Abstract
Distraction osteogenesis (DO) is a limb-lengthening procedure that combines mechanical tension stress with fracture healing to provide a unique opportunity for detailed histological examination of bone formation. Osteopontin (OPN) is a multifunctional matricellular protein believed to play a key role in wound healing and cellular response to mechanical stress. We studied the expression of OPN during DO using standard immunohistochemical (IHC) staining techniques. In addition, we compared the expression of OPN to proliferation (PCNA-positive cells) in the DO gap. After 14 days of distraction in the rat, these stains revealed variations in OPN expression and its relationship to proliferation according to the cell type, tissue type, and mode of ossification examined. Fibroblast-like cells within the central fibrous area exhibited intermittent low levels of OPN, but no relationship was observed between OPN and proliferation. In areas of transchondral ossification, OPN expression was very high in the morphologically intermediate oval cells. During intramembranous ossification, osteoblasts appeared to exhibit a bimodal expression of OPN. Specifically, proliferating pre-osteoblasts expressed osteopontin, but OPN was not detected in the post-proliferative pre-osteoblasts/osteoblasts that border the new bone columns. Finally, intracellular OPN was detected in virtually all of the mature osteoblasts/osteocytes within the new bone columns, while detection of OPN in the matrix of the developing bone columns may increase with the maturity of the new bone. These results imply that the expression of OPN during DO may be more similar to that seen during embryogenesis than would be expected from other studies. Furthermore, the biphasic expression of OPN during intramembranous ossification may exemplify the protein's multi-functional role. Early expression may facilitate pre-osteoblastic proliferation and migration, while the latter downregulation may be necessary for hydroxyapatite crystal formation.
Keywords
D
Beyond 7 days of distraction in the rat, the distraction gap is bridged by five biologically active, spatially oriented zones (Figure 1) (Aronson et al. 1997b). The central zone, termed the fibrous interzone, is dominated by fibroblast-like cells and mesenchymal cells associated with parallel collagen bundles. The fibrous interzone is bound on both sides by the primary matrix front where mesenchymal stromal cells proliferate and differentiate into osteoblasts and where bone matrix deposition first occurs. The primary matrix fronts are, in turn, bound by the zones of microcolumn formation where bone matrix expands around osteoblasts/osteocytes into parallel microcolumns alternating with developing sinusoids. These areas have been previously described and provide a spatially oriented in vivo model of bone formation analogous to the growth plate for chondrogenesis. The temporospatial organization of the distraction gap enables one to study cellular and molecular events in several cell types at various stages of bone formation and cell development. Within a single histological section, one can observe pre-osteoblasts, proliferating osteoblast progenitors, fully differentiated osteoblasts, and osteocytes spatially oriented in a manner that is typically representative of their function and degree of maturity.
Bone formation during DO is extremely rapid, approximately seven times that of embryonic development. It is believed that the mechanical tension stress provided by the external fixator is responsible for the increased rate of osteogenesis. However, the exact mechanism by which the mechano-signaling stimulates osteoblastogenesis during DO is unknown.
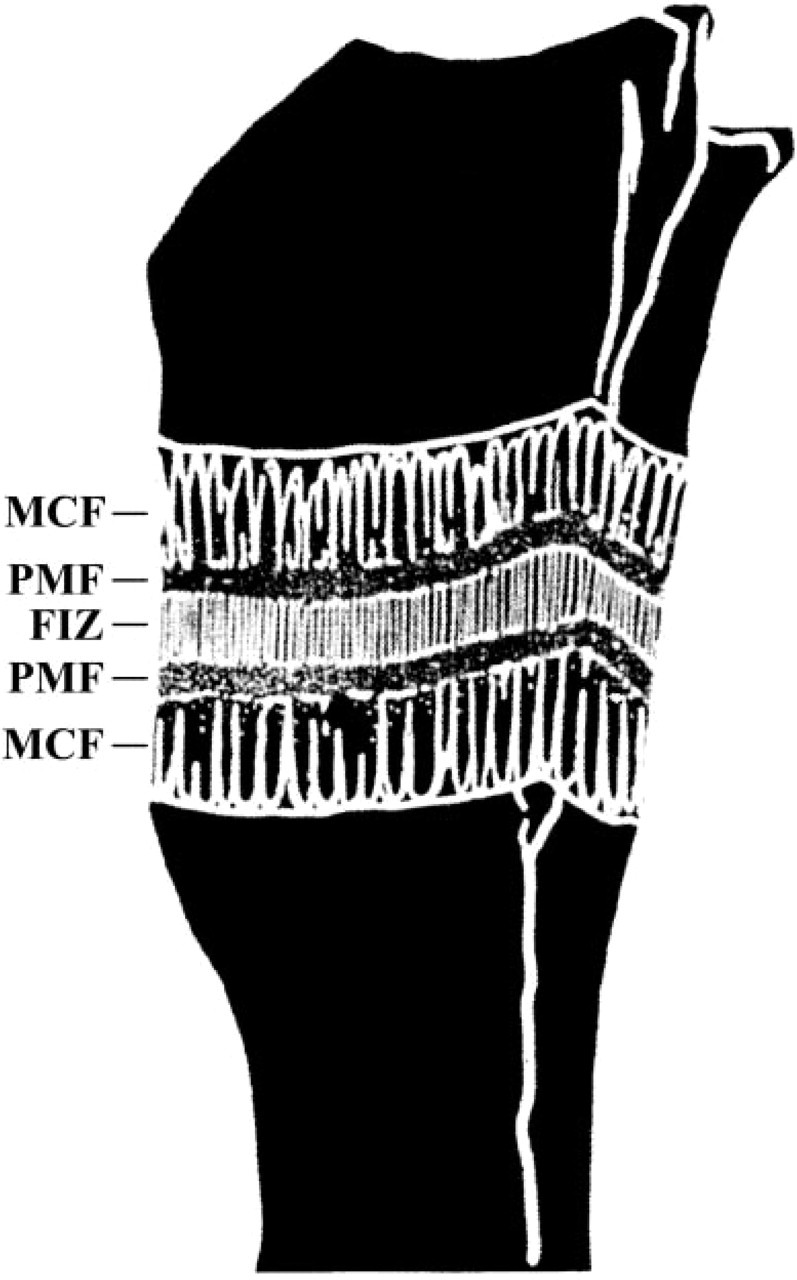
Schematic depicting the five biologically active zones formed in the rat tibia after ten days of distraction. The centrally located fibrous interzone (FIZ) is bounded on both sides by the proximal and distal primary matrix front (PMF). Each primary matrix front is bounded additionally by the respective proximal and distal areas of microcolumn formation (MCF), which extend from the cortical host bone and intermedullary canals.
Osteopontin (OPN) is a non-collagenous multifunctional glycoprotein routinely present in mineralized tissues (McKee and Nanci 1996), which is believed to play an integral role in cellular responses to mechanical stimuli. One recent study in OPN knockout mice revealed that OPN is necessary for the increase in osteoclastic bone resorption and decrease in osteoblastic bone formation normally associated with skeletal unloading (Ishijima et al. 2001). Moreover, OPN appears to be necessary for mechano-stimulation of osteogenesis in the developing cranial suture. Conversely, mechanical stimuli are known to induce and/or increase the expression of OPN in osteoblasts and osteocytes (Sodek et al. 1995; Toma et al. 1997; Miles et al. 1998; Terai et al. 1999; You et al. 2001). These studies imply that OPN may play an integral role in the osteogenic response to mechanical stress during DO.
An immunohistochemical (IHC) study of OPN expression in developing rat mandible reported various degrees of OPN expression by pre-osteoblasts, osteoblasts, and osteocytes (Pinero et al. 1995). Furthermore, this study noted a progressive increase in OPN detection in the membranous bone matrix that accompanied maturation of the new bone. Other studies have noted a biphasic pattern of OPN expression corresponding to proliferation and differentiation of osteoblastic cells during bone formation (Stein and Lian 1993; Aubin et al. 1995). This area is comparable to the intramembranous primary matrix front during DO. In contrast, an in situ hybridization (ISH) study of OPN expression in a rat model of DO reported detection of mRNA transcripts only in the pre-osteoblasts and osteoblasts of the primary matrix front and the leading edge of the microcolumns with no report of a biphasic expression pattern (Sato et al. 1998). The differences in these reports are somewhat surprising because bone formation in the mandible occurs by intramembranous ossification and previous studies in the rat model of DO have demonstrated a dense line of proliferating osteoblast progenitors (the proliferative front) within the primary matrix front (Aronson et al. 1997a), similar to that seen in the developing rat mandible. The reasons for these discrepancies are not clear, and further investigation is needed to determine if they are technique- or model-dependent. Furthermore, examination of OPN's presence in the extracellular matrix during DO may provide new insights into its role during bone formation under mechanically stressed conditions in vivo. To our knowledge, no IHC study of OPN protein localization during DO has been reported. Therefore, the present study was performed to examine OPN expression during DO by standard IHC methods.
In this study, osteopontin protein expression during DO in the rat was examined after 14 days of distraction, focusing on areas of intramembranous and transchondral ossification. To examine any potential relationship between OPN expression and proliferation of osteoblastic cells, OPN-stained sections were also compared to serial sections stained for PCNA. The working hypotheses for these studies were (a) that OPN would be detected at various levels in the extracellular matrix throughout the distraction gap, (b) that OPN would be detected at different intensities in fibroblasts of the central fibrous interzone, pre-osteoblasts, and osteoblasts of the primary matrix front, and osteoblasts and osteocytes within the forming microcolumns, and (c) that a relationship would be observed between proliferation of pre-osteoblasts and OPN expression in the primary matrix front where intramembranous ossification occurs.
Materials and Methods
Surgical Procedures
The following procedures were approved by the institutional animal care and use committee and performed as previously described (Aronson et al. 1997a). Briefly, under sodium pentobarbital anesthesia, 4-month-old male Sprague-Dawley (Harlan; Indianapolis, IN) rats were fitted with external fixators on the left tibia. A low-energy mid-diaphysial osteotomy was created by drilling three small holes through the mid-diaphysis and applying direct lateral pressure. The fibula of the fixated leg was also fractured by direct pressure. The rats were given 0.1 mg/kg buprenex for analgesia and returned to their cages for observation during recovery from anesthesia. Distraction was initiated the following morning (1-day latency) at a rate of 0.2 mm b.i.d. (0.4 mm/day) for 14 days. Immediately after distraction (Day 14 post-op), the animals were sacrificed under sodium pentobarbital anesthesia. The left distracted tibiae were removed by disarticulation at the knee and ankle, and the soft tissues were dissected away. The specimens were then placed in 10% neutral buffered formalin, with the external fixators intact, for 48 hr. This was done to minimize shrinkage of the fibrous tissue of the distraction callus after removal of the fixator. Tibiae were removed from the fixators using a manual saw and returned to 10% NBF before histological processing (approximately 3 additional days).
Specimen Preparation
Specimens were processed as described (Aronson et al. 1997a,2001; Skinner et al. 1997). These procedures have been used previously for IHC labeling of PCNA (Aronson et al. 1997a,2001) and are preferred for labeling of OPN in decalcified bone (Frank et al. 1993). After fixation, specimens were decalcified in 5% formic acid, dehydrated, and embedded in paraffin. Coronal sagittal sections were cut at 5 μm thick and mounted on Ultrastick glass slides (Becton-Dickinson; Franklin Lakes, NJ).
Immunohistochemistry for OPN
Sections were deparafinized and rehydrated through xylene and serial dilutions of EtOH to distilled H2O. They were then incubated in Antigen Retrieval Citra (Biogenex; San Ramon, CA) at 95C for 15 min. Sections were washed in PBS, pH 7.4, with 0.02% Triton X-100 (PBS) twice for 3 min each. All incubations were performed in a humidity chamber at room temperature and followed by two, 3-min washes in PBS. For labeling of OPN, the specimens were incubated with levamisol (Vector Laboratories; Burlingame, CA) for 30 min to block endogenous alkaline phosphatase activity. Primary antibody OP-199 (Liaw et al. 1994), generously provided by Dr. Giachelli, was applied at a concentration of 74 μg/ml, diluted in PBS with 1% normal goat serum, for 90 min. OP-199 is known to recognize full-length OPN as well as the thrombin and MMP cleavage fragments. Its activity does not appear to be impaired by phosphorylation, glycosylation, or sulfation (Liaw et al. 1994; Rittling and Feng 1998; personal communication with Dr. Giachelli). For negative control, the primary antibody was replaced with normal goat IgG. All sections were incubated with biotinylated anti-goat IgG for 20 min. Vectastain ABC-AP (Vector Laboratories) prepared according to the manufacturer's instructions was applied for 30 min. Finally, color was developed using NBT/BCIP (Dako; Carpinteria, CA). Development of the blue substrate was monitored under a microscope and reactions were quenched when the signal-to-noise ratio was observed to be at an optimum. The sections were dehydrated in EtOH and Xylene and mounted using permanent medium.
Immunohistochemical Labeling of PCNA
To compare the relationship of OPN expression to proliferation, serial sections to those stained for OPN were immunohistochemically labeled for PCNA expression as previously described (Aronson et al. 1997a). Briefly, PC-10, a mouse monoclonal antibody against PCNA, (Biogenex; ready to use) or normal mouse IgG for negative control (Dako) was applied for 60 min. The sections were incubated with biotinylated horse anti-mouse antibody for 20 min, followed by a 30-min incubation with peroxidase-conjugated streptavidin. Color was developed by application of DAB (Zymed; South San Francisco, CA). Development of the brown substrate was quenched after 3 min. Slides were then dehydrated and mounted as above.
Semiquantitation of OPN- and PCNA-positive Cells
Semiquantitation of PCNA+ and OPN+ cells was performed as previously described, with slight modifications (Aronson et al. 2001). A standardized grid of 0.36-mm2 squares was overlaid on the labeled sections. Each square was examined and assigned to one of the previously defined DO zones: fibrous interzone, primary matrix front, or microcolumn formation. Positively stained cells in each square were counted and the totals for each zone or region were determined. Three mid-coronal sections were scored by three independent observers with <3% differences in cell counts. Positively stained cell counts were expressed in concentrations (number of cells/mm2 per DO zone). The positive cell counts in each region were averaged per specimen. The zonal histology was less representative in more peripheral sections. Therefore, only central sections with all four cortices and both marrow spaces were counted. In addition, the intensity of the positively stained cells within each zone was qualitatively noted by each of the three observers where “+” indicated that the overall staining was weakly positive and “+++++” indicated intense staining.
Results
Sections incubated with OP-199 or PC-10 displayed several novel staining patterns, and no significant signal was seen in the control sections. Proliferating cells (PCNA+) were distributed in typical fashion throughout the central callus as described (Aronson et al. 1997a). Briefly, proliferating cells were scattered throughout the centrally located fibrous interzone in an apparently random and unorganized pattern (Figure 2C). At the primary matrix front, a dense line of proliferating cells (proliferation front) was seen outlining the microcolumns where intramembranous ossification occurred. These cells are believed to be osteoblast progenitors and immature osteoblasts (Figure 2H). In areas of transchondral ossification, the number and concentration of proliferating cells in the primary matrix front appeared to be significantly less than in the intramembranous primary matrix front (Table 1). No PCNA+ cells were seen in the zone of microcolumn formation.
Qualitatively, OPN expression appeared to vary according to the tissue and cell type examined. OPN was detected in the matrix and in some cells in each of the three zones. The intensity of the stain in both positive cells and matrix was weakest in the fibrous interzone compared to the other tissue types examined (Figure 2A). OPN-positive fibroblast-like cells were scattered throughout the fibrous interzone in an apparently random fashion, and matrix staining was relatively even throughout.
In the primary matrix front, OPN expression varied according to the type of ossification examined. In areas of transchondral ossification, the intensity of the matrix staining was weak and relatively even throughout. Positively stained cells in the transchondral primary matrix front appeared to express moderate to high levels of OPN (Figure 2D). In fact, the overall intensity of OPN in the positive oval cells was higher than in any other cell type (Table 1). OPN appeared to be evenly distributed throughout the cytoplasm of transchondral cells, and unstained nuclei were common (Figure 2D).
In the intramembranous primary matrix front, a distinct biphasic pattern of OPN expression was observed (Figure 2F). A thin dense line of OPN+ cells was seen where the primary matrix front borders the fibrous interzone. In addition, a second line of OPN- cells, corresponding to nonproliferative differentiating preosteoblasts, was present between the OPN+ line and the edge of the developing microcolumns where OPN appeared to be upregulated again. Comparison of OPN-stained sections with those stained for PCNA revealed close relationships between the OPN+ line and the proliferation front as well as the OPN- line and the postproliferative cells of the primary matrix front immediately adjacent to the microcolumns (Figure 2H). Matrix staining in the intramembranous primary matrix front also appeared to follow this pattern.
The concentration of OPN+ cells in the microcolumns was notably higher than in columns formed by transmembranous ossification (Table 1). However, this probably reflects differences in total cell density in these columns rather than differences in the incidence of OPN expression. No other notable differences in OPN expression were seen in the transchondral and intramembranous microcolumns. In this region of the callus, the matrix and cells stained with an intensity that varied from moderate to high, whereas sinusoids between the microcolumns showed little or no OPN+ staining (Figure 2I). At the leading edge of the microcolumns, where they border the primary matrix front, the matrix was moderately positive for OPN. However, the intensity of the matrix staining in the microcolumns grew progressively greater with increasing proximity to the host cortex (Figure 2K). This may indicate that the concentration of OPN in the new bone matrix increases with the maturity of the new osteoid.
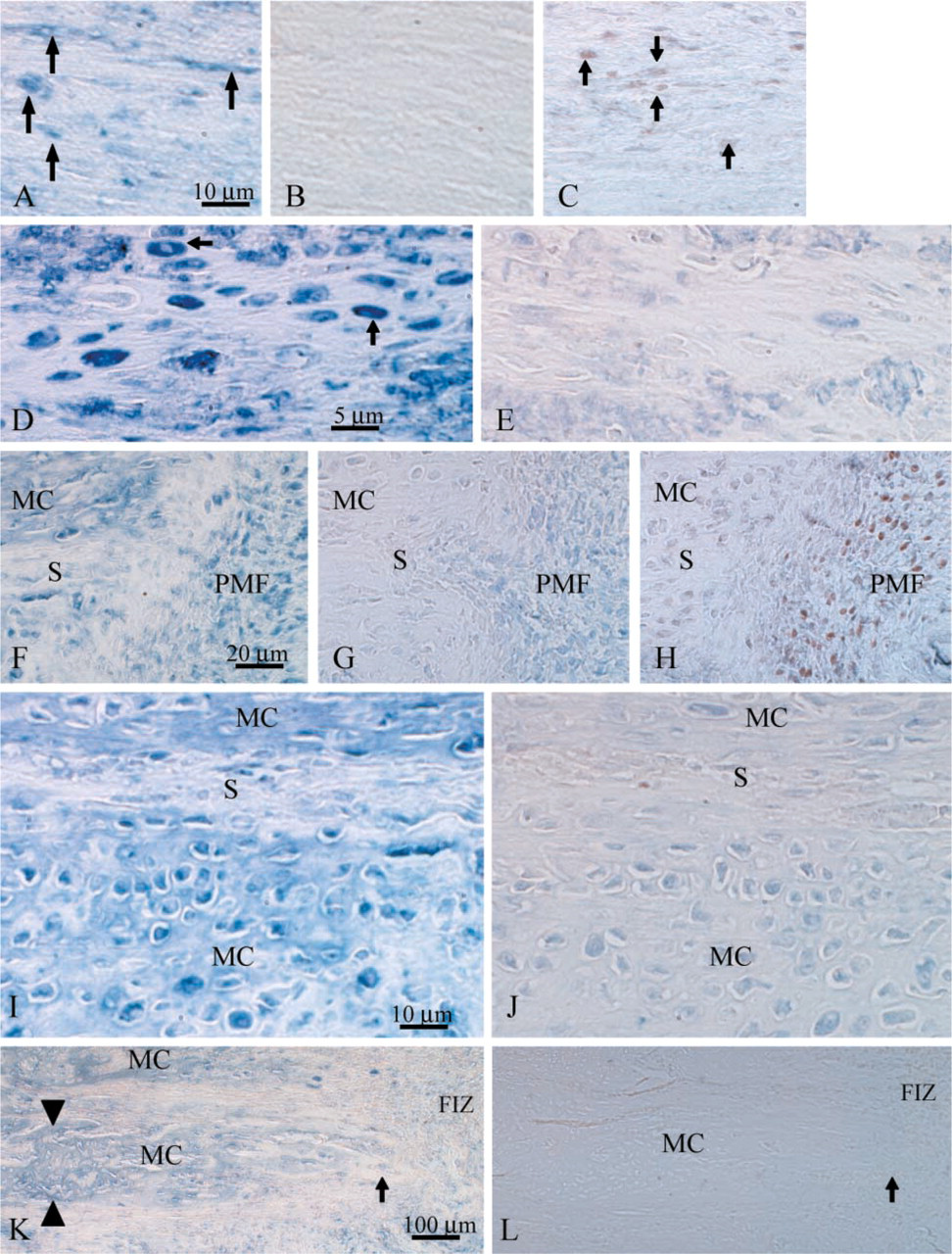
Immunohistochemical labeling of OPN and PCNA. (
Semiquantitation of OPN and PCNA labeling in the DO gap
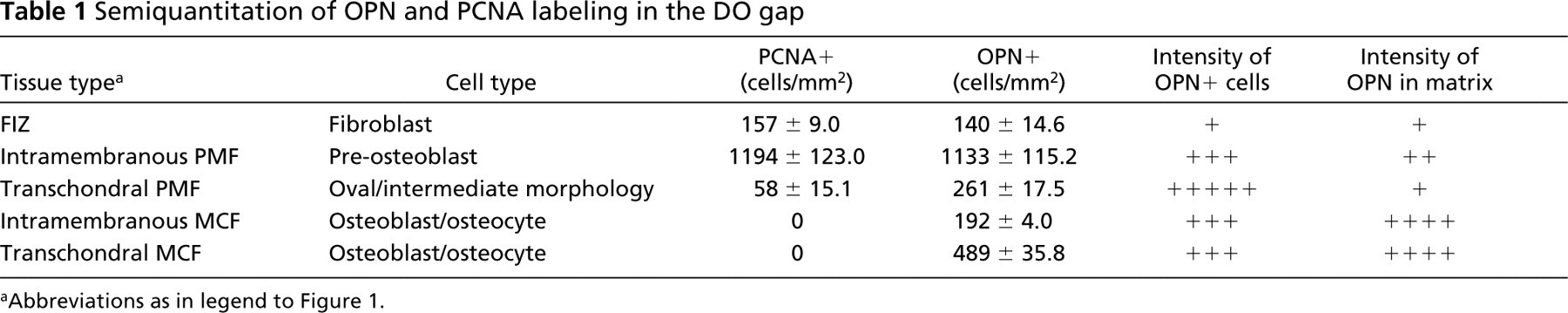
Abbreviations as in legend to Figure 1.
Semiquantatively, the concentration of both OPN+ and PCNA+ cells varied according the tissue and cell type examined (Table 1). The concentration of PCNA+ and OPN+ cells was highest in the intramembranous primary matrix front and lowest in the fibrous interzone. The concentration of OPN+ cells appeared comparable to the number of PCNA+ cells in both the fibrous interzone and the intramembranous primary matrix front. Interestingly, OPN+ cells were significantly more concentrated than PCNA+ cells in the transchondral primary matrix front.
Discussion
This study demonstrates the expression of OPN during DO in adult (4-month-old) rats by IHC. It provides evidence that cells in the fibrous interzone, intramembranous and transchondral primary matrix front, and the developing microcolumns express OPN. The intracellular and extracellular intensity of the OPN stain and concentration of OPN+ cells is highly variable between the three zones. Comparison of OPN-labeled sections to those labeled for PCNA revealed a close relationship between proliferation and OPN expression only in the primary matrix front where intramembranous ossification occurred.
During DO, the tissue composition of the distraction callus, or gap, undergoes three major changes. As in typical fracture healing, a hematoma is formed at the time of osteotomy. During the first 7–10 days of distraction, the hematoma is gradually replaced by fibrous tissue and initial bone formation begins at the proximal and distal host cortices and marrow spaces. As the stretching continues, the new bone columns continue to form along the tension vector, growing towards the center of the gap. When the desired amount of lengthening is achieved, the distraction is stopped and the external fixator remains in place to stabilize the callus until the new bone is bridged/fused, consolidated, and the medullary canal is formed in the lengthened segment.
The work presented here utilized specimens harvested immediately after 14 days of active distraction. These specimens provide the opportunity for detailed histological examination of the osteogenesis in a temporospatially organized manner isolated from processes of bone resorption. The concentration of OPN+ cells, intensity of intracellular staining, and intensity of matrix staining in the FIZ were all notably lower than in the other tissue types examined. In contrast, an ISH study during DO in the rat did not detect OPN in the cells of the fibrous interzone (Sato et al. 1998). This discrepancy may be due to differences in the sensitivity of mRNA vs protein detection techniques. Furthermore, in an IHC study of OPN in the developing rat mandible, OPN was not detected in the fibroblasts adjacent to areas of intramembranous bone formation (Pinero et al. 1995). The induction of OPN by mechanical stress in fibroblasts and osteoblasts is well documented (Kubota et al. 1993; Sodek et al. 1995; Toma et al. 1997; Carvalho et al. 1998; Miles et al. 1998; Nomura and Takano-Yamamoto 2000). Therefore, it is plausible that the mechanical tension of DO is responsible for the presence of OPN in fibroblasts seen here.
Intracellular OPN appeared to be localized in discrete foci either within the cytoplasm or on the membrane of the fibroblast-like cells during DO (Figure 2A). An in vitro study of fetal calvarial fibroblasts reported two patterns of intracellular OPN expression (Zohar et al. 1997). In less differentiated migratory cells OPN was present in a perimembranous pattern, whereas labeling was perinuclear in more mature matrix-producing cells. The chromogenic technique used here does not allow detailed examination of intracellular staining patterns. However, in the fibrous interzone it may be necessary for some cells to produce new matrix to fill the expanding gap while others migrate either to the primary matrix font or other locations. Therefore, both perinuclear and perimembranous expression of OPN may be present in the fibrous interzone.
The comparison of OPN to PCNA in the fibrous interzone did not reveal any relationship between proliferation and OPN expression. This was expected because of the apparent random and unorganized pattern of proliferation in the fibrous interzone. A double labeling technique is needed to draw any firm conclusions about the relationship of OPN to proliferation in this zone.
Two types of ossification were examined in the primary matrix front: transchondral and intramembranous. There were distinct differences in both OPN and PCNA expression patterns between the two modes of ossification. The concentration of both OPN+ and PCNA+ cells was dramatically higher in areas of intramembranous bone formation, whereas the intensity of intracellular staining for OPN was dramatically higher in the morphologically intermediate transchondral cells than in the pre-osteoblasts/osteoblasts of the intramembranous matrix front. There appeared to be little difference in the intensity of OPN labeling in the extracellular matrix. Therefore, OPN may have a different role(s) during initial matrix formation according to the mode of ossification.
The biphasic pattern of OPN expression seen in areas of intramembranous bone formation is similar to that seen in other models (Stein and Lian 1993; Aubin et al. 1995; McKee and Nanci 1995; Sodek et al. 1995). However, this pattern is significantly different from the mRNA pattern reported during DO where OPN mRNA was present only in the leading edge of the microcolumns (Sato et al. 1998). Most importantly, the biphasic expression of OPN seen here was not reported at the mRNA level. This indicates that although mRNA transcripts are at high concentrations in the primary matrix front, the protein is actually expressed at lower levels compared to cells in the microcolumns, where mRNA was below detectable levels. These findings suggest that the abundance of OPN mRNA transcripts is not reflective of the intracellular abundance of the protein during DO.
Biphasic OPN expression during intramembranous osteogenesis is probably representative of its multifunctionality during osteogenesis. The binding of extracellular OPN by αvβ3 or other integrins on the cell surface may activate several signaling cascades leading to increased proliferation, cell motility, and survival signals (Sodek et al. 2000), all of which may be necessary to sustain the proliferation throughout DO. Therefore, it is not surprising to see OPN expressed in the proliferative front, where it may serve both as an adhesive and migratory element for non-proliferating cells and simultaneously stimulate proliferation in other cells. In addition, OPN is known to inhibit hydroxyapatite crystal formation (Boskey et al. 1993; Hunter et al. 1994; Wada et al. 1999; Hoyer et al. 2001). Hence, extracellular OPN in the proliferation front may also serve to prevent premature crystalline matrix formation that would embed the osteoblast progenitors and prevent future column growth. Conversely, elimination of OPN expression by the post-proliferative osteoblastic cells between the proliferation front and the edge of the microcolumns may be necessary for initiation of hydroxyapatite and hence matrix formation.
In the transchondral primary matrix front, the intense intracellular OPN stain combined with the low extracellular OPN signal suggests that OPN may play a different role compared to intramembranous ossification. The pattern of OPN expression in these cells was identical to the mRNA pattern reported (Sato et al. 1998). Comparison of OPN and PCNA labeling failed to reveal any clear association during transchondral bone formation. However, no firm conclusion can be drawn due to the apparent disorganized pattern of proliferation and heterogenous expression of OPN in these cells. The extremely low intensity of the extracellular OPN signal suggests that OPN interactions with cell surface membranes may play a less prominent role in transchondral ossification. In contrast, the apparent high concentration and even distribution of OPN within the morphologically intermediate oval cells suggests that the protein may have an important function in intracellular processes. Recent studies have suggested a role for intracellular OPN in the CD44-ERM complex involved in cell migration (Zohar et al. 2000), and there has been some speculation that this complex may be associated with cell transformation (Sodek et al. 2000). The oval cells found in areas of transchondral ossification during DO are believed to be intermediate between chondrocytic and osteoblastic phenotypes. Therefore, there may be a high potential for OPN's involvement in this transdifferentiation process.
The similarity of OPN expression in the microcolumns formed by either intramembranous or transchondral ossification indicates that the mode of initial matrix formation has little or no effect on the expression of OPN by osteoblasts/osteocytes after they become embedded in new osteoid. OPN was detected in osteoblasts and the extracellular matrix of the microcolumns approximately 200 μm from the border with the primary matrix front. This suggests that the protein is produced and secreted by embedded osteoblasts relatively soon after initial matrix formation. At the leading edge of the microcolumns the intensity of the extracellular stain is relatively weak but continues to grow with proximity to the host cortex and/or marrow space (reflective of the maturity or relative age of the new bone during DO), indicating that secretion of OPN continues throughout the maturation of the new bone. This high level of extracellular OPN is probably a chemotaxic and adhesive agent for osteoclasts during the subsequent formation of the medullary canal within the lengthened segment. However, it may also be important for continued maturation of the new bone as other matrix components bind to OPN. Finally, the apparent high level of OPN in the developing bone matrix indicates that although OPN may be able to impair hydroxyapatite crystsallization, it may not interfere with crystallization in the later stages of bone maturation. Such a change in protein function could be achieved by enzymatic cleavage or other post-translational modifications that would limit OPN's ability to inhibit crystal formation.
In agreement with our hypotheses, the results reported here indicate that OPN is present at some level in fibroblasts, pre-osteoblasts, osteoblasts, transchondral cells, and osteocytes in the distraction callus of the rat tibia after 14 days of lengthening. However, this expression appears to vary greatly among cell types and stages of osteogenesis. Comparison of OPN to PCNA on serial sections revealed a distinct relationship between OPN expression and proliferation where intramembranous ossification occurred. In this area OPN expression was biphasic with one peak corresponding to proliferation and a second corresponding to bone matrix maturation. Labeling of OPN was also seen in the extracellular matrix of each zone examined. Taken in the context of other published data, these results suggest that OPN may play distinctly different roles in each of the three callus zones and according to the mode of bone formation. This implies that OPN plays an integral role in the mechanical induction and maintenance of rapid bone formation during DO. Future studies addressing the questions raised here may further our understanding of these mechanisms.
Footnotes
Acknowledgements
Supported by NIH grants AR44987 and AA12223, by the Brooks Medical Research Fund, by the Laboratory for Limb Regeneration Research-Arkansas Children's Hospital Research Institute, and by the Departments of Orthopedics and Pediatrics, University of Arkansas for Medical Sciences.
We especially thank Dr Ceclia Giachelli for her generous provision of the OP-199 antibody used in this experiment.
