Abstract
The histochemical distribution of sialoglycoconjugates in the CA1 region in the hippocampus formation of 9-week-old rats and 30-month-old rats was examined using electron microscopy in combination with two lectins, Maackia amurensis lectin, specific for Siaα2–3Gal, and Sambucus sieboldiana agglutinin, specific for Siaα2–6Gal. Each lectin stained the plasma membranes of pyramidal cells, indicating that the Siaα2–3Gal and Siaα2–6Gal groups were expressed on their plasma membranes. These lectins also bound to synapses in the stratum lacunosum moleculare. The staining intensity of the lectins in the synapses in these layers was downregulated in the 30-month-old rats. These results indicated that both the Siaα2–3Gal and Siaα2–6Gal groups are expressed on these synapses and that the expression of these sialyl linkages decreases in the aged brain.
G
Although many studies have demonstrated the importance of structural change of glycans during development, very little information is available on changes in glycans during aging. Because the biosynthesis of glycans is not controlled by the interaction of a template and depends on the concerted action of glycosyltransferases, the structures of glycans are much more variable than those of proteins and nucleic acids. Therefore, the structures of glycans can be easily altered by physiological conditions of the cells. Accordingly, age-related alterations of the glycans are relevant to the understanding of physiological changes found in aged individuals. It is important to determine the molecular events that occur in glycoconjugates during aging. As a step in this direction, we have recently found differences in glycoproteins between young adult and older rat brains by a combination of SDS-PAGE and lectin blotting analyses (Sato et al. 1998). Subsequently, in a histochemical analysis using two lectins, Maackia amurensis lectin (MAL) and Sambucus sieboldiana agglutinin (SSA), we showed that the expression levels of α2–3 and α2–6 sialoglycoproteins were downregulated in several regions of the aged rat hippocampus (Sato and Endo 1999). In this study we attempted to determine the precise region at which the change of α2–3 and α2–6 sialoglycoproteins occurs in the aged rat hippocampus. The experiments were based on a histochemical analysis of MAL and SSA using electron microscopy.
Materials and Methods
Animals
Five each of 9-week-old and 30-month-old Fischer female rats were used in this study. All experimental procedures using laboratory animals were approved by the Animal Care and Use Committee of the Tokyo Metropolitan Institute of Gerontology. Because the same results were found in all five rats in each group, only the data obtained for one sample in each group is described in detail here.
Lectin Staining for Light Microscopy
Under pentobarbital anesthesia, the brains were dissected out and immediately frozen in an OCT compound (Sakura; Tokyo, Japan) with dry ice. Rat brain coronal sections, perpendicular to the septotemporal axis at the approximate mid-point, were cut into 6-μm-thick sections on a cryostat and mounted on gelatin-coated slides. The sections were fixed by incubation in 4% formaldehyde in 10 mM Tris-HCl (pH 7.4), 0.14 M NaCl, 1 mM CaCl2, 1 mM MgCl2, and 1 mM MnCl2 (TBS) for 10 min. They were then immersed in 0.3% H2O2 in methanol for 30 min at room temperature (RT). Then the sections were washed three times for 10 min at RT with TBS and incubated with the biotinylated lectins (SSA 33.3 μg/ml; MAL 13.3 μg/ml) (Seikagaku; Tokyo, Japan) diluted in 3% bovine serum albumin (BSA)–TBS for 1 hr at RT. The sections were washed again with TBS three times for 10 min and incubated for 1 hr at RT with horseradish peroxidase (HRP)-conjugated streptavidin (20 μg/ml) (DAKO; Carpinteria, CA). After further washes three times with TBS for 10 min, the sections were incubated with 3,3′-diaminobenzidine solution (0.5 mg/ml in TBS) containing 0.3% H2O2 for 10 min at RT and then washed with distilled water.
Lectin Staining for Electron Microscopy
Under pentobarbital anesthesia, the rats were perfused transcardially with 10 mM PBS, pH 7.4, and then with 4% formaldehyde–PBS at RT 20 min. The brains were immediately dissected out and placed in 4% formaldehyde–PBS at 4C for 2 hr. Lectin staining for electron microscopy was carried out according to Miyata et al. (1996) with a minor modification. Sections (40 μm thick) were cut with a microslicer DTK-1500 (DSK; Kyoto, Japan). The sections were washed three times (10 min each) with PBS and treated with 5% BSA–PBS for 1 hr. They were then incubated with biotinylated lectins (SSA 33.3 μg/ml; MAL 13.3 μg/ml) in 0.1% BSA–PBS for 24 hr at 4C, washed three times (20 min each) with PBS, and then incubated with HRP-conjugated streptavidin (0.5 μg/ml) (Jackson Immunoresearch; West Grove, PA) in 0.1% BSA–PBS for 24 hr at 4C. As cytochemical controls, the sections were incubated with SSA or MAL in the presence of 0.2 M N-acetylneuraminic acid (Nacalai Tesque; Kyoto, Japan), or incubated with the HRP–streptavidin alone. After washing three times with PBS, the specimens were immersed in 3,3′-diaminobenzidine solution (0.5 mg/ml in PBS) containing 0.005% H2O2 for 5 min at RT and then washed with distilled water. Labeled specimens were postfixed with 2% glutaraldehyde and then with 1% OsO4 in 0.1 M phosphate buffer, dehydrated through a graded series of ethanol and propylene oxide, and then embedded in Epon 812. The ultrathin sections were post-stained with lead citrate and observed under a transmission electron microscope (JEM-1010; JEOL, Tokyo, Japan).
Morphological Study with Electron Microscopy
After perfused fixation with 4% formaldehyde–PBS, the brains were dissected out and fixed in 2.5% glutaraldehyde–PBS for 1 hr at 4C. They were postfixed in 1% OsO4–0.1 M phosphate buffer for 1 hr at 4C and dehydrated through a series of graded ethanol. After passage through propylene oxide, the specimens were embedded in Epon 812. Ultrathin sections were cut and stained with uranyl acetate and lead citrate and then examined with a transmission electron microscope (JEM-1010).
Results
Alteration of Granule Cells in Dentate Gyrus in the Aged Brain
Two populations of granule cells were observed in the granule cell layer of the hippocampal formation of the 9-week-old rats (Figure 1A). Cells with high electron density were observed in the innermost portion of the granule cell layer (Figure 1A, indicated with H) and cells with low electron density were in the remaining portion (Figure 1A, indicated with L). On the other hand, the cells with low electron density were mainly observed in the granule cell layer of the 30-month-old rats (Figure 1B, indicated with L), suggesting that the cells with high electron density almost disappeared during aging. It is interesting that the cells with high electron density were observed not only in the granule cell layer but also in the stratum pyramidale of the CA1 region of the 9-week-old rats, and the numbers of these cells were downregulated in the 30-month-old rats (data not shown). The same results were found in all five rats in each group, indicating that the difference was not due to differences among individuals.
Lectin Histochemistry
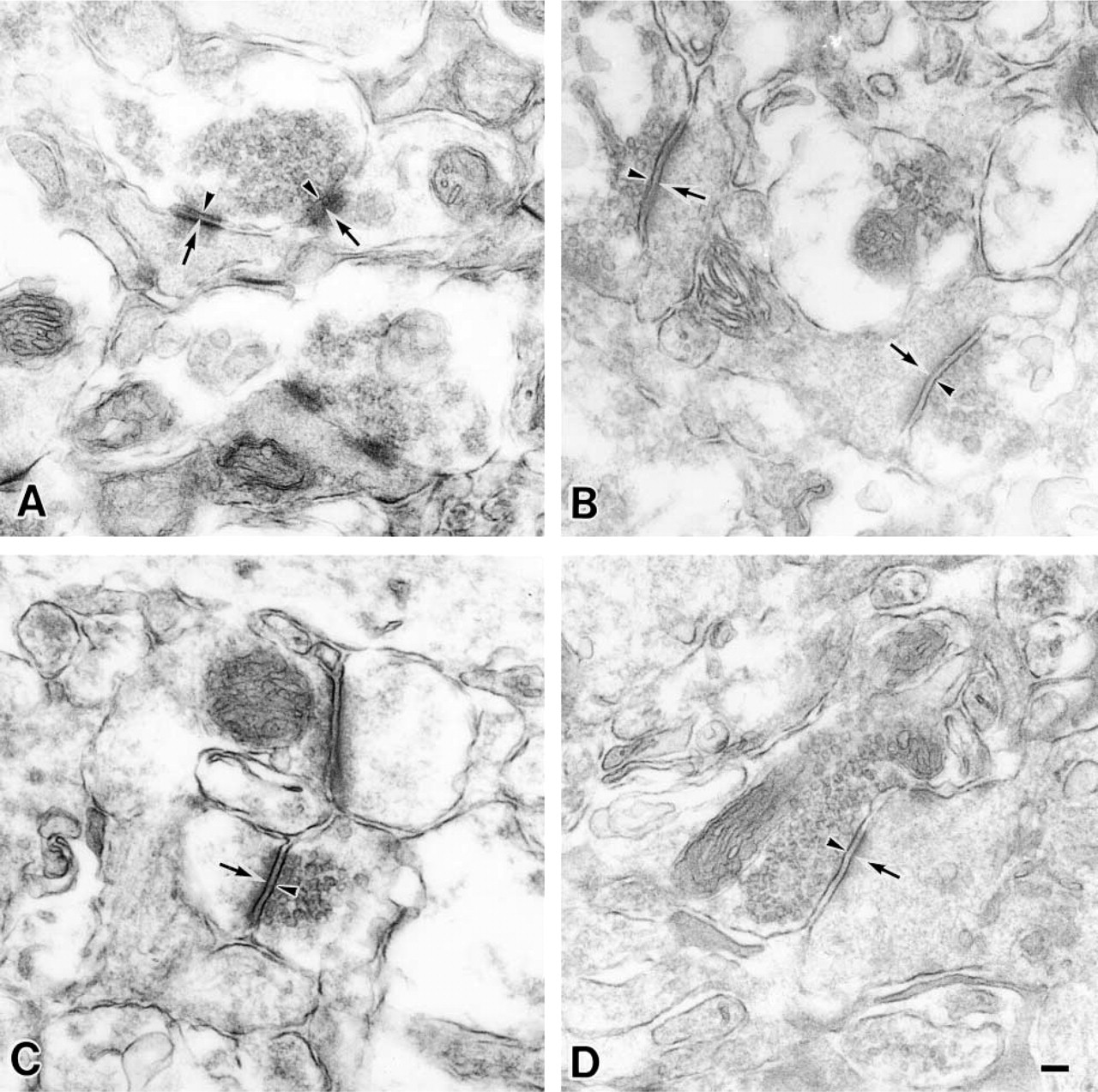
Cell Bodies of the Neurons in the Pyramidal Cell Layer and Their Dendrites in the Stratum Radiatum. Because a histochemical study of the CA1 region at the light microscopic level revealed reactivity to the stratum radiatum (sr in Figures 2F–2I) and low reactivity to the pyramidal cells (sp in Figures 2B–2E), we analyzed both areas in detail by electron microscopy. The plasma membranes of neuronal cell bodies in the stratum pyramidale from 9-week-old (Figure 3A) and 30-month-old rats (Figure 3B) were stained by MAL (indicated by arrowheads). The stratum radiatum is next to the stratum pyramidale, and many dendrites that originated from the pyramidal cells in the stratum pyramidale were observed there. The plasma membranes of the dendrites in stratum radiatum were also reactive with MAL in the 9-week- and 30-month-old rats (arrowheads in Figures 4A and 4B, respectively). Although all plasma membranes of cell bodies and dendrites were stained by MAL, no difference in staining between the 9-week-old and 30-month-old rats was observed. On the other hand, SSA showed similar reactivities to these plasma membranes (Figures 3C, 3D, 4C, and 4D). The lectin binding was inhibited by the addition of a hapten sugar, 0.2 M N-acetylneuraminic acid. Nonspecific binding of HRP–streptavidin conjugates and endogenous peroxidase activity were scarcely detected (data not shown). These results indicated that the Siaα2–3Gal and Siaα2–6Gal groups were expressed on the plasma membranes of the neuronal cell bodies and dendrites of pyramidal cells and suggested that the expression of these sialyl linkages did not change significantly during aging.
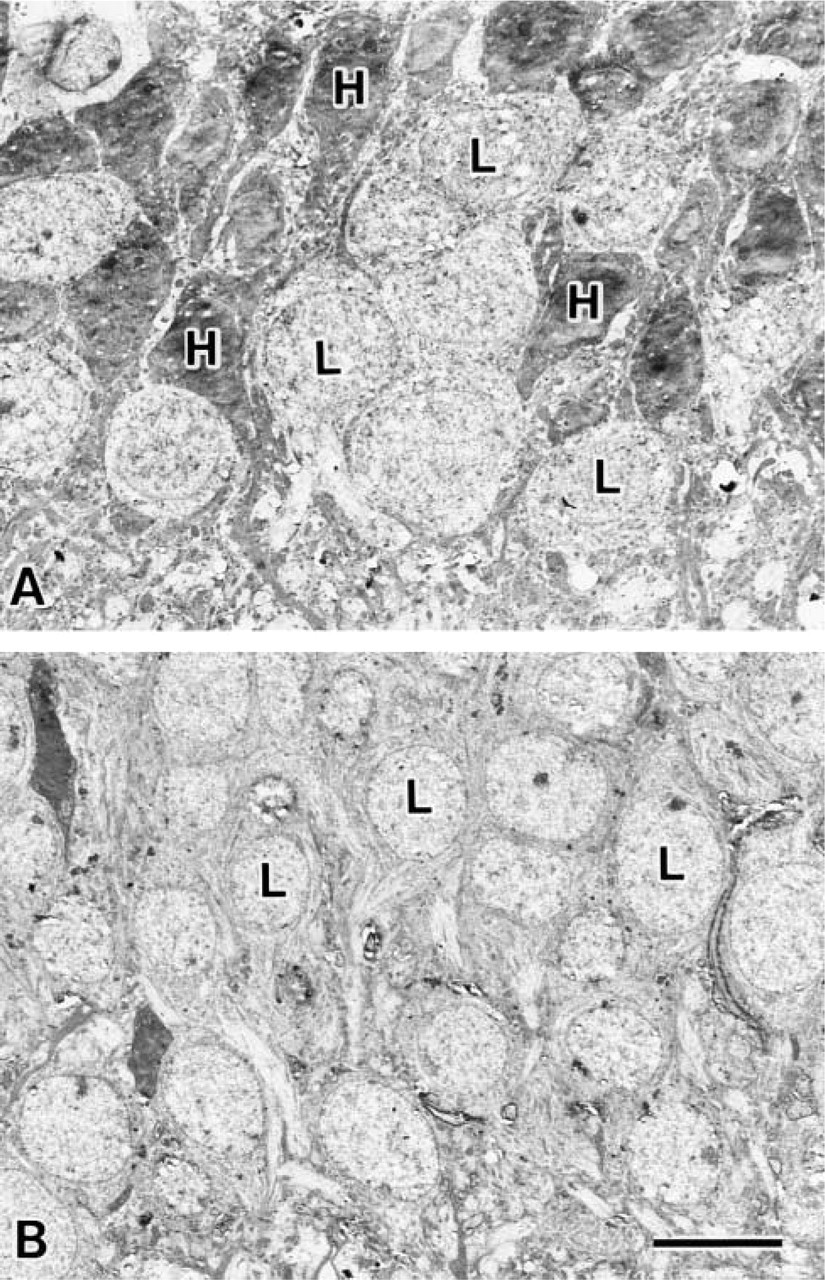
Transmission electron micrographs of granule cells in the dentate gyrus. Top of the picture is the hilar side and bottom is the granule cell layer (the orientations of these figures are indicated in Figure 2A). (
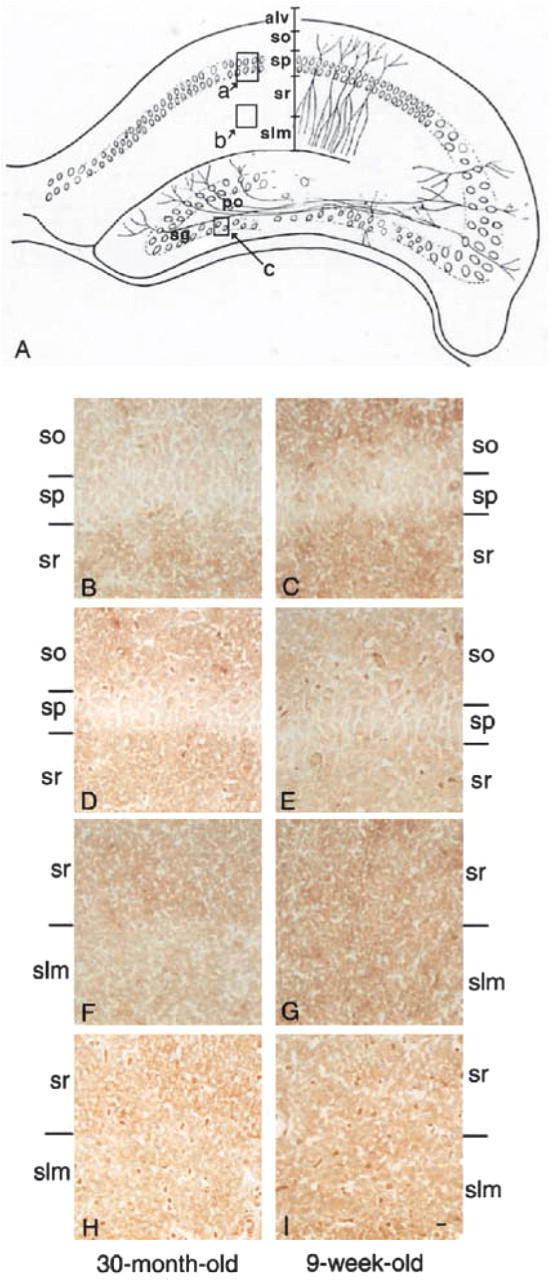
Light micrographs of the CA1 region of hippocampal formations from a 30-month old-rat (
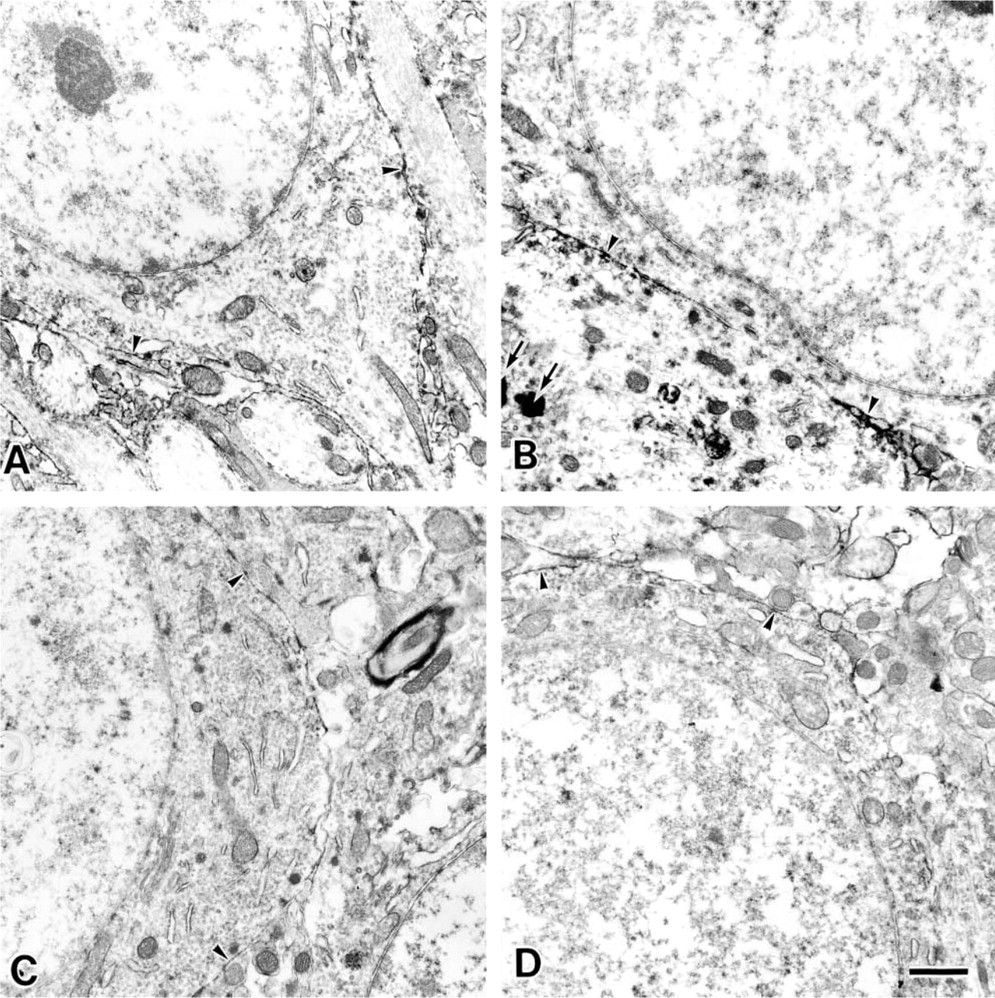
Electron micrographs of the neuronal cell body in the stratum pyramidale of the CA1 region from a 9-week-old rat (
It should be noted that conspicuous lipofuscin accumulation was observed only in the cytoplasm of the 30-month-old rats (arrows in Figure 3B). Lipofuscin is known to accumulate in the cytoplasm of aged cells only (Amenta et al. 1988).
Synapses in the Stratum Lacunosum Moleculare of the CA1 Region. The stratum lacunosum moleculare of the CA1 region is composed of stratum lacunosum and stratum moleculare, and is known to contain many synapses (Johnston and Amaral 1988). For example, in the stratum lacunosum, Shaffer collaterals, which originate from the CA2 or CA3 region, form synapses on the dendrites that originate from pyramidal cells in the CA1 region. Perforating fibers, which originate from the entorhinal area, also form synapses on the same dendrites in the stratum moleculare of the CA1 region. These synaptic formations are believed to play crucial roles in learning and memory. A histochemical study at the light microscopic level showed the reactivities of MAL and SSA to the stratum lacunosum moleculare in the 9-week-old rats (Figure 2G and 2I). On the other hand, their reactivities in the 30-month-old rats were slightly downregulated (Figure 2F and 2H). Therefore, we analyzed this area in detail with electron microscopy.
Electron micrographs of the dendrites in the stratum radiatum of the CA1 region from a 9-week-old rat (
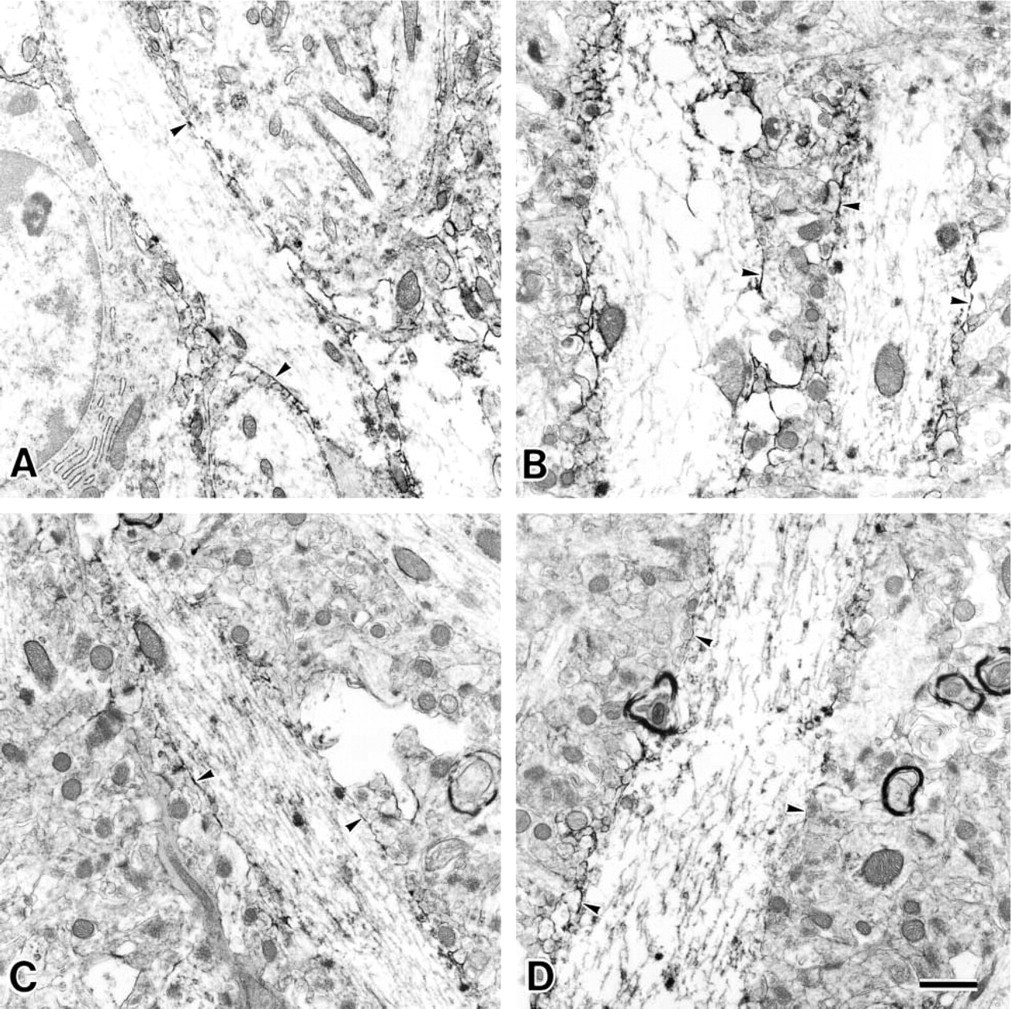
At first, the presynaptic membranes could be easily identified because of the presence of synaptic vesicles (arrowheads in Figures 5, 6, and 7). The reactivity of MAL was observed at the presynaptic membranes (arrowheads in Figures 5A and 6A) and postsynaptic membranes (arrows) in the stratum lacunosum moleculare of the 9-week-old rats. On the other hand, when the synaptic membranes of the 30-month-old rats were stained by MAL, its reactivity was less intense than that in the synaptic membranes of the 9-week-old rats (Figures 5B and 6B, indicated with arrowheads for the presynaptic membranes and arrows for the postsynaptic membranes). It is noteworthy that, in the 30-month-old rat samples, they showed similar intensity when they were stained with or without MAL (Figure 7). These results indicated that the reactivity of MAL decreased in the aged synapses. SSA also showed the same reactivity with these pre- and postsynaptic membranes (Figures 5C, 5D, 6C, and 6D, indicated with arrowheads for the presynaptic membranes and arrows for the postsynaptic membranes). Binding of these lectins was also inhibited by the addition of 0.2 M N-acetylneuraminic acid (data not shown). On the basis of these results, we conclude that, in these regions, the Siaα2–3Gal and Siaα2–6Gal groups were expressed on the pre- and postsynaptic membranes and that aging affected their expression.
Electron micrographs of synapses in the stratum lacunosum of the CA1 region from a 9-week-old rat (
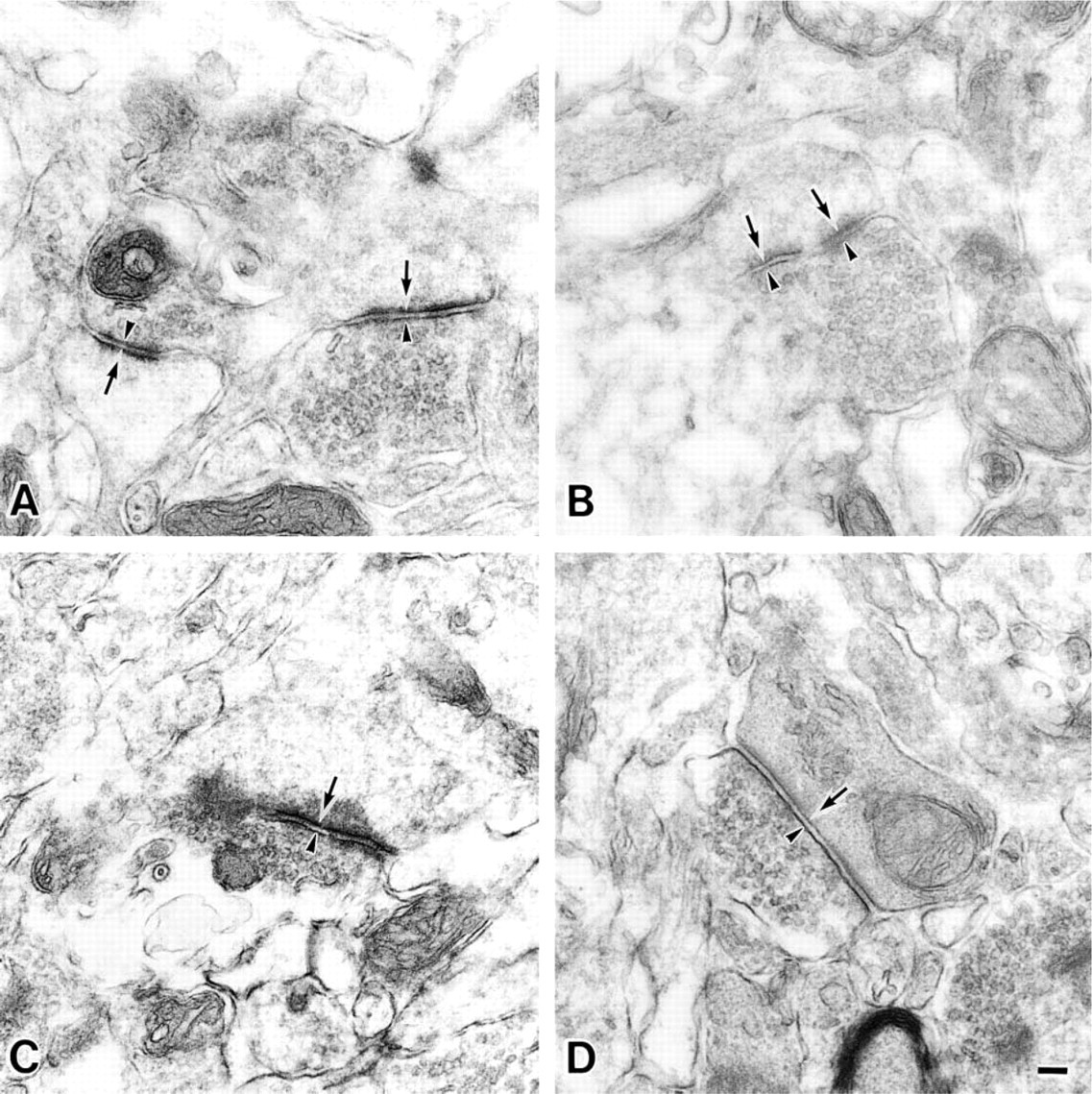
Electron micrographs of synapses in the stratum moleculare of the CA1 region from a 9-week-old rat (
Discussion
Alteration of Granule Cells in the Dentate Gyrus During Aging
In this study we observed two kinds of cells (low and high electron density) in the granule cell layer of the 9-week-old rats, while in the 30-month-old rats we observed mainly low electron density cells (Figure 1). These results suggest that the high electron density cells almost disappeared during aging. These high electron density cells were localized at the innermost portion in the granule cell layer. The granule cells in the innermost region are known to be newly generated (Kaplan and Hinds 1977; Kuhn et al. 1996; Eriksson et al. 1998) and are also stained by anti-PSA antibody (Seki and Arai 1991). PSA is known to be attached mainly to NCAM, to be highly expressed during neuronal development, and to affect the plasticity of neurons (Rutishauser and Jessell 1988; Landmesser et al. 1990; Seki and Arai 1993; Miller et al. 1994). However, the expression of PSA in the innermost portion in the granule cell layer disappeared during aging and the regeneration of granule cells was diminished (Seki and Arai 1995; Sato and Endo 1999). On the basis of these results, it was suggested that the high electron density cells detected in the 9-week-old rats may have been actively regenerating and then may have disappeared during aging. It is interesting that the cells with high electron density were observed not only in the granule cell layer but also in the stratum pyramidale of the CA1 region of the 9-week-old rats, and the numbers of these cells were downregulated in 30-month-old rats (data not shown). These results suggest that regeneration of neurons may also occur in the stratum pyramidale of the CA1 region.
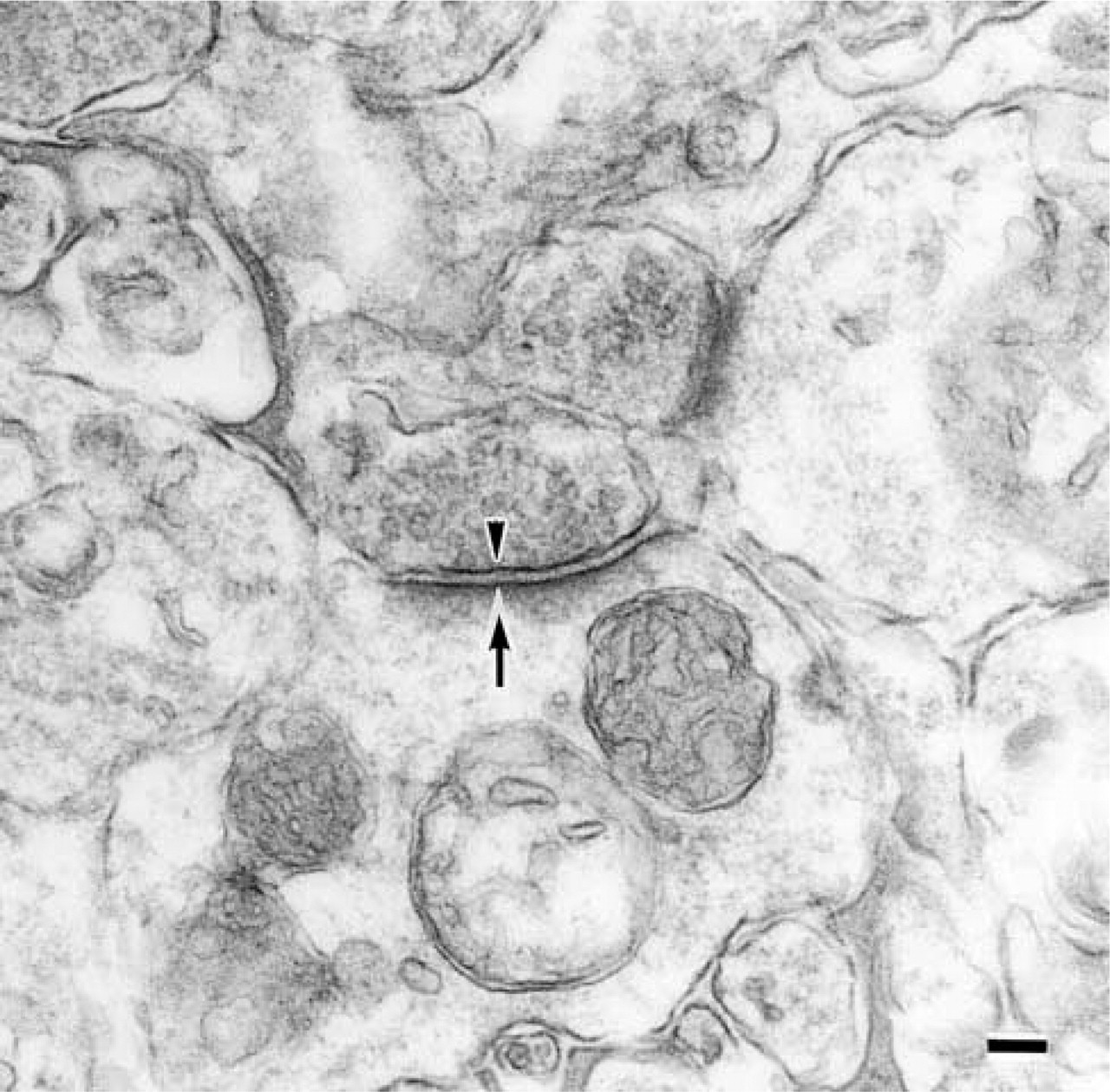
Cytochemical control of an electron micrograph of a synapse in the stratum moleculare of the CA1 region from a 30-month-old rat. The staining of this sample was carried out without lectin. The presynaptic membrane is indicated by an arrowhead and the postsynaptic membrane by an arrow. Bar = 0.1 μm.
Lectin Histochemistry
The stratum lacunosum moleculare of the CA1 region is known to contain many synapses (Johnston and Amaral 1988). In the present study, we observed that MAL and SSA bound to the synaptic membranes and that their reactivity was downregulated in the aged brain (Figures 5 and 6). These results indicated that the young adult rats had many glycoconjugates carrying the Siaα2–3 and Siaα2–6 groups at synapses in the stratum lacunosum moleculare, and suggested that the expression of these sialyl groups decreased during aging. On the other hand, the expression of these sialyl groups did not change on the plasma membranes of pyramidal cells (Figures 3 and 4). Therefore, the decrease in expression of sialyl linkages may be specific for synaptic membranes. The decrease of sialyl groups in the synapses in the aged brain could be due to a depression of the activity of sialyltransferases, such as α2–3 sialyltransferases which are involved in the formation of the Siaα2–3 group and/or in α2–6 sialyltransferases which are involved in the formation of the Siaα2–6 group (Tsuji 1996). Another possibility is that the decrease in sialyl groups is due to an increase in the activity of sialidases. Three mammalian sialidases have been described thus far: intralysosomal, cytosolic, and membrane-associated sialidases (Miyagi and Tsuiki 1984, 1985, Miyagi et al. 1990; Hata et al. 1998).
In the cerebral cortex, the amounts of gangliosides (sialic acid-containing glycolipids) in the synaptosomes decrease during aging (Ando et al. 1986; Waki et al. 1994). Therefore, in the hippocampus, the decrease in gangliosides in the synapses may also have caused the decrease staining of MAL and SSA during aging. However, it is not clear whether the amount of gangliosides in the hippocampus also decreases during aging. Furthermore, the activity of sialidase, which can hydrolyze only the gangliosides, decreased in synaptic membranes during aging (Saito et al. 1995). Therefore, it is very unlikely that a membrane-associated sialidase contributes to the decrease of gangliosides in the synapses during aging. These results suggest that the expression of Siaα2–3 and 2–6Gal groups in the synapses may be regulated in a complex manner, although the details remain unclear.
It was reported that cell adhesion molecules are crucially involved in the assembly and restructuring of synapses during development and that they are involved in synaptic plasticity (Edelman and Crossin 1991; Schachner 1991). Sialic acid residues are known to act as ligands for several adhesion molecules, such as those in the Siglec family. The Siglecs are a subfamily of I-type lectins that specifically recognize sialic acids and function as endogenous lectins (Crocker et al. 1994; Powell and Varki 1995). Our present study clearly revealed a change in sialic acids that occurs specifically in synapses. Therefore, it is probable that during aging the decrease in sialoglycoconjugates affected the intercellular adhesion properties via a sialic acid-binding molecule, like Siglecs, and the disturbance of adhesion then affected the synaptic activity. Such a change may induce deterioration of brain functions in aged people.
Footnotes
Acknowledgements
Supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology and by the Mizutani Foundation.
We thank Mr M. Fukuda and Ms T. Shibata (Laboratory for Electron Microscopy and Department of Anatomy, Kyorin University School of Medicine) for technical assistance.
