Abstract
Hepatocyte growth factor (HGF) is a potent inducer of hepatocyte proliferation and is expressed during liver failure. In this study we used the in situ reverse transcriptase-polymerase chain reaction (RT-PCR) method to detect HGF mRNA expression in normal rat livers and cirrhotic rat livers induced by treatment with N-nitrosodimethylamine (DMN). In normal control livers, in situ RT-PCR detected HGF mRNA expression in Ito cells and Kupffer cells, both of which showed rounded morphologies. However, in the cirrhotic livers induced by DMN, HGF mRNA-positive cells were spindle-shaped and surrounded the hepatocytes located around the sinusoids. These cells appeared to be sinusoidal endothelial cells as well as Ito and Kupffer cells. Because it has been suggested that HGF expression is related to transforming growth factor-β (TGF-β) levels that may play an essential role in disease progression in cirrhotic livers, TGF-β mRNA expression in normal and cirrhotic livers was also compared using in situ RT-PCR. Our results confirmed that expression of TGF-β mRNA co-localized with HGF mRNA expression in the cirrhotic liver.
H
Although HGF is present at low concentrations in the serum of apparently normal individuals (Zarnegar and Michalopoulos 1989; Tsubouchi et al. 1991), serum concentrations of HGF rise in response to liver injury, and increased HGF mRNA levels have been shown in partially hepatectomized rats (Zarnegar et al. 1991). In animal models, the administration of recombinant human HGF to rats with hepatic fibrosis/cirrhosis prevented the onset and progression of hepatic disease (Ishii et al. 1995; Matsuda et al. 1995). It was also reported that HGF may be important for tissues other than the liver, such as kidney, placenta, brain, lung, pancreas, and hemopoietic tissues, (Michalopoulos and Zarnegar 1992). Recent data have suggested that HGF may be involved in the protection of the kidney and stomach against injury (Kawaida et al. 1994; Takahashi et al. 1996; Kinoshita et al. 1998). Therefore, it was recently proposed that HGF gene therapy might be potentially useful in the treatment of patients with fatal liver diseases or liver conditions otherwise untreatable by conventional therapy (Ueki et al. 1999). Experiments in that study showed that introduction of the HGF gene suppressed the expression of TGF-β induced by N-nitrosodimethylamine (DMN). TGF-β plays an essential role in the progression of fibrosis in various organs including the liver. An in vitro study using lung fibroblasts showed that TGF-β negatively regulated HGF gene expression (Matsumoto et al. 1992). Therefore, it is possible that HGF may be beneficial in liver cirrhosis by inhibiting TGF-β.
Studies using in situ hybridization methods have suggested that HGF mRNA is localized to Kupffer, Ito, and sinusoidal endothelial cells in the cirrhotic livers of rats and humans (Noji et al. 1990; Yamaguchi et al. 1996). However, it has also been reported that, in normal rat liver, only Ito cells expressed HGF mRNA, and not other cells, including parenchymal, endothelial, and Kupffer cells (Schirmacher et al. 1992). With regard to TGF-β mRNA expression, mesenchymal cells, especially desmin-positive perisinusoidal cells (e.g., fat-storing cells and myofibroblasts), as well as liver periductal and periportal cells, appear to be the principal sources of TGF-β in normal, regenerative, and fibrogenic liver states (Nakatsukasa et al. 1990a, b, 1991).
In the present work we studied both normal and cirrhotic rat livers. Cirrhosis was induced by administration of DMN (Jenkins et al. 1985; Jezequel et al. 1987). We then performed in situ RT-PCR to demonstrate the localization of HGF and TGF-β mRNA transcripts.
Materials and Methods
Animal Treatments
Male Sprague-Dawley rats (Slc:SD; Japan SLC, Shizuoka, Japan) weighing 170–210 g were used in all experiments. For the development of hepatic fibrosis/cirrhosis, 10 μl/ml DMN (Wako; Osaka, Japan) dissolved in saline was given IP at 1 ml/kg body weight three times per week for 5 weeks. Ten rats were treated with DMN and five were treated with saline as controls. After the 5 weeks, surviving rats were anesthetized with sodium pentobarbital (Dainihon Pharmaceuticals; Osaka, Japan) (50 mg/ml), sacrificed by bleeding from the carotid artery, and the livers removed immediately afterwards.
For measurement of HGF concentration, we prepared 10 rats as a different group. In this satellite group, five rats were administered DMN and five were treated as controls.
Specimen Preparation
Liver sections were fixed in Streck Tissue Fixative (STF) Molecular Biology Grade (Streck Laboratories; Omaha, NE) overnight as a substitute for formalin. STF included diazoidinyl urea, 2-bromo-2-nitropropane-1,3-diol, zinc sulfate, sodium citrate (Nace et al. 1999; Pietrzak-Johnston et al. 2000). Paraffin-embedded sections were first stained using Azan-Mallory dyes followed by hematoxylin and eosin. Unstained specimens were used for immunohistochemical and in situ RT-PCR examination. For immunohistochemical examination, single sections were placed on each glass slide. For in situ RT-PCR examination, three sections were placed on each in situ PCR glass slide (Perkin Elmer; Branchburg, NJ), with one section used as the positive control, one as the negative control, and one as the sample for detection.
Measurement of HGF Concentration
Plasma samples were collected from cervical veins of conscious rats at 1, 2, 3, 4, and 5 weeks after the start of the course of injections. Each sample consisted of approximately 250 μl plasma collected after centrifugation with heparin solution. Collection was performed just before injection and at 4 and 24 hr after. Plasma HGF concentrations were measured using a rat HGF EIA kit (Institute of Immunology; Tokyo, Japan).
Immunohistopathological Examination
Sections pretreated with 0.1% trypsin for 10 min at RT were immunohistochemically stained using a Histofine PO kit (Nichirei; Tokyo, Japan) as previously described (Ishidoya et al. 1995). Incubations were performed with goat anti-human collagen (types I, III, and IV) polyclonal antibodies (1:20 dilution, 60 min, RT) (SBA; Birmingham, AL) or mouse anti-human α-SMA monoclonal antibody (1:100 dilution, 60 min, RT) (DAKO; Tokyo, Japan) as primary antibodies. Diaminobenzidine substrate was used for signal detection. Sections were counterstained with hematoxylin (Muto Industrial; Tokyo, Japan).
In Situ RT-PCR Examination
Detection of HGF and TGF-β mRNA was carried out using a two-step in situ RT-PCR procedure as previously reported (Lee et al. 1993; McMillan et al. 1996; Nuovo 1996, 1997; Iwasaka et al. 2000; Sinigalliano et al. 2001) with the following modifications. Sections were pretreated with 10 μg/ml proteinase K (Sigma Aldrich; St Louis, MO) in 0.01 M PBS, pH 7.4, at RT for 10 min. Rat HGF sequence (Tashiro et al. 1990), obtained from Oligoexpress (Amersham Pharmacia Biotech; Tokyo, Japan) was used to design forward (5′-AGCTCAGAACCGACCGGCTTGCAACAGGAT-3′, nucleotides 61–90) and reverse (5′-TTACCAATGATGCAATTTCTAATATAGTCT-3′, nucleotides 511–540) primers for HGF mRNA amplification. For TGF-β mRNA, primers 5′-CTTCAGCTCCACAGAGAAGAACTGC-3’ (nucleotides 1267–1291, forward) and 5′-CACGATCATGTTGGACAACTGCTCC-3’ (nucleotides 1564–1540, reverse) were used (Clontech Laboratories; Palo Alto, CA).
Positive controls consisted of performing in situ RT-PCR in the absence of DNase. RT reactions were performed by the application of 2.5 U/μl MuLV-reverse transcriptase (Perkin Elmer; Norwalk, CT) to the slides. Buffer without MuLV-reverse transcriptase was applied one section on each slide as a negative control. Slides were incubated at 42C for 30 min, then 94C for 5 min.
PCR amplification was performed using the hot-start procedure in a GeneAmp In Situ PCR System (PE Biosystems; Tokyo, Japan) thermal cycler. Slides were incubated first at 94C for 5 min (initial denaturation), followed by 30 cycles at 94C for 1 min and 55C for 3 min for denaturing and annealing, respectively.
After PCR amplification, slides were washed in 0.1 M Tris-buffered saline, pH 9.5, containing 0.15 M NaCl and 50 mM MgCl2 (washing buffer), and soaked in 0.1 X SSC containing 0.4% bovine serum albumin at 45C for 5 min to block nonspecific binding activity (stringent wash). Immunohistochemical signal detection was carried out using mouse anti-digoxigenin monoclonal antibody Fab fragments conjugated to alkaline phosphatase (1:200 dilution, 30 min, RT) (Roche; Mannheim, Germany), and signals visualized by nitroblue tetrazolium chloride (NBT) (Sigma Aldrich Japan) and bromochloroindoxyl phosphate (BCIP) (Sigma Aldrich Japan).
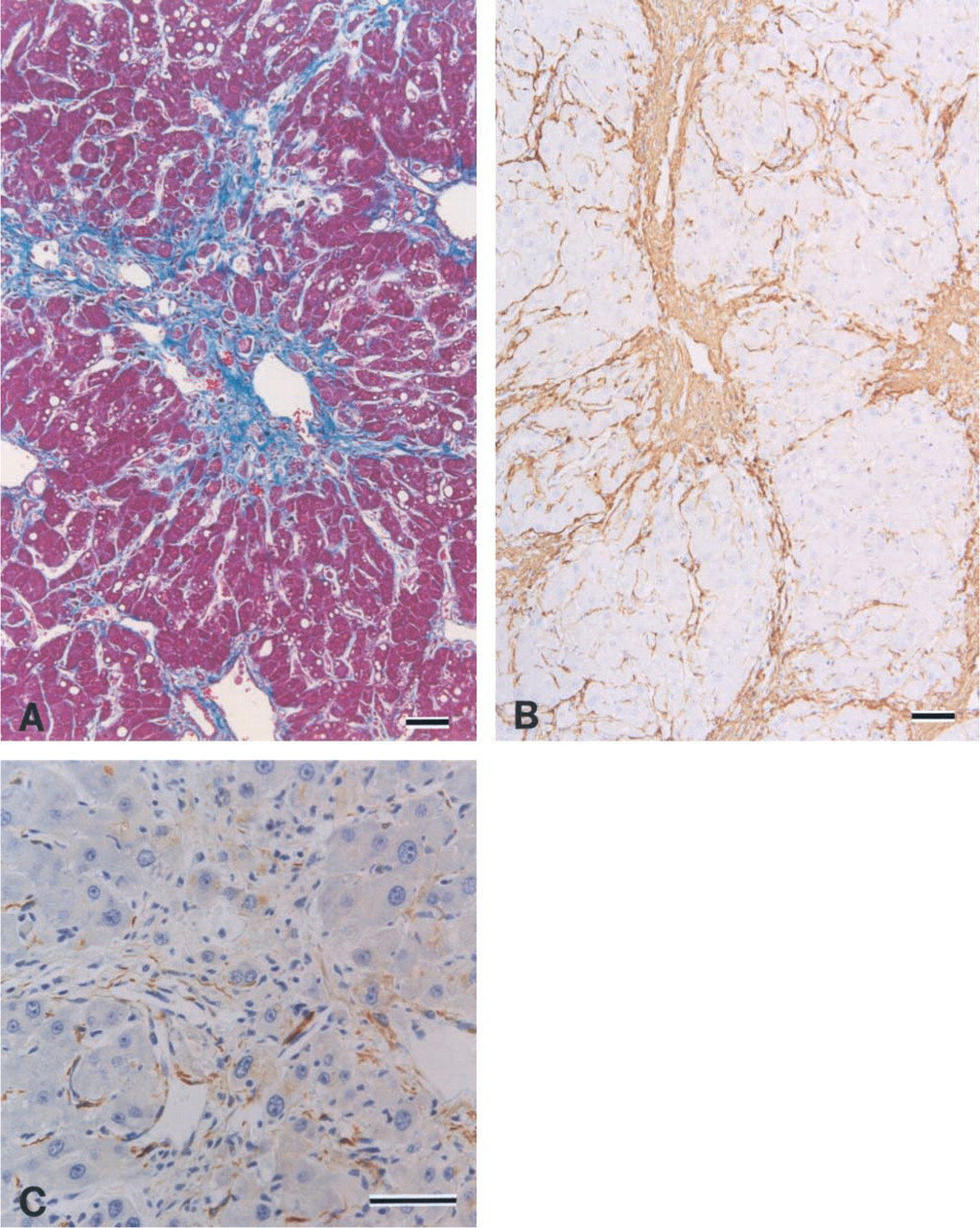
Paraffin sections of cirrhotic livers from rats administered DMN for 5 weeks, stained with Azan-Mallory dyes (
Results
Preparation of Cirrhotic Livers in Rats by DMN Administration
Fifty percent of DMN-treated rats died within 5 weeks as a result of the DMN administration. Surviving rats were sacrificed and the livers harvested for histopathological examination. Cirrhosis was observed in all DMN-treated rats, both grossly and histopathologically (Figure 1A). The presence of fibrous areas was confirmed by staining with goat anti-collagen (types I, III, and IV) polyclonal antibodies (Figure 1B, type III) and mouse anti-α-SMA monoclonal antibody (Figure 1C). In controls, α-SMA-positive cells were detected mainly in the portal spaces, either as elements of vascular walls or as fibroblast-like cells scattered in the connective tissue, or finely apposed to bile ductules. In the lobule, actin immunoreactivity was detected in the walls of large- and medium-sized terminal hepatic veins. After the DMN treatment, widened and irregular septa containing actin-positive cells joined centrilobular veins, resulting in a micronodular cirrhosis pattern.
Determination of HGF Concentrations in the Plasma of Rats During DMN Administration
We used ELISA to determine plasma HGF concentrations in satellite group. In the treated rats of the satellite group, three rats died 4 weeks after treatment. Although concentrations at 4 hr after administration were higher than those just before administration, concentrations at 24 hr were lower than those at 4 hr after administration. However, serum HGF levels increased as the number of DMN administrations increased and reached a maximum just before death (Table 1).
Detection of HGF and TGF-β mRNA in Normal and Cirrhotic Livers Using In Situ RT-PCR
Using the in situ RT-PCR method, DNA signals were detected in cell nuclei on positive control slides (data not shown) that were not pretreated with DNase. However, no signals were detected on the negative control slides, in which RT-PCR was performed without MuLV-reverse transcriptase (Figures 2A and 2C).
In situ RT-PCR results confirmed that HGF mRNA was expressed in Kupffer cells located in the sinusoid and in Ito cells located in the space of Disse in normal livers (Figure 2B). HGF mRNA expression was also detected in sinusoidal endothelial cells in cirrhotic livers (Figures 2D and 2E). Whereas the morphology of these cells was rounded in control livers, the cells were spindle-shaped and surrounded the hepatocytes in cirrhotic livers. Hemangioendothelial cells and hemangiopericytes expressed HGF mRNA in both normal and cirrhotic livers.
Concentration of the HGF in the plasma collected from rats treated with DMN for 5 weeks
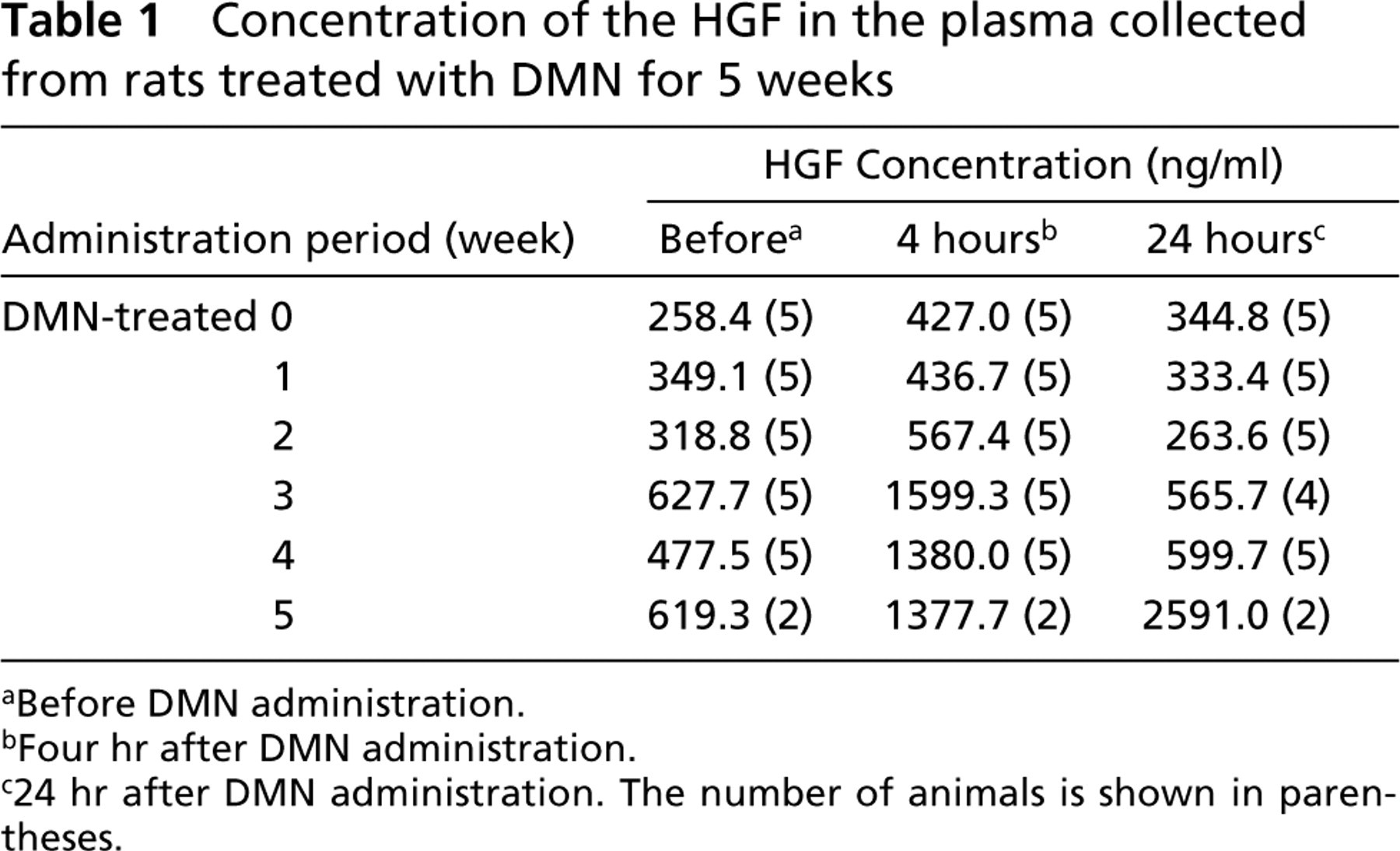
aBefore DMN administration.
bFour hr after DMN administration.
c24 hr after DMN administration. The number of animals is shown in parentheses.
TGF-β mRNA signals were almost undetectable in normal livers except the hemangioendothelial cells and hemangiopericytes (Figure 3A). Some mesenchymal cells, including Ito cells and Kupffer cells, slightly expressed TGF-β mRNA. However, in cirrhotic livers, results showed that cells expressing TGF-β mRNA corresponded to areas of HGF mRNA expression (Figure 3B).
Discussion
It was recently reported that HGF mRNA appeared to be produced by mesenchymal cells, including Ito, Kupffer, and sinusoidal endothelial cells, in cirrhotic livers in both rats and humans (Noji et al. 1990; Yamaguchi et al. 1996). However, other research, using Northern blotting analyses and in situ hybridization in normal rat livers, suggested that HGF was expressed in fat-storing cells rather than in the myofibroblast-like cells derived from these fat-storing cells that are linked to the fibrogenesis seen in chronic liver disease (Schirmacher et al. 1992).
A large number of studies have examined TGF-β expression. Reports showed that mesenchymal cells, especially desmin-positive perisinusoidal cells (e.g., fat-storing cells and myofibroblasts), as well as periductal and periportal cells of the liver, are the principal sources of TGF-β transcripts in normal, regenerative, and fibrogenic liver states by in situ hybridization (Nakatsukasa et al. 1990a, b, 1991).
Recently, it was shown that the TGF-β appeared to inhibit HGF synthesis through suppression of HGF gene expression (Matsumoto et al. 1992), indicating a close association between TGF-β and HGF. In this study, we used in situ RT-PCR to demonstrate the localization of HGF and TGF-β mRNA in normal and lethally cirrhotic livers. The use of in situ RT-PCR allowed greater sensitivity compared to Northern blotting analysis or in situ hybridization.
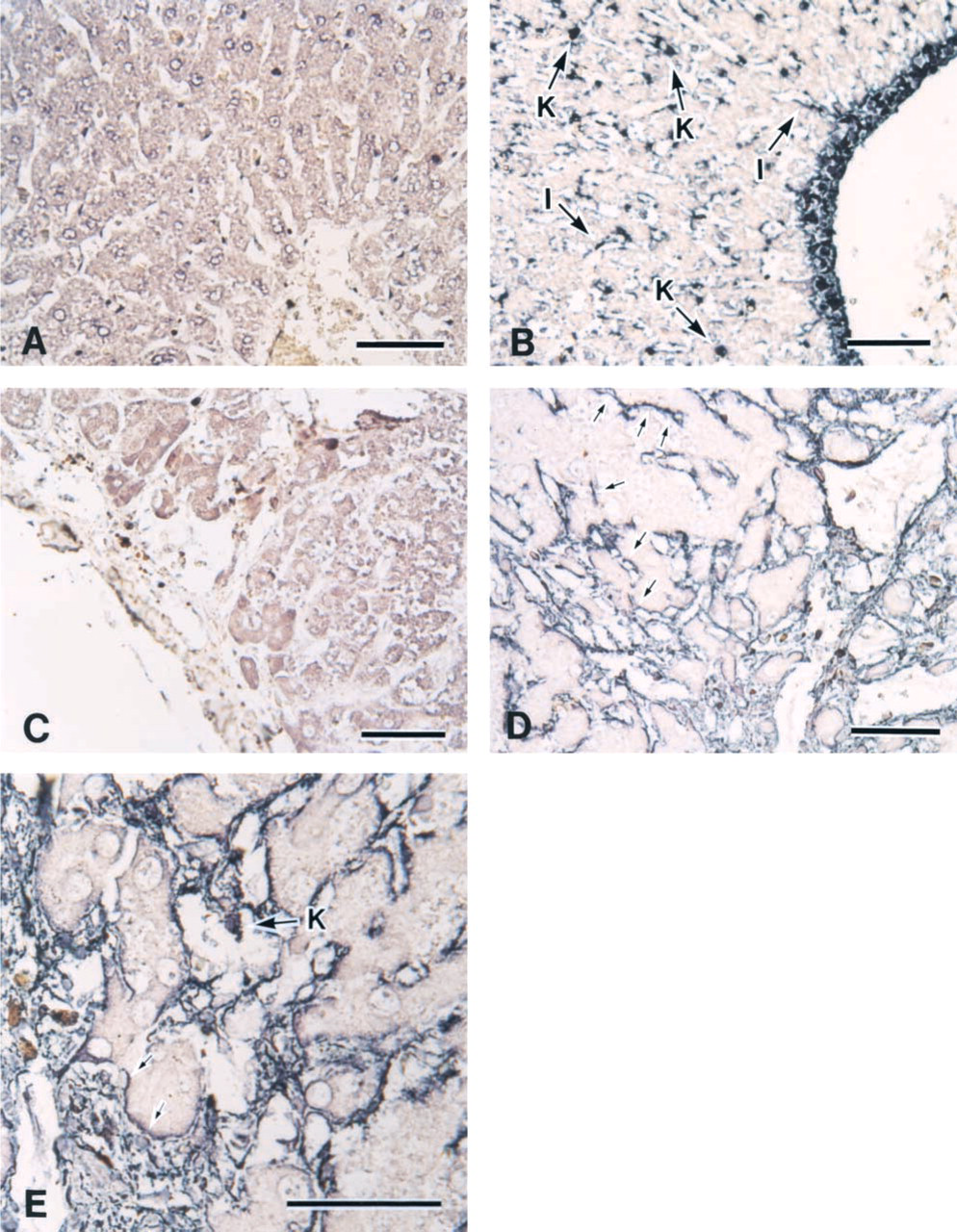
In situ RT-PCR detection of rat HGF in normal (
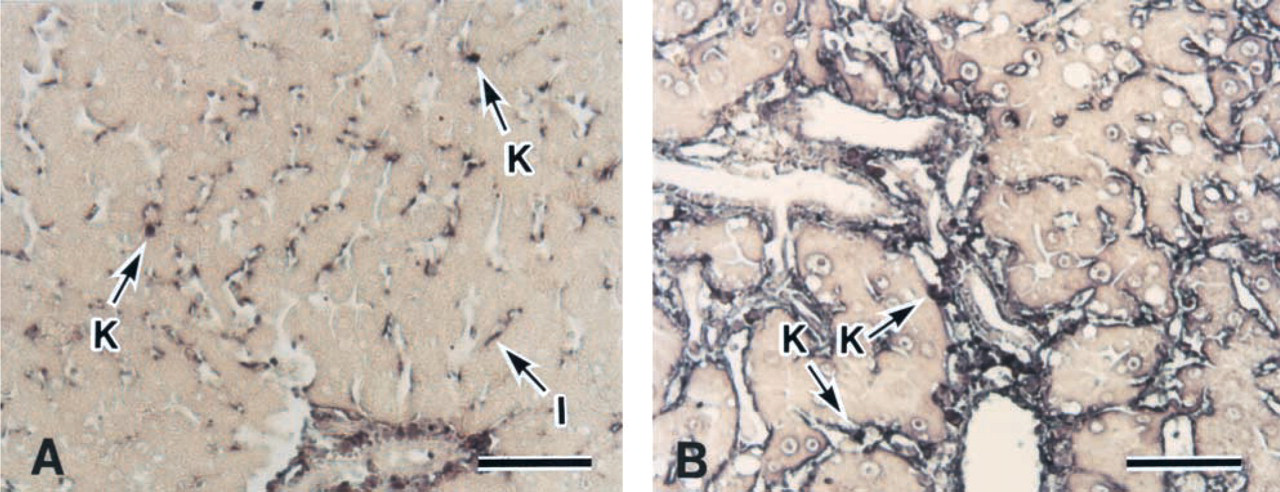
In situ RT-PCR detection of rat TGF-β in normal (
In this study for detection of the HGF mRNA with certainty, we performed determination of plasma HGF concentrations. As the result of the ELISA analyses, the concentration peaked at 4 hr after administration during the periods of administrations except just before death. At 5 weeks after the dosing of DMN, plasma HGF concentrations reached a maximal level continually. In human liver disease, it is known that plasma HGF levels of patients in acute hepatitis are higher than those in chronic hepatitis. In this model of DMN, however, it was observed that the higher plasma HGF levels, the more severe the fibrosis in the liver cirrhosis. Therefore, we could investigate the gene expression of the HGF and TGF-β in the more severe fibrosis producing HGF by using the in situ RT-PCR method.
As the result of in situ RT-PCR, HGF mRNA expression was observed in mesenchymal cells around blood vessels and bile ducts in both normal and cirrhotic livers. In the normal liver, HGF mRNA was also detected in rounded mesenchymal cells, thought to be Ito cells and Kupffer cells, located in the space of Disse (Wake 1980). Although in previous reports only weak HGF mRNA expression was observed by in situ hybridization method, HGF protein can be detected in plasma from normal rats and humans. Therefore, the relatively strong HGF mRNA signals detected in our study may be the result of using a more sensitive detection technique compared to previous studies. We also detected TGF-β mRNA expression that corresponded to areas that expressed HGF mRNA, which provided clear evidence of HGF and TGF-β mRNA co-localization in normal rats.
We also examined HGF and TGF-β mRNA levels in cirrhotic livers in rats given DMN. In this study, half of the rats treated with DMN were dead within 5 weeks due to lethal DMN-induced liver cirrhosis. Histopathological analysis showed excessively expanded fibrous areas in the cirrhotic livers, with pseudonodule formation and most hepatocytes undergoing degeneration or necrosis. Immunostaining for α-SMA was used to identify myofibroblasts. It has been reported that, in DMN-treated cirrhotic livers, activated hepatic stellate cells (also known as fat-storing or Ito cells) are also α-SMA-positive, such that α-SMA-positive cells are thought to be derived from Ito cells and from a subset of mesenchymal cells (Baroni et al. 1996; Shimizu et al. 2001; Tada et al. 2001). In this study, α-SMA-positive cells were observed around the blood vessels in both normal and cirrhotic livers, and some spindle-shaped cells were also stained in cirrhotic liver only, in agreement with previous reports. Although HGF mRNA was detected in the spindle-shaped cells, it was located mainly in the surrounding hepatocytes. With regard to TGF-β mRNA in the cirrhotic liver, we clearly confirmed findings that cells positive for TGF-β mRNA co-localized with cells expressing HGF mRNA. It appeared that the regions expressing both HGF and TGF-β mRNA were sinusoidal endothelial cells and Kupffer cells, as well as Ito cells (α-SMA-positive cells). HGF and TGF-β mRNA-expressing cells corresponded to mesenchymal regions stained by anti-collagen (especially type IV collagen) polyclonal antibodies. As a result in the immunohistochemical examination, the positive area stained by anti-collagen type IV was larger than those areas stained by anti-collagen type I or III. Hepatic fibrosis, regardless of cause, is characterized by increased extracellular matrix components that collectively form hepatic scar tissues consisting of fibril-forming collagens (types I and III). Type IV collagen is known to be a major component of the basement membrane, and type IV collagenase is secreted by myofibroblast-like cells derived from lipocytes (also known as stellate, fat-storing, perisinusoidal, or Ito cells) (Friedman 1993). It is generally believed that myofibroblast-like cells derived from fat-storing (Ito) cells and Kupffer cells have the ability to secrete type IV collagenase and thus to repair the damaged liver. Recently, it was reported that the degree of HGF receptor (c-met) gene expression changed during myogenesis to fat-storing (Ito) cells (Kastner et al. 2000). However, TGF-β appears to be the main fibrogenic mediator, and dose-dependently increases the synthesis and secretion of collagen type IV (Knittel et al. 1996). Therefore, the results of the present study suggest that the cells expressing HGF mRNA were not only Ito and Kupffer cells but also sinusoidal endothelial cells associated with type IV collagen surrounding the hepatocytes, and were involved in TGF-β production. HGF is highly effective in repairing the cirrhotic liver through suppression of TGF-β, which mediates fibrogenesis. Therefore, it is possible that sinusoidal endothelial cells also become able to express HGF, which may have a therapeutic impact.
In conclusion, our results suggest that HGF and TGF-β mRNA, believed to be important factors in liver injury repair, are expressed in mesenchymal cells (Kupffer cells and Ito cells) in the normal liver. In the cirrhotic liver, our results show that sinusoidal endothelial cells also express HGF and TGF-β mRNA. Mesenchymal cells, including sinusoidal endothelial cells, produce TGF-β as a signal of liver failure and are responsible for collagen production during fibrogenesis. We also believe that these same cells, including sinusoidal endothelial cells, secrete HGF, which promotes fibrosis repair.
Footnotes
Acknowledgements
We wish to thank Mr Eiji Obana (Teijin SRL Lab. Inc., Japan) and Ms Ayumi Namiki for excellent tissue preparations.
