Abstract
Large-scale mouse mutagenesis experiments now under way require appropriate screening methods. An important class of potential mutants comprises those with defects in the development of normal cerebellar patterning. Cerebellar defects are likely to be identified often because they typically result in ataxia. Immunohistochemistry (IHC) is commonly used to reveal cerebellar organization. In particular, the antigen zebrin II (=aldolase C), expressed by stripes of Purkinje cells, has been valuable in revealing cerebellar pattern abnormalities. The development of whole-mount procedures in Drosophila, chick, and Xenopus embryos allows complex patterns to be studied in situ while preserving the integrity of the structure. By combining procedures originally designed for embryonic and early postnatal tissue analyses, we have developed a whole-mount IHC protocol using anti-zebrin II, which reveals the complex topography of Purkinje cells in the adult mouse cerebellum. Furthermore, the procedure is effective with a number of other antigens and works well on both perfusion-fixed and immersion-fixed tissue. By use of this approach, normal adult murine cerebellar topography and patterning defects caused by mutation can be studied without the need for three-dimensional reconstruction.
THE MAMMALIAN CEREBELLUM is an ideal model in which to explore pattern formation in the CNS. Although cerebellar histology appears uniform, functional, anatomic, and molecular studies have all revealed an elaborate topography. The cerebellum is organized around an array of transverse zones (reviewed in Ozol et al. 1999; Armstrong and Hawkes 2000; Eisenman 2000) and parasagittal stripes (Hawkes and Leclerc 1987; Armstrong et al. 2000) of Purkinje cells. The most thoroughly studied molecular marker of adult cerebellar topography is zebrin II (Hawkes et al. 1985, 1992; Hawkes and Leclerc 1987; Brochu et al. 1990; Hawkes and Gravel 1991; Ahn et al. 1994; Hawkes and Herrup 1996; Hawkes 1997; Hawkes and Eisenman 1997; Walther et al. 1998). Isolation of zebrin II cDNA clones from cerebellar expression libraries has revealed a 98% identity to the metabolic isoenzyme aldolase C (Ahn et al. 1994). Zebrin II is a 36-kD polypeptide expressed strongly by a subset of Purkinje cells arranged in parasagittal stripes that are reproducible in detail between individuals and conserved across species [e.g., rat (Leclerc et al. 1990), opossum (Doré et al. 1990), and mouse (Eisenman and Hawkes 1993)]. Purkinje cells that are immunoreactive for zebrin II express the antigen in their dendrites, somata, and axons but not in the nucleus (Brochu et al. 1990). A detailed map of zebrin II expression in the mouse has been published (Eisenman and Hawkes 1993). Almost every feature of cerebellar structure and function is influenced by the positional information encoded in the Purkinje cells. For example, both climbing and mossy fibers terminate in discrete parasagittal stripes that align with the Purkinje cell stripes (reviewed in Hawkes 1997) and, as a result, the cerebellar functional map also consists of stripes and patches (e.g., Chockkan and Hawkes 1994; Hallem et al. 1999).
Pattern formation is crucial to normal cerebellar function. If Purkinje cell stripe formation is disrupted, the consequences are abnormal topography and severe motor control problems [e.g., disabled (mdab1-1) (Howell et al. 1997; Gallagher et al. 1998)]. Neurological mutants are an important tool in which to dissect the interplay between genes, the nervous system, and behavior. Interestingly, one of the most common phenotypes described among spontaneous mouse mutants is motor ataxia. This is not surprising because animal handlers can readily detect the abnormal motor behavior of a mouse while changing cages or during casual observation of the animal's behavior. As a result, a large number of mutants are known with ataxic phenotypes that correlate with defects in cerebellar lobulation, histology, and molecular composition (e.g., Sidman et al. 1965; Goldowitz et al. 1997; Goldowitz and Hamre 1998). These strains have served as important tools to examine genetic determinants of cerebellar development and the effects of these cerebellar sensitive gene mutations on events downstream of the primary genetic insult.
With the advent of large-scale murine mutagenesis experiments, there is a need to rapidly screen for patterning defects in the cerebellum. However, although zebrin II expression is a sensitive way to detect abnormal Purkinje cell patterns, the three-dimensional (3D) reconstruction of cerebellar topography is far too slow for routine screening purposes. To address this problem, we have developed a whole-mount IHC staining procedure for the adult mouse cerebellum. The method is a combination of a staining technique developed for the embryonic and neonatal opossum brain (Luqué et al. 1998a,b) and procedures to stain mouse, chick, and frog embryos in whole mount (Dent et al. 1989; Davis et al. 1991; Davis 1993). The protocol is reliable and robust and can also be applied to mice without overt behavioral symptoms. Although designed initially to reveal the zone and stripe distribution of zebrin II in Purkinje cells, it has readily adapted to a range of IHC markers and therefore should have wide utility.
Materials and Methods
Mice
All animal procedures conformed to institutional regulations and the Guide to the Care and Use of Experimental Animals from the Canadian Council for Animal Care. CD1 mice were purchased from Charles River Laboratories (Montreal, PQ, Canada). Cerebellum-deficient folia mice (cdf: Cook et al. 1997) and mice with a spontaneous mutation in the NPC1 gene [Balb/c NPCnih, a model of human Niemann-Pick type C disease (Pentchev et al. 1986)] came from the Jackson Laboratory Mouse Mutant Resource (Bar Harbor, ME), and are maintained in our animal colony. Lurcher (Lc/+) cerebella were a gift from Dr. M. Vogel (University of Maryland, Baltimore, MD). All mice were older than postnatal Day 20 (P20) and weighed 25–35 g. Cerebella were obtained from a variety of postnatal ages. No differences in the results were observed for different weights or ages.
Antibodies
Anti-zebrin II, a mouse monoclonal antibody (MAb) produced by immunization with a crude cerebellar homogenate from the weakly electric fish Apteronotus (Brochu et al. 1990), was diluted 1:150. It recognizes an antigen expressed selectively by a subset of Purkinje cells that form a reproducible array of transverse zones and parasagittal stripes.
Mouse MAb10B5 was raised against neurospheres from the embryonic murine striatum (Williams et al. 1993) and was diluted 1:100. The 10B5 antigen is expressed in the somata of interneurons, and in the adult cerebellum primarily stains the granular layer.
Both anti-zebrin II and MAb10B5 were produced in our laboratory and used directly from spent hybridoma culture medium.
Mouse monoclonal anti-calbindin (anti-CaBP, IgG1 isotype: Swant, Bellinzona, Switzerland) was used diluted 1:500. In the adult mouse cerebellum, CaBP is expressed exclusively by Purkinje cells. All Purkinje cells are immunoreactive.
Horseradish peroxidase-conjugated rabbit anti-mouse immunoglobulins (Jackson ImmunoResearch Laboratory; West Grove PA) was diluted 1:200.
Fixation and Sectioning
For perfusion-fixation, mice were injected IP with 100 mg/kg somnotol. When the toe-pinch reflex was undetectable, the mice were transcardially perfused with ice-cold 0.9% saline until all saline flushing out of a laceration made in the right atrium was void of blood. Ice-cold 4% paraformaldehyde in 0.1 M PBS, pH 7.4 (Sigma; St Louis, MO) was then perfused for approximately 6 min to allow complete fixation of the cerebellum. The cerebellum was removed from the calvaria and immediately placed in ice-cold fixative without removal of the meninges. The tissue was allowed a further 24–48 hr of postfixation at 4C. No difference in staining due to fixation time was seen. For immersion-fixation, mice were anesthetized as above and the brains removed from the calvaria. The cerebellum was then separated from the rest of the brain and immersion-fixed, ice-cold, for 24–48 hr in two or three changes of 4% paraformaldehyde in PBS.
Whole-mount Immunohistochemistry
Throughout the protocol, gentle rocking or nutation is necessary for proper reagent penetration. Ensure that the tissue is completely covered in solution at all times to prevent drying out. We have used 2.0-ml cryotube vials (Nalge Nunc International; Naperville, IL) filled to 1.5 ml. Each cerebellum was processed separately.
Steps
After allowing the cerebellum to bathe in fixative for 24–48 hr, it is postfixed in Dent's fixative [4 parts absolute methanol (MeOH), 1 part dimethysulfoxide (DMSO) (Dent et al. 1989)] overnight at 4C. Initial fixation in paraformaldehyde was used to ensure preservation of antigenicity and cell morphology, and postfixation with methanol to initiate dehydration. The addition of DMSO increases methanol penetration.
Incubate the tissue in Dent's bleach [4 parts MeOH:1 part DMSO:1 part 30% hydrogen peroxide (H2O2)] until the tissue is completely white (~8 hr) to block endogenous peroxidase activity.
Dehydrate the cerebellum twice for 30 min in 100% MeOH. This is important because large amounts of water trapped in the tissue may form ice crystals on subsequent freezing.
Subject the tissue to four or five cycles of freezing in 100% MeOH to −80C in a freezer and thawing to room temperature (RT) on the bench. The frozen tissue is brittle and care must be taken to avoid damage, especially while it thaws. Five cycles of freezing ensure that antigens in the deeper parts of the cerebellum are rendered accessible. However, we have successfully revealed zebrin II expression in Purkinje cell dendrites with only two cycles of freezing.
Incubate the tissue overnight in 100% MeOH at −80C to ensure good reagent penetration. The tissue can be stored for prolonged periods at this stage.
Rehydrate the tissue for 90 min each in 50% MeOH, 15% MeOH, and PBS at RT.
Enzymatically digest the tissue in 10 μg/ml proteinase K (>600 U/ml: Boehringer Mannheim, Laval, PQ, Canada) in PBS for 5 min at RT to permit free penetration of reagents.
Rinse the cerebellum three times for 10 min in PBS at RT.
Block nonspecific antibody binding by incubating the cerebellum in 2% non-fat skim milk powder, 0.1% Triton X-100 in PBS (PBSMT; Davis 1993) for 6–8 hr at RT, changing the PBSMT two or three times during this period. Although incubation times and the number of changes can be decreased, elimination of background staining is difficult. The procedure presented here has worked well for a variety of antibodies (monoclonal and polyclonal) and may be regarded as the starting point for a novel antibody.
Incubate the tissue for 48 hr in primary antibody diluted in PBSMT containing 5% DMSO at 4C (to improve penetration).
Rinse the tissue in PBSMT two or three times for a period of 2–3 hr each at 4C.
Incubate the tissue overnight at 4C in secondary antibody diluted in PBSMT + 5% DMSO.
Once again rinse the tissue in PBSMT three or four times for 2–3 hr each at 4C, followed by a final overnight incubation at 4C.
Rinse the cerebellum in 0.2% bovine serum albumin, 0.1% Triton X-100 in PBS (PBT; Davis 1993) for 2 hr at RT. Thorough rinsing (Steps 11,13,14) is crucial to ensure that unbound antibodies do not remain trapped within the tissue and give an unacceptably high background staining.
Peroxidase binding sites are revealed by incubating in a freshly-prepared 3,3′-diaminobenzidine (DAB; Sigma, St Louis, MO) solution consisting of 1 × 10 mg DAB tablet in 20 ml PBT and 10 μl 30% H2O2.
The peroxidase reaction can be stopped once the desired color intensity is reached by washing the cerebellum in PBT + 0.04% sodium azide. (Even though sodium azide is an effective inhibitor of bacterial and fungal growth, it should be avoided until this step because it is also an inhibitor of horseradish peroxidase.)
If clearing is desired, the tissue is dehydrated completely in sequential rinses of 30%, 50%, 80%, and 100% MeOH for 30–60 min each at RT then transferred to 1 part benzyl alcohol in 2 parts benzyl benzoate for clearing (BABB). The clearing solution should be stored in glass or polypropylene containers because it readily dissolves polystyrene.
Section Immunocytochemistry
Cerebella to be sectioned were cryoprotected through graded sucrose solutions: 10% (2 hr), 20% (2 hr or until the cerebellum sank), and 30% (overnight) at 4C. The tissue was then frozen in OCT embedding compound (VWR; Mississauga, ONT, Canada). Serial sections were cut at 50 μm in the transverse plane and mounted on gelatin-coated slides. Sections for IHC staining were processed by using slide-mounted (IHC) and mounted in DPX (BDH Chemicals; Toronto, ONT, Canada). Briefly, slide-mounted sections were rinsed two times for 5 min each in PBS, incubated in 1% H2O2 for 10 min, and then rinsed three times for 5 min each in PBS. The sections were then incubated in 10% normal goat serum (NGS) in PBS and 0.5% Triton X-100 for 2 hr at RT. The tissue was then left in primary antibodies diluted in 10% NGS, PBS, and 0.5% Triton X-100 overnight at 4C. After rinsing three times in PBS for 5 min each, the tissue was incubated in secondary antibody diluted in NGS, PBS, and 0.5% Triton X-100 for 2 hr at RT. Antibody concentrations were the same as those used for whole-mount IHC. A freshly prepared DAB solution was used to visualize antibody binding, as for the whole mounts. Sections from cerebella previously immunoperoxidase-stained in whole mount were mounted directly.
Whole-mount photomicrographs were captured with a SPOT digital camera (Diagnostic Instruments; Sterling Heights, MI) mounted on a Zeiss Stemi SV6 microscope. Cerebella were photographed immersed in PBT, with incident illumination. Montages were assembled in Adobe Photoshop 4.0 (Tucson, AZ). The images were cropped and corrected for brightness and contrast but not otherwise manipulated.
Results
Zebrin II Expression in Normal Mouse Cerebellum
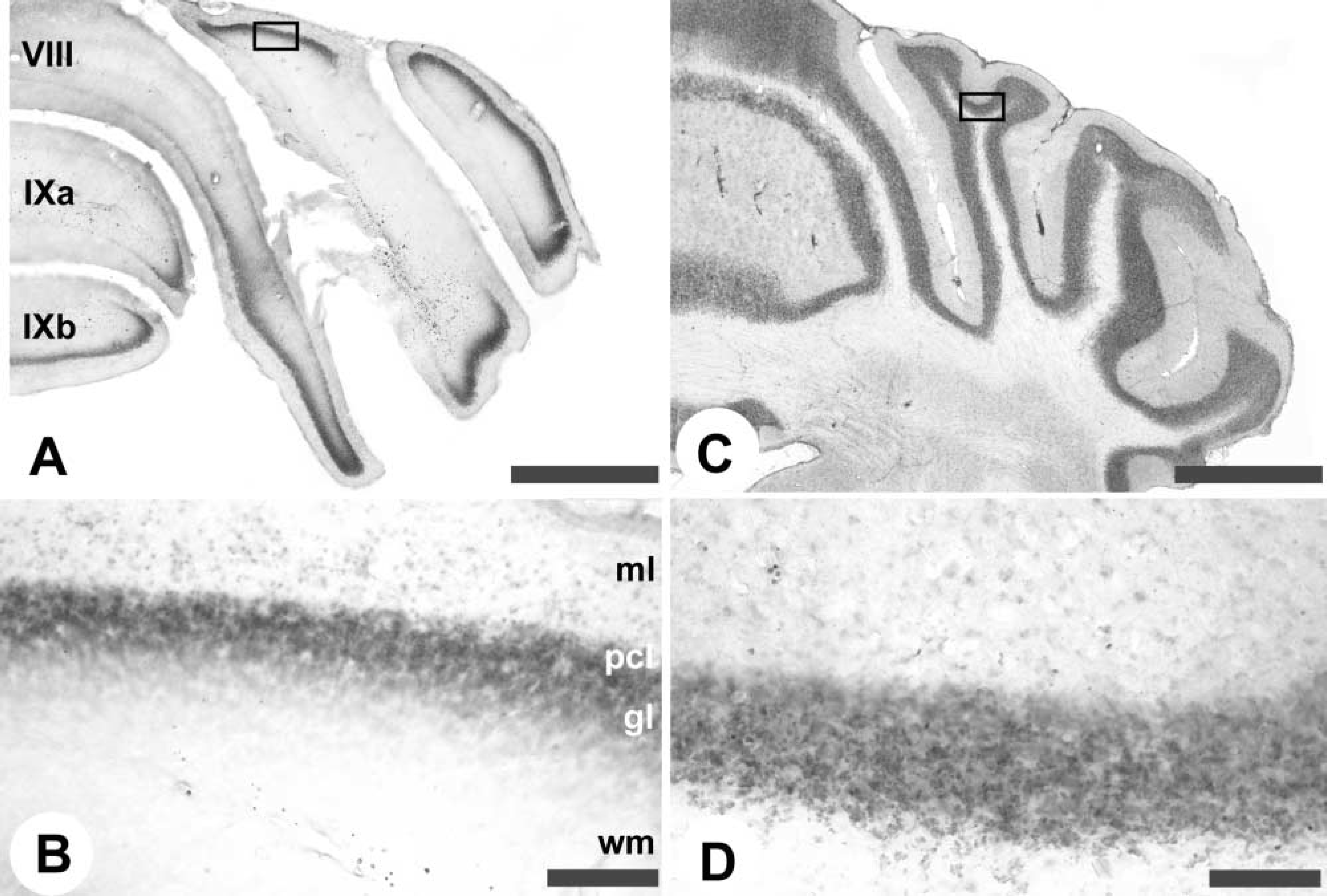
The vermis of the cerebellar cortex is constructed around four transverse zones: the anterior zone (AZ; ~lobules I–V), central zone (CZ; ~lobules VI–VII), posterior zone (PZ; ~lobules VII–VIII), and nodular zone (NZ; ~lobules IX–X), each of which is further subdivided into parasagittal stripes (e.g., Hawkes 1997; Ozol et al. 1999; Armstrong and Hawkes 2000; Armstrong et al. 2000). A similar arrangement pertains in the hemispheres. By using anti-zebrin II whole-mount IHC, the transverse zones and parasagittal stripes in the cerebellar cortex are clearly revealed (Figures 1 and 2). Purkinje cells expressing zebrin II show immunoreactivity in their dendrites, somata, and axons. The data shown come from both immersion- and perfusion-fixed animals. Similar results were obtained in each case. No staining was obtained when the primary antibody was omitted or replaced by myeloma-conditioned culture medium. The expression pattern revealed in whole mount can be compared to that derived previously from serially stained sections and 3D reconstruction (Figure 1) (Eisenman and Hawkes 1993; Ozol et al. 1999). Figure 2A shows lobule IV of the AZ in a whole mount-stained cerebellum and Figure 2B shows a similar view in a transverse section. Most Purkinje cells in the AZ are zebrin II-negative. The exceptions form a prominent, narrow P1+ at the midline and the P2+ stripes ~500 μm laterally to either side. More lateral still, a broader, weakly immunoreactive P3+ is seen. [Zebrin-immunoreactive stripes are numbered from P1+ medially to P7+ laterally: zebrin-negative (P-) stripes are numbered according to the immediately medial P+ stripe (Eisenman and Hawkes 1993).]
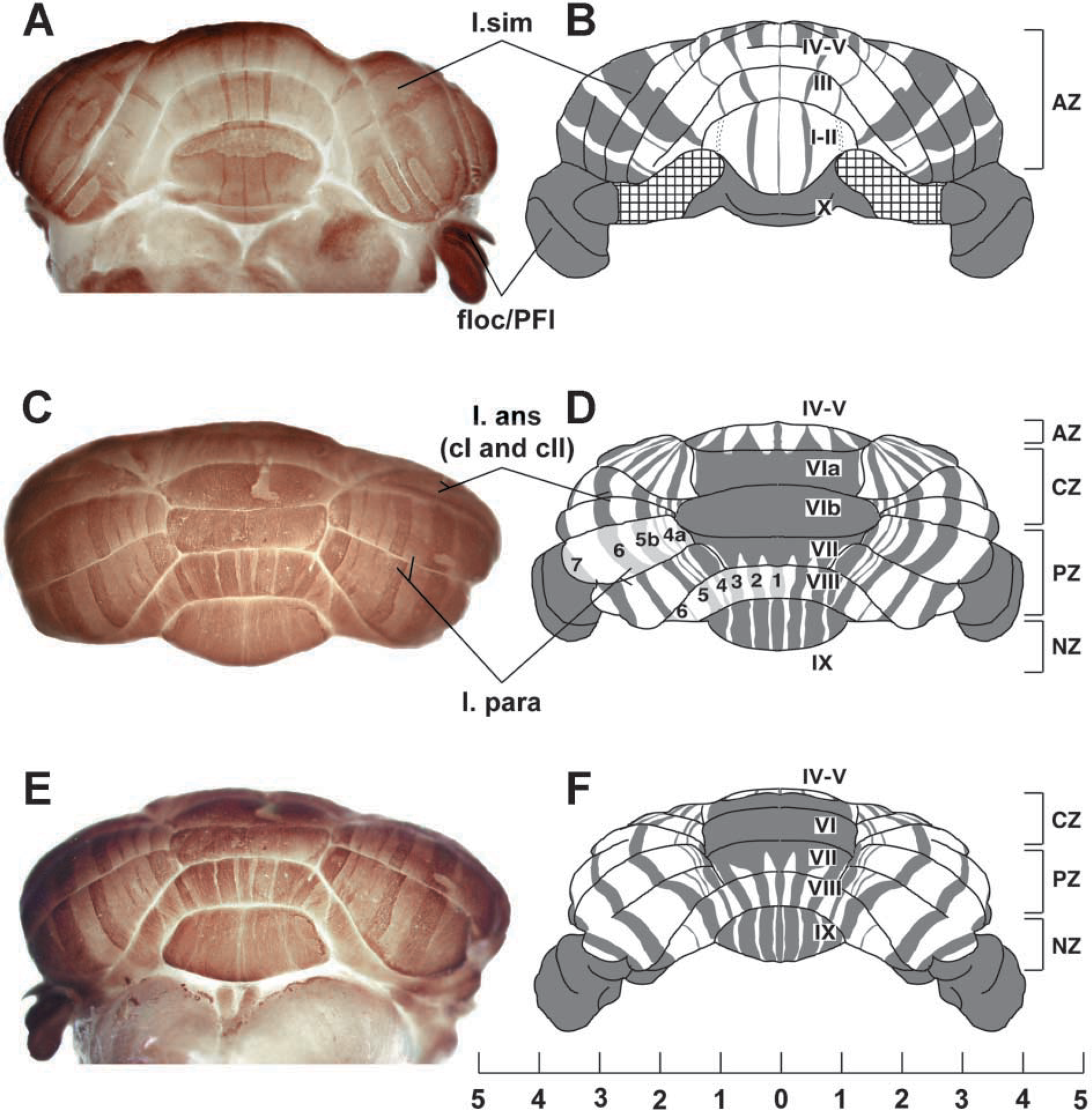
Whole-mount zebrin II peroxidase immunohistochemistry of the adult mouse cerebellum. (
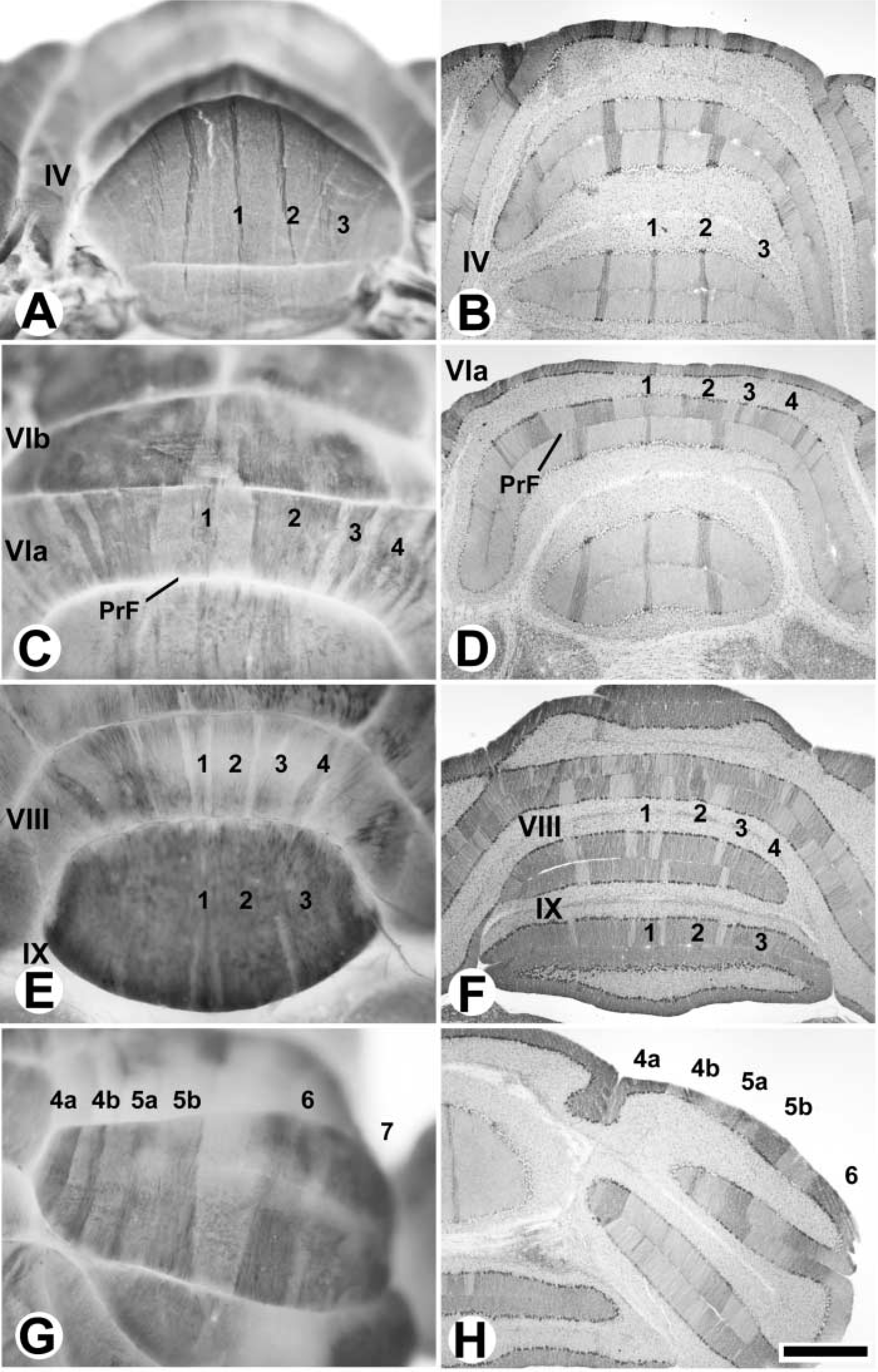
Compartmentation of the adult mouse cerebellum as revealed by whole-mount zebrin II peroxidase immunohistochemistry. Whole-mount views are shown at left; matched transverse sections from another individual, immunoperoxidase-stained on the section, are shown at right. All panels show zebrin II selectively expressed in subsets of Purkinje cells. Because of the strong dendritic immunoreactivity, staining is prominent in the molecular layer. The P+ stripes are labeled (as 1–7 for clarity). (
The AZ extends caudally to the dorsal aspect of the primary fissure, where it interdigitates with stripes derived from the CZ (Ozol et al. 1999). This region is shown in whole mount in Figure 2C and in transverse section in Figure 2D. In the CZ (e.g., lobule VIb in Figure 2C), all Purkinje cells are zebrin II-immunoreactive except for interdigitating spurs of zebrin II-negative Purkinje cells near the midline, which terminate in lobule VIb (Ozol et al. 1999). In the posterior lobe vermis, alternating stripes of zebrin II+/- Purkinje cells reappear in the PZ (lobule VIII; Figures 2E and 2F). In the PZ, the stripes of zebrin II-immunoreactive Purkinje cells (P1±P4+) are much wider than in the AZ. Finally, in lobule IX, the PZ ends and the NZ appears, as the zebrin II-negative Purkinje cells disappear. The pattern of alternating stripes seen in the AZ and PZ of the vermis is also present in the hemispheres. Figures 2G and 2H compare a whole-mount view and a transverse section through the ansiform lobule of the anterior lobe hemisphere: the P4±P7+ stripes are labeled.
Zebrin II Expression in the Cerebellum Deficient Folia Mutant
To demonstrate the utility of zebrin II whole-mount IHC on a mutant background, we have stained cerebella from the ataxic cerebellum deficient folia (cdf/cdf) mouse (Cook et al. 1997), in which an embryonic Purkinje cell dispersion defect, restricted primarily to the zebrin II-negative population, results in abnormal patterning of the anterior lobe vermis (inter alia: Beierbach et al. 2001). Figure 3 compares whole-mount views of the anterior lobe vermis from wild-type (+/+; Figures 3A and 3C) and homozygous cdf/cdf mutant (Figures 3B and 3D) cerebella. The +/+ AZ shows the normal narrow, well-defined P1+ and P2+ stripes. In contrast, the cdf/cdf cerebellum has an AZ with ragged, ill-defined stripes that are significantly wider than normal. Subcellular localization of zebrin II to dendrites, axons, and somata is maintained in cdf/cdf mutant cerebella (Beierbach et al. 2001).
Other Cerebellar Antigens
Calbindin (CaBP) expression in the cerebellum is restricted entirely to Purkinje cells [rat (De Camilli et al. 1984; Wassef et al. 1985) mouse (Ozol et al. 1999)]. CaBP is an especially useful cerebellar marker because a number of known mutations in mice lead to progressive Purkinje cell death [e.g., lurcher (Lc/+) (Caddy and Biscoe 1979) NPC (Higashi et al. 1993) tottering (tg) (Heckroth and Abbott 1994); Purkinje cell degeneration (pcd) (Wassef et al. 1987)]. To illustrate the general applicability of the staining technique, anti-CaBP whole-mount IHC has been used to reveal patterned Purkinje cell death in two ataxic strains of mutant mice, Lc/+ and NPCnih (Figure 4).
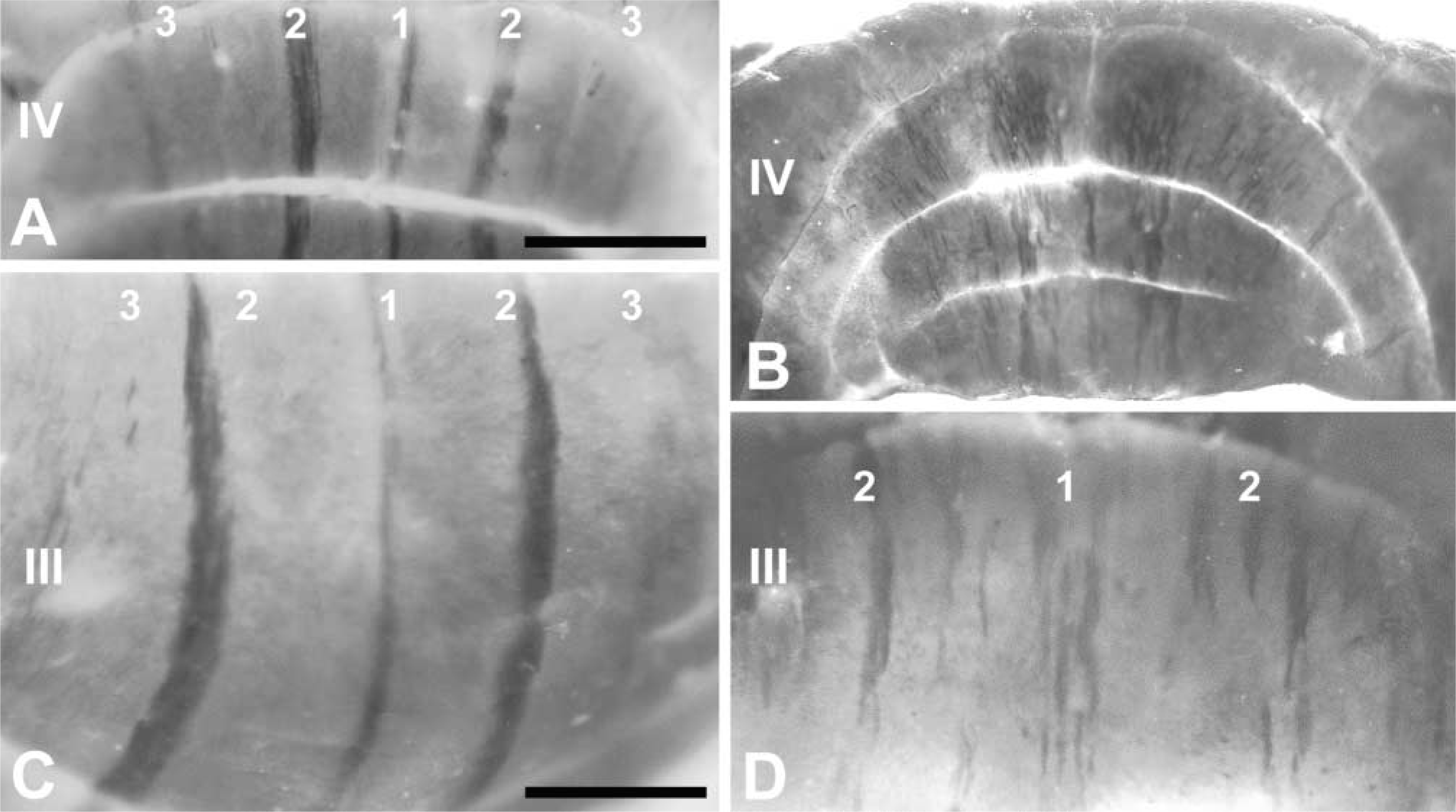
Zebrin II expression in the AZ of the normal mouse (
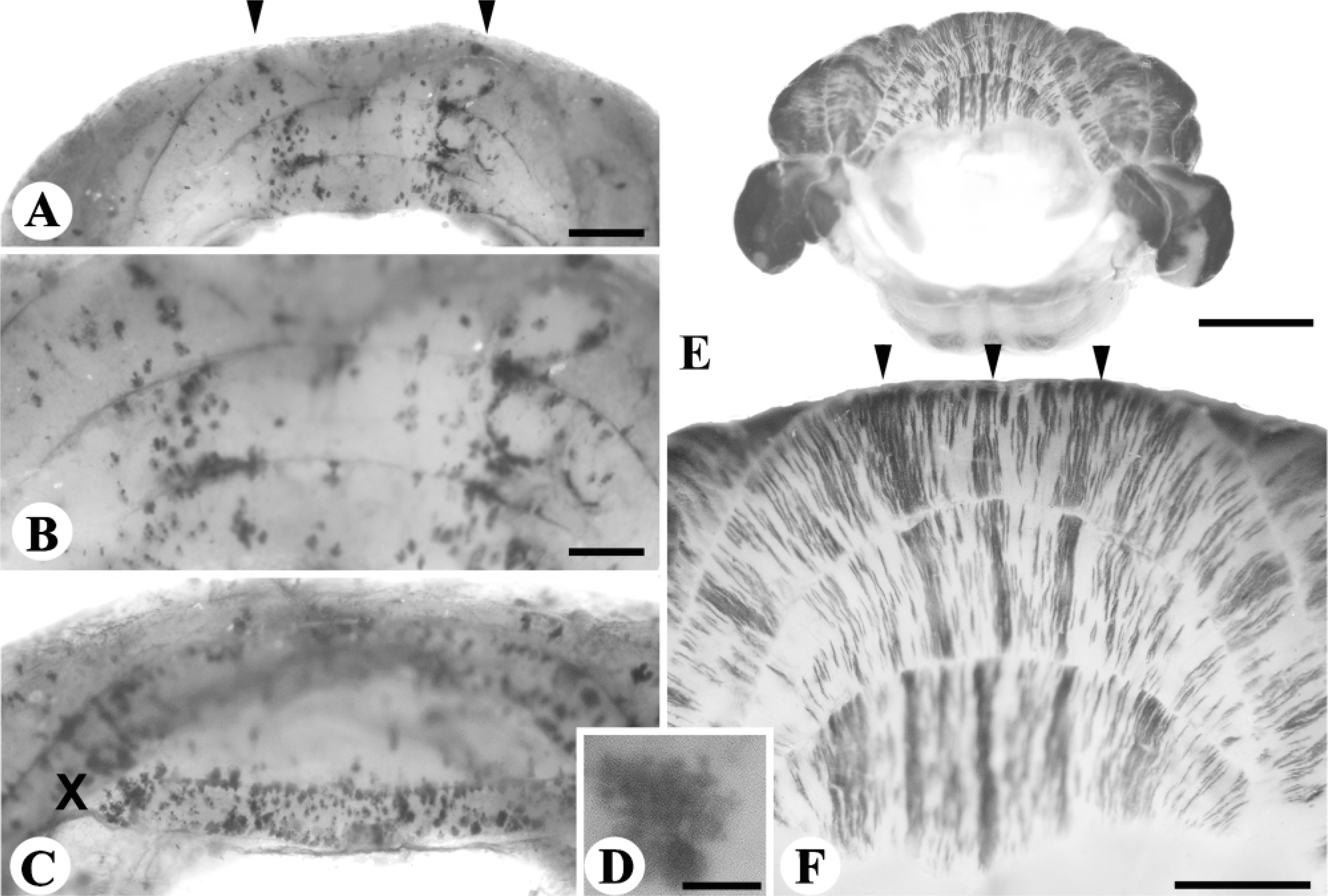
Patterned cell death in the cerebellum revealed by whole-mount IHC with the Purkinje cell-specific marker anti-CaBP. (
Figures 4A-4D show anti-CaBP immunoreactivity in the Lc/+ cerebellum at postnatal day (P)43, at which age the majority of Purkinje cells have degenerated (Caddy and Biscoe 1979). The remaining Purkinje cells are concentrated in two regions, a pair of parasagittal stripes in the AZ of the anterior lobe vermis (Figures 4A and 4B), and the NZ (Figure 4C). Because the surviving Purkinje cells are relatively sparse, it is possible to directly image individual somata and dendritic arbors in situ (Figure 4D). Patterned Purkinje cell death can also be demonstrated in whole mount in the NPCnih mouse (Figures 4E and 4F). In the homozygous mutant at P60, Purkinje cell death is well under way (Higashi et al. 1993) to revealing an array of parasagittal stripes of surviving Purkinje cells (e.g., in the AZ and anterior lobe hemispheres: Figures 4E and 4F).
Antibody Penetration in Whole Mount-immunostained Cerebella
In whole-mount staining it appears likely that the density and thickness of the adult cerebellar tissue poses a major barrier to antibody penetration. To determine the extent of antibody penetration, cerebella stained in whole mount were cryostat-sectioned and examined (Figure 5). Zebrin II and CaBP are Purkinje cell antigens and therefore might be detected even if antibody penetration into the tissue is limited. Indeed, the standard recipe described above favors the detection of antigens in the molecular layer of the cerebellar cortex. However, by increasing the incubation period in the primary antibody from 48 to 96 hr (Step 10) and in the secondary antibody from overnight to 2 days (Step 12), antigens can be detected in all layers of the cerebellum. As an example, we have used the anti-granule cell antibody MAb10B5 (Williams et al. 1993; Hawkes et al. 1999) to test the flexibility of the whole-mount protocol (Figure 5). The 10B5 antigen is expressed weakly by all cerebellar interneurons but predominantly by granule cells. In whole-mount views, DAB reaction product is seen faintly beneath the cerebellar surface. Sections through the whole mounts show that reaction product is deposited appropriately in the granular layer (Figures 5A and 5B). However, reagent penetration into the tissue is incomplete in two respects. First, only the granular layer near the exposed cerebellar surface is well labeled (Figure 5A) and staining is incomplete or absent in the depths of the sulci. Second, even in regions where granular layer staining is strong the density of the reaction product fades towards the white matter tracts (e.g., Figure 5B). Neither of these artifacts is seen when MAb10B5 is used to immunoperoxidase-stain cryostat sections (Figures 5C and 5D). For analysis of antigen expression patterns within the cerebellar fissures, even longer incubation times, or careful postfixation dissection of the obstructing tissue, are plausible options.
Penetration of immunoperoxidase staining into the tissue in whole mount compared to staining on the section as revealed by using the granule cell marker MAb10B5. (
Discussion
The whole-mount IHC protocol described here has several advantages for routine screening for cerebellar patterning defects. By use of anti-zebrin II, a reliable high-resolution topographical map can readily be demonstrated. Previous studies have shown zebrin II IHC to be a powerful tool. Its expression reflects the fundamental molecular architecture of the cerebellum and is sensitive to genetic manipulations of cerebellar development [e.g., Lc/+ (Tano et al. 1992); weaver (Eisenman et al. 1998); disabled (Gallagher et al. 1998); cdf (Beierbach et al. 2001; Figure 3) or patterns of Purkinje cell death [e.g., the acid sphingomyelinase null (ASMKO) mouse (Sarna et al. 2001)] but insensitive to secondary influences such as general health status, age, or gender. It is highly reproducible among individuals, is insensitive to the genetic background against which a mutation is seen, and is accurately quantifiable, both for the number of stripes and their widths.
Whole-mount IHC combines the advantages of zebrin II IHC with the ability to see the pattern without sectioning and 3-D reconstruction. Not only is it much more rapid but it is at least as reliable and requires far less effort. As a result, whole-mount IHC can easily be automated to generate a routine screen for cerebellar patterning mutants. We have also successfully used whole-mount fluorescence IHC (unpublished data), which suggests that double-label protocols could be developed straightforwardly. Because the protocol works well with immersion-fixed tissue (e.g., Figure 2A), it is also unnecessary to include a time-consuming transcardiac perfusion. In addition, zebrin II IHC is compatible with many fixatives and, if necessary, the cerebella can be subsequently sectioned for histological analysis.
Before sectioning the tissue, it is also possible to clear the stained cerebella to reveal patterning deeper within the tissue. The clearing agent that is chosen is usually determined by matching its refractive index as closely as possible with that of the tissue. For embryos, BABB appears to work best after sequential methanol dehydration (reviewed in Klymkowsky and Hanken 1991). Zebrin II Purkinje cell bands in BABB-cleared adult cerebella are easily identified (data not shown). This being the case, it is possible that the more classical methods, such as xylene or toluene clearing after ethanol dehydration, may produce better results. For all antibodies that we have tested, the staining patterns are clearly visible throughout all layers of the cerebellum without clearing and photograph with sufficient contrast.
This protocol was developed on the basis of IHC principles already widely used. The major departure from previously published whole-mount protocols (Dent et al. 1989; Davis 1993; Luqué et al. 1998a) was the need to accommodate the size, density, and thickness of adult tissue. Essentially, we have extrapolated the approaches for embryonic and early postnatal tissue treatment to the adult. In addition, however, extensive measures are taken to penetrate the tissue while at the same time maintaining the integrity and morphology of structures. Although the primary impetus to develop this staining technique was for zebrin II IHC, we have also used it successfully here to reveal the distributions of CaBP in Purkinje cells and 10B5 in granule cells. In addition, we have used the method with several other antigens (data not shown), including the small heat-shock protein HSP25, which is expressed in the adult cerebellum by narrow stripes of Purkinje cells confined to the CZ, the NZ, and the paraflocculus (Armstrong et al. 2000), and tyrosine hydroxylase, which is expressed ectopically by Purkinje cell subsets in several mutant strains [e.g., tottering and leaner (Austin et al. 1992; Abbott et al. 1996; Fletcher et al. 1996) dilute-lethal (Sawada et al. 1999) pogo (Jeong et al. 2001); NPCnih (Sarna et al. unpublished data). We have found that the whole-mount staining procedure adapts readily to a range of species (our unpublished data), thereby opening the door to comparative studies of cerebellar patterning. Finally, by using this whole-mount IHC approach, we are confident that antigen distributions can be revealed elsewhere in the nervous system, such as barrels in the primary somatosensory cortex and ocular dominance stripes and columns in the primary visual cortex. With luck, the laborious task of sectioning and 3D reconstruction can be minimized.
Footnotes
Acknowledgements
Supported by grants from the Canadian Institutes of Health Research and the Ara Parseghian Foundation for Medical Research (RH).
We thank Robert Stack and Matthew Larouche for husbandry and genotyping of the NPCnih mice, Estrella Gonzales for technical assistance, Dr Michael Vogel for the gift of Lc/+ cerebella, and Drs Len Eisenman (Philadelphia) and Cairine Logan (Calgary) for discussions and advice.
