Abstract
Aging affects oxidative metabolism in liver and other tissues. Carnitine acyltransferases are key enzymes of this process in mitochondria. As previously shown, the rate of transcription and activity of carnitine palmitoyltransferase CPT1 are also related to carnitine levels. In this study we compared the effect of dietary L-carnitine (100 mg L-carnitine/kg body weight/day over 3 months) on liver enzymes of aged rats (months 21–24) to adult animals (months 6–9) and age-related controls for both groups. The transcription rate of CPT1, CPT2, and carnitine acetyltransferase (CRAT) was determined by quantitative reverse transcription real-time PCR (RTQPCR) and compared to the activity of the CPT1A enzyme. The results showed that the transcription rates of CPT1, CPT2, and CRAT were similar in aged and adult control animals. Carnitine-fed old rats had a significant (p<0.05) 8–12-fold higher mean transcription rate of CPT1 and CRAT compared to aged controls, adult carnitine-fed animals, and adult controls, whereas the transcription rate of CPT2 was stimulated 2–3-fold in carnitine-fed animals of both age groups. With regard to the enzymatic activity of CPT1 there was a 1.5-fold increase in the old carnitine group compared to all other groups. RNA in situ hybridization also indicated an enhanced expression of CPT1A in hepatocytes from L-carnitine-supplemented animals. These results suggest that L-carnitine stimulates transcription of CPT1, CPT2, and CRAT as well as the enzyme activity of CPT1 in the livers of aged rats.
Keywords
THE AGING PROCESS is often associated with altered mitochondrial function and decreased energy production by oxidative metabolism (Chiu et al. 1999). In rat hepatocytes there is an increasing rate of mitochondrial fragility as the animal ages (Hagen et al. 1997). Consequently, mitochondrial decay appears to be one of the principal underlying causes that lead to cell decline in the aging process (Shigenaga et al. 1994).
Mitochondrial energy production from the main oxidative substrates, i.e., fatty acids and glucose, is regulated by carnitine acyltransferases such as CPT1 and carnitine acetyltransferase (CRAT), the latter regulating the level of acetyl CoA, which is a potent physiological inhibitor of the pyruvate dehydrogenase complex (McGarry and Brown 1997).
The best described function of CPT is its role in the carnitine-dependent fatty acid transport system through the inner mitochondrial membrane. This consists of the malonyl-CoA-sensitive CPT1 localized in the outer mitochondrial membrane, carnitine acylcarnitine translocase, an integral inner membrane protein, and carnitine palmitoyltransferase 2 (CPT2), which is localized on the matrix side of the inner membrane (Kerner and Hoppel 2000).
Various isoforms of CPT1 are also part of the peroxisomal and microsomal acyltransferase system, the latter appearing to influence whether triglycerides accumulate in the cytosol or whether they are exported via lipoproteins (very low-density lipoproteins; VLDLs) to peripheral tissues (Jin et al. 1992; Abo-Hashema et al. 1999; Broadway et al. 1999).
However, it should be mentioned that a complex metabolic equilibrium exists among the various carnitine pools in the different body compartments. In aged rats there is a significant decrease of total carnitine levels in the brain, serum, heart, and skeletal muscle, accompanied by an increase in the liver (Costell et al. 1989; Maccari et al. 1990). Furthermore, several studies (Odiet et al. 1995; Wang et al. 2001) have shown that activity and transcription of the muscle isoform of CPT1 (CPT1B), which is the dominant isoform in skeletal muscle, heart and fat cells (Brown et al. 1997), are downregulated in aged rats and cannot be restored to the level found in young animals by leptin or dietary carnitine. On the other hand, dietary carnitine restored the function of liver mitochondria in old rats (Hagen et al. 1998). As shown in a recent study, a significant increase in CPT1 activity after 10 days of dietary supplementation with L-carnitine was detected in liver but not in skeletal muscle (Heo et al. 2000). Therefore, liver can be considered as a primary target organ for effects of dietary carnitine administration. In the present study, an animal model with livers from aged rats (21 months at the start of a 3-month feeding experiment) and a control group of adult animals (6 months at start) was used. To the best of our knowledge, this is the first report to describe an age-dependent effect of dietary L-carnitine on the transcription rates of CPT1, CPT2, and CRAT and on the activity of CPT1 in liver.
Materials and Methods
Animals
Male Sprague-Dawley rats (OFA-17; Versuchstierzuchtanstalt der Universität Wien, Himberg), aged 7 months (adult, n = 12) and 21 months (old, n=24) were used in this study. They were housed separately in Makrolon-III cages under standard conditions (25C; 50–60% atmospheric humidity; light from 0600–1800 hr; Altromin-R rat diet and tapwater ad libitum). All procedures were performed strictly in accordance with the Austrian law for animal tests and approved by the Austrian Commission for Animal Test Affairs (TVNr:GZ 68.205/31-Pr/4/2000).
Experimental Assay for Validating the Effect of Dietary Carnitine
L-Carnitine was obtained from Lonza (Basel, Switzerland). After overnight (12-hr) water deprivation, animals were weighed and received an L-carnitine solution (60 mg L-carnitine/ml water) to achieve a total dosage of 100 mg/kg body weight/day. After animals had drunk the L-carnitine solution, bottles were filled with water. Controls had drinking water ad libitum. After 3 months the animals were sacrificed.
Briefly, after application of narcosis with a mixture of oxygen:nitrous oxide (1:2) and 4% halothane per inhalation, the thorax was opened. Animals were exsanguinated by withdrawal of blood from the right ventricle using sterile heparin- and EDTA Vacuettes. Livers were excised and frozen at −180C.
RNA In Situ Hybridization (RISH)
Ten-μm frozen sections were mounted on poly-L-lysine-coated slides and fixed in 3% phosphate buffered formaldehyde, dehydrated in ethanol, and air-dried. For each specimen, 50 ng of a digoxigenin-labeled PCR product for rat CPT1A was diluted in a commercially available hybridization buffer (Hybrisol IV; Oncor, Gaithersburg, MD). To ensure perfect permeabilization and denaturation of probes and targets, slides were placed in a metal box and heated for 5 min in an 80C water bath and then incubated overnight at 37C. After hybridization, slides were washed in PBS, three times for 10 min and incubated with anti-digoxigenin antibodies conjugated to rhodamine (Roche Diagnostics; Mannheim, Germany) diluted 1:10 in blocking solution (Roche) for 30 min at 37C. Afterwards, slides were washed with PBS (three times for 10 min), rinsed with water, and counter-stained with 2 μg/ml DAPI (Sigma; St Louis, MO) in McIlvaine's buffer (0.1 M citric acid monohydrate + 0.2 M disodium hydrogen phosphate) for 20 min at room temperature, rinsed with distilled water, air-dried, and covered with 50% glycerol in PBS. For evaluation of slides a Zeiss Axiophot Microscope in combination with a Filterset 40 (according to D. Pinkel) and the Power Gene 760 Karyotyping & Probe System PSI (Halladale, UK) was used (G65, G63) (Nowotny et al. 1996).
A fluorescein-labeled PCR product for β-actin was used as positive control probe. As negative controls, hybridization buffer without probe was added to the reference sections.
Quantitative Reverse Transcription Polymerase Chain Reaction (RTQPCR)
For preparation of mRNA, 0.5-cm3 pieces of rat liver were covered with 4 M guanidine isothiocyanate in sterile Petri dishes and minced with sterile scalpels. mRNA preparation and subsequent cDNA preparation were done with commercially available systems (Amersham Pharmacia Biotech Europe; Freiburg, Germany).
RT-PCR was carried out using a Lightcycler System (Roche Diagnostics) that allows amplification and detection (by fluorescence) in the same tube, using a kinetic approach. Lightcycler PCR reactions were set up in microcapillary tubes using 2 μl cDNA (10 ng) with 18 μl of a LightCycler-Fast Start DNA Master Mix SYBR Green I reaction mix (Roche Diagnostics) as described (Wittwer et al. 1999) containing upstream and downstream PCR primers, MgCl2, and SYBR Green. The final concentrations of the reaction components were 0.5 μM each primer, 4 μM MgCl2, and 1 × SYBR Green Master mix. Primers used were either synthesized as described for β-actin and G6PDH (Harrison et al. 2000) or designed by a commercial supplier (Sequlab; Göttingen, Germany). We used a rat-specific 416-bp amplimer of CPT1 (sense 5′-ggAgAcAgAcAccATccAAcATA-3′, antisense 5′-AggTgATggAccTTgTcAAacc-3′) and primers for a 173-bp CPT2 amplimer (sense 5′-gggAAgggAAgg-gAgAcgAg-3′, antisense 5′-ccAAgAcAcTgcgTcAggAc-3′) and a 173-bp CRAT (sense 5′-gAAgcccTTcTccTT-3, antisense 5′-gAAgcccTTcTccTT-3′) amplimer derived from the human sequence. For RTQPCR a hot-start protocol (10 min/94C) followed by a minimum of 30 cycles of denaturation (10 sec/94C), annealing 64C/5 sec, and elongation 72C/34 sec was employed. Acquisition temperature for quantification was 81C for β-actin and G6PDH, 89C for CPT1, and 87C for CPT2 and CRAT.
For calibration, standards ranging from 10 pg down to 1 fg were prepared from RT-PCR products purified after electrophoresis on 2% agarose gels.
As a negative control, samples with water instead of target cDNA were used.
Evaluation of quantitative RT-PCR was made after converting the amount of target amplified (fg per 20-ul assay containing 10 ng cDNA) to copy numbers. Transcription rate was calculated as number of copies of CPT1, CPT2, or CRAT/100 copies of β-actin or G6PDH, which were used as genes for standardization (Harrison et al. 2000).
Determination of CPT1 Activity
CPT1 activity was analyzed as described (Bieber et al. 1972). Briefly, pieces of frozen liver (about 1 g) were minced and suspended in buffer (0.25 M sucrose, 1 mM EDTA, 0.1% ethanol, 1 mg/liter antipain, 2 mg/liter aprotinin, 1 mg/liter leupeptin, 0.7 mg/liter pepstatin, 0.2 mM phenylmethylsul-fonilfluoride) at a ratio of 1:5 (w/v). These suspensions were then homogenized on ice using an Ultra-Turrax T8 (IKA Laboratory Technics; Vienna, Austria) at maximum speed for 30 sec and centrifuged at 300 × g at 4C for 10 min. One ml of the precleared supernatant was transferred to a 1.8-ml reaction vial (Eppendorf) and centrifuged at 12,000 × g at 4C for a further 5 min.
CPT activity was assayed in these supernatants spectrophotometrically by following the release of CoA-SH from palmitoyl-CoA using the general thiol reagent DTNB (5,5′-dithio-bis-(2-nitrobenzoic acid)); 850 μl Tris-HCl-DTNB buffer (116 mM Tris, 2.5 mM EDTA, 2 mM DTNB, 0.2% Triton X-100, pH 8.0). Fifty μl homogenization buffer and 50 μl cleared supernatant was added to four semi-microcuvettes (Greiner). After 5-min preincubation at 30C, 50 μl palmitoyl-CoA (1 mM dissolved in double distilled water) was added to three cuvettes. The fourth cuvette was used as a blank, adding 50 μl water instead of palmitoyl-CoA. The reaction was then started by adding 5 μl L-carnitine solution (1.2 mM dissolved in 1 M Tris, pH 8), immediately followed by photometric measurement (Hitachi U-2010; 30C, 412 nm) for 180 sec (Bremer and Norum 1967). Activity was defined as nmol CoA-SH released/min/mg protein.
The protein content of the cleared supernatants was determined according to the method of Bradford (1976).
Statistics
Statistical comparison between the groups used ANOVA followed by Dunnet's t-test for multiple comparison (Dunnet 1964).
Results
Characteristics of Experimental Animals
Mean body weight of male Sprague-Dawley rats, aged 21 months, was significantly higher than the body weight of 7-month-old rats (568 g vs 490 g). Oral pretreatment with L-carnitine did not significantly affect body weight. To ensure that experimental animals consumed the entire amount of L-carnitine, animals received an adequate amount of an L-carnitine solution (60 mg L-carnitine/ml water) after overnight (12-hr) water deprivation to achieve a total dosage of 100 mg/kg body weight/day. After animals had consumed the L-carnitine solution completely, bottles were refilled with pure tapwater. Controls received drinking water ad libitum.
Influence of Age and Carnitine Supplementation on Transcription of Carnitine Acyltransferases
Our first goal was to determine whether there is an age-related difference in the transcription rates of liver carnitine acyltransferases similar to those described for CPT1B in muscle and heart and to see how dietary L-carnitine might influence this. Two methods, i.e., RTQPCR and RISH, were used for these investigations.
Results of RTQPCR
As shown in Figure 1 and Table 1, dietary L-carnitine resulted in a 10-fold increase of the mean transcription rate for CPT1 and CRAT and a twofold increase for CPT2 in livers from the carnitine group of old animals in contrast to the age-matched control group. In livers of adult animals, dietary L-carnitine also significantly stimulated the transcription rate of CPT2 but not of CPT1 and CRAT. Furthermore, there were no significant differences in the transcription rates of CPT1, CPT2, and CRAT between old and adult controls.
For quantitating transcription of CPT1, CPT2, and CRAT, RTQPCR using the LightCycler SYBR Green technique was employed, which has been shown to produce more precise results than other approaches for mRNA quantification such as probe hybridization (Northern blots) or band densitometry (Schmittgen et al. 2000).
The level of gene expression was measured by relative or absolute RTQPCR. For absolute quantitation, a standard curve relates the PCR signal to the input copy numbers of a defined template (Winer et al. 1997), while relative quantification measures the relative change in mRNA expression in comparison to housekeeping genes (Schmittgen et al. 2000). Relative quantitation was adequate to study the changes in gene expression. Because the use of β-actin as the only control gene has been critically discussed (Suzuki et al. 2000), G6PDH was chosen as a second internal control. In rat livers, transcription of β-actin proved to be more constant than G6PDH. Therefore, a higher degree of significance was obtained when transcript copy numbers of CPT1, CPT2, and CRAT were normalized to β-actin (Table 1).
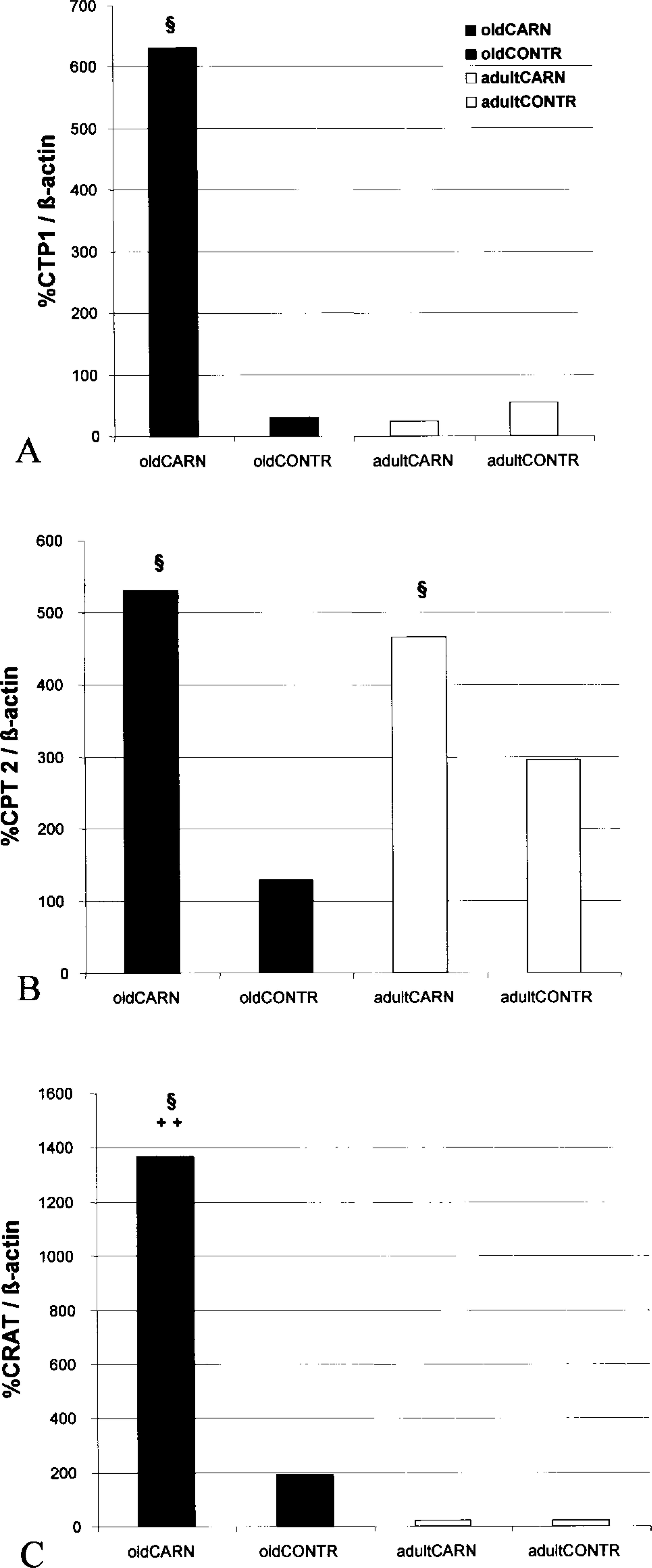
Transcription rates of carnitine acyltransferases showing the effect of L-carnitine supplementation in aged rats (oldCARN and oldCONTR) and adult animals (adultCARN and adultCONTR).
Effect of age and dietary L-carnitine on CPT1, CPT2, and CRAT a
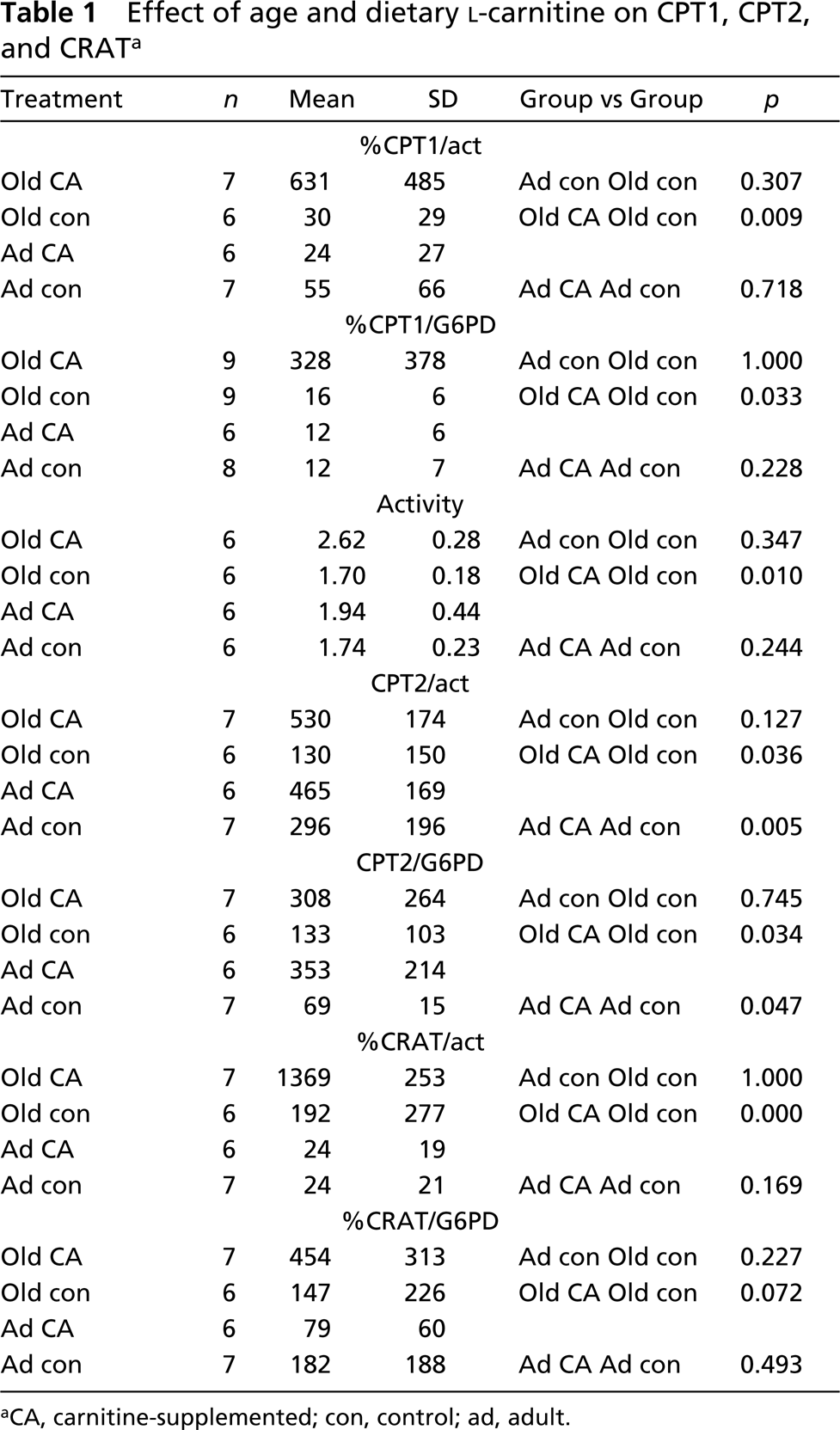
CA, carnitine-supplemented; con, control; ad, adult.
Influence of Age and Carnitine Supplementation on Activity of CPT1A
Our second goal was to test if there was an age-related effect of dietary L-carnitine on enzyme activity of CPT1A in rat hepatocytes. As shown in Table 2 and Figure 2, the enzyme activity of CPT1 in aged carnitine-supplemented rats was 1.5-fold higher than in the age-matched control group (p<0.01). Again, there was no significant difference between aged and adult control animals. L-Carnitine supplementation had a slight but not significant stimulating effect on the activity of CPT1A in livers of adult animals. Furthermore, the results displayed in Table 2 show that there was a good correlation between enzyme activity and transcription rate as defined by the CPT1A/β-actin ratio (p<0.001) or the CPT1A/G6PD ratio (p = 0.01) in old carnitine-treated rats and in age-related controls but not in the corresponding groups of adult animals.
Correlation between relative gene expression of CPT1 and enzyme activity [nmol (palmitate)/min/mg protein]

Results of Fluorescent RISH
The third goal of this study was to test if RISH would be a suitable tool for measuring the effect of L-carnitine supplementation on hepatic tissue. Results of fluorescent RISH of livers from carnitine-supplemented rats compared to controls are shown in Figure 3. Especially in the border zones of cryostat sections of both old and young carnitine-fed animals, more hepatocytes appeared to be positive for CPT1 expression compared to the age-related controls.
The RISH performed in this study showed a higher density of hybridization signals for CPT1 in the border zones of liver parenchyma in both aged and adult carnitine-supplemented animals compared to unsupplemented controls (Figure 3). This supports a previous report showing a great heterogeneity of CPT activity in various hepatocyte fractions of liver parenchyma (Hagen et al. 1998).
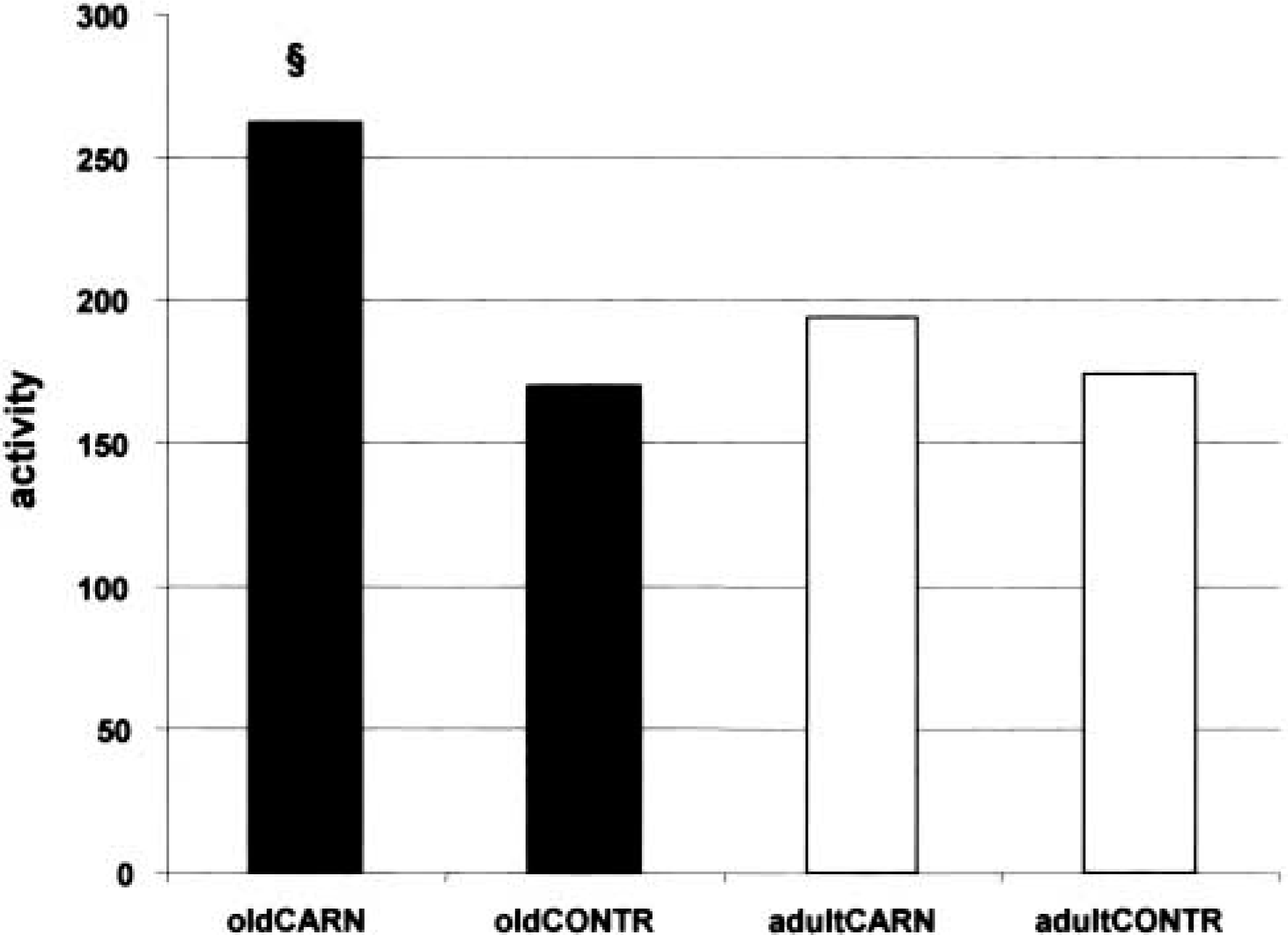
Activity of CPT1-enzyme (nmol palmitate/min/mg protein × 100) showing the effect of L-carnitine supplementation in aged rats (oldCARN and oldCONTR) and adult animals (adultCARN and adultCONTR). § p<0.05.
Discussion
The present study is the first report demonstrating that dietary L-carnitine causes an alteration in the expression of CPT1A, CPT2, and CRAT at the mRNA level and for CPT1A also on the level of enzymatic activity in livers of old rats in contrast to livers from adult animals. This work therefore provides evidence for a significant correlation between altered enzyme activity and transcription rate of CPT1, especially in livers of carnitine-supplemented old animals, but no evidence for a significant age-correlated alteration of the transcription rate and enzyme activity of CPT1A without carnitine supplementation.
The results obtained in liver, where CPT1A is the predominant isoform, were in sharp contrast to findings in heart, skeletal muscle, and adipose tissue (Odiet et al. 1995; Wang et al. 2001), which contain the CPT1B isoform. This might partly be explained by the fact that the uptake of carnitine is much slower in muscle compared to liver, particularly in senescent animals. Therefore, carnitine supplementation for 2 weeks (Odiet et al. 1995) may be less adequate in comparison to one for 3 months as in the present study. Furthermore, it should be mentioned that the two CPT1 isoforms are encoded by completely different genes, which are localized on different chromosomes (van der Leij et al. 2000) and regulated individually (Cook et al. 2001). CPT1B is not at least regulated by PPARa (peroxisome proliferator-activated receptor alpha), whereas CPT1A has a TRE (thyroid hormone-responsive element) and a CCAAT box, which is frequently observed in glucocorticoid-responsive genes.
This might be caused by different sensitivity to carnitine stimulation due to downregulated thyroid hormones in senescent organisms. (Maebashi et al. 1982; Gadaleta et al. 1990). In addition, it has recently been shown (Saheki et al. 2000) that L-carnitine has the potential to downregulate Fos/Jun, of which high levels are known to inhibit the GRE (glucocorticoid-responsive element) and TRE (e.g., Jonat et al. 1990; Bodenner et al. 1992). Thus, L-carnitine has the potential to upregulate genes such as CPT1A and carbamoylphosphate synthetase which have TRE and GRE in their promoter regions (eg. Jansen et al. 2000; Saheki et al. 2000). These results support previous findings reporting an additive effect of carnitine-betamethasone and carnitine-thyroxine (Lohninger et al. 1986a,b, 1996).
An age-associated slower mRNA degradation does not appear to be involved because mitochondrial CPT2 was stimulated by dietary L-carnitine independently of age, although previous studies (e.g., Thakur and Chaurasia 1997) demonstrated that nuclease resistance of mRNA increases with age. In addition, the possibility that variations in transcriptional accuracy might occur as a consequence of age-associated molecular misreading (e.g., van Leeuwen et al. 2000) can be excluded because all amplicons had exactly the same melting profile, which is automatically determined in the course of each analysis using RTQPCR with the LightCycler SYBR green technology.
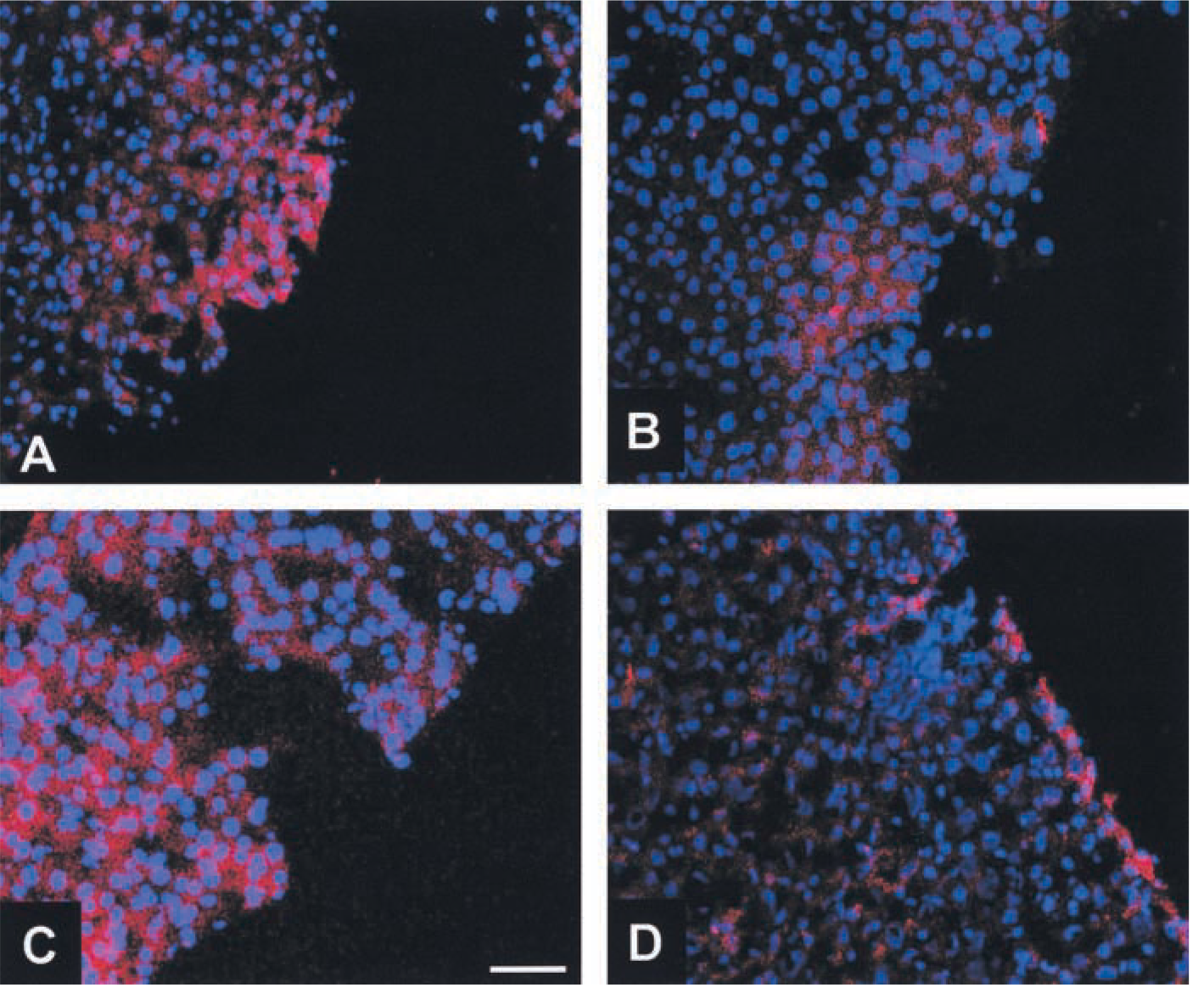
Results of RISH on cryostat sections of livers using CPT1-specific digoxigenin-labeled oligonucleotides as detected by rhodamine-labeled anti-DIG antibodies and counterstaining with DAPI.
Furthermore, there was a significant correlation between CPT1A mRNA content and activity, whereby the enhancement of mRNA transcription was more pronounced. It is well established that long-term changes in CPT1 activity are based on changes at the transcriptional level (McGarry and Brown 1997). Therefore, the increase in CPT1 activity is most probably caused by an increased CPT1 protein content and not by short-term regulation of the activity, e.g., by the activation of a Ca2+/calmodulin-dependent signal transduction cascade involving protein kinase II activation followed by cytokeratin phosphorylation (Velasco et al. 1998). One of the reasons for the smaller relative increase in CPT1 activity compared to CPT1 mRNA might be the well-known age-dependent increase of oxygen radicals rather than a diminished translation rate. Oxygen radicals are known to damage cellular macromolecules including lipids, DNA, and proteins. The accumulation of oxidatively damaged proteins, the extent of which varies among tissues, increases markedly with age (Shigenaga et al. 1994). In mitochondria of mouse hearts, treatment with oxygen-derived radicals resulted in a decrease of CPT1B activity (Odiet et al. 1995). In addition, other cellular mechanisms might be involved. Malonyl-CoA appears to be an unpromising candidate because, compared to CPT1B, the CPT1A activity is less sensitive to malonyl-CoA, the physiological allosteric inhibitor (Starritt et al. 2000).
Methodological pitfalls, causing the observed differences between transcription rate and activity of CPT, can be mostly excluded for the following reasons. (a) Due to the necessary dilution steps in the in vitro assay of CPT activity, an inhibition of the enzyme by malonyl-CoA is negligible. (b) The affinity of CPT1 for the acyl-CoA substrate alters under ketogenic conditions and phosphorylation as induced by, e.g., glucagon or forskolin (Harano et al. 1985). Such effects can be excluded because the measurements were all carried out under the same in vitro conditions. (c) The sensitivity of CPT1 to inhibition may alter (Cook et al. 1980; Cook 1984; Grantham and Zammit 1988) due to a change in membrane fluidity rather than an attenuation in numbers of malonyl-CoA-binding sites. Because all animals had the same diet, such dietary effects can be excluded. (d) Changes in hepatocyte cell volume (Guzman et al. 1994) or cytosolic pH (Moir and Zammit 1995), which may be responsible for modulation of CPT1 activity, were avoided, because extractions were performed under isotonic conditions and buffering at physiological pH.
Comparison of isolated enzyme activities may be misleading in general, since in vitro assays are non-physiological compared to the in vivo environment of the enzyme in the cell. Although other factors such as long-chain fatty acids, clofibrate, the plasticizer DEHP [=2-(diethyl-hexylphthtalate], and glucagon (e.g., Brady and Brady 1989a,b; Brady et al. 1991; Chatelain et al. 1996) increase mRNA levels of CPT1A and CRAT in rat hepatocytes, carnitine supplementation appears to be most effective in livers of aged rats.
Further studies on key enzymes of other metabolic pathways may show whether dietary L-carnitine can cause changes in the utilization of fuel in aged organisms.
Footnotes
Acknowledgements
Supported by the Herzfelder Stiftung and the Austrian Federal Chancellery.
Thanks are due to Jennifer Hirsch for expert help with preparation of cryostat sections.
