Abstract
We examined the distribution of sialoglycoconjugates in the cerebellum of 9-week-old and 30-month-old rats using light microscopy and electron microscopy in combination with two lectins, Maackia amurensis lectin (MAL) for Siaα2-3Gal and Sambucus sieboldiana agglutinin (SSA) for Siaα2-6Gal. Each lectin showed characteristic staining patterns. In young adult rats, MAL stained a strongly granular layer, a weakly molecular layer, and the medullary lamina, while SSA more strongly stained the medullary lamina than the molecular and granular layers. After aging, different staining patterns were obtained. Intense SSA reactivity was observed in the granular layer and intense MAL reactivity was observed in the medullary lamina of the aged groups. The reactivity of Purkinje cells with MAL was downregulated in the aged rats. These results indicated that Siaα2-3Gal and Siaα2-6Gal were expressed in distinct regions of the rat cerebellum and that their expression patterns changed in the aged brain.
S
Cerebellar neurons are arranged in a highly regular manner that results from repetition of the same basic circuit module (Voogd 1995). Such neuronal networks have important roles in modulating the force and range of movement and in the learning of motor skills. The cerebellum is a simple structure consisting of many cerebellar folds, or folia. Each single folium contains three cell layers. The outermost molecular layer is composed primarily of the axons of granule cells, known as parallel fibers, that run parallel to the long axis of a folium. It also contains scattered stellate and basket cells, which function as interneurons, as well as the dendrites of the underlying Purkinje cells. Beneath the molecular layer is the Purkinje cell layer, which consists of a single layer of the large cell bodies of Purkinje cells arranged side by side. Purkinje cells have extensive dendritic trees that extend up into the molecular layer in a single plane perpendicular to the main axis of a folium. The axons of Purkinje cells extend down into the underlying medullary lamina. The granular layer contains a vast number of densely packed small neurons, mostly small granule cells. The granular layer contains small clear spaces called cerebellar glomeruli, in which cells in the granular layer form complex synaptic contacts with the bulbous expansions of mossy fibers. The innermost medullary lamina (white matter) contains many myelinated axons, including climbing fibers, Purkinje cell axons, and mossy fibers.
Because the biosynthesis of glycans is not controlled by the interaction of a template and depends on the concerted action of glycosyltransferases, the structures of glycans are much more variable than those of proteins and nucleic acids. Therefore, the structures of glycans can be easily altered by the physiological conditions of the cells (Lowe 2001). Accordingly, age-related alterations of the glycans are relevant to the understanding of physiological changes found in aged individuals. It is important to determine the molecular events that occur in glycoconjugates during aging. As a step in this direction, we recently found age-related differences in brain glycoproteins by lectin blotting analyses (Sato et al. 1998). Subsequently, in a histochemical analysis using two lectins, Maackia amurensis lectin (MAL) and Sambucus sieboldiana agglutinin (SSA), we showed that the expression levels of α2-3 and α2-6-sialoglycoproteins were downregulated in several regions of the aged rat hippocampus (Sato and Endo 1999; Sato et al. 2001).
In this study we examined the distribution of sialoglycoconjugates in the cerebellum of 9-week-old rats and 30-month-old rats using light microscopy and electron microscopy in combination with two lectins, MAL and SSA.
Materials and Methods
Animals
Five each of 9-week-old and 30-month-old female Fischer rats were used in this study. All experimental procedures using laboratory animals were approved by the Animal Care and Use Committee of Tokyo Metropolitan Institute of Gerontology. All efforts were made to minimize the number of animals used and their suffering. Because the same results were found in all five rats in each group, only the data obtained for one sample in each group is described in detail here.
Lectin Staining for Light Microscopy
Lectin staining was carried out as previously described (Sato et al. 2001). Under pentobarbital anesthesia, the brains were dissected out and immediately frozen in an OCT compound (Sakura; Tokyo, Japan) with dry ice. Rat brain coronal sections, perpendicular to the septotemporal axis at the approximate mid-point, were cut into 6-μm-thick sections on a cryostat and mounted on gelatin-coated slides. The sections were fixed by incubation in 4% formaldehyde in 10 mM Tris-HCl (pH 7.4), 0.14 M NaCl, 1 mM CaCl2, 1 mM MgCl2, and 1 mM MnCl2 (TBS) for 10 min. They were then immersed in 0.3% H2O2 in methanol for 30 min at room temperature (RT). Thereafter, the sections were washed three times for 10 min at RT with TBS, blocked with 3% bovine serum albumin (BSA)-TBS, and then incubated with biotinylated lectins (SSA, 33.3 μg/ml; MAL, 13.3 μg/ml) (Seikagaku; Tokyo, Japan) diluted in 3% BSA-TBS for 1 hr at RT. The sections were washed again three times with TBS for 10 min and incubated for 1 hr at RT with horseradish peroxidase (HRP)-conjugated streptavidin (20 μg/ml) (DAKO; Carpinteria, CA). After further washes three times with TBS for 10 min, the sections were incubated with 3,3′-diaminobenzidine (DAB) solution (0.5 mg/ml in TBS) containing 0.3% H2O2 for 10 min at RT and then washed with distilled water. Sialidase treatment was performed as follows. The sections were incubated with sialidase (4 U/ml) from Arthrobacter ureafaciens (highly purified grade; Nacalai Tesque, Kyoto, Japan) in 0.5 M sodium acetate buffer (pH 5.0) for 2 hr at RT. After washing three times with TBS, they were incubated with the biotinylated lectins and then with HRP-conjugated streptavidin, and finally with DAB as above.
Lectin Staining for Electron Microscopy
Under pentobarbital anesthesia, the rats were perfused transcardially with 10 mM PBS, pH 7.4, and then with 4% formaldehyde-PBS at RT over 20 min. The brains were immediately dissected out and placed in 4% formaldehyde-PBS at 4C for 2 hr. Lectin staining for electron microscopy was carried out according to Miyata et al. (1996) with a minor modification. Sections (40 μm thick) were cut with a microslicer DTK-1500 (D.S.K.; Kyoto, Japan). The sections were washed three times (10 min each) with PBS and treated with 5% BSA-PBS for 1 hr, and they were incubated with biotinylated lectins (SSA, 33.3 μg/ml; MAL, 13.3 μg/ml) in 0.1% BSA-PBS for 24 hr at 4C, washed three times (20 min each) with PBS, and then incubated with HRP-conjugated streptavidin (0.5 μg/ml) (Jackson Immunoresearch; West Grove, PA) in 0.1% BSA-PBS for 24 hr at 4C. As cytochemical controls, the sections were incubated with SSA or MAL in the presence of 0.2 M N-acetylneuraminic acid (Nacalai Tesque), or incubated with the HRP-streptavidin alone. Sections incubated with no lectin and no HRP-streptavidin were also prepared. After washing three times with PBS, the specimens were immersed in DAB solution (0.5 mg/ml in PBS) containing 0.005% H2O2 for 5 min at RT, and then washed with distilled water. Labeled specimens were post-fixed with 2% glutaraldehyde, and then 1% OsO4 in 0.1 M phosphate buffer, dehydrated through a graded series of ethanol and propylene oxide, and then embedded in Epon 812. The ultrathin sections were post-stained with lead citrate and observed under a transmission electron microscope (JEM-1010; JEOL, Tokyo, Japan).
Results
Light Microscopic Study
Staining with MAL and SSA, which specifically bind to Siaα2-3Gal and Siaα2-6Gal groups, respectively, showed that the Siaα2-3Gal and Siaα2-6Gal groups were expressed in distinct regions of the rat cerebellum. MAL staining was intense in the granular layer and weak in the molecular layer and the medullary lamina (Figure 1A), whereas SSA staining was intense in the medullary lamina and weak in the granular and molecular layers (Figure 1D).
We next examined the effect of age on the staining patterns and found two differences. MAL staining in the medullary lamina was slightly stronger in 30-month-old rats than in 9-week-old rats (Figures 1A and 1B), whereas SSA staining in the granular layer was much stronger in the aged rats than in the young adult rats (Figures 1D and 1E). The staining intensity in the remaining parts did not change drastically with aging. MAL and SSA staining disappeared after sialidase treatment (Figures 1C and 1F, respectively), indicating that the binding of each lectin to the sialic acid residue was highly specific. To examine in detail the staining of the rat cerebellum with SSA and MAL, we analyzed the whole sections with electron microscopy.
Electron Microscopic Study
In the aged group, SSA strongly stained the space between the cell bodies and neurites, where an extracellular matrix appeared to be present (Figure 2D, arrows). When the same section was examined at higher magnification, the extracellular matrix of the granular layer was confirmed to be stained with SSA (Figure 2E, asterisk). The extracellular matrix filled up the space between the membranes of cell processes. These membranes are indicated by arrowheads in Figure 2E. These results are consistent with those obtained at the light microscopic level. However, the glycoproteins whose expression patterns change with age remain to be identified. On the other hand, MAL stained the extracellular matrix of 9-week-old rats and 30-month-old rats (Figures 2A and 2B, respectively, arrows), but no difference in staining intensity between them was observed.
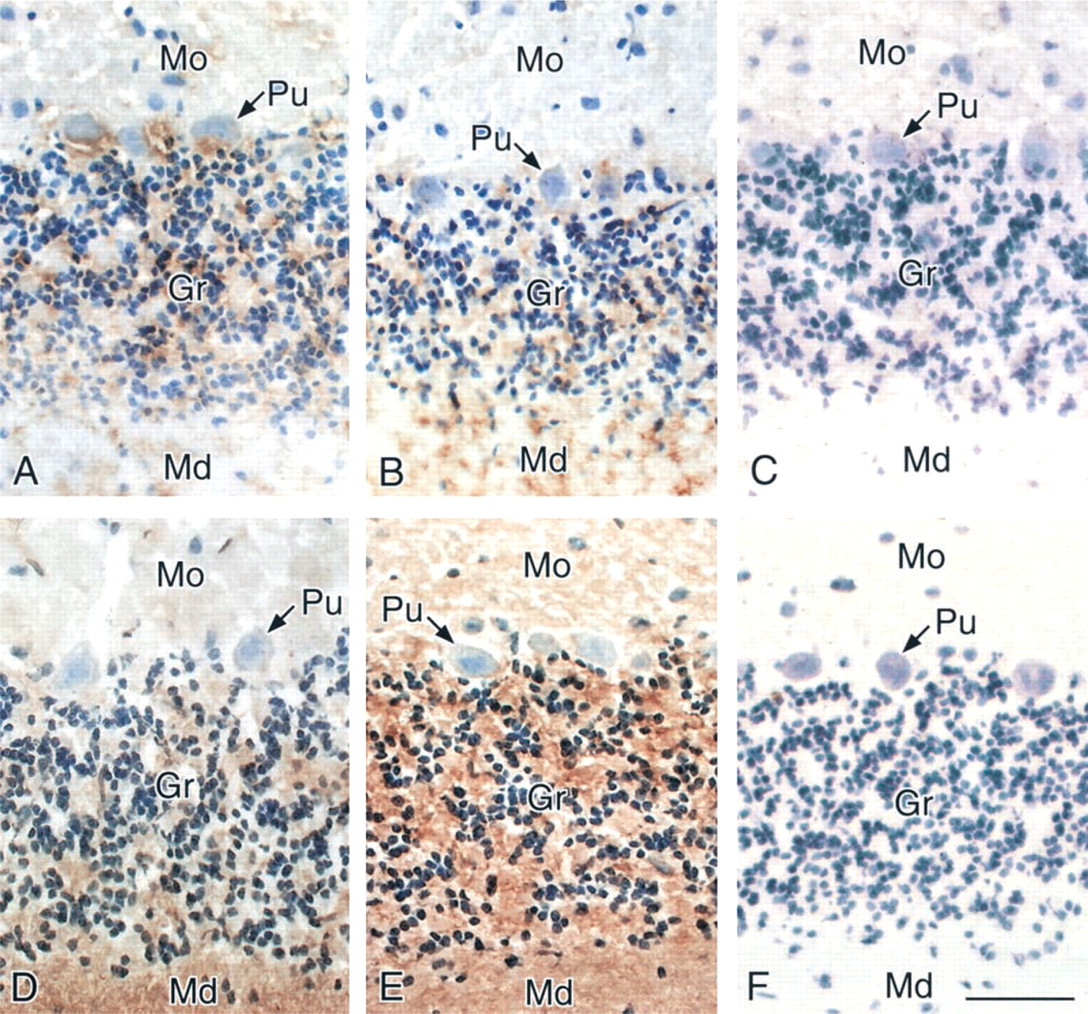
Light micrographs of the cerebellum from a 9-week-old rat (
The cerebellar glomeruli are indicated by Gl in Figure 2. The plasma membranes of the cerebellar glomeruli and the granule cells in the granular layer from 9-week-old and 30-month-old rats were stained by MAL and SSA, although an age-associated change was not observed (Figures 2A–2D, arrowheads). Nonspecific binding of HRP-streptavidin conjugates and endogenous peroxidase activity were scarcely detected under our experimental conditions because there was no difference when the sections were subjected to the staining procedure without lectin (Figure 2F) or HRP-streptavidin conjugates (data not shown). Because the electron density of myelin was high, the intensity of staining of myelin (indicated by M in Figures 2B and 2F) was similar when the samples were subjected to the staining procedure with or without lectin.
The plasma membranes of the Purkinje cells were stained by both lectins at the light microscopic level, as shown in Figure 1. MAL appeared to react more weakly in aged rats than in young adult rats, although details were not clear at the light microscopic level. Therefore, we examined the staining at higher resolution with electron microscopy. The plasma membranes of the Purkinje cells from the 9-week-old (Figure 3C) and 30-month-old rats (Figure 3D) were stained by SSA (arrowheads). The Golgi apparatus of the Purkinje cells from both groups was weakly stained (Figures 3C and 3D). However, no difference in staining between the 9-week-old and 30-month-old rats was observed. On the other hand, MAL staining of Purkinje cell membranes of the 30-month-old rats was less intense than that of the 9-week-old rats (Figures 3A and 3B, arrowheads). Observations at the light microscopic level could not reveal whether the dendrites of Purkinje cells reacted with the lectins (Figure 1). At the electron microscopic level, the dendrites of Purkinje cells from both young and aged rats were stained by both SSA and MAL (data not shown).
MAL staining of the Golgi apparatus of the Purkinje cells of the 9-week-old rats was stronger than that of 30-month-old rats (Figures 3A and 3B). Binding of the lectins was inhibited by the addition of a hapten sugar, 0.2 M N-acetylneuraminic acid (data not shown). It is also noteworthy that nonspecific binding of HRP-streptavidin conjugates and endogenous peroxidase activity were scarcely detected (Figures 3E and 3F). These results indicated that the Siaα2-3Gal and Siaα2-6Gal groups were expressed on the plasma membranes of the Purkinje cells and suggested that the expression of the Siaα2-6Gal linkage did not change significantly during aging but that of the Siaα2-3Gal was downregulated.
It should be noted that conspicuous lipofuscin accumulation was observed only in the cytoplasm of the 30-month-old rats (Figures 3B and 3D–3F, indicated by L). Lipofuscin, which has high electron density, is known to accumulate in the cytoplasm of aged cells only (Amenta et al. 1988).
Discussion
The present study demonstrates that the expression patterns of sialoglycoconjugates containing the Siaα2-3Gal and Siaα2-6Gal groups are region-specific in the rat cerebellum and change in the aged brain (Table 1).
The granular layer contains a vast number of densely packed small neurons, mostly small granule cells. The granular layer also contains small clear spaces called cerebellar glomeruli, in which the bulbous terminal of a mossy fiber makes synaptic contact with Golgi and granule cell axons (Voogd 1995). In this study we observed that SSA binds to one or more extracellular matrix components and that its reactivity is upregulated in the aged brain (Figure 2). Which molecule carries the Siaα2-6 group at the extracellular matrix in the granular layer remains to be solved. The increase of the Siaα2-6 group in the granular layer in the aged brain may be due to an increase of the activity of α2-6-sialyltransferases, which are involved in the formation of the Siaα2-6 group (Tsuji 1996). Another possibility is that the increase in sialyl groups is due to a depression in the activity of sialidases. Three mammalian sialidases have been described so far: membrane-associated, intralysosomal, and cytosolic sialidases (Miyagi and Tsuiki 1984,1985; Miyagi et al. 1990; Hata et al. 1998).
It should be noted that the expression levels of α2-3 and α2-6-sialoglycoproteins are downregulated in the aged rat hippocampus (Sato and Endo 1999; Sato et al. 2001). On the other hand, the expressions of the Siaα2-6 group at the extracellular matrix in the granular layer and the Siaα2-3 group in the medullary lamina are upregulated. These results suggest that the expression of sialylglycoconjugates is regulated in a region-specific manner.
The Purkinje cells are characterized by their rich and extensive dendritic tree in one plane. This structure is designed to accommodate an enormous synaptic input. The Purkinje cells are responsible for the only output signal from the cerebellar cortex, and the activity of the Purkinje cells is highly regulated by several excitatory and inhibitory afferent inputs. In this study we found that the staining intensity of the Purkinje cells by MAL was downregulated in the aged rats. Because such a decrease in the sialic acid residue reduces cell-cell repulsion and enhances cell-cell contact, the transmission efficiency of synaptic input and output signals may be affected. Interestingly, the expression of the Siaα2-3 group decreased and that of the Siaα2-6 group did not change during aging. One possible explanation for this is that there is an increment in the activity of a novel sialidase that acts on the Siaα2-3 group but not on the Siaα2-6 group. Although we cannot completely rule this explanation out, it seems unlikely because such a sialidase has not been identified in mammals. Another possibility is that the decreased expression of Siaα2-3 group is due to a depression in the activity of α2-3-sialyltransferase. Recently, the mRNAs of seven sialyltransferases in the mouse hippocampus were found to be differentially expressed. Furthermore, some sialyltransferase mRNAs were observed to be upregulated and others were found to be downregulated (Okabe et al. 2001). These results suggest that expressions of sialyltransferases are regulated by physiological activity. However, it is unclear whether sialoglycoconjugates are synthesized actually in proportion to the expression levels of each sialyltransferase mRNA, because glycan biosynthesis is regulated in a complicated manner (i.e., it depends on such things as glycosyltransferase activity, the availability of an acceptor glycan, and the availability of a nucleotide sugar donor).
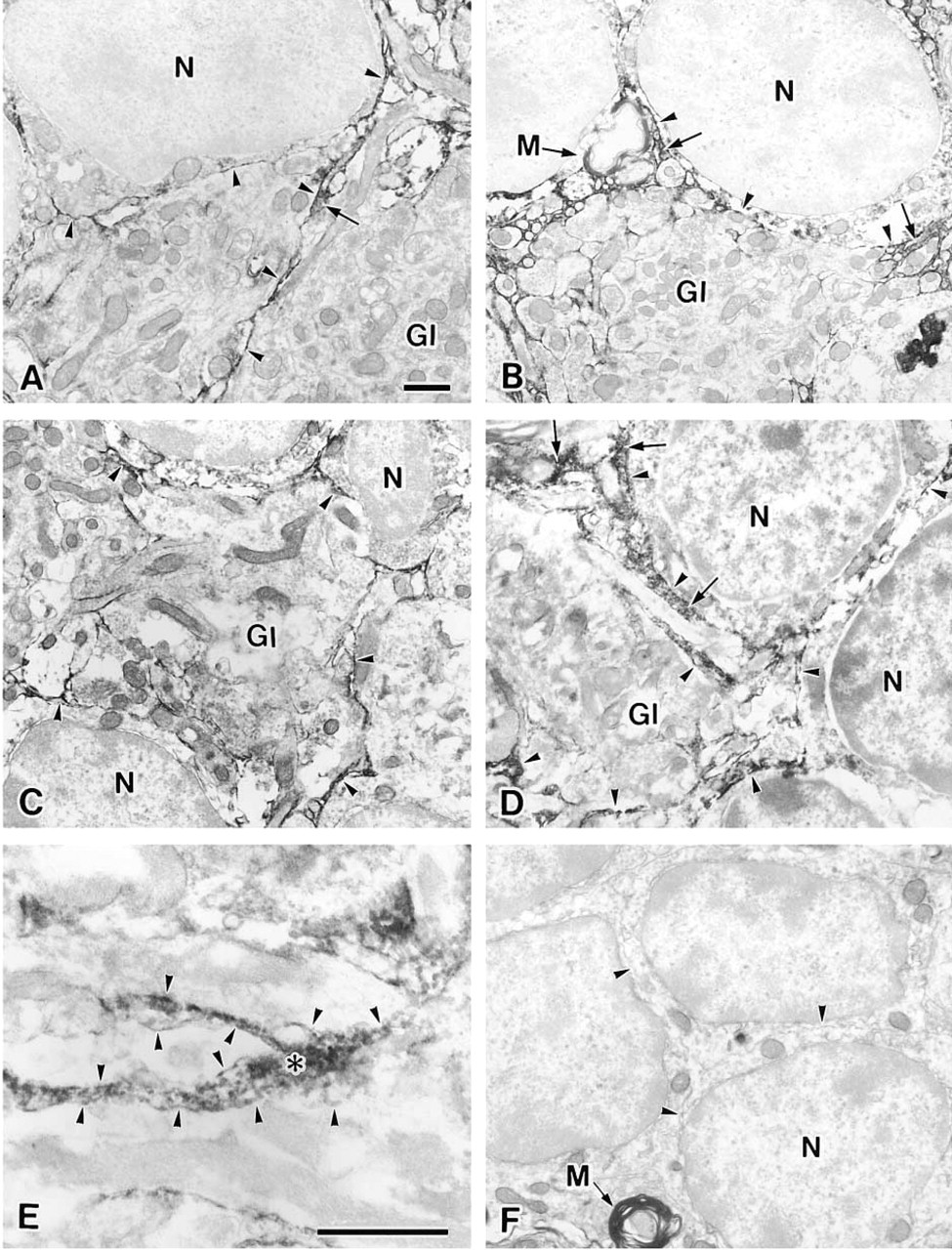
Electron micrographs of the granular cell layer from a 9-week-old rat (
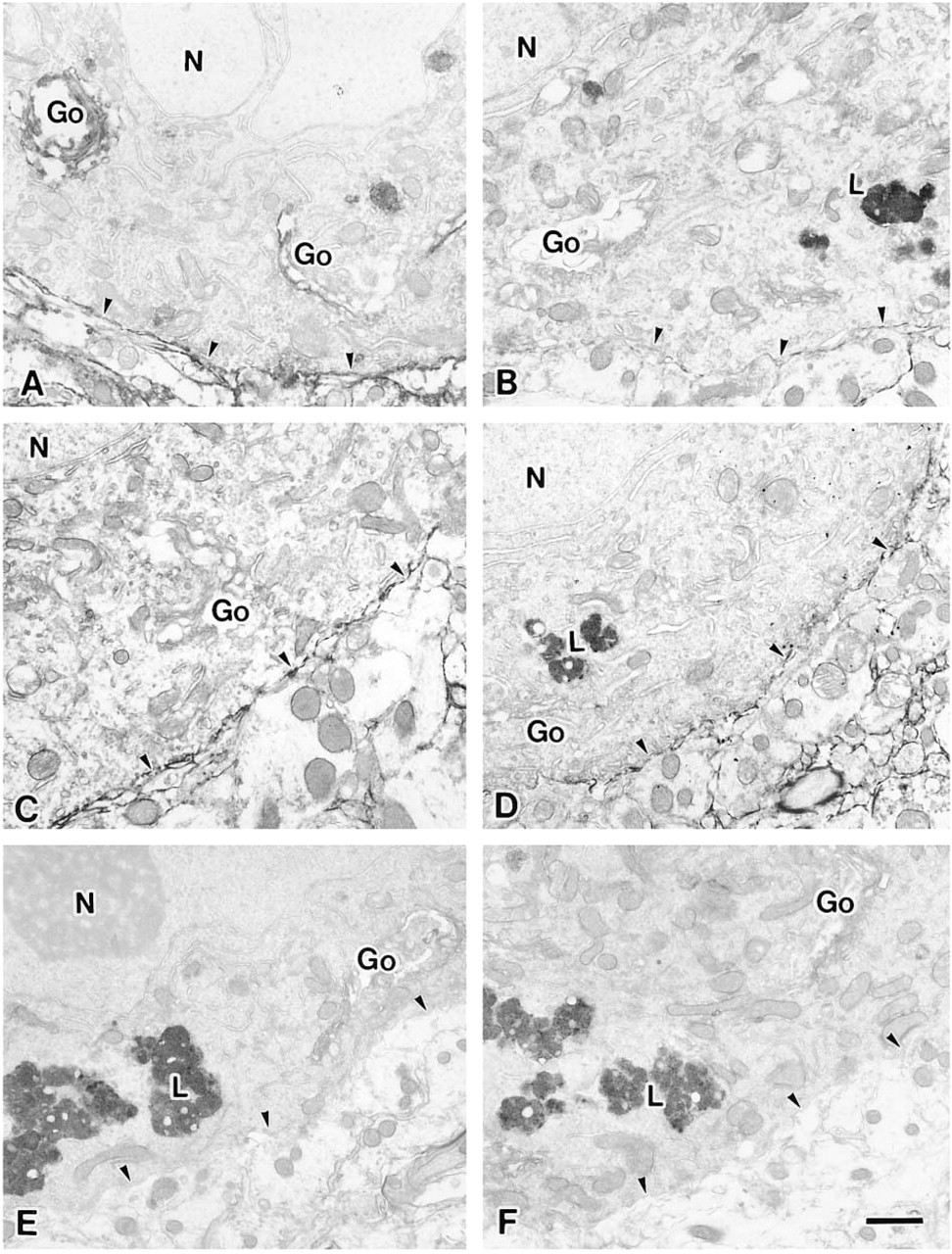
Electron micrographs of Purkinje cells from a 9-week-old rat (
Summary of lectin binding patterns in rat cerebellum a
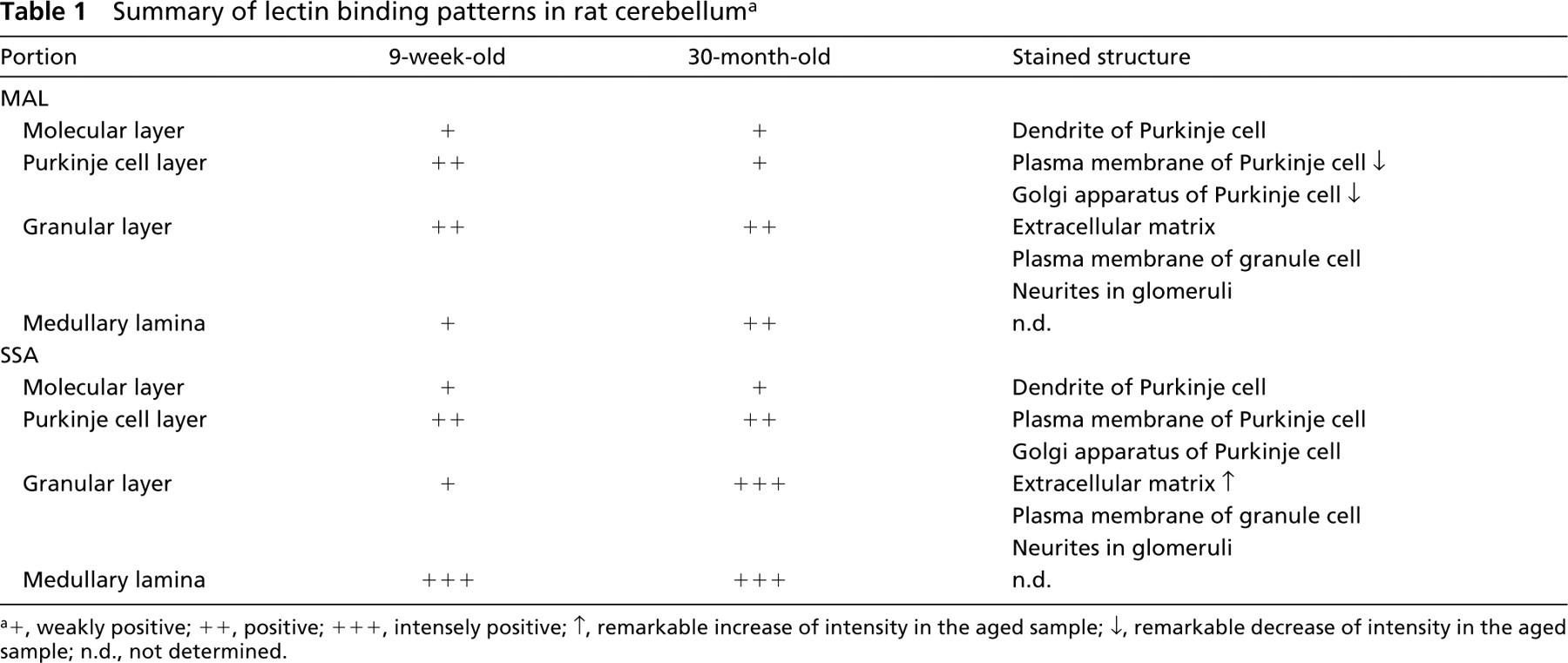
+, weakly positive; ++, positive; +++, intensely positive; ↑, remarkable increase of intensity in the aged sample; ↓, remarkable decrease of intensity in the aged sample; n.d., not determined.
Our finding that the expression of the Siaα2-3Gal group and Siaα2-6Gal group changed during aging is biologically significant for three reasons. First, because sialic acids are ubiquitous components of cell surface glycoconjugates, a change of sialic acid expression during aging may modulate molecular and cellular interactions by changing the electrostatic potential of cells. Second, a change of expression of sialic acid during aging may disturb cell-cell recognition via various sialic acid-binding molecules. One type of such molecules are the siglec proteins, which are endogenous lectins that recognize specific sialic acid linkages (Crocker et al. 1994, Powell and Varki 1995). Of the 10 designated siglecs described in the literature (Munday et al. 2001; Li et al. 2001), only siglec-4a (myelin-associated glycoprotein, or MAG) is present in the brain. Siglec-4a, which recognizes only the Siaα2-3Galβ1-3GalNAc linkage (Powell and Varki 1995), helps to maintain myelin function (Kelm et al. 1996) and stabilizes developing axo-glial interactions (Marcus et al. 2002). Another sialic acid-binding lectin is L1, which binds to α2-3-linked sialic acid residues on CD24, which, in turn, induces neurite outgrowth (Kleene et al. 2001). Third, a change in the sialic acid content of some glycoproteins during aging may reduce their normal function by changing their physical properties, their vulnerability to enzymatic digestion, and the ability of lectins or antibodies to recognize their underlying structures (Rossenberg 1995).
We previously compared young and aged rat cerebellums using the lectins SSA and MAL after SDS-PAGE (Sato et al. 1998). Staining with MAL revealed a distinct 115-kD band that decreased in the aged rats. Although the identity of the 115-kD protein is unknown, it may be the same protein that reacts with MAL in Purkinje cells, and decreases in this protein with aging may explain the decreased reactivity of these cells with MAL in aged rats. On the other hand, the staining intensity of several glycoprotein bands with SSA increased in brain samples from the aged rats, indicating an increase of the Siaα2-6 linkage after aging. We are starting to purify and identify the glycoprotein bands detected by SSA. Future studies are required to determine the full significance of the change in sialic acid during aging.
The present results clearly reveal a change of sialic acids during aging. Therefore, the change of sialoglycoconjugates during aging probably affects intercellular adhesion via a sialic acid-binding molecule, such as one of the siglec proteins, and the disturbance of adhesion then affects brain activity. Such a change may induce a deterioration of navigational brain functions, such as those involved in reaching an object, exploring a new environment, and acquiring knowledge of one's spatial orientation. These functions are believed to require undisturbed cerebellar activity (Petrosini et al. 1996).
Footnotes
Acknowledgments
Supported in part by a grant-in-aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, the Mizutani Foundation, and the Naito Foundation.
We thank Mr M. Fukuda and Ms T. Shibata (Laboratory for Electron Microscopy and Department of Anatomy, Kyorin University School of Medicine) for their technical assistance.
