Abstract
The impact of progesterone on neuronal tissues in the central (CNS) and peripheral (PNS) nervous system is of significant scientific and therapeutic interest. Glial and neuronal cells of vertebrates express steroidogenic enzymes, and are able to synthesize progesterone de novo from cholesterol. Progesterone is described to have neuroprotective, neuroreparative, anti-degenerative, and anti-apoptotic effects in the CNS and the PNS. Thus, the first clinical studies promise new therapeutic options using progesterone in the treatment of patients with traumatic brain injury. Additionally, experimental data from different animal models suggest further positive effects of progesterone on neurological diseases such as cerebral ischemia, peripheral nerve injury and amyothropic lateral sclerosis. In regard to this future clinical use of progesterone, we discuss in this review the underlying physiological principles of progesterone effects in neuronal tissues. Mechanisms leading to morphological reorganizations of neurons in the CNS and PNS affected by progesterone are addressed, with special focus on the actin cytoskeleton. Furthermore, new aspects of a progesterone-dependent regulation of neurosteroidogenesis mediated by the recently described progesterone binding protein PGRMC1 in the nervous system are discussed.
Keywords
Introduction
Progesterone was first isolated from the ovaries of female rats in 1935. Since then, the scientific interest in this hormone has been constantly high. Progesterone is a C21-steroid hormone that plays an elementary role in the female menstruation cycle and is essential for the establishment and maintenance of pregnancy. Nevertheless, gestagenes such as progesterone are also produced in male organisms. Since the beginning of the 1990’s, the already heightened interest in progesterone continued to increase when Baulieu and Robel (1990) first described that steroidogenic enzymes are also expressed in the central nervous system (CNS). In the following years, different laboratories were able to show that both neuronal and glial cells in the CNS and peripheral nervous system (PNS) are able to synthesize progesterone from cholesterol (Mellon et al. 1993; Compagnone et al. 1995; Guennoun et al. 1997; Furukawa et al. 1998; Tsutsui et al. 2000). From that time, neuronal-derived steroids were termed neurosteroids, which continue to be of high scientific and therapeutic interest.
Previous research had provided evidence for the high significant physiological function of progesterone in the nervous system. Furthermore, progesterone might also be an effective option in the therapy against different severe neurological diseases (Deutsch et al. 2013). Indeed, numerous preclinical and early clinical studies have revealed the protective effects of progesterone in patients with traumatic brain injury (TBI) (Roof et al. 1994; Djebaili et al. 2004, 2005; Wright et al. 2007; Xiao et al. 2008). TBI is a common and severe disorder and the up-to-date therapy for patients with TBI cannot adequately reduce morbidity and mortality rates; thus, progesterone represents a new promising approach in TBI therapy. Additionally, experimental data from various animal models suggest further benefits of progesterone against other neurological disorders. Progesterone might also be a potential therapeutic option for treating apoplexies, as the post-ischemic application of progesterone leads to a better outcome in different rat models (Sayeed et al. 2007; Ishrat et al. 2009; Wang et al. 2010). Furthermore, the anti-inflammatory protective effects of progesterone in animal models of autoimmune encephalomyelitis might suggest a future role for the hormone for patients with multiple sclerosis (Garay et al. 2007, 2008, 2009; Yates et al. 2010; Yu et al. 2010; Giatti et al. 2012).
Thus, progesterone seems to be a promising option in the therapy of a variety of neurological disorders. With regards to the molecular mechanisms that underlie the physiological and pathological effects and functions of progesterone in the nervous system, research is still in its early stages. This review provides an overview of the current scientific knowledge about progesterone effects and the underlying molecular mechanisms in cerebellar Purkinje cells of the CNS (Fig. 1) and dorsal root ganglia neurons of the PNS (Fig. 2), with a special focus on its influence on neurosteroidogenesis and the modulation of the actin cytoskeleton (Fig. 3).
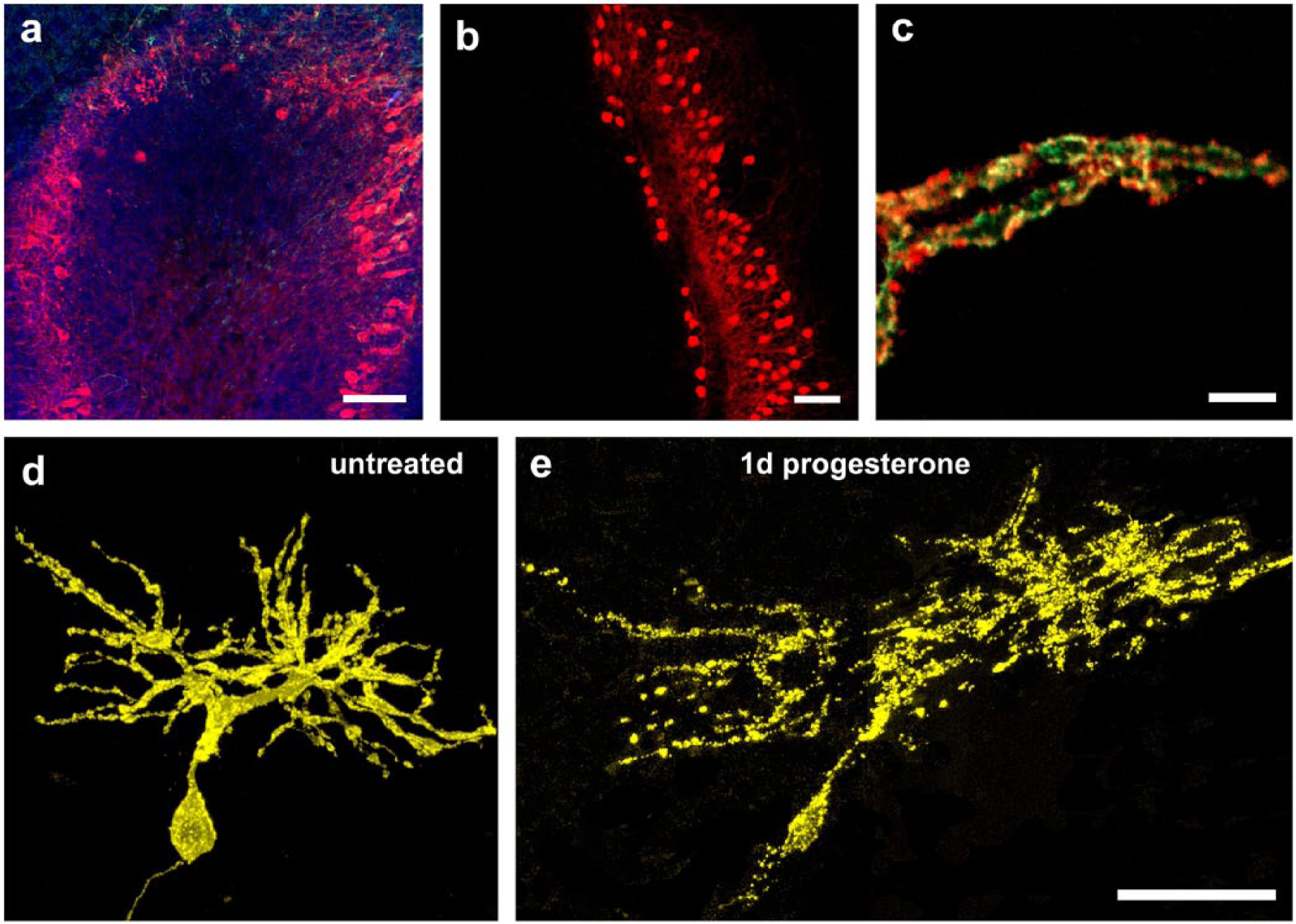
(a) The three-layered cortex structure of the cerebellum, with Calbindin- (red) labeled Purkinje cell bodies located in the Purkinje cell layer, and their dendritic arbors, which extend to the outer molecular layer of the cerebellar cortex. The inner granule layer is characterized by incoming neuronal axons (green) and large numbers of granule cell nuclei (blue). (b) Purkinje cell bodies are linearly arranged within the Purkinje cell layer. (c) Dendritic spines (green, pEYFP-actin transfected) of Purkinje cells are co-localized with PSD95-labeled functional synapses (red, anti-PSD95-AK, IHC). (d) Untreated Purkinje cell [15 days in vitro, (div)] at a time point of 1 day after transfection with pEYFP-actin (yellow). (e) The same Purkinje cell after progesterone treatment for 1 day. Progesterone significantly increased the dendritic length and cell area of Purkinje cells within 1 day of cultivation. Scale, (a, b) 100 µm; (c) 5 µm; (d, e) 50 µm.
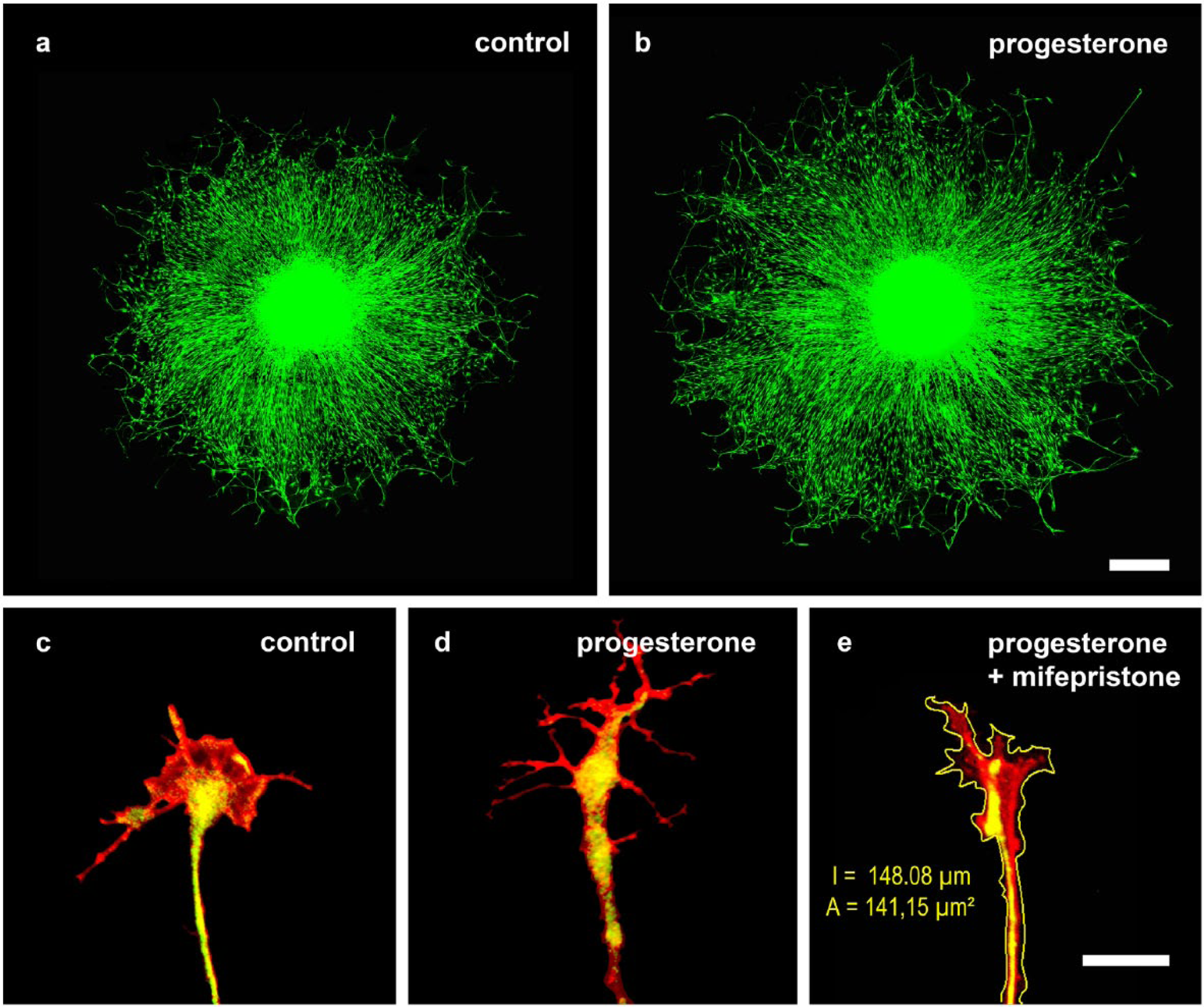
Immunostaining of heavy neurofilaments (NF-H) in chicken dorsal root ganglia using confocal laser scanning microscopy after cultivation for 3 days in (a) nutrient medium lacking any stimulating factor and (b) with addition of 10 nM progesterone. Progesterone treatment was followed by a significantly enhanced outgrowth and crosslinking. Scale bar: 500 µm. Changes in cytoskeletal properties within the neuronal growth cone show similar variances. (c) Controls showed typical distribution of neurofilaments (anti-NF-M-FITC) and actin (phalloidin-TRITC). Incubation with progesterone (d) led to a distinct enlargement of the peripheral zone, with additional lamellipodia and filopodia. (e) Morphological analysis to confirm the staining, measuring the area and circumference starting from the base of the neuronal growth cone, which is defined as the maximum distance perpendicular to the axonal axis measured at 2 µm from the beginning of the axonal enlargement forming the growth cone. Scale, 5 µm.
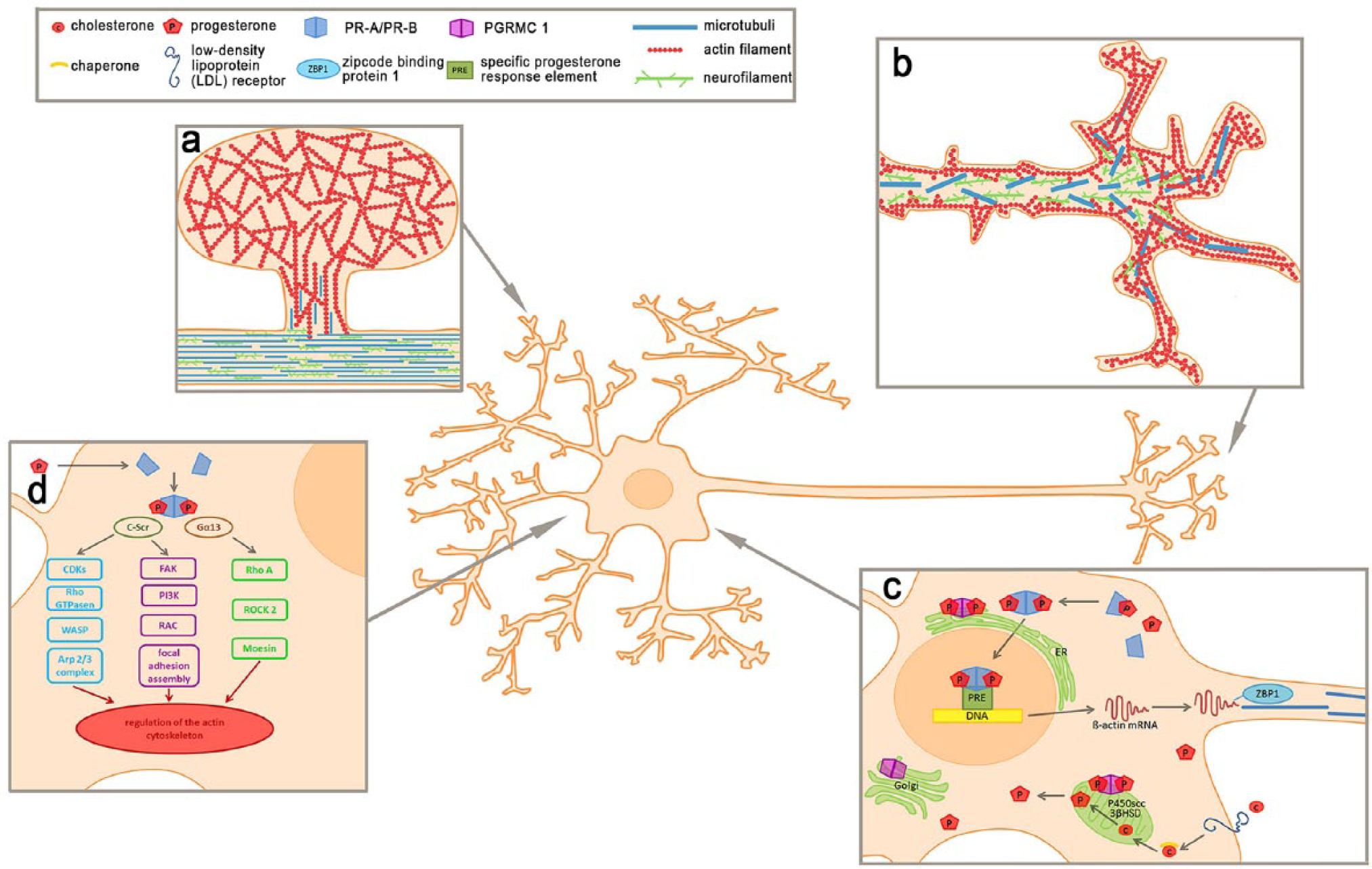
Signaling pathways within the neuronal growth cone of DRG neurons. Neurons were shown to undergo cytoskeletal rearrangements especially within the neuronal growth cones and (a) spine due to progesterone treatment. Spines are mainly composed of actin units. Whereas monomer G-Actin is mostly located in the periphery, polymeric F-actin can be found in the center. In contrast, the cytoskeleton of growth cones (b) is classically divided into three domains based on the distribution of cytoskeletal proteins. The central domain is mainly characterized by neurofilaments and microtubules, reaching through the arc of actomyosin, representing the transition zone, into the peripheral zone build of actin filaments. (c) The underlying mechanism of cytoskeletal reorganization after progesterone incubation includes genomic, long-term effects following activation of the classic progesterone receptors A and B. These progesterone receptors undergo conformational changes after progesterone binding. Thereupon, the receptors dissociate from chaperone proteins, dimerize, translocate into the nucleus, and interact with specific progesterone response elements (PRE) to modulate the transcription of target genes involved in dendritogenesis, spinogenesis, and synaptogenesis. Contrarily, PGRMC1 is mainly known to be part of the regulation of neurosteroidogenesis, the cerebellar maturation process, and neuroprotective mechanisms. It is associated with membranes of different cell organelles, including the Golgi-apparatus, endoplasmic reticulum and mitochondria. (d) For short-term effects of progesterone, various interactions of sex steroids with different, non-classical signaling cascades have been described, directly activating kinase cascades within the cytoplasm or at the cell membrane. Both signaling pathways might lead to changes in the cytoskeletal composition of the neuronal growth cones following progesterone incubation.
Progesterone Effects in Immature Purkinje Cells Are Mediated through Classical Progesterone Receptors
Purkinje cells, named after Jan Evangelista Purkynĕ, are large GABAergic neurons, located in the cortex of the cerebellum (Fig. 1a), which is divided into three layers: the outer, high-fiber molecular layer, the Purkinje cell layer and the inner granule layer. The Purkinje cell bodies are 50–70 µm in size and characteristically reveal a linear arrangement in the Purkinje cell layer (Fig. 1b). The stem dendrites arise from the Purkinje cell body and usually divide into two or three main dendrites, which branch into numerous smaller secondary and tertiary dendrites (Fig. 1). The emergent dendritic arbor is completely covered with dendritic spines, which represent the postsynaptic component of excitatory and inhibitory synapses (Fig. 1c, Fig. 3a) and are therefore crucial for the proper development and function of both immature and mature Purkinje cells.
Purkinje cells are subjected to a complex process of maturation, which starts prenatally and proceeds into the late postnatal period. Simultaneously, these cells were shown to express the steroidogenic enzymes cholesterol side-chain cleavage enzyme (commonly referred to as P450scc) as well as 3-β-hydroxysteroid dehydrogenase (3β-HSD) (Do Rego et al. 2009) and are able to synthesize progesterone de novo from cholesterol (Ukena et al. 1998, 1999) (Fig. 3c). As the synthesis of progesterone in the cerebellum seems to be restricted to neonatal Purkinje cells, it is well justified to consider that progesterone is probably involved in the development of a precise cerebellar circuit (Ukena et al. 1998, 1999). Indeed, application of progesterone to neonatal Purkinje cells was shown to induce an increase in the formation and development of dendritic spines (spinogenesis), synapses (synaptogenesis) and dendrites (dendritogenesis) in vitro and in vivo (Sakamoto et al. 2001, 2002, Wessel et al. 2014). Furthermore, our recent study confirmed these findings by directly visualizing—for the first time—a significant increase in dendritic length and dendritic area in the same Purkinje cells before and after stimulation with progesterone for 24 hours (Fig. 1d, 1e). Single Purkinje cells within the complex cerebellar tissue of rollertube slice cultures were specifically transfected with the aid of microinjection (pEYFP-actin) to allow for the non-overlapping imaging of these cells (Fig. 1d, 1e) (Wessel et al. 2014).
In different experimental studies, the aforementioned effects of progesterone on spinogenesis and dendritogenesis in Purkinje cells were absent when progesterone application was combined with mifepristone (RU486) (Sakamoto et al. 2001, 2002; Wessel et al. 2014). Even though the anti-progesterone activities of mifepristone are well-known and established aspects of clinically therapy, mifepristone has also been shown to act as an agonist for classical progesterone receptors under specific cellular conditions (Cadepond et al. 1997; Liu et al. 2002) dependent on the cellular bioavailability of certain co-activators and co-repressors (Meyer et al. 1990; Katzenellenbogen and Katzenellenbogen 2002). For this reason, mifepristone is now more appropriately referred to as a selective progesterone receptor modulator (SPRM) (Leonhardt et al. 2003). In addition to its interaction with progesterone receptors, mifepristone has anti-glucocorticosteroid activity (Bertagna et al. 1984; Gaillard et al. 1984; Cadepond et al. 1997). Indeed, both glucocorticoid (GR) and mineralocorticoid (MR) receptors are expressed in developing as well as in adult Purkinje cells (Ahima et al. 1992; Lawson et al. 1992). Nevertheless, in the cerebellum, the anti-progesterone functions of mifepristone compensate for the inhibition of glucocorticosteroid receptors as in Purkinje cells. It was also demonstrated that mifepristone-induced inhibition of GR is dose-dependent, thus GR are probably not affected by the single dose used of 2 μM mifepristone (Cadepond et al. 1997). Furthemore, PCR-based analyses have demonstrated an increase in the amount of classical progesterone receptor-mRNA subsequent to progesterone treatment (Wessel et al. 2014). Therefore, it can be assumed that progesterone induces its effects on spinogenesis and dendritogenesis in immature Purkinje cells via classical progesterone receptors.
Indeed, PCR and western blot analyses confirm the expression of classical progesterone receptors in neonatal as well as in mature Purkinje cells (Sakamoto et al. 2003; Wessel et al. 2014). The so-called classical progesterone receptors were first described in the 1970’s (O’Malley et al. 1970). These nuclear receptors are defined as ligand-activated transcription factors and are localized in the cytoplasm, linked to chaperone molecules (Smith and Toft 1993). After binding of progesterone to the receptors, they dissociate from the chaperone molecules and dimerize. Following dimerization, the complexes translocate to the nucleus and modulate transcription through their interaction with specific progesterone-response elements (Tsai and O’Malley 1994; Leonhardt et al. 2003; DeMarzo et al. 1991; Edwards et al. 1991) (Fig. 3c).
Through these genomic mechanisms, classic progesterone receptors are likely to induce the observed progesterone effects on dendritogenesis and spinogenesis. Taken together, the observed induction of cerebellar development induced by progesterone might implicate promising options for the clinical use of progesterone in patients with traumatic, degenerative or ischemic deficits in the cerebellum.
Unfortunately, though, recent data indicate that progesterone effects attributed to the classic progesterone receptors seem to be restricted to the neonatal period (Wessel et al. 2014). Thus, a recent investigation of changes in the dendritic spine density in neonatal [7 days in vitro (div)], juvenile (15 div) and matured (30 div) Purkinje cells after incubation with progesterone confirm that progesterone indeed induces an increase in the number of dendritic spines in immature and young Purkinje cells. But, interestingly, these new data strongly indicate that, in mature Purkinje cells, the effects of progesterone are no longer detectable (Wessel et al. 2014). Furthermore, incubation with mifepristone or mifepristone plus progesterone failed to show measurable effects on dendritic length or dendritic spine density in mature Purkinje cells (Wessel et al. 2014). Thus, progesterone seems to induce neuronal growth in immature Purkinje cells by interaction with classical progesterone receptors, without affecting mature Purkinje cells. These results implicate that progesterone might be a therapeutic option for children with cerebellar disorders; in contrast, the simple application of progesterone might not be helpful in the therapy of cerebellar diseases in adults. Here, new perceptions will probably be brought about by the US-American PECARN-Study, which concerns progesterone application to children with TBI and is currently in the planning phase.
PGRMC1 Induces Spinogenesis Even in Mature Purkinje Cells
Other studies have demonstrated that progesterone effects are also mediated by another progesterone binding protein, and that progesterone effects in Purkinje cells attributed to this receptor may continue into adulthood. The expression of this different type of progesterone receptor class was shown in the cerebellum over the past decade (Sakamoto et al. 2004, 2008). Progesterone receptor membrane component 1 (PGRMC1) is a progesterone binding protein associated with the membranes of the endoplasmic reticulum, the Golgi apparatus and mitochondria (Selmin et al. 1996; Sakamoto et al. 2004; Xu et al. 2011) (Fig. 3c). Its exact mechanism of action is still unclear, but various studies have indicated an interaction with steroidogenesis (Hughes et al. 2007; Brinton et al. 2008; Rohe et al. 2009). Indeed, current results strongly suggest that PGRMC1 directly interacts with the production of neurosteroids in Purkinje cells (Wessel et al. 2014). Other research in different neuronal tissues revealed that increased PRGMC1 activation in response to progesterone leads to an increase in receptor expression. Progesterone-induced up-regulation of PGRMC1 mRNA was observed after a spinal cord injury in rats (Labombarda et al. 2003), whereas PGRMC1 expression was significantly lowered in the hypothalamic tissue of mice harboring a double knockout of progesterone receptor A and B (Krebs et al. 2000). In the hippocampus, an increase in PGRMC1 was induced by cyclic progesterone exposure (Zhao et al. 2012), and we, too, described a similar progesterone-dependent up-regulation of PGRMC1 in Purkinje cells (Wessel et al. 2014). Not only did we observe an increase in PRGMC1 protein but also an increase in dendritic spine density when classical progesterone receptors were antagonized by mifepristone for more than 72 hours (mifepristone long-time incubation, MLTI) (Wessel et al. 2014). Even though the accompanied increase in the amount of PGRMC1 strongly indicates that the increase in dendritic spine density after MLTI is mediated by endogenous progesterone via PGRMC1, it is important to note that mifepristone itself can have a protective effect on different neuronal cells in experimental studies (Behl et al. 1997; McCullers et al. 2002; Ghoumari et al. 2003; Rakotomamonjy et al. 2011). Mifepristone significantly improves Purkinje cell survival in cerebellar roller tube cultures because of its influence on neuronal depolarization, which is independent of the classical progesterone receptors (Ghoumari et al. 2003, 2006; Rakotomamonjy et al. 2011). Nevertheless, none of these studies have demonstrated an influence of mifepristone on Purkinje cell morphology and dendritic spine density; but they have demonstrated an increase in primary Purkinje cell survival during the critical postnatal week (Ghoumari et al. 2003, 2006; Rakotomamonjy et al. 2011). In contrast, MLTI was shown to increase the dendritic spine density and area without influencing Purkinje cell survival (Wessel et al. 2014); the slice cultures used were from 10-day pups in order to avoid apoptosis in cerebellar slice cultures during the first postnatal days. Given that (i) mifepristone incubation started after the critical period of apoptosis; (ii) the PGRMC1 amount was shown to increase under MLTI; (iii) Purkinje cell morphology was affected; and (iv) these effects were absent when PGRMC1 was blocked, it is well-justified to consider that endogenous progesterone and PGRMC1 are critical for the increase in dendritic spine density and area induced by MLTI.
Thus, classical progesterone receptors and PGRMC1 probably compete for progesterone. If the classical progesterone receptors are (i) absent (Krebs et al. 2000) or (ii) blocked by mifepristone for a longer period (Wessel et al. 2014) or (iii) if PGRMC1 expression is increased through different mechanisms (Labombarda et al. 2003; Zhao et al. 2012), progesterone binding to PGRMC1 might be enhanced. This hypothesis is confirmed by the fact that the increase in dendritic spine density following MLTI in Purkinje cells was absent when MLTI incubation was combined with the PGRMC1 receptor blocker (AG 205). Thus, progesterone probably mediates its effects on the development, function and morphology of Purkinje cells not only through the classic progesterone receptorsbut also through PGRMC1.
Others have shown that PGRMC1 is involved with neurosteroidogenesis (Craven et al. 2007; Hughes et al. 2007; Rohe et al. 2009). Hughes et al. (2007) demonstrated that PGRMC1 is required for P450 activity, whereas Min et al. (2004) reported a specific increase in the hydroxylation of progesterone induced by an overexpression of the inner zone antigen (IZA) in the adrenal cortex, which is identical to PGRMC1. Also in the cerebellum, we previously showed that the increase in spinogenesis mediated through PGRMC1 was significantly decreased when 3β-HSD was blocked with trilostane (Wessel et al. 2014). Thus, a progesterone-induced up-regulation of PGRMC1 in Purkinje cells seems to increase the activity of cytochrome P450scc and 3β-HSD, which in turn raises the internal progesterone concentration leading to the observed spinogenesis (Fig. 3c). Interestingly, this effect was not just observed in immature and juvenile Purkinje cells but also in mature Purkinje cells (Wessel et al. 2014). This is especially promising for potential therapeutic use, as PGRMC1 expression has already been shown to increase after TBI (Guennoun et al. 2008), which renders PGRMC1 a highly interesting target for further clinical research.
Progesterone Induces Neuronal Outgrowth in Dorsal Root Ganglia (PNS)
Progesterone might also be a prospective therapeutic option in the PNS, as different studies suggest that progesterone improves at least the sensory function after peripheral nerve injury. For example, injury-induced allodynia was improved after progesterone treatment (Coronel et al. 2011; Dableh and Henry 2011). Even though positive effects, for example, on myelination (Koenig et al. 1995) and on the survival of motoneurons (Yu 1989) have already been described, further information about the molecular mechanisms beyond the effects of progesterone in the PNS is still missing. A recent in vitro study dealt with the basic effects of progesterone on primary cultures from chicken dorsal root ganglia (DRG). Interestingly, a highly significant increase in the outgrowth of neuronal DRG processes was detected 24 hours after treatment with progesterone as compared with untreated controls (Fig. 2a, 2b) (Olbrich et al. 2013). Thus, progesterone also seems to induce neuronal outgrowth in developing neurons of the PNS. Because DRG neurons express the necessary enzymes for progesterone synthesis—P450scc and 3β-HSD (Guennoun et al. 1997; Do Rego et al. 2009; Schaeffer et al. 2010)—there is strong evidence to consider that DRG neurons themselves are able to synthesize progesterone. Taken together, progesterone probably has a crucial function for the physiological development of DRG neurons.
Classical progesterone receptors are expressed in DRG neurons (Chan et al. 2000), and, like in the adult cerebellum, progesterone effects are absent in the DRG when progesterone treatment is combined with mifepristone (Olbrich et al. 2013). Interestingly, classical progesterone receptors are expressed not only in the soma but over the entire surface of the neuronal growth cone (Olbrich et al. 2013). Such an extraneuronal expression of classical progesterone receptors was previously described by Waters et al. (2008) in neurites and synapses of the rat hippocampus. Beside this, the expression of classical progesterone receptors in DRG neurons was found using RT-PCR-analysis (Olbrich et al. 2013). Thus, it is reasonable to suggest that neuronal outgrowth following progesterone incubation is mediated by genomic mechanisms of the classical progesterone receptors. As progesterone seems to be involved in the formation of the peripheral neuronal circuits, and because axonal navigation plays a key role in the proper formation of neuronal connections during development, an influence of progesterone on axonal growth cones can be considered. The neuronal growth cone is crucial for axon navigation and acts as a sensory motile machine (Goldberg and Burmeister 1986; Dent and Gertler 2003; Lowery and Van Vactor 2009; Dent et al. 2011). Different guidance signals lead the axon to the final target and allow for the formation of proper synaptic connections (Dickson 2002; Lowery and Van Vactor 2009; Marín et al. 2010). Indeed, it could be shown, that progesterone induces an increase in the size and motility of neuronal growth cones in DRG neurons (Olbrich et al. 2013) (Fig. 2c, 2d).
In the study by Olbrich and colleagues (2013), progesterone increased the appearance of lamellipodia and, to a lesser extent, that of filopodia, which resulted in augmented growth cone circumference and growth cone area. No effects of progesterone on growth cone size were observed when progesterone treatment was combined with mifepristone (Fig. 2e), whereas mifepristone administration alone resulted in a growth cone morphology comparable to that of the controls. Consequently, exogenous stimulation with progesterone seems to provide functional benefits for peripheral neuronal tissues like the DRG. Further studies are needed to determine whether and how the observed progesterone effects can be used therapeutically to generate positive effects on impaired peripheral neuronal tissues.
Rapid Interactions of Progesterone With the Actin Cytoskeleton
Both endogenous and exogenous progesterone seem to induce morphological changes in neuronal tissues. As the morphology and, particularly, the motility of cells are determined by the cytoskeleton (Bentley and Toroian-Raymond 1986; Chien et al. 1993; Lafont et al. 1993; Dent and Kalil 2001), it is well justified to consider that progesterone induces such morphological changes by modulating the cytoskeleton.
The cytoskeleton of Purkinje cells reveals a characteristic distribution. Microtubules and neurofilaments (EYFP-Tub und GFP-NFM) build the scaffold of the cell body and the dendritic arbor of the Purkinje cells, whereas actin-filaments (pLife-Act Tag RFP), in particular, accumulate in the periphery of dendritic arbors where the dendritic spines are located (Fig. 1d, Fig.3a). With the aid of time-series analyses in combination with Fluorescence Recovery After Photobleaching (FRAP) and confocal laser scanning microscopy (CLSM), potential changes in this typical cytoskeletal distribution can be investigated. In our previous study, fluorescence in a defined region of the dendritic arbor, the so-called region of interest (ROI), was bleached and the time for fluorescence recovery in this area was measured. In untreated controls, the local fluorescence recovery was measurable within 4 minutes (Wessel et al. 2014). Because no transport of the fluorescence-labeled β-actin was detectable but the fluorescence recovered within the dendritic spines of the ROI, it is reasonable that there is local protein biosynthesis in the dendritic arbor of Purkinje cells. This morphological evidence of local protein biosynthesis in dendrites is in line with results described by Bramham and Wells (2007). As local synthesis of proteins is much faster than transport of proteins from soma into the dendritic tree, it probably allows Purkinje cells to react quickly to internal and external stimulations. Thus, progesterone might induce morphological changes by increasing actin biosynthesis within dendrites.
Indeed, Okabe and Hirokawa revealed that an anterograde flux of actin monomers induces the polymerization of actin filaments in growth cones (Okabe and Hirokawa 1991). Thus, an increase of actin monomers might lead to an enhanced dendritogenesis and spinogenesis in Purkinje cells. Nevertheless, our recent study could not detect any increase in the time of the fluorescence recovery after photobleaching in progesterone-treated Purkinje cells (Wessel et al. 2014). A possible explanation might be that progesterone has an impact on the amount of actin monomers within the dendritic arbor without changing the rate of the protein biosynthesis. This could be explained by the fact that FRAP is suitable for the detection of variations in the time to fluorescence recovery, for example, based on transport mechanisms, but not suitable for detecting changes in the intensity of protein biosynthesis. Therefore, further studies are needed to clarify the role of the actin cytoskeleton in the genomic mechanisms of progesterone receptors in cerebellar Purkinje cells.
In DRG neurons, neurofilaments are prominently expressed in the neurites and build up the central region in the neuronal growth cone. Actin filaments instead are detectable in the peripheral zone of the neuronal growth cone forming lamellipodia and filopodia, even though some scattered neurofilaments also penetrate the periphery (Fig. 3b) (Marsh and Letourneau 1984; Schaefer et al. 2002; Dent and Gertler, 2003). Interestingly, treatment with progesterone leads to significant morphological alterations in the growth cone shape and its characteristic distribution, which is brought about by a clear shift in cytoskeletal components. Thus, the amount of lamellipodia and filopodia—densely packed with actin-filaments—increases after progesterone treatment in comparison to the controls. The shift in the ratio of actin to neurofilaments favors that of actin filaments, and leads to a considerably enlarged peripheral zone induced by progesterone treatment (Olbrich et al. 2013). This result suggests that the interaction with the actin cytoskeleton is indeed an important mechanism of progesterone to effect neuronal tissues.
Previous studies postulated a correlation between size and motility of neuronal growth cones. Indeed, Argiro and colleagues (1984) supposed that a reduced growth cone size leads to a slower neurite extension, probably mediated by either a decreased protein synthesis in the cell body or a reduced axonal transport of proteins towards the leading edge (Argiro et al. 1984). Thus, a stimulant like progesterone, which enlarges the growth cone size, might also enhance growth cone motility. A recent study examined this hypothesis. Growth cones were transfected via microinjection and observed for a couple of hours with the aid of CLSM. In controls, active and motile neurons with apparently normal organelle movement could be monitored. Actin filaments, but also neurofilaments, showed a constant motion and turnover. Actin filaments in the peripheral zone formed new motile cell protrusions, which constantly changed shape as the cones explored the environment (Olbrich et al. 2013). It is remarkable that both filopodia and lamellipodia were newly formed, but lamellipodia are not as dynamic as filopodia. Additionally, a turnover in actin was observed within the growth cone, which was increased prior to spreading. Actin turnover seemed to initiate new filopodia and lamellipodia, whereas neurofilament expression simply followed new actin branches to consolidate such new protrusions. Whereas control growth cones seemed to scan their environment without moving forward, progesterone-treated growth cones showed increased cytoskeletal motility and changes in their morphology, induced by a rapid turnover of filopodia and lamellipodia within minutes. These progesterone-treated growth cones started growing out after approximately 30 minutes. All observed effects of progesterone were completely blocked by progesterone with mifepristone. Thus, progesterone does not just increase the size of DRG neuronal growth cones but also seems to increase the rate of motility and outgrowth of the growth cone by interacting with the actin cytoskeleton.
As Argiro et al. (1984) concluded, slow neurite extension might be mediated by a decrease in protein synthesis in the cell body or a reduced axonal transport of proteins toward the leading edge. In turn, progesterone might induce outgrowth and motility of the growth cone by (i) increasing neuronal protein biosynthesis, (ii) inducing axonal protein transport, or (iii) increasing protein biosynthesis and axonal mRNA transport into the dendrite, followed by local protein biosynthesis in the dendrite. Indeed, Gu et al. (2002) described a direct axonal transport of β-actin mRNA from the soma to the neuronal growth cone, mediated by zona pellucida B1 (ZPB1) (Fig. 3c). A progesterone-induced increase in the β-actin transcription, in line with an enhanced mRNA transport into the growth cone, followed by local protein biosynthesis of β-actin, might be an explanation for the observed changes in the actin cytoskeleton after progesterone incubation (Fig. 3c, 3d). Interestingly, some of the effects were detected within a few minutes after progesterone administration, which indicates that there is different progesterone mechanisms of interaction with the actin cytoskeleton, mechanisms which are not all mediated genomically. Indeed, progesterone receptors have been identified to mediate the rapid activation of kinase cascades within the cytoplasm (Simoncini et al. 2003; Fu et al. 2007). Such extra-genomic progesterone-induced mechanisms probably include different molecular cascades, which submit fast actin rearrangements via the direct interaction with actin binding proteins (Giretti and Simoncini 2008) (Fig. 3c).
Further studies are needed to get a better idea of the exact mechanisms involved in the interaction between progesterone and the cytoskeleton. Nevertheless, it seems very likely that different mechanisms and receptors are involved in the mediation of progesterone effects.
Thus, progesterone is a multilateral neurosteroid with varying functions in divergent regions of the CNS and PNS. Progesterone acts through a couple of specific and non-specific receptors, inducing genomic and non-genomic receptor mechanisms. This degree of versatility gives rise to a broad range of potential therapeutic options for the use of progesterone in different neurological disorders.
Footnotes
Acknowledgements
We thank Jonathan Hakim, Discipline of Anatomy & Histology, Faculty of Medicine, University of Sydney, for critically reading the manuscript. Furthermore, we gratefully thank Claudia Grzelak, Anke Lodwig, and Thuan Nguyen for excellent technical work as well as Aja Lenz for secretarial work.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.O., L.W., A.B.-R., B.B.-S., and C.T. have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully thank FoRUM (Forschungsförderung Ruhr Universität Bochum Medizinische Fakultät) and RUB (Ruhr Universität Bochum) for financial support (F670–2009). L.O. especially thanks the Heinrich und Alma Vogelsang-Stiftung and L.W. the Manfred Lautenschlaeger-Stiftung for financial support in line with a graduation scholarship.
