Abstract
While the surgical procedure of distraction osteogenesis (DO) is very successful in the treatment of orthopedic conditions, its major limitation of slow bone formation in the distracted gap has prompted numerous attempts to understand and accelerate this slow bone formation. Interestingly, WNT/FZD signaling has been identified as a critical pathway in mediating bone formation and regeneration but has not yet been studied in the context of DO. The objective of this study was to determine the spatial and temporal localization of endogenous WNT signaling proteins at various times of bone formation in a wild-type mouse model of DO. In this study, the DO protocol performed on mice consisted of three phases: latency (5 days), distraction (12 days), and consolidation (34 days). Our immunohistochemical findings of distracted bone specimens show an increased expression of WNT ligands (WNT4 and WNT10A), receptors (FZD1 and 2, LRP5 and 6), β-catenin, and pathway antagonizers (DKK1; CTBP1 and 2; sFRP1, 2, and 4) during the distraction phase, which were then down-regulated during consolidation. This is the first published report to show an activation of the WNT pathway in DO and could help identify WNT as a potential therapeutic target in accelerating bone regeneration during DO.
Distraction osteogenesis (DO) is a surgical technique developed by Ilizarov (1989a) for the treatment of limb length discrepancies, limb deformities, and bone loss secondary to trauma, infections, and postresection of tumors. It is considered the best type of in vivo tissue engineering. The procedure consists of application of an external fixator to the two ends of the bone to be lengthened or aligned, transverse osteotomy of that bone, followed by gradual and controlled distraction of the bone ends until the desired lengthening is obtained. The whole process consists first of a latency phase of 5 to 7 days following the osteotomy, during which no distraction occurs. This is followed by the distraction phase, where actual distraction is performed until the desired lengthening is obtained. Then follows the consolidation phase, where the fixator is kept in place without distraction, until the newly formed bone in the distracted gap becomes mechanically strong enough so that the external fixator can be safely removed. This consolidation phase is very long—about 1 month for every centimeter lengthened (Ilizarov 1989b; Aronson et al. 1990). This lengthy period during which the external fixator has to be kept can exacerbate or lead to many problems and numerous medical, social, and financial complications to the patient, the family, and the health system (Paley 1990). The question then arises: how to accelerate the consolidation of newly formed bone in the distracted gap so that the length of time the fixator has to be kept in place can be shortened?
To better understand the limiting factors involved in DO and how this bone consolidation phase can be accelerated, we and other groups have extensively studied the cellular and histologic features involved in DO. We have used animal models of DO to understand the expression profile of important molecular pathways involved in DO, including BMP, FGF, IGF, TGF-β, PDGF, and VEGF molecular signaling (Eingartner et al. 1999; Rauch et al. 2000; Haque et al. 2006, 2007; Ai-Aql et al. 2008; Han et al. 2008; Haque et al. 2008).
While the canonical WNT signaling pathway has been recently identified as a critical regulator in modulating bone formation and bone mass (Chen and Alman 2009; Secreto et al. 2009) and even shown to direct osteogenesis during bone healing (Chen et al. 2007a), to our knowledge there are no published data of its role in the context of DO. WNT molecules belong to a family of 19 secreted proteins that are implicated in various biological processes, particularly osteogenesis. In WNT/FZD signaling, WNT proteins bind their membrane receptors, Frizzled (FZD) 1 and 2, and their coreceptors, low-density lipoprotein receptor protein (LRP) 5 or 6 to initiate the canonical WNT signaling cascade. This results in the accumulation and nuclear translocation of β-catenin, which then interacts with other factors (e.g., TCF/LEF) to activate the transcription of WNT downstream target genes (Chen and Alman 2009).
The osteogenic potential of WNT is achieved by inducing the expression of bone-specific genes (e.g., Runx2, Osterix) and by reducing adipogenic genes (e.g., PPARγ, C/EPB), which ultimately causes mesenchymal cells to commit toward the osteoblastogenesis lineage rather than adipogenesis. As an important and critical biological pathway, the balance of WNT signaling is fine-tuned by signal cross talk with other pathways, such as BMP and FGF, and is tightly regulated by negative feedback mechanisms that antagonize WNT ligand–receptor interactions. Such WNT antagonizers include secreted frizzled-related proteins (sFRPs), Dickkopf 1 (DKK1) secreted molecule, and the transcriptional corepressor C-terminal binding proteins (CTBP). sFRPs and DKK1 are soluble proteins that inhibit WNT signaling by directly binding WNT ligands and interfering with WNT/LRP/FZD interactions (Bovolenta et al. 2008), whereas CTBPs are believed, depending on the context, to act as transcriptional corepressor or coactivators by modulating interactions with APC/β-catenin/TCF (Hamada and Bienz 2004; Fang et al. 2006; Bhambhani et al. 2011).
Previous reports have indicated that WNT is involved in both osteogenesis (intramembranous bone formation) and chondrogenesis (endochondral bone formation) but appears to be more powerful in modulating osteogenesis (Day et al. 2005; Hill et al. 2005). We have shown in a mouse model of DO that both mechanisms of bone formation are involved in bone repair and that this dynamic process depends on many factors, specifically the rigidity of the fixator (Haque et al. 2008). Therefore, understanding the temporal and spatial expression of the WNT pathway and characterizing its endogenous role in directing osteogenesis during DO may help pave the way in developing a therapeutic target to enhance bone regeneration during DO. The aim of this study is to describe the immunohistochemical localization of members of the canonical WNT signaling pathway in mouse tibial bone specimens that have undergone limb lengthening in DO. Because of the very large numbers of different molecules involved in the WNT signaling pathway, we have chosen to analyze the temporal and spatial expression of only some of the prototypical components representing agonists (WNT4, WNT10A), antagonists (sFRP1, 2, and 4; DKK1), transcriptional regulators (β-catenin, CTPB1, 2), and receptors/coreceptors (LRP5, 6, FZD1, 2). Some have been selected because they have been identified and studied in the context of fracture healing (WNT4; β-catenin; LRP5, 6; sFRP1; FZD1, 2, 4; DKK1) (Hadjiargyrou et al. 2002; Zhong et al. 2006; Chen et al. 2007a; Bajada et al. 2009; Gaur et al. 2009; Komatsu et al. 2010), while others (WNT10A, CTBPs) have not been studied at all in bone repair and healing.
Materials and Methods
DO was performed on 40 healthy adult female wild-type C57BL/6J mice (Charles River, Montréal, Canada). Mice were all 2–3 months of age, with average weight of 22g. All protocols were approved by the McGill University Animal Care Committee.
Surgery Protocol
The protocols for surgery and distraction were identical to those of our previous studies (Haque et al. 2008; Alam et al. 2009). DO surgery was performed using a miniature Ilizarov fixator on the right tibia of the mice, as described (Tay et al. 1998). Mice were anesthetized using isoflurane and injected with buprenorphine for pain management. Two small pins were transfixed 90° apart in the distal and proximal ends of the tibia using a handheld drill. Two rings were fixed to the pins, and 3 screws connected the rings at the proximal and tibial ends. Transverse osteotomy was then performed on the tibia using a scalpel, and the bones were aligned before suturing the incision.
DO procedure was then divided into three phases: latency, distraction, and consolidation (Fig. 1). After a delay of 5 days postosteotomy to allow for healing of the surrounding inflamed tissue and hematoma (latency phase), distraction was started whereby the tibia was distracted at a rate of 0.2 mm every 12 hr for a period of 12 days (distraction phase). The bone was then allowed to consolidate for 34 days (consolidation phase) where the distractor remained in place but distraction was ceased.
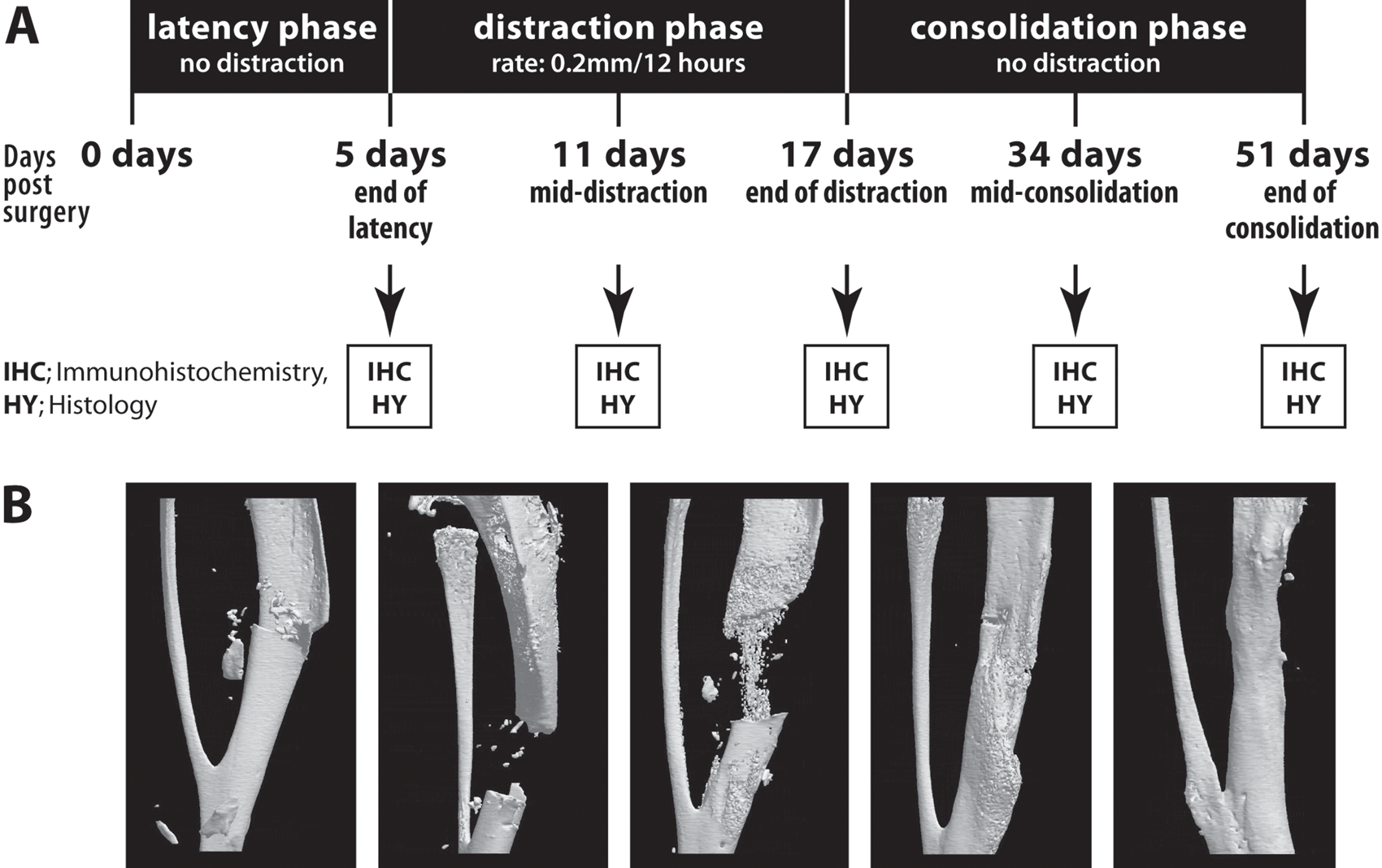
Distraction osteogenesis (DO) surgical procedure and timeline in mouse model. Following osteotomy, the fixator is left in place, and DO is divided into three phases: latency phase (5 days total), distraction phase (days 5 to 17, for a total of 12 days), and consolidation phase (days 17 to 51, for a total of 34 days). (A) Summarizing schema of DO time-course study in mouse model. Tibial specimens were collected and analyzed postosteotomy at 5, 11, 17, 34, and 51 days. (B) Radiologic images of distracted mouse tibiae collected at 5, 11, 17, 34, and 51 days postosteotomy (images obtained from our previously published study, with permission from Bone; Haque et al. 2008).
Sample Collection
Mice were divided into five groups according to a specific time of sacrifice: day 5 (end of latency phase), days 11 and 17 (middle and end of distraction phase), days 34 and 51 (middle and end of consolidation phase). Mice were euthanized; the external fixator device was carefully removed; and the lengthened tibia was dissected. The tibias were then fixed overnight in 4% paraformaldehyde and processed for histologic and immunohistochemical analysis, as described below. For the samples undergoing radiologic analysis, X-ray views of the fixed operated tibiae were taken using a Faxitron X-ray machine (Faxitron MX-20, McGill Center for Bone and Peridontal Research).
Histology
The fixed tibias were embedded in 4.5% methylmethacrylate. Sections of 6 µm were deplastified and stained by Trichrome Goldner for comparative histology. The defined region of interest was the entire distraction gap, and cross-sectional slides were analyzed to distinguish between mineralized and unmineralized tissue. Pictures were taken under various magnifications using a Leica microscope (Leica Microsystems, Richmond Hill, Canada) attached to a Q-Imaging camera (Olympus DP70).
Immunohistochemistry
The fixed tibias were embedded in methylmethacrylate. Sections of 6 µm were deplastified and hydrated, then endogenous peroxidase was blocked with 3% hydrogen peroxide for 10 min. Nonspecific binding was blocked by incubation in 10% normal horse serum in phosphate-buffered saline for 20 min, followed by 15-min incubation with avidin and then 15-min incubation with biotin. For immunostaining, sections were incubated with polyclonal primary goat antibodies (Santa Cruz Biotechnologies, Santa Cruz, CA) in a humidified chamber at 4°C overnight to detect for the various proteins (see supplemental online table): WNT4 (catalog no. sc-5214), WNT10a (sc-69135), FZD1 (sc-30428), FZD2 (sc-68327), LRP5 (sc-21390), LRP6 (sc-17984), CTBP1 (sc-5963), CTBP2 (sc-5967), DKK1 (sc-30785), sFRP1 (sc-7425), sFRP2 (sc-7426), sFRP4 (sc-22498), and β-catenin (sc-1496). Antibodies were used at a final concentration of 2 µg/ml diluted in phosphate-buffered saline with 1% normal horse serum. For the second antibody, a biotinylated horse anti-goat antibody (Vector Labs, Burlington, CA) for 30 min at a dilution of 1:400. Sections were stained using the avidin–biotin complex method (Vector Labs) for 30 min, followed by DAB–peroxidase revelation. Finally, sections were counterstained with Hematoxylin QS and mounted with Permount. For every slide tested, we also performed a negative control by omitting the primary antibody.
Analysis of Immunohistochemistry
While immunohistochemistry is traditionally used as a qualitative assessment of cell and extracellular components that stain positively, we have developed a special technique to semiquantitatively grade positive cell staining. As described by our own laboratory (Rauch et al. 2000; Hamdy et al. 2003; Haque et al. 2006, 2007, 2008) and others (Tavakoli et al. 1999; Yeung et al. 2001), we have used this grading technique in the present study to evaluate positively stained cells. The number of cells stained positive and expressing the various proteins is evaluated by cell counting, as a percentage of total cells. Each immunostained section was graded by a blind observer, with the following scores for signal intensity and percentage of positively stained cells: –, no staining in the majority of cells; +, staining in less than 25% of cells; + +, staining in 25%−50% of cells; + + +, staining in 50%−75% of cells; + + + +, staining in more than 75% of cells. The analyses were performed for the callus and center of the distracted zone. Chondrocytes and fibroblastic cells were identified morphologically. Osteoblastic cells were very low in number and/or showed very weak signal intensity in immunostaining, specifically in the distraction phase, and were therefore not taken into account for immunohistochemical grading.
Results
To assess the bone composition and WNT expression profile of the distracted zone at different times of de novo bone formation, distracted tibiae were collected at various intervals for histologic and immunohistochemical analysis (see Fig. 1 for schema of DO timeline).
First, histologic sections of bone specimens stained with Goldner-Trichrome revealed the cellular and mineral composition of the distracted tibiae at different times of bone formation (Fig. 2). At day 5 postosteotomy (end of latency), sections revealed mostly hematoma forming at the osteotomy site with little to no callus or calcified tissue (Fig. 2, top left panel, Day 5). By distraction days 11 and 17, early bridging of the osteotomized site occurred with formation of soft callus and cartilage tissue and with the presence of chondrocytes and fibroblastic cells. These constituents correspond mostly to early endochondral ossification. As mentioned earlier, bone repair during DO involves a dynamic interplay of intramembranous and endochondral bone formation and so results in different cell types in the distracted site: mesenchymal cells, chondrocytes, fibroblasts, chondroblast-like cells, and, later in osteogenesis, osteoblasts and osteocytes. As shown in Fig. 2 at days 17 and 34, chondrocytes and elongated fibroblastic cells were identified morphologically in the distracted gap and appeared to be growing in the direction of distraction (bottom panel, white arrows). By the consolidation phase, bridging of the distal and proximal tibial bone fragments took place, with visible bone and calcified tissue deposition in the distracted gap (Fig. 2, green-stained mineralized patterns at days 34 and 51).
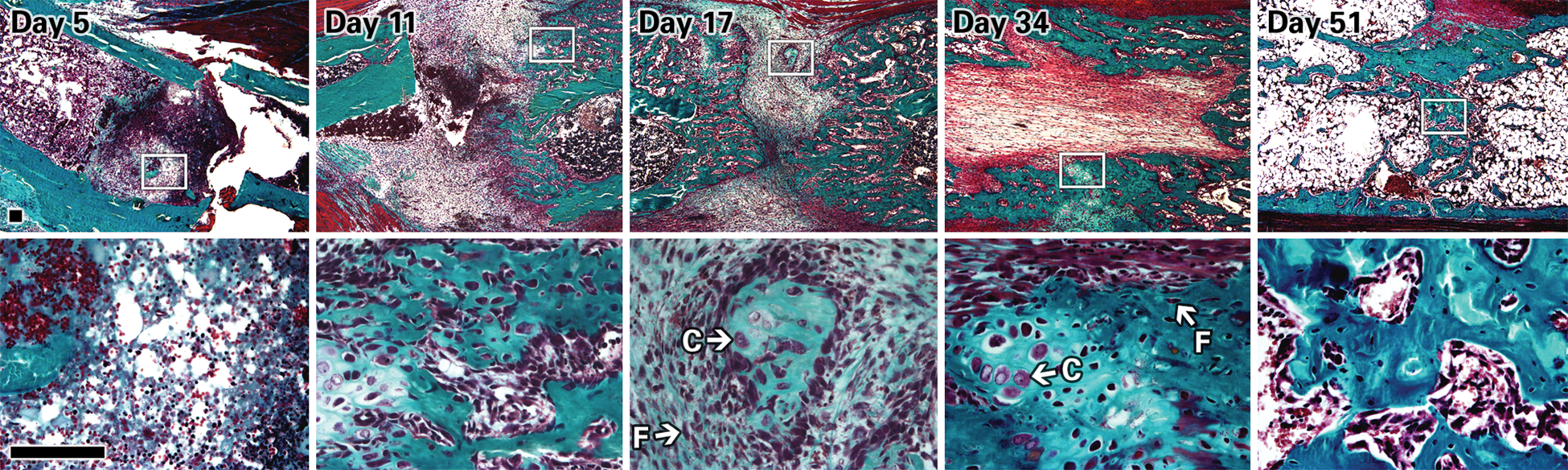
Histology images (5× top row, 40× lower row) of distracted tibiae sections stained with Goldner-Trichrome at days 5, 11, 17, 34, and 51. Images were taken at the center of the callus area. Chondrocytes and fibroblastic cells can be identified morphologically, as indicated by the white arrows and letters C and F, respectively. At day 5 postosteotomy, no bone or cartilage has formed in the distracted gap between the bone fragments (green segments). At day 11, there is soft callus and cartilage formation (red and purple area), and by day 17, the start of bone bridging and union can be seen. At days 34 and 51, there is calcified tissue and matrix deposition (green-stained patterns) in the distracted gap. Scale bar = 100 μm.
Next, immunostaining was performed in the distracted gap for proteins involved in the WNT signaling pathway, including WNT4 and WNT10a ligands; receptors FZD1, FZD2, LRP5, LRP6; and WNT antagonists CTBP1, CTBP2, sFRP 1, sFRP2, sFRP4, DKK1. Overall, immunostaining for most of the molecules analyzed showed an intense signal during the distraction phase (day 17). Figures 3, 4, and 5 show representative images of tibial sections that stained positive for WNT ligands, receptors, and antagonists, respectively, and at three time points of DO (days 17, 34, and 51). A summary of the immunohistochemistry grading is shown in Fig. 6.
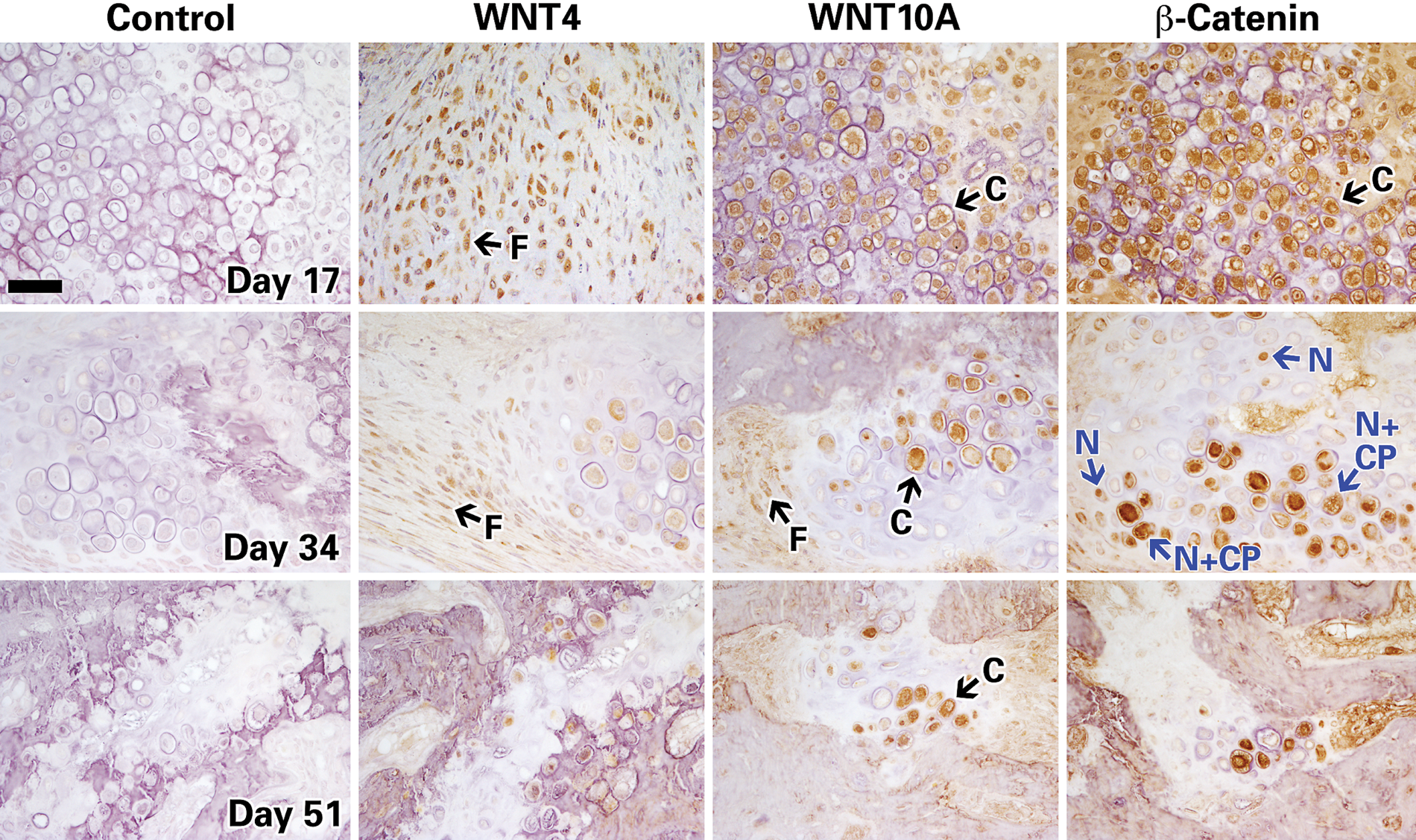
Histochemistry images of distracted mouse tibiae immunostained for positive regulators of WNT pathway (WNT4, WNT10a, and β-catenin) at days 17, 34, and 51. Representative images taken at 40×; scale bar represents 100μm. Chondrocytes and fibroblastic cells are indicated by the black arrows and letters C and F, respectively. In the middle-row panel of β-catenin, the blue arrows and N indicate nuclear staining, whereas N + CP indicates both nuclear and cytoplasmic staining.
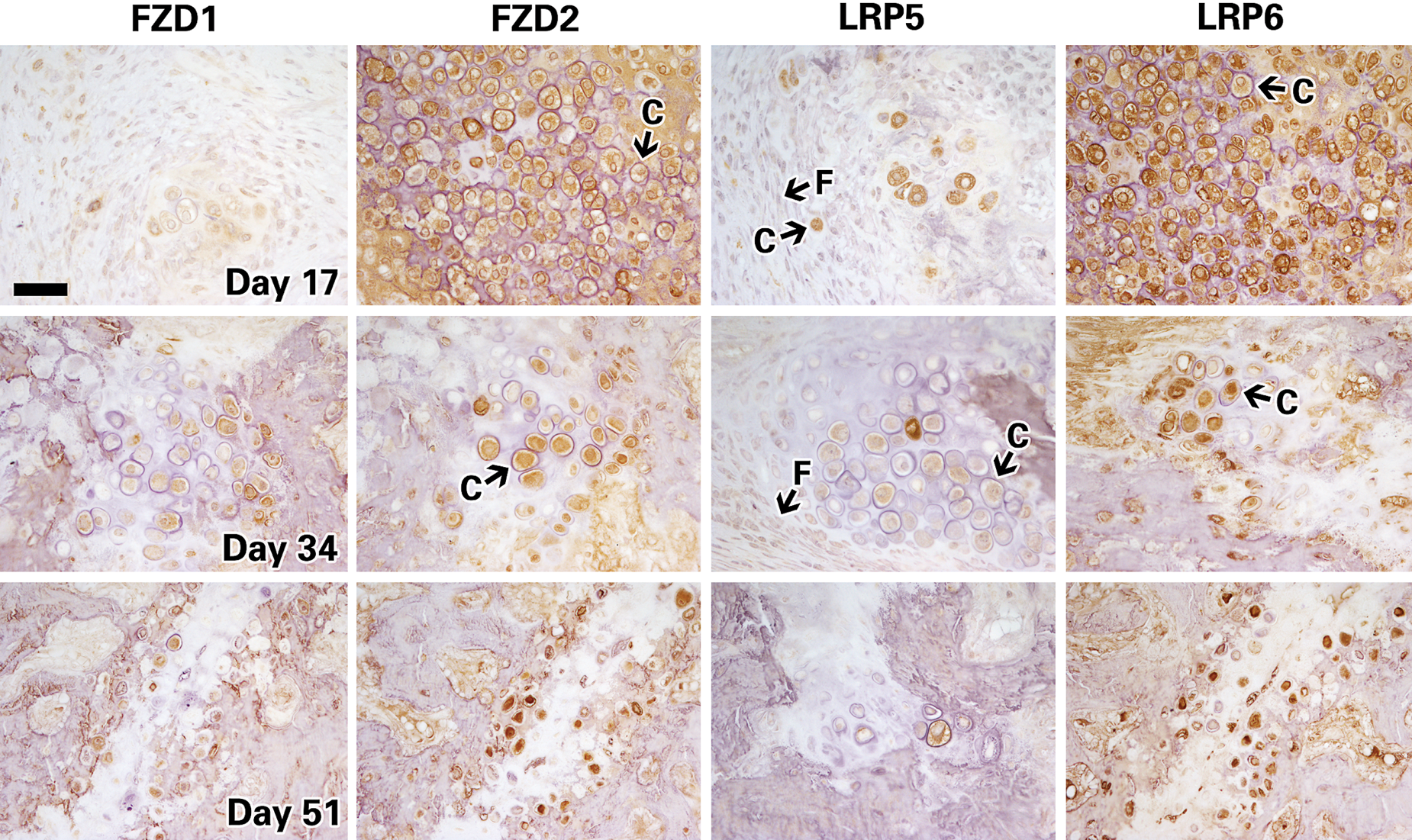
Histochemistry images of distracted mouse tibiae immunostained for receptors (FZD1 and 2) and coreceptors (LRP5 and 6) of the WNT pathway at days 17, 34, and 51. Chondrocytes and fibroblastic cells are indicated by the black arrows and letters C and F, respectively. Representative images taken at 40×; scale bar represents 100μm.
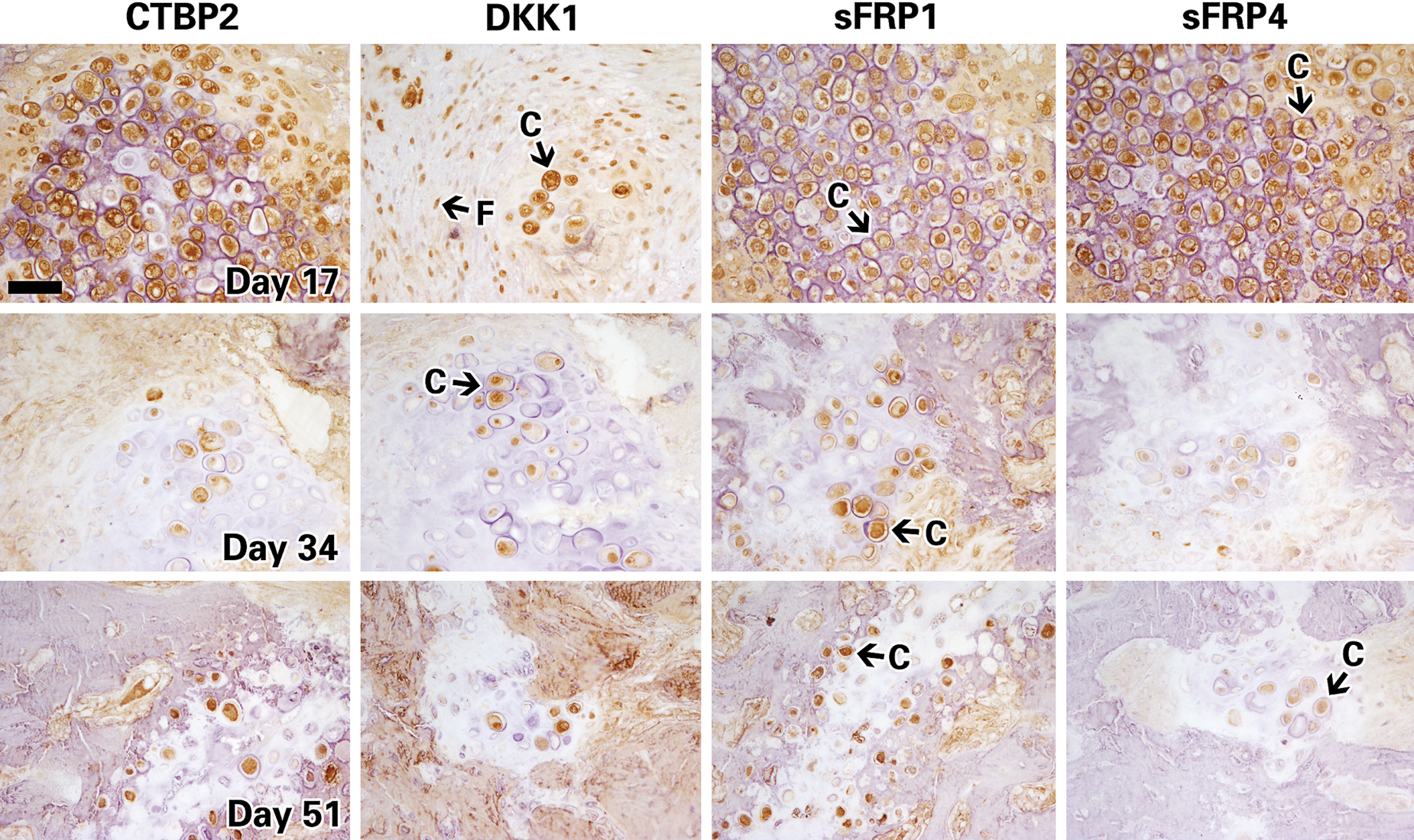
Histochemistry images of distracted mouse tibiae immunostained for known inhibitors of the WNT pathway (CTBP2, DKK1, sFRP1, and sFRP4) at days 17, 34, and 51. Chondrocytes and fibroblastic cells are indicated by the black arrows and letters C and F, respectively. Representative images taken at 40×; scale bar represents 100μm.
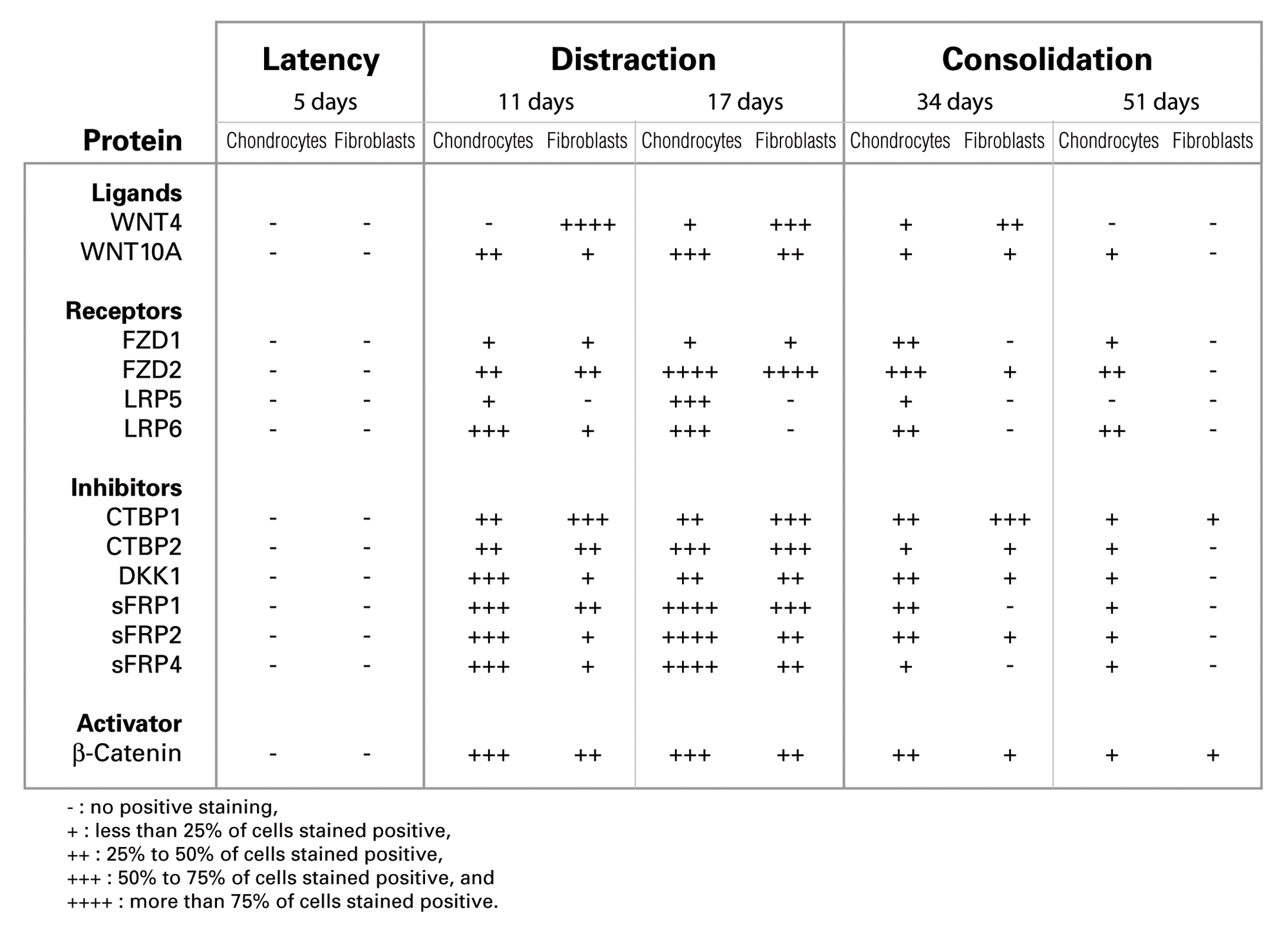
Immunohistochemistry results of WNT signaling protein expression in the distracted zone.
While most of the proteins peaked in expression at day 17, their expression progressively decreased at later stages of bone and callus repair during middle and end of bone consolidation (days 34 and 51). Specifically, expression of WNT ligands (4 and 10a), receptors (FZD2 and LRP6), inhibitors (CTBP1, 2; DKK1; sFRP1, 2, and 4), and β-catenin were detected at high levels during the distraction phase at days 11 and 17 (summarized in Fig. 6). By mid and end bone consolidation (days 34 and 51), the expression of these same proteins was greatly reduced. Peak expression of WNT10A and FZD2 was detected at 17 days, then reduced dramatically by 34 and 51 days, respectively.
The expression of β-catenin, the most important downstream target of the WNT pathway, was upregulated at days 11 and 17, particularly in chondrocytes whereby 50%−75% cells stain positive, compared to 25%−50% in fibroblasts (as summarized in Fig. 6). However, during the bone consolidation phase at days 34 and 51, β-catenin expression was downregulated in fibroblasts and chondrocytes, mostly to less than 25% positive cell staining (Figs. 3 and 6). Furthermore, β-catenin was detected almost exclusively intracellularly, in which some cells stained positive in the nucleus only and some in the cytoplasm and nucleus both (Fig. 3, indicated with blue arrows in β-catenin panel). This localized expression pattern concurs with β-catenin’s accepted status as an intracellular and cytoplasmic protein, which upon activation by the canonical WNT cascade, translocates to the nucleus (Hino et al. 2005; Chen et al. 2007a).
Note that the subcellular localization may not be as evident for some proteins in the immunodetection images. For instance, immunoimages of known extracellular soluble proteins such as WNT4, WNT10A (Fig. 3), and DKK1 (Fig. 4) may give the impression of intracellular/nuclear staining, but this may be a result of a limitation of the immunohistochemical technique (Walker 2006), which represents in two dimensions what is really occurring in three dimensions.
Overall, our immunohistochemical findings of proteins in the WNT pathway show that the proteins are upregulated specifically during the early stages of distraction (days 11 and up to day 17), then downregulated at later stages of bone formation and calcification (days 34 and 51). Interestingly, the same temporal expression profile was observed in inhibitors of WNT—namely, CTBP1 and 2; DKK1; and sFRP1, 2, and 4. These proteins exhibited maximal expression at day 11 and 17 in chondrocytes and fibroblasts, with dramatic decrease in expression at days 34 and 51 (as shown in Fig. 6).
While expression of most proteins was detected in similar levels in fibroblasts and chondrocytes, a difference in signal intensity was sometimes observed between these cells. For instance, the WNT4 ligand was maximally expressed in more than 75% of fibroblasts at day 11, when no positive staining was detected in chondrocytes at this same time interval. Conversely, the sFRP4 inhibitor was detected in 50%−75% of fibroblasts and in less than 25% of chondrocytes at day 11. Therefore, modulation of expression of some WNT molecules can vary between chondrocytes and fibroblasts even within the same distracted area, which indicates that the expression of WNT molecules may be contingent on cell type.
Discussion
It is well established that activation of the WNT/FZD pathway is associated with increased bone formation, density, and volume (Zhong et al. 2006; Liu et al. 2009; Augello and De Bari 2010 ). We postulated that because the WNT pathway is critical for bone formation during bone development and fracture healing, it may be equally essential for bone repair during DO.
Indeed, this time-course immunohistochemical study revealed that the WNT pathway is at maximal activation during the distraction phase in mouse tibiae, at the time when de novo bone is constantly being generated. Our findings also show that chondrocytes and fibroblasts express increased WNT signaling molecules at the proliferative stage when early de novo bone is being formed (mid distraction day 17), then decrease in expression later, when the bone has consolidated (day 34 and 51). This maximal activation of the WNT pathway was observed in known positive regulators of the pathway—namely, WNT4, WNT10A, β-catenin, FZD1 and 2, LRP5 and 6—and negative regulators alike—namely, CTBP1 and 2; DKK1; and sFRP1, 2, and 4.
Our findings show a preponderance of chondrocytes during the early stages of DO (day 11 and 17), which coincided with an upregulation of WNT signaling. These findings are in agreement with other studies, as reviewed by Deschaseaux et al. (2009). The authors showed that a high level of WNT molecules can favor chondrocytic proliferation instead of osteogenic differentiation and that the WNT/β-catenin pathway is activated in committed progenitors in the early stages of chondrogenesis and osteogenesis, then downregulated with osteoblastic maturation (Chen et al. 2007a, 2007b).
Furthermore, we found a dramatic upregulation in all inhibitors of WNT members studied during mid and end distraction: CTBP1 and 2; DKK1; and sFRP1, 2, and 4. Our results show that expression of negative regulators of WNT are increased almost concurrently, at mid and end distraction, with that of positive regulators of WNT/FZD (see Fig. 6). In the case of our murine DO model, the osteogenic WNT/FZD pathway is likely stimulated due to the distraction and mechanical strain on the bone. As a result, the endogenous negative feedback machinery “kicks in” to induce WNT inhibitors, to regulate the activated WNT/FZD pathway. This would explain the timely increase in WNT inhibitors during distraction, then likely acting to downregulate positive activators of WNT/FZD, as observed later during consolidation. Other authors have reported similar findings of WNT antagonists during osteogenesis—namely, sFRPs (Boland et al. 2004), DKK1 (Liu et al. 2009), and Sclerostin by osteoblasts/osteocytes (Winkler et al. 2003). Interestingly, the presence of specific inhibitors of WNT (e.g., DKK1) has also been shown to potentiate osteogenesis by decreasing WNT signaling (Jones et al. 2002; Liu et al. 2009). These cellular events seem to align with the mechanistic principle of “more WNT = early chondrogenesis and osteogenesis” and “less WNT = osteoblast maturation and differentiation.”
Our findings in a mouse model of DO provide further evidence that endogenous WNT inhibitors tightly regulate the balance of WNT pathway during osteogenesis and outline the significance of endogenous negative regulators of WNT in maintaining the WNT signaling pathway in tight homeostasis. In fact, secreted WNTs have been shown to have the capacity to induce the self-renewal of vicinal mesenchymal stem cells in vivo (Liu et al. 2009), which suggests that in the absence of negative regulators, WNT could potentiate bone overgrowth.
Specifically regarding LRP5 and LRP6 staining, our results revealed two very interesting findings: there was a much stronger signal detected for LRP6 compared to LRP5 and a decreased expression of LRP5 but not LRP6 after the mechanical forces of distraction are stopped. Evidence in the literature indicates that LRP6 appears a stronger signaling receptor and possesses a more prominent role than LRP5 in WNT signal transduction (Macdonald et al. 2011), although genetic studies have shown that they have redundant roles during skeletal development (Joeng et al. 2011). However, fracture repair in LRP5 knockout mice was clearly delayed (Komatsu et al. 2010), implying that at least LRP5 is important for this process. Whether LRP6 would contribute to a greater extent than LRP5 to the regeneration events as observed here in DO is still speculative and will require further corroborative evidence. In addition, we and others have shown that the mechanical forces applied during the distraction phase trigger a cascade of events that lead to upregulation of several osteogenic and angiogenic signaling pathways and the expression of numerous cytokines and growth factors. Transient upregulation of LRP5 during the mechanotransduction phases could provide the initial cues during DO, while LRP6 would provide the long-lasting effects. Osteogenic responses upon a mechanical load were almost totally abrogated in the LRP5 knockout mice (Sawakami et al. 2006), suggesting that LRP5 is essential for mature osteoblasts activity. Thus, it is still unclear whether LRP5 and LRP6 could have different or overlapping roles during different phases of DO.
In the search for therapeutic strategies and regenerative medicine for accelerating bone regeneration in DO, it is important to outline the precise role of the osteogenic WNT/FZD/β-catenin pathway in the different phases of DO. As such, this study is very useful as it delimits when and where endogenous WNT molecules are switched on as a result of distraction strain. A major strength of the study is that the time-course expression profile of WNT signaling is examined at the protein level in a well-established in vivo model of DO. As in our study, Zhong et al. (2006) found that in a rat model of fracture repair, expression levels of WNT signaling members (i.e., FZD2, WNT4, and LRP5, β-catenin) were increased as detected by RT-PCR. Therefore, our findings align with previous reports showing that the spatial and temporal localization of WNT signaling is increased at the site of injury and during the early stages of osteogenesis, due to distraction strain. Because there is a clinical need to accelerate bone consolidation during DO, future studies should focus on whether manipulating the WNT/FZD pathway during early and mid distraction can have a positive effect on bone repair during DO.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received financial support from the Shriners of North America.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
