LeX antigen is closely related to the series of SSEA carbohydrate antigens extensively studied in embryogenesis, differentiation of embryonic stem cells and germ cells to endodermal, mesodermal, and ectodermal lineages, and during neoplasia (Solter and Knowles 1978; Kannagi et al. 1983; Fenderson et al. 1987; Damjanov et al. 1994; Dabelsteen 1996; Ito et al. 1997; Shamblott et al. 1998; D'Costa and Petitte 1999; Kurahara et al. 1999; Yoshido-Noro et al. 1999). This FORSE-1 carbohydrate antigen is considered a marker of progenitor cells in the earliest embryonic forebrain region of the mammalian CNS (Tole et al. 1995). The antigen is present on cell surface proteoglycans and glycolipids and varies with cell type and stage of differentiation (Allendoerfer et al. 1995). Because FORSE-1 antigen is one of the cell surface molecules, its expression may be regulated by temporally and spatially restricted transcription factors with corresponding patterns of expression. For example, a known transcription factor, BF-1 (brain factor-1) was found to have a remarkable pattern of expression that overlapped with FORSE-1 immunolocalization (Tole and Patterson 1995). Although FORSE-1 expression during embryogenesis has been characterized in rat CNS (Allendoerfer et al. 1995), there are no previous studies of FORSE-1 expression during lung development. To determine whether the FORSE-1 MAb recognizes cells within the PNEC/NEB phenotype, identified by expression of 5HT, a definitive neuroendocrine marker was studied coordinately. We report immunohistochemical studies on FORSE-1 expression in developing rabbit lung, using tissue slice preparations, and in primary cultures established from different stages of lung development using immunocytochemical labeling and confocal microscopy. Immunoblotting identified a 286-kD carrier protein of FORSE-1 antigen. Co-immunoreactivity of SSEA antigens and FORSE-1 in 5HT-expressing cells makes a link with embryonic patterns observed in other tissues and organs. Therefore, FORSE-1 is expressed on a specific subpopulation of pulmonary cells and appears to identify a developmental lineage of progenitors of PNECs/NEBs.
Materials and Methods
Animals
New Zealand White does at 16, 18, 21, 24, 26, and 28 days’ gestation (E16–E28) and postnatal days 1, 3, and 7 (P1–P7) litters were sacrificed by lethal injection. The fetuses were removed and the entire lungs aseptically dissected. For primary culture, excised lungs were kept immersed in cold CO2 Independent Medium (Gibco BRL; Burlington, Ontario, Canada). The parenchyma and vasculature were teased away from airways under a dissecting microscope, leaving tracheal/bronchial trees largely intact. To perform immunochemical labeling, lung slices were fixed with 4% paraformaldehyde and tissue slices (200–400 μm) were prepared using a Leica Vibratome 1000S.
Cell Culture
Primary cultures of fetal rabbit lung cells were established by enzymatic dissociation of whole lungs (E16–E21 gestation) and dissected bronchial trees (E24–P7). On average, 2 million cells/well were maintained in α-MEM supplemented with 10% FBS and cultured on Lab-Tek slides at 37C in 5% CO2. To label proliferating cells, cultures were incubated in medium containing 10 μM 5-bromo-2'-deoxyuridine (BrdU) for 3 hr and then fixed in 3.8% paraformaldehyde after 3, 5, 7, or 10 days in culture.
Immunocytochemistry, Antibodies, Signal Amplification, and Confocal Microscopy
Specific antibodies recognizing variant epitopes on LeX, FORSE-1, and SSEA-1, SSEA-3, and SSEA-4 were obtained from the Developmental Studies Hybridoma Bank (University of Iowa; Iowa City, IA). All other antibodies were purchased from commercial sources and applied as previously described (Yeger et al. 2001). Immunocytochemical labeling was enhanced with the catalyzed reporter deposition (CARD) amplification method as previously described (Yeger et al. 2001). Briefly, hydroxysuccinimide ester of rhodamine (TRITC-NHS) and biotin (Biotin-NHS) were coupled to tyramine according to Hopman et al. (1998) to produce TRITC-tyramine and biotin-tyramine substrates for peroxidase reaction, The conjugate was utilized in the CARD system to amplify FORSE-1 immunofluorescent and immunohistochemical labeling, respectively. Sections of paraffin-embedded lung tissue were cut at 3-μm thickness, deparaffinized, rehydrated and treated with 0.1 M citrate buffer, pH 6.0, for 5 min for antigen retrieval before being immunostained using FORSE-1 and anti-5HT antibodies. We used a CARD system method (King et al. 1997) with biotin-tyramine conjugate to amplify FORSE-1 signal. Essentially, after FORSE-1 incubation and application of the secondary antibody-peroxidase conjugate the signal was amplified by biotin-tyramine substrate. After reaction with HRP-avidin conjugate, signals were visualized with DAB substrate. The NOVA Red alkaline phosphatase (AP) substrate (Vector Labs; Burlingame, CA) was applied for 5HT visualization by fluorescence, which was compatible with organic dehydration and mounting in Permount.
In triple-labeling experiments after incubation with both FORSE-1 and anti-5HT antibodies, donkey anti-mouse IgM-HRP conjugate and donkey anti-goat IgG-AMCA conjugate antibodies were applied to lung slices and cultured cells. The TRITC-tyramine CARD system was also used for signal amplification of FORSE-1 (Chao et al. 1996), while goat anti-BrdU-FITC antibody conjugate was used to detect BrdU incorporation. Controls included performing labeling experiments without primary antibodies. No or insignificant nonspecific labeling was observed even after CARD amplification.
Fluorescent images (dual labeling, FITC/TRITC, or triple staining, FITC/TRITC/AMCA) of FORSE-1, 5HT, and/or BrdU in cultured cells and paraffin section were obtained using an Olympus fluorescent microscope with a cooled 3CCD color camera. A Leica confocal laser scanning microscope with SCANWARE software was used to visualize FITC/TRITC dual signal in tissue slices. Separate color images of FORSE-1, 5HT, and BrdU from either immunofluorescence microscope or confocal scanner were merged to form a composite FORSE-1/PNEC/NEB marker image. Image processing was done using Adobe Photoshop 4.0 software.
Quantitation of Labeled Cells
FORSE-1- and 5HT-positive cells/clusters and co-labeled cells/clusters were counted in the entire areas of 22 × 22 mm Lab-Tek chambers, using a grid scale and in triplicate, in the developmental series, E16 to P7 and in cultures maintained for 3, 5, 7, and 10 days. To determine PNEC/NEB proliferation, BrdU-positive cells were counted in association with either 5HT, FORSE-1, or co-labeled cell populations. These data were normalized for the total number of keratin-positive epithelial cells. Cell numbers of different mixed cell populations in culture, were compared to epithelial cells expressing cytokeratin and calculated as follows:
The data were calculated as mean ± SD using Prism Graph-Pad statistical software.
Immunoblotting
Lungs from different developmental stages (embryonic and newborn rabbits) were freshly dissected and homogenized in a homogenization buffer plus protease inhibitors as described by Allendoerfer et al. (1995). The homogenates were centrifuged at 1000 rpm for 20 min to remove nuclei and cell debris. The resulting supernatants were then centrifuged at 100,000 X g for 60 min in a Beckman 70 Ti rotor. After two consecutive washes, the final pellets were re-suspended in homogenization buffer. This material, referred to as the membrane fraction, was quantified for protein concentration using the Bio-Rad DC protein assay kit (Bio-Rad Laboratories; Mississauga, Ont, Canada) and frozen at −80C until further use. Equal amounts (80 μg/lane) of various membrane protein fractions were resolved by 6% SDS-PAGE with 3% stacking gel and electroblotted onto PVDF membranes using a Bio-Rad minigel transfer system. Blots were blocked for 1 hr at room temperature in TBS containing 0.05% Tween-20 (TBS-T), 2% BSA, and 5% nonfat milk. Blots were washed three times in TBS-T and then incubated in 1:20 of FORSE-1 hybridoma supernatant for 16 hr at 4C. The membrane was washed three times in TBS-T and incubated with 1:5000 of donkey anti-mouse IgM conjugated with peroxidase (Jackson Laboratories; BIO/CAN Scientific, Mississauga, Ont, Canada). Specific FORSE-1 bands were visualized by chemiluminescence detection (Amersham Pharmacia Biotech; Baie d'Urfe, PQ, Canada).
Results
Frequency and Distribution of FORSE-1 Immunoreactivity in Developing Lung
In tissue slice preparations and paraffin sections of gestational stage E16 rabbit fetal lungs, the FORSE-1 MAb strongly labeled airway epithelium in both large and small airways but not in the surrounding mesenchyme (Figures 1A and 1B). At this early stage, no 5HT-immunoreactive cells were identified within the primitive epithelium. At E18, while FORSE-1 epitope expression was still prominent on airway epithelial cells, co-localization of FORSE-1 antigen and positive 5HT immunoreactivity became apparent in some cells (Figures 1C and 1C'). Whereas FORSE-1 antigen was localized mostly on the apical surface of epithelial cells, 5HT expression was cytoplasmic (Figures 1C and 1C'). Between E21 and E28, FORSE-1 immunoreactivity became restricted to NEB cells, as identified by co-expression with 5HT (Figures 1D-1G). The FORSE-1 labeling exhibited a mostly apical and/or basolateral cell membrane localization. At E28 a low level of FORSE-1 epitope expression was detectable on most NEB cells and on surrounding cells in the vicinity of NEBs. However, a small number of cells within NEBs retained strong membrane FORSE-1 immunoreactivity. By comparison, 5HT immunoreactivity appeared diffusely distributed throughout the cytoplasm of NEB cells (Figures 1G and 1G'). During later gestational stages only trace amounts of FORSE-1 antigen were detected in some NEBs (not shown). Therefore, FORSE-1 epitope expression became gradually restricted to maturing NEBs, reinforcing the notion of developmentally regulated expression.
FORSE-1 epitope expression in situ in gestational series of fetal rabbit lung. (
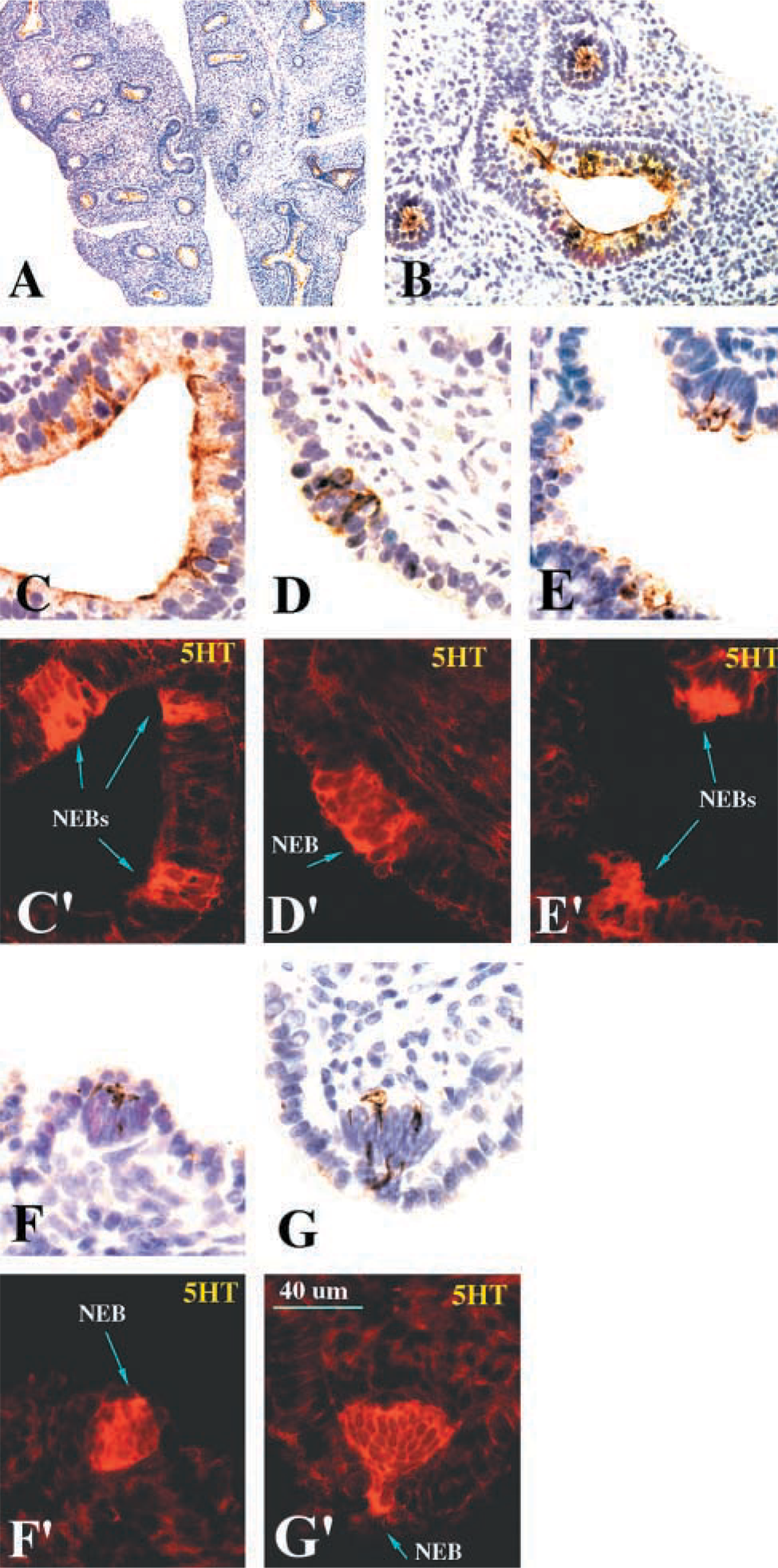
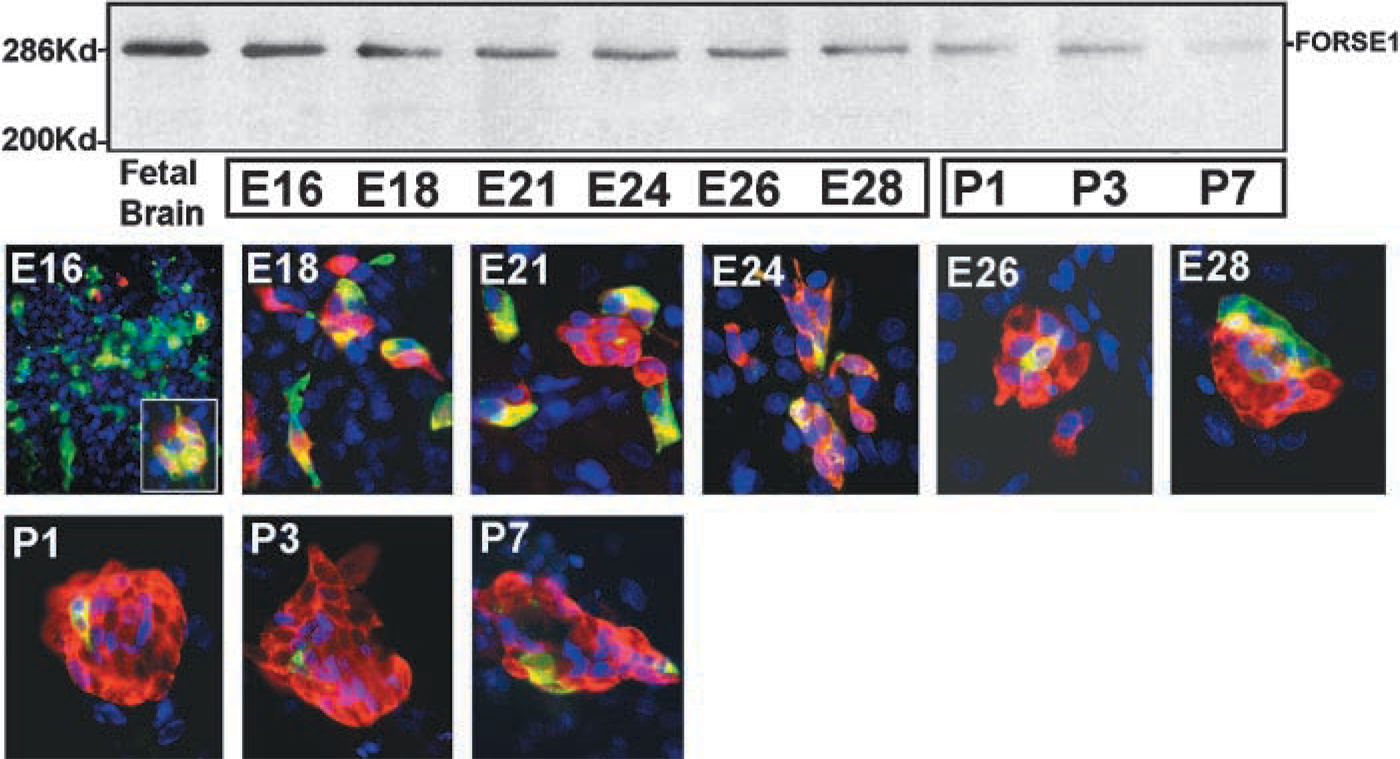
Primary cultures established from E16 lungs were maintained in vitro for up to 3 days to identify co-expression of the FORSE-1 epitope and 5HT. These cultures were also preincubated with BrdU to assess the proliferative capacity of the cells expressing the FORSE-1 epitope and 5HT immunoreactivity. The results of these experiments recapitulate the developmental patterns observed in situ. Most primitive epithelial cells were FORSE-1-immunopositive at E16, with evidence of rare cell clusters exhibiting coordinate expression with 5HT (Figure 2 and inset). The number of cells showing concomitant expression of FORSE-1 epitope and 5HT (Figure 2 and inset) increased between E18 and E21. However, this ratio then shifted; most cells expressed 5HT only by E28 (Figure 2). The number of NEB clusters increased significantly between E18 and E28. At E28, large NEBs contained cells with strong immunoreactivity for both the FORSE-1 epitope and 5HT. In cultures of lung on postnatal days 1(P1), P3, and P7, cells of large NEBs were predominantly 5HT-positive. Occasional FORSE-1-immunoreactive cells were observed. Cultures of cells from lungs at E16 and E24 maintained for up to 3, 5, 7, and 10 days in vitro and labeled with BrdU revealed that the number of BrdU-positive cells within NEBs and/or cells expressing FORSE-1 and 5HT also gradually decreased with gestational age (Figure 3A). Furthermore, in day 3 cultures of E24 lung the cells exhibiting concomitant expression of FORSE-1 and 5HT were BrdU-positive (Figure 3B), as were those cells expressing 5HT only. The fact that the BrdU-labeled 5HT-positive cells showed restricted growth, i.e., did not form colonies, suggests that this cell population has a long doubling time, as previously reported using [3H]-thymidine labeling (Cutz et al. 1985b). Overall, the confocal analysis of the FORSE-1/5HT-expressing cell population in culture corroborated the in situ observations, supporting the notion of a developmental sequence from FORSE-1-epitope-positive cells through FORSE-1/5HT immunoreactivity, followed by reduced FORSE-1 expression and a marked increase in 5HT-expressing cells exhibiting morphological features of mature NEBs.
Quantitation of FORSE-1- and 5HT-expressing Cell Populations
The numbers of single cells and cell clusters (four to ten cells and >10 cells) staining for FORSE-1, 5HT, and FORSE-1/5HT were counted both in short-term (4-day) cultures established from the gestational series lungs (Figures 4 and 5) and E16 lung maintained for up to 10 days in vitro (Figures 6 and 7). The numbers of FORSE-1-positive single cells declined dramatically with increasing gestational age or with time in culture, whereas the number of FORSE-1 positive cell clusters varied little. In contrast, there was a dramatic increase in 5HT expression in both single cells and NEB cell clusters. BrdU labeling of short-term cultures established from the gestational series of lungs showed a significant decrease in BrdU incorporation in single cells and cells within clusters co-expressing FORSE-1 and 5HT and a minimal decrease in cells or clusters expressing only FORSE-1 or 5HT (Figure 5). Therefore, quantitation of labeled cells revealed at least two subpopulations of cells, suggesting that co-expression of FORSE-1 and 5HT was associated with developmental maturation.
(
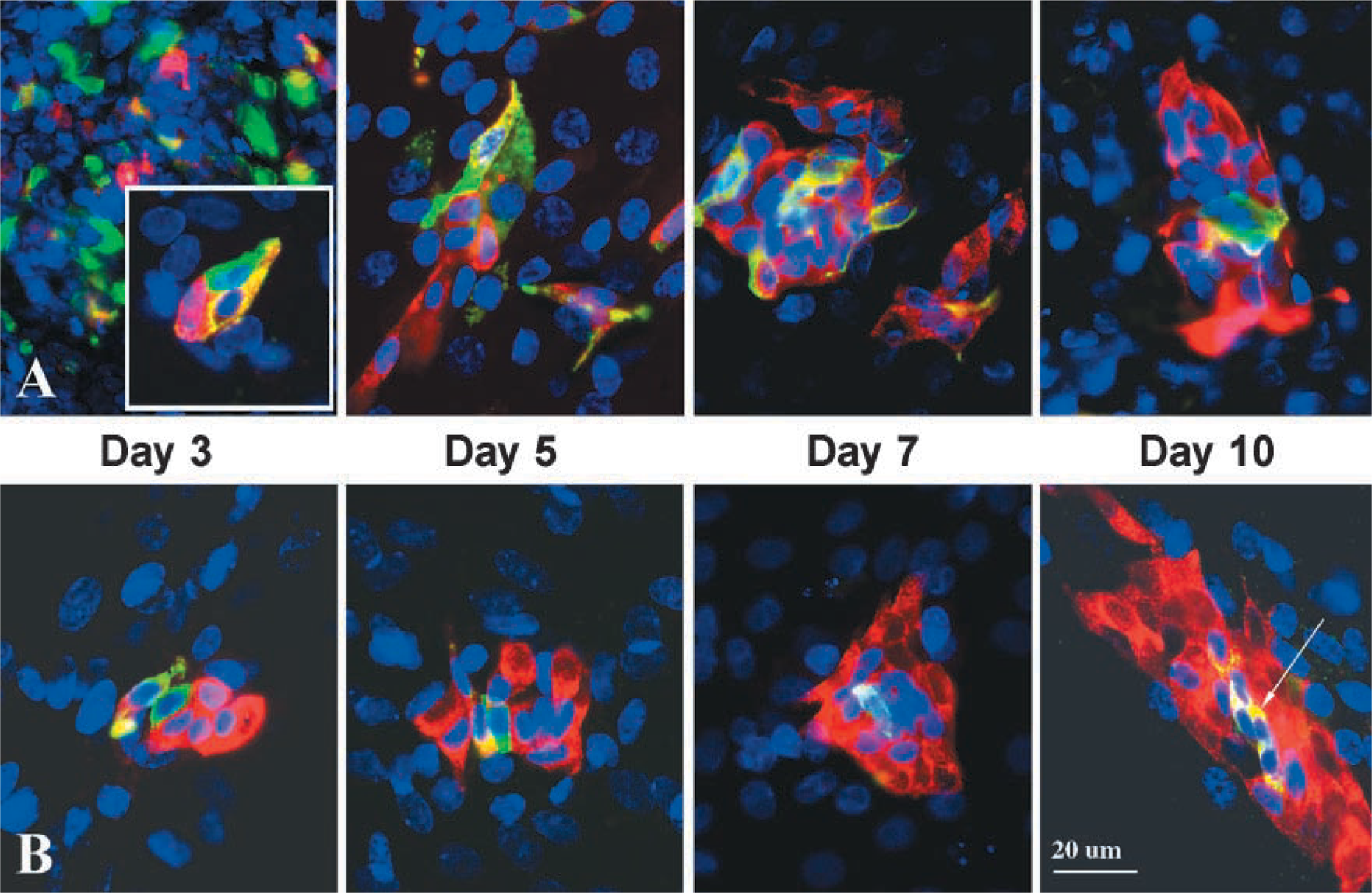
Confocal microscopy of FORSE-1 (green fluorescence, cytoplasmic) and 5HT (red fluorescence, cytoplasmic) immunoreactivity and BrdU incorporation (blue fluorescence, nuclear) in E16 (
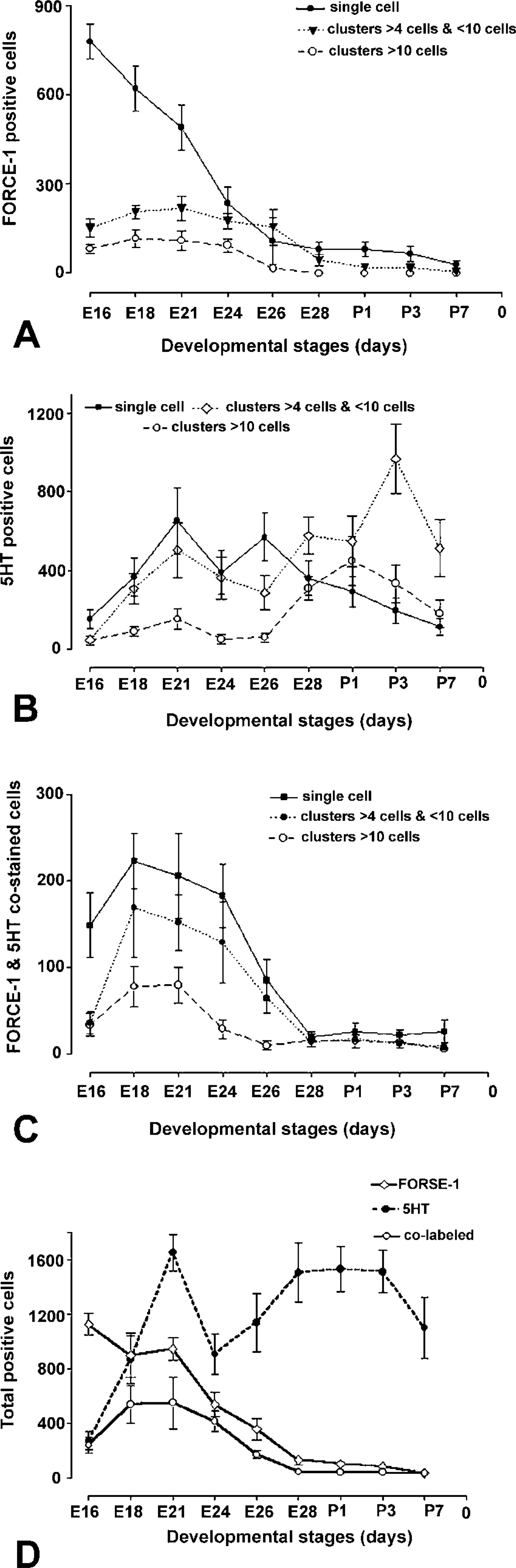
Fetal lung from gestational days E16–E28 and postnatal days P3 and P7 were cultured for 4 days and immunolabeled for FORSE-1 and 5HT. Positive-staining cells were counted and the results normalized for the total number of keratin-positive epithelial cells as described in Materials and Methods. (
This quantitative pattern of FORSE-1/5HT expression and correlation with BrdU labeling was essentially recapitulated in cultures of E16 lungs maintained for up to 10 days in culture (Figure 6). In this case, single cells, small clusters, and large clusters were scored for expression of FORSE-1, 5HT, and for incorporation of BrdU. Because FORSE-1 epitope expression diminished linearly with time in culture, 5HT expression increased and peaked in the day 5–7 culture period for single cells, in the 7–10 day period for small (4–10 cells) clusters, and by 10 days for large (>10 cells) clusters (Figure 6D). In contrast, cells co-expressing FORSE-1/5HT cells decreased gradually with time in culture, coordinate with decrease in the number of FORSE-1 only expressing cells and a significant increase in 5HT only expressing cells. Cells expressing 5HT appeared to cease proliferation only with increased time in culture, as observed in cultures labeled with BrdU (Figure 7). Overall, our data demonstrate a developmental sequence of coordinated expression of FORSE-1 and 5HT coinciding with phenotypic maturation of NEBs.
FORSE-1 Epitope Is Associated with a 286-kD Carrier Glycoprotein
Extracts of lungs from E16 to E28 and postnatal days 1, 3, and 7 were resolved by SDS-PAGE under non-reducing conditions and FORSE-1 epitope-containing proteins were immunoblotted with the FORSE-1 MAb and detected by the chemiluminescence method. The FORSE-1 epitope was associated with a major 286-kD protein band (Figure 2). This protein probably represents a glycoprotein and/or proteoglycan, based on polysaccharide composition of the FORSE-1 epitope previously established in brain studies (Tole et al. 1995). In the latter study, the FORSE-1 epitope was localized to a proteoglycan of >200 kD residing on the cell surface and was also associated with glycolipids. Because SDS-PAGE cannot resolve the possible association of the FORSE-1 epitope with potential glycolipids, it is possible that in the lung a number of other molecules could carry this epitope. Significantly, the expression level of the 286-kD glycoprotein diminished markedly with gestational age, corresponding closely to the reduction observed in immunolocalization studies.
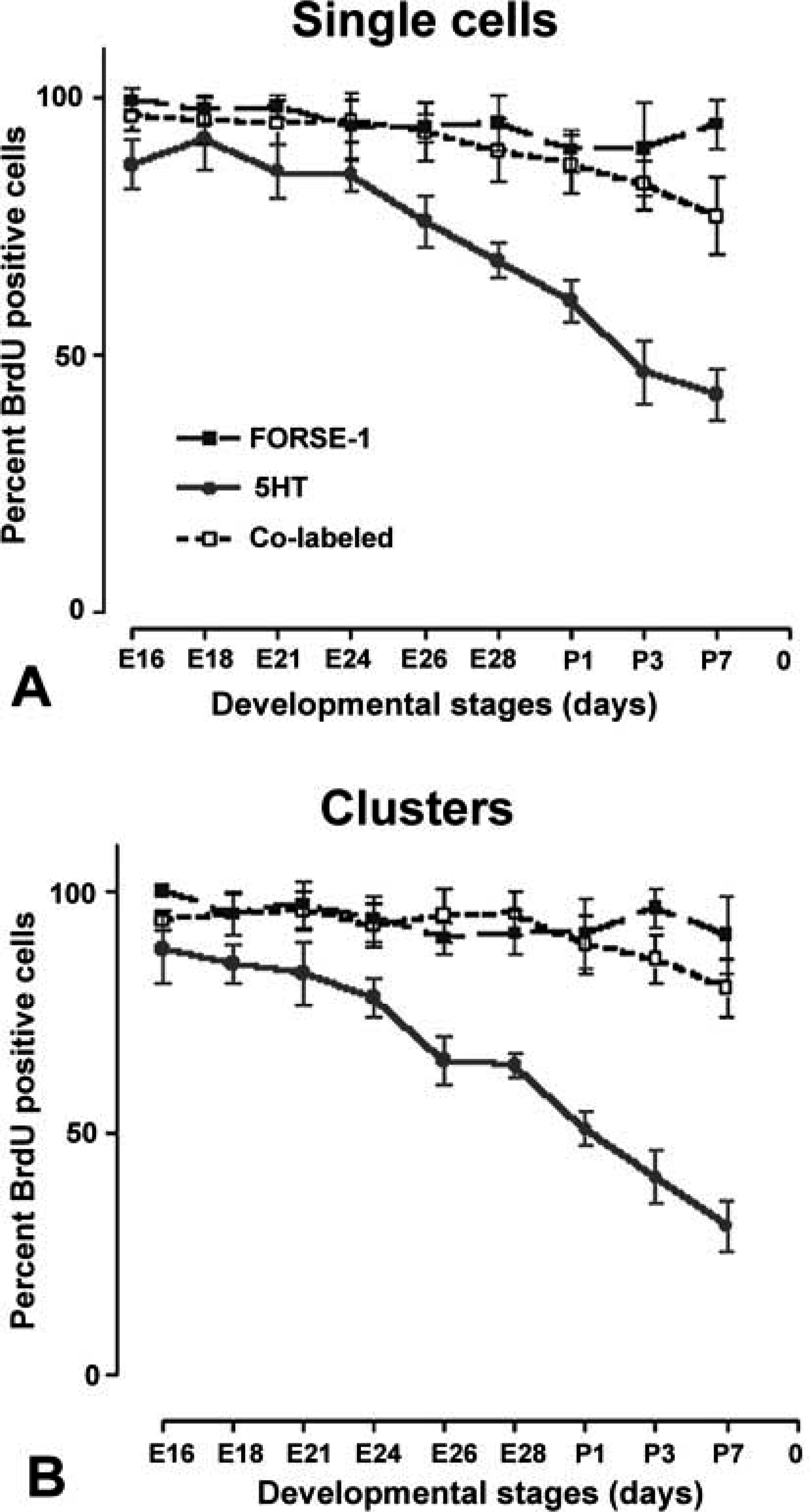
Fetal lungs from gestational days E16–E28 and postnatal days P3 and P7 were cultured for 4 days, labeled with BrdU and for FORSE-1 and 5HT. (
FORSE-1 Is Co-expressed with Stage-specific Embryonic Antigens (SSEAs)
The FORSE-1 epitope is a component of the LeX antigen, as demonstrated in studies on brain (Allendoerfer et al. 1995), and is related to the SSEA series of antigens. We therefore asked whether developing lung cells also express antigenic epitopes recognized by the series of antibodies specific for SSEA-1, SSEA-3, and SSEA-4 previously described in studies of developing embryos and embryonal carcinoma cells (Solter and Knowles 1978; Kannagi et al. 1983; Fenderson et al. 1987). Cultured cells were triple labeled with antibodies to FORSE-1, SSEA series, and 5HT, and were examined by confocal microscopy (Figure 8). Cells expressing only FORSE-1, co-expressing FORSE-1 and 5HT, and co-expressing SSEA antigens were found. Interestingly, as shown in Figure 8 (middle panel) SSEA-3 antigen was found on epithelial cells with the obvious morphological neuroendocrine feature of neurite-like processes. SSEA-3 antigen was concentrated in the terminal button, while FORSE-1 was localized to the cell body. In other cells, the FORSE-1 epitope and SSEA antigen-rich regions were found adjacent to each other, suggesting a possible association with different membrane components. The fact that individual cells concomitantly expressed 5HT and antigenic epitopes of FORSE-1, SSEA-1, −3, and −4 could implicate these carbohydrate epitopes in neuroendocrine cell development. These observations could also be simply markers of specific developmental stages.
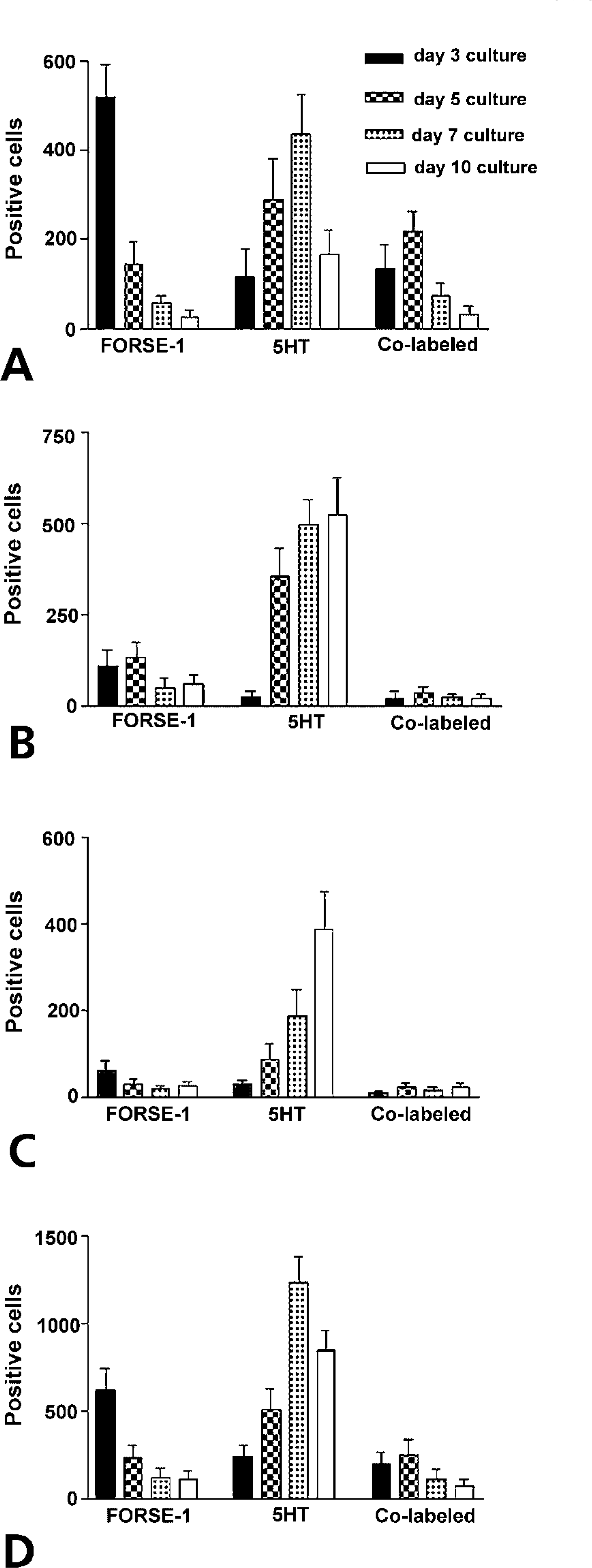
E16 fetal lungs were cultured for 3, 5, 7, or 10 days and immunolabeled. The graphs represent counts of FORSE-1 and 5HT positively labeled single cells, and co-labeled (
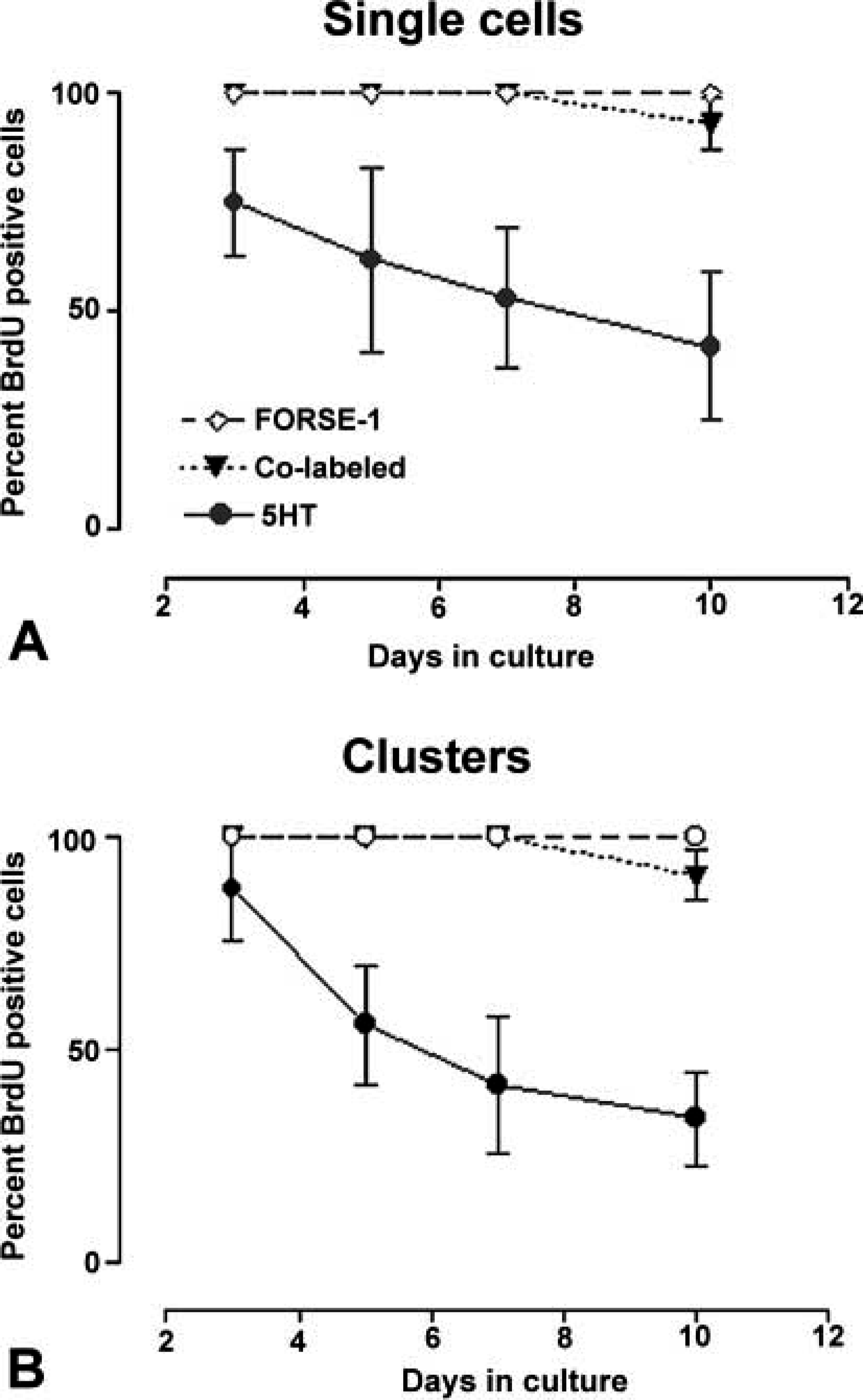
E16 fetal lungs were cultured for 3, 5, 7, or 10 days and BrdU labeled before immunostaining for FORSE-1 and 5HT. (
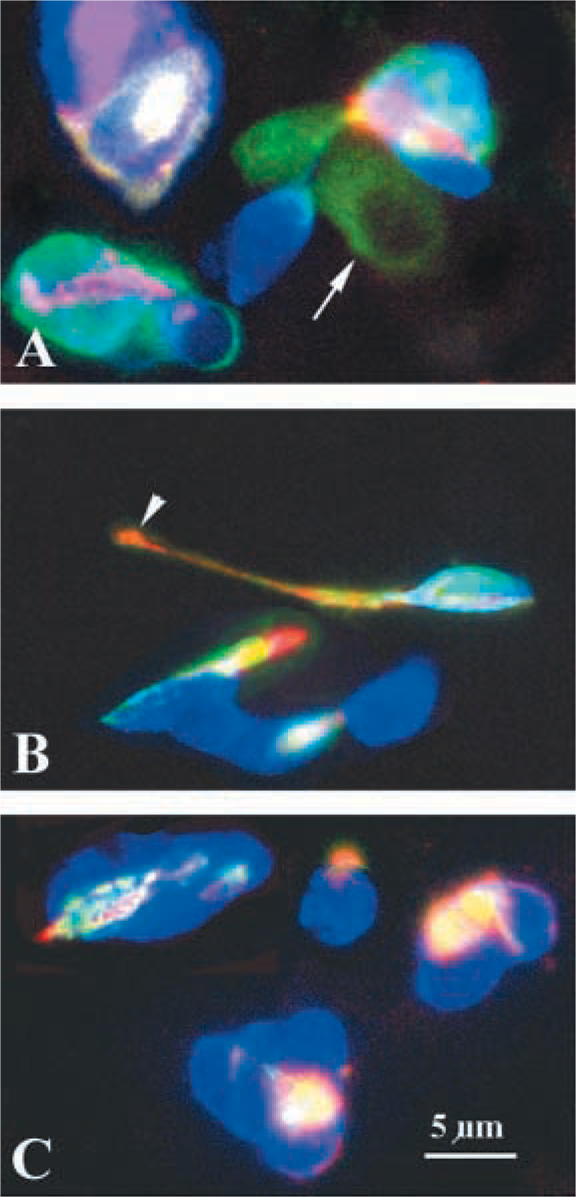
Confocal microscopy of E18 lung cultured for 3 days and triple labeled with antibodies against FORSE-1 (green fluorescence,
Discussion
Current thinking in the PNEC/NEB field supports the notion that the primitive undifferentiated pulmonary epithelium expresses a pluripotent phenotype before further commitment and differentiation into the respective specialized cell subtypes of the lung (Sorokin et al. 1993; Wuenschell et al. 1996; Sunday and Cutz 2000). In this regard, expressions of PGP9.5 and NSE are taken as indicators of the neuroendocrine phenotype (Haley et al. 1997). For example, expression of PGP 9.5 becomes restricted to differentiating PNECs and is lost in differentiating Clara cells, serous cells, and other cells of mucociliary epithelium. A similar pattern of expression has been observed for CGRP (Wuenschell et al. 1996). Our observation of FORSE-1 epitope expression in primitive undifferentiated epithelium before expression of 5HT supports this idea and also confirms earlier studies on SSEA antigens (Miyake et al. 1988; Itai et al. 1990). Because these antigens appear before PNEC differentiation, the inference might be that the pulmonary epithelium resembles a regionally restricted neuroepithelium (not unlike that observed in developing CNS) (Tole et al. 1995), which then comes under the developmental influence of the surrounding mesoderm. It is known that the mesenchyme in proximal and distal regions of the lung can dictate the ultimate phenotype of the pulmonary epithelium, whether Clara cell-like or mucociliary (Warburton et al. 1998). Expansion of the pulmonary tubules into the surrounding mesenchyme would trigger different mesenchymal-epithelial interactions that could then play a critical role in determining the temporal and spatial patterns of neuroendocrine differentiation. It is noteworthy that NEBs predominate in the bronchiolar regions and upper airways admixed with PNECs, whereas single PNECs are found only in the tracheal epithelium (van Lommel et al. 1999). Furthermore, a number of studies in different species have linked bombesin/GRP expression of PNECs/NEBs with mitogenic effects on surrounding epithelium and mesenchyme that express the bombesin/GRP receptor (Li et al. 1994; Wang et al. 1996; Emanuel et al. 1999). This probably constitutes a critical molecular mechanism in the epithelial-mesenchymal interactions that govern tubulogenesis, branching morphogenesis, and ultimate functional differentiation of the pulmonary epithelium.
Here we show that FORSE-1-positive cells acquire expression of 5HT in a temporal manner during gestation corresponding to the development of the airways (Figures 4 and 5). Cells co-expressing the FORSE-1 epitope and 5HT were replaced by mainly 5HT-expressing cells, signifying PNEC/NEB differentiation. Residual expression of a small number of the FORSE-1-positive cells was detectable in maturing NEBs. Recent studies have implicated NEB loci as the site for retention of a cell population with regenerative potential in the distal airways (Reynolds et al. 2000). Cells expressing both Clara cell marker and neuroendocrine cell markers appeared to reside with the NEBs or in close proximity (Reynolds et al. 2000). Subsequent [3H]-thymidine and BrdU labeling studies have identified a slowly proliferating cell population (label-retaining cells, LRCs) associated with NEBs and thought to constitute the “stem cells” of the lung (Hong et al. 2001). It is conceivable that cells retaining expression of the FORSE-1 epitope, residing nearby or within NEBs, may be equivalent to this slowly proliferating LRC population.
Studies on developing hamster lung have provided some additional insights into factors involved in the control of neuroendocrine differentiation and the role of NEBs in pulmonary epithelial proliferation (Hoyt et al. 1990, 1993; Ito et al. 1997). Essentially, more than 40% of labeled cells concentrated around NEBs that were lightly labeled, suggesting that neuroendocrine cells were added to mature NEBs through differentiation from adjoining airway epithelial cells. The FORSE-1 epitope expression pattern appears to support this idea and now identifies this potential progenitor population of airway epithelium.
The observation that the FORSE-1 epitope, equivalent to LeX, is gradually lost from PNECs/NEBs coincident with maturation of PNEC/NEB cells and acquisition of 5HT, suggests a potential role for FORSE-1 in this developmental programming. Co-labeling with antibodies to SSEA antigens supports the embryonic phenotype of the neuroendocrine-programmed cells that ultimately lose SSEA antigens as neuroendocrine maturation progresses towards term. Therefore, we found co-expression and patterns suggesting a sequential progression with NEB maturation. Earlier studies of SSEA-1-related antigens in developing human lung found that LeY, LeX, and sialyl LeX-i were strongly expressed in cells in lung buds, bronchial buds, and terminal buds, coincident with formation of the corresponding lung structures (Miyake et al. 1988; Itai et al. 1990). Tumors arising from lung epithelium re-expressed these antigens to high levels, becoming easily detectable in plasma, and suggested retrodifferentiation, of the cancer cells to stages of immature embryonic lung cells. In support of this idea of retrodifferentiation we have noted expression of FORSE-1 on small-cell lung carcinoma cell lines, the presumed counterpart to PNECs/NEBs in normal lung (unpublished observation). Therefore, our studies expand on previous studies demonstrating the association of the LeX/SSEA antigens with lung development and now with neuroendocrine cell differentiation.
Glycosylation patterns of proteins and lipids residing on the cell surface have been extensively investigated as markers of embryogenesis and fetal development and as determinants of tumor-associated antigens and tumor properties, particularly metastasis (Dabelsteen 1996; Hakamori 1996). Stage-specific embryonic antigens, specifically the lacto-series SSEA-1, globo-series SSEA-3 and −4 oligosaccharides were first identified in studies on embryonal carcinomas and embryonic stem cells, where it was found that they define spatial and temporal differentiation of the ectoderm, mesoderm, and endoderm (Solter and Knowles 1978; Kannagi et al. 1983). Expression of SSEA-3 antigen is considered a surface marker for primitive and visceral endoderm (Damjanov et al. 1994). Our observation of SSEA-3 expression on cells that express 5HT supports the notion of derivation of PNECs (and, for that matter, the primitive pulmonary epithelium) from an endodermal lineage (Haley et al. 1997).
Interestingly, our immunolabeling results suggest that the FORSE-1 epitope may be expressed exclusive of SSEA antigens or may precede SSEA antigen expression (Figure 6). Because FORSE-1 is essentially equivalent to LeX and the globo-series SSEA antigens must become fucosylated to become the lactoseries antigens as defined by Lewis X (LeX) and variants of LeX, such as sialylLeX, which represent terminal carbohydrate structures, the latter possibility is less likely. Therefore, FORSE-1 epitope expression may still constitute a unique antigen within the Lewis antigen group.
The Lewis antigens are identifiable with specific monoclonal antibodies and are strongly associated with a number of different tumors (Kurahara et al. 1999; Liu et al. 2001), particularly lung carcinomas, in which their expression correlates with tumor burden and metastatic potential (Miyake et al. 1988; Itai et al. 1990). Although expression of Lewis antigens has been described in development of normal lung (Miyake et al. 1988; Itai et al. 1990), the studies presented here are the first demonstration of expression of the LeX/SSEA-1 antigenic epitope in the pulmonary neuroendocrine lineage. The function of the Lewis antigens (sialyl Lex, Lea) includes that of ligands for selectins (P-selectin, E-selectin) required for cell-cell adhesion and of ligands governing rolling attachment of immune cells and tumor cells to endothelium (Dabelsteen 1996; Fukuda et al. 2000; Luo et al. 2001). Considering their roles in differentiation, cell-cell and cell-matrix interactions, we postulate a similar requirement of the FORSE-1/LeX antigen for interactions of differentiating and maturing committed PNEC/NEB cells.
Confocal microscopy revealed localization of the FORSE-1 epitope to the plasma membrane of this progenitor subpopulation of PNECs/NEBs, suggesting probable involvement in cell-cell interactions and/or cell-matrix interactions. Western blotting analysis demonstrated the FORSE-1 epitope on a 286-kD carrier protein which decreased significantly by postnatal day 7. Allendoerfer et al. (1999) have recently shown that, in developing brain, the FORSE-1 epitope is carried primarily by glycolipids during embryonic development and on glycoproteins and proteoglycans in postnatal and adult tissue. Our observations suggest, although not exclusively, that in developing lung the FORSE-1 epitope is associated with a large protein throughout the gestational period. Because the FORSE-1 epitope has been associated with cell adhesion and differentiation in the CNS (Allendoerfer et al. 1995, 1999; Tole et al. 1995), we propose that the temporally and spatially restricted expression of FORSE-1 epitope carrier protein in developing lungs may signify similar roles in pulmonary epithelium and vis-a-vis PNEC/NEB differentiation.
Among the molecules that are involved in cell-cell interactions, we previously identified NCAM (neural cell adhesion molecule) on PNECs/NEBs using the MOC-1 monoclonal antibody (Spiers et al. 1992). In an organ culture system derived from E11 hamster fetal lung, Ito et al. (1997) used the NCAM marker to show that neuroendocrine cells differentiated from non-neuroendocrine epithelial cells devoid of mesenchyme but with a requirement for basement membrane (Matrigel). This appears to indicate pre-programming of neuroendocrine cell differentiation dependent on the extracellular matrix (ECM) or on factors associated with the ECM. Whether NCAM- and FORSE-1-containing glycoproteins are developmentally interrelated is not known, although studies in developing brain suggest that they may be mutually exclusive (Tole et al. 1995). In fact, NCAM expression is prominent in mature NEBs (Spiers et al. 1992) when FORSE-1 expression appears to be greatly restricted to a few cells. Antibody to NCAM inhibited the formation of NEB in explant cultures of hamster lung (Ito et al. 1997). Therefore, the FORSE-1 carrier protein (potentially a proteoglycan) may serve as an inhibitory signal for NEB cell maturation, thereby providing a reservoir of PNEC progenitors. As PNEC/NEB cells undergo functional maturation most of the FORSE-1 immunoreactivity disappears. This could mean a downregulation of the glycoproteins containing the specific FORSE-1 epitope or possibly masking of the epitope by sialylation or other modifications (Fenderson et al. 1987; Dabelsteen 1996). Tentatively, the finding that FORSE-1 epitope is an antigen associated with a high molecular weight glycoprotein, presumably a proteoglycan, supports the notion of a “receptor” for an ECM-localized differentiation signal.
Taken together, these earlier studies and our present data favor the idea of a potential progenitor cell type that is capable of multipotent differentiation depending on requirements of the pulmonary epithelium for repair after injury or in response to environmental influences (e.g., hypoxia). Retention of FORSE-1-expressing cells residing within mature NEBs suggests that this may be the cell type that is programmed for regeneration of pulmonary epithelium after toxic injury (Reynolds et al. 2000; Hong et al. 2001). Whether the FORSE-1 antigen-expressing cells in developing rabbit lung correspond to a developmentally committed putative progenitor of the PNEC/NEB cell system remains to be determined. The fact that a residual FORSE-1/5HT-immunopositive cell subpopulation could still be detected postnatally suggests possible retention of progenitors into adulthood. Whether this FORSE-1-expressing cell is capable of pluripotent differentiation requires further investigation.
Footnotes
Acknowledgements
Supported by funding from Canadian Institutes of Health Research (CIHR), operating and Group Grant on Lung Development (EC and HY, MOP12742 and MGP15270).
We thank Ms Elizabeth Bienkowski and Ms Veronica Wong for technical assistance. The FORSE-1 monoclonal antibody developed by Dr P.H. Patterson was obtained from the Developmental Studies Hybridoma Bank developed under auspices of the NICHD and maintained by the University of Iowa (Department of Biological Sciences; Iowa City, IA).