Abstract
Ultracryotomy of fixed specimens in combination with immunogold labeling is widely used for ultrastructural localization of many interesting molecules. Since the introduction of this technique, vast improvements in techniques and machinery have been established and the entire process has been made easier and more accessible. Normally, sections are cut and labeled within 1 day to prevent possible loss or redistribution of soluble antigens within the sections. An increasing demand for more sections and multiple labeling protocols prompted us to investigate the extent to which ultrathin cryosections can be stored. This would render the time spent behind an ultracryomicrotome more efficient and would allow immunogold labeling at a later stage. We investigated whether gelatin plates, 2.3 M sucrose, or 1.0% methyl cellulose/1.2 M sucrose can be used to store thawed frozen sections for a longer period of time. Ultrathin sections of mildly fixed tissue and cultured cells were stored for up to 6 months before immunogold labeling. The preservation of the ultrastructure of stored sections was excellent and was similar to that of immediately processed sections. Importantly, prolonged storage did not affect the labeling intensity.
I
To investigate whether stored ultrathin thawed cryo-sections can be used for immunolabeling we first evaluated the preservation of the ultrastructure after prolonged storage in two tissues and cultured cells. Whether the number of detectable immunodeterminants changed during storage was measured for a soluble protein at low and high intracellular concentrations and for a transmembrane protein.
We demonstrate that thawed ultrathin cryosections can be stored on a Formvar/carbon-coated copper grid in 1.0% methylcellulose/1.15 M sucrose mixture at 4C for more than 6 months without noticeable effects on ultrastructure and labeling efficiency.
Materials and Methods
Tissue Preparation and Cryosectioning
Male Wistar rats were anesthetized using 60 mg/kg (body weight) sodium pertobarbital. Whole body perfusion was performed through the abdominal aorta using 4% formaldehyde (FA) in 0.1 M phosphate buffer, pH 7.4 (PB). The pancreas and liver were removed, cut into small pieces, and postfixed for 2 hr in 4% FA in buffer.
The spleen-derived murine DC line D1 (hence referred to as D1 cells) has been described previously and was cultured under standard conditions (Winzler et al. 1997). Non-stimulated D1 cells were fixed with a mixture of 2% FA and 0.2% glutaraldehyde in PB for 2 hr at room temperature (RT), rinsed with PBS/0.15 M glycine, and embedded in gelatin.
Ultrathin cryosections were prepared according to Liou et al. (1996). Briefly, the specimens were infused with 2.3 M sucrose overnight, placed on a copper or aluminum specimen holder, and frozen in liquid nitrogen. Ultrathin sections (50–100 nm) were cut with a diamond knife (Drukker International; Cuijck, the Netherlands) on a Leica Ultracut T equipped with a cryo-attachment (Leica Microsystems; Vienna, Austria). The sections were picked up from the diamond knife using either 2.3 M sucrose or 1.0% methyl cellulose 25CP/1.15 M sucrose (MCS) and transferred to a Formvar/carbon-coated copper grid mounted on a microscope slide with a self-adhesive label.
Storage of Ultrathin Thawed Frozen Sections and Immunolabeling
Two different storage procedures were used. After retrieving the sections in the pick-up solution, the thawed sections were transferred to a carbon/Form-var-coated copper grid on a microscope slide in a closed petri dish at RT. Thereafter, the grids were either transferred to 2% gelatin in PB and stored at 4C or allowed to dry for 30 min at RT and next stored at 4C in the pick-up solution.
For quantitative evaluation, the grids were stored for different time intervals and then immunolabeled the same day under identical circumstances. Before immunolabeling the grids were placed on 2% gelatin in PB at 40C for 20 min. Immunolabeling of pancreas tissue was performed with rabbit anti-rat amylase serum (Posthuma et al. 1987). Rat liver sections were labeled with rabbit anti-rat albumin (5 μg/ml; Nordic Immunological Laboratories, Tilburg, The Netherlands). D1 cells were labeled with rabbit anti-mouse invariant chain (Ii) luminal part (S23; a kind gift of Dr. N. Koch, University of Bonn, Bonn, Germany; Koch et al. 1987). The antibodies were visualized with 10-nm protein A-gold as described previously (Slot et al. 1991).
Quantification
The labeling density (LD) for amylase in zymogen granules was measured in 25 different cells in different acini from the pancreas using a JEOL EM 1200 electron microscope (JEOL; Tokyo, Japan) equipped with a TV camera. At a final magnification of ×60,000 the gold particles were counted in the center of four secretory granules per cell on the TV screen using an onscreen overlay of 0.5 × 0.5 μm. The LD for amylase in the ER was determined in 25 different cells by measuring the surface of the ER cisternae using the point hit method with a standard grid lattice (d = 0.1 μm; Weibel 1979) on electron micrographs (×60,000) and counting the gold particles in the same area.
Background levels were measured in a similar way after immuno-incubation with a non-relevant antibody from the same species. The background appeared to be neglectable (<0.5 gold particles/μm2).
The average zymogen granule diameter was measured by using Qwin (Leica) on digitized EM negatives. Approximately 100 granules from 25 different cells were measured.
To compare the labeling densities after different storage intervals for the Ii membrane protein in D1 cells, the LD was expressed as gold particles/intersection. Intersection points were counted in 20 micrographs (×60,000) using a standard grid lattice (d = 0.1 μm) (Weibel 1979). Gold particles were counted over the same membranes.
Results
Ultrastructure of Stored Thawed Ultrathin Cryosections
The grids that were stored on the gelatin plates at 4C turned dark within a week and became progressively dirty, perhaps due to oxidation of the copper. This storage method was not further pursued.
Grids stored for a prolonged time in sucrose or MCS did not show any salt deposits, bacterial growth, or other disturbing artifacts. An advantage of preparing the Formvar-coated grids on a microscope slide with a self-adhesive label is that information about the specimens can be written on the label (Figure 1). The slides can be stored very efficiently in a normal plastic slide box. During storage in sucrose or MCS at 4C, the volume of the drop of pick-up solution becomes smaller due to the evaporation of water. This led occasionally to some crystal formation.

Ultrathin cryosections on Formvar/carbon-coated grids stored in MCS on a microscope slide provided with a label. Sections in MCS can be identified by the light reflection.
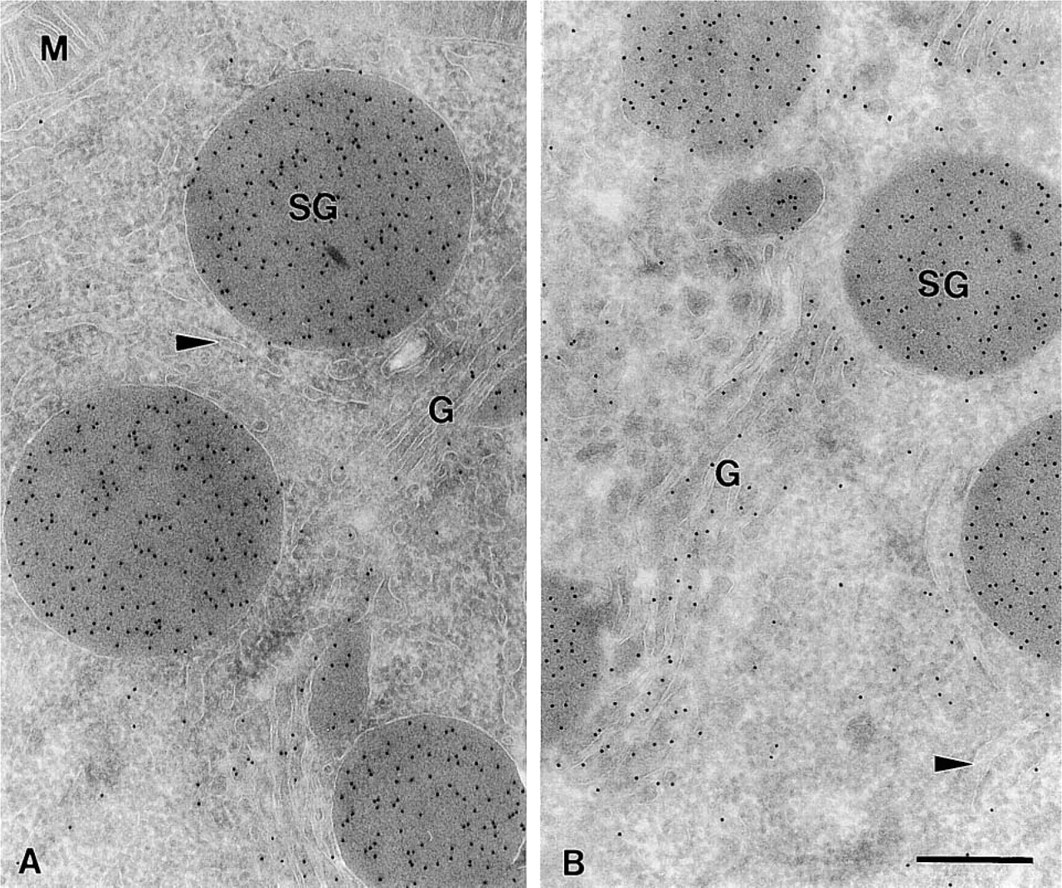
Electron micrograph of an ultrathin cryosection of rat exocrine pancreas retrieved with MCS. (
As can be judged from Figures 2 and 3, the ultrastructure of stored sections was excellent, irrespective of the time they were stored. The typical differences in morphological appearance after sucrose and MCS pick-up, as described by Liou et al. (1996), did not change after storage. Examination of the fine structure at higher magnification showed that all the fine details were clearly visible even after 32 weeks of storage at 4C in MCS (Figures 2 and 3).
Labeling Densities in Stored Thawed Ultrathin Cryosections
Soluble Proteins. To assess whether prolonged storage of thawed ultrathin frozen sections affects the LD, we used mildly fixed pancreas (4% FA) as a model system. Two different situations were evaluated: the LD for amylase in the secretory granules (SG), which contain a high protein and amylase concentration, and in the ER, which has a fairly low protein and amylase concentration (Posthuma et al. 1987; Oprins et al. 1994). The LDs were determined after storage in either sucrose or MCS up to 32 weeks.
Figure 4 shows that the LD for amylase in SGs from sections picked up and stored in 2.3 M sucrose is about 20% lower than in MCS. This was not due to changes in size of the SGs in the different pick-up solutions.
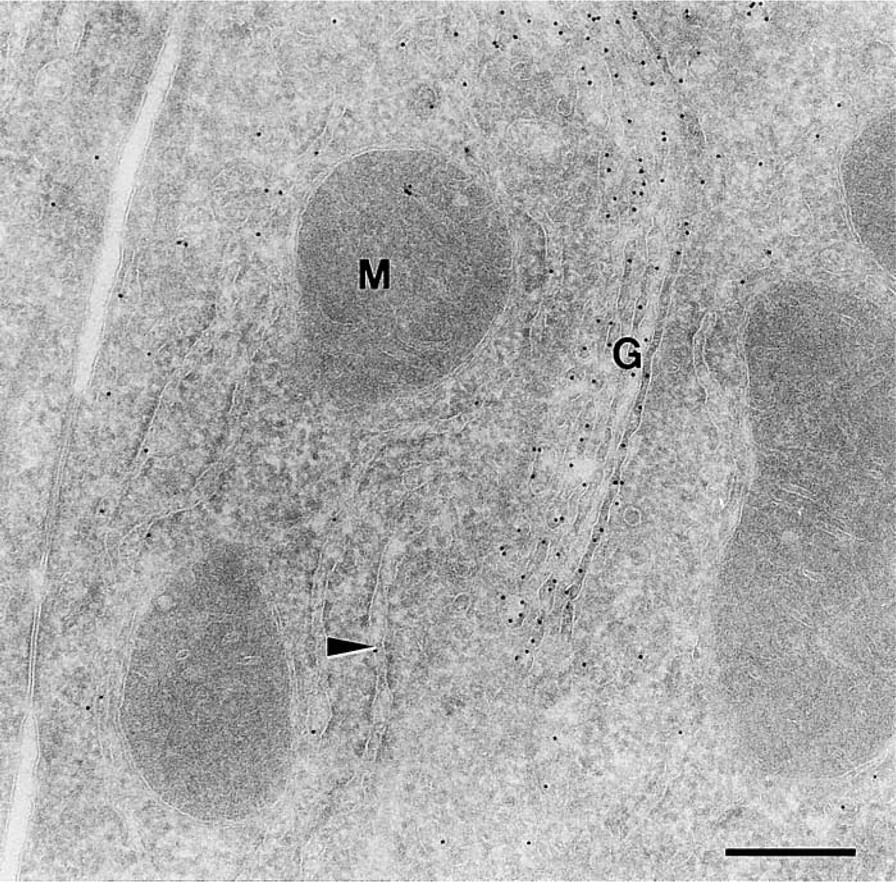
Electron micrograph of an ultrathin cryosection of rat liver retrieved with MCS and stored for 3 weeks in MCS. The section was immunogold-labeled for rat albumin. G, Golgi complex; M, mitochondrion; arrowhead, RER. Bar = 300 nm.
The average SG diameter is the same: MCS pickup, 0.71 ± 0.06 μm; sucrose pick-up, 0.68 ± 0.05 μm. Although some variations in LD could be measured, the LD stabilized at 90–95% of the LD measured in immediately processed grids and remained at that level for more than 6 months. The same holds true for the sucrose-stored grids, although the variation in LD was much larger, ranging from 72% to 121%.
Knowing that pick-up and storage in MCS yielded higher LDs than in sucrose, we measured the LD in the ER of amylase only after MCS pick-up. Figure 5 shows that the variation in LD was considerably higher in the ER compared to the SGs. However, no significant changes in LD could be observed after different times of storage.
Membrane-bound Proteins. In Figure 6 the LD (expressed as gold particles per intersection) of the Ii transmembrane molecule is shown after storage up to 14 weeks in MCS at 4C. No significant differences could be measured.
Discussion
Storage of ultrathin cryosections is very convenient from an experimental point of view. In a standard sectioning session, on average 20% of the time is spent in trimming and finding the right location in the specimen. Next, ultrathin sections are prepared and immunolabeled the same day. This limits the number of grids produced. When the sectioning time can be extended to a full day, the efficiency of sectioning in terms of production will be greatly enhanced. Furthermore, when a second evaluation of the same specimen is at hand, it is very convenient when the sections are already available and no additional sectioning session is necessary. In a recent review, Tokuyasu (Tokuyasu 1997) suggests that sections can be stored on sucrose or sucrose supplemented with albumin for 1–7 days. Whether storage affected the preservation of the ultrastructure and the maintenance of antigenicity was not discussed.
We used amylase of the rat exocrine pancreas as a model for a soluble constituent present in an organized matrix of proteinaceous nature. A wide range of matrix densities are covered. In the RER, low concentrations of proteins are present, whereas in the SGs a very high concentration is present (Posthuma et al. 1987; Oprins et al. 1994).
In a standard procedure the sections are picked up and transferred to a Formvar/carbon-coated grid.
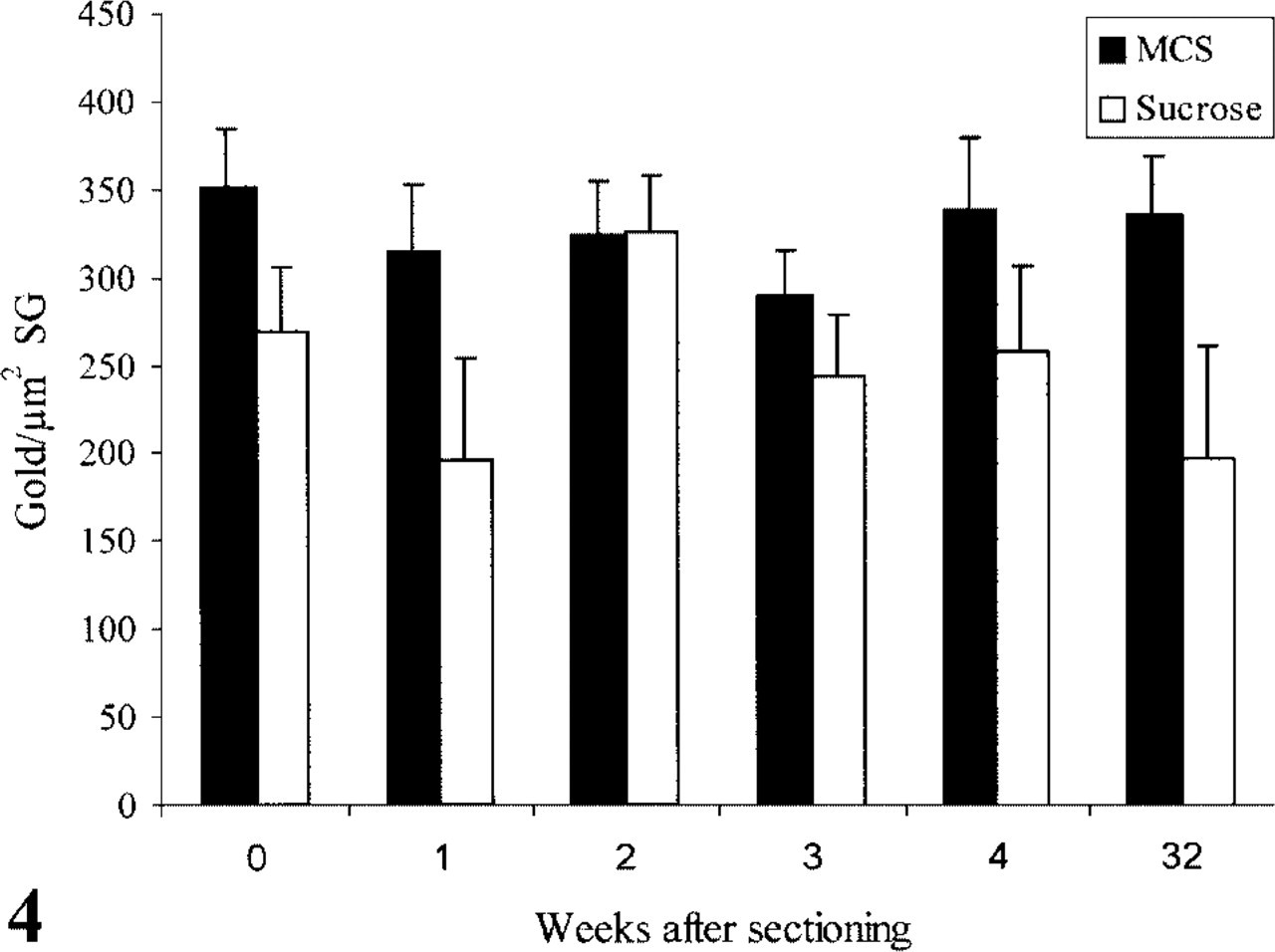
Number of gold particles/μm2 secretory granule surface (SG) after immunolabeling with anti-amylase and protein A-gold at different time points after sectioning. The sections were stored at 4C in 2.3 M sucrose or methylcellulose/sucrose solution (MCS).
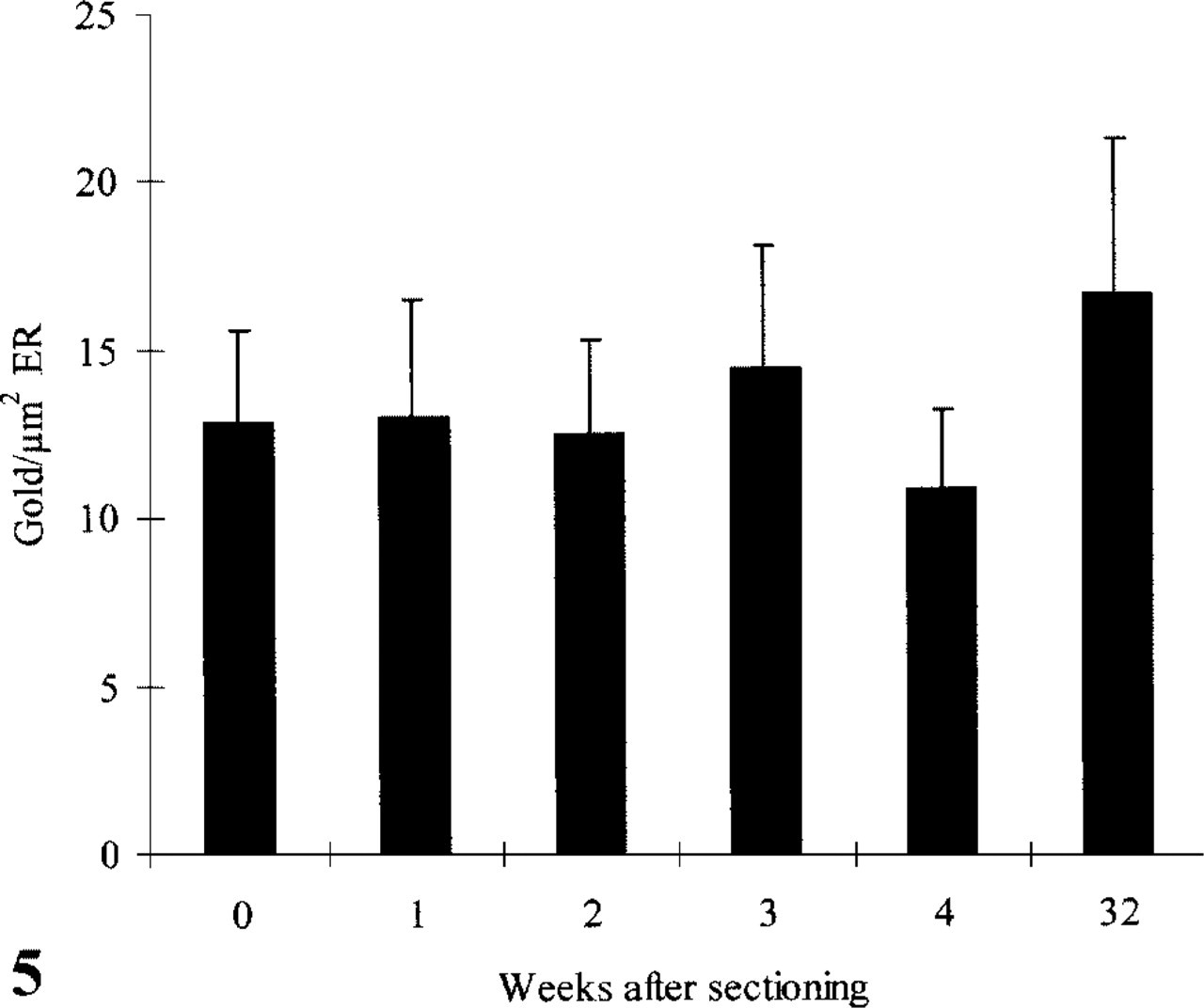
Number of gold particles/μm2 ER after immunolabeling with anti-amylase and protein A-gold at different time points after sectioning. The sections were stored at 4C in MCS solution.
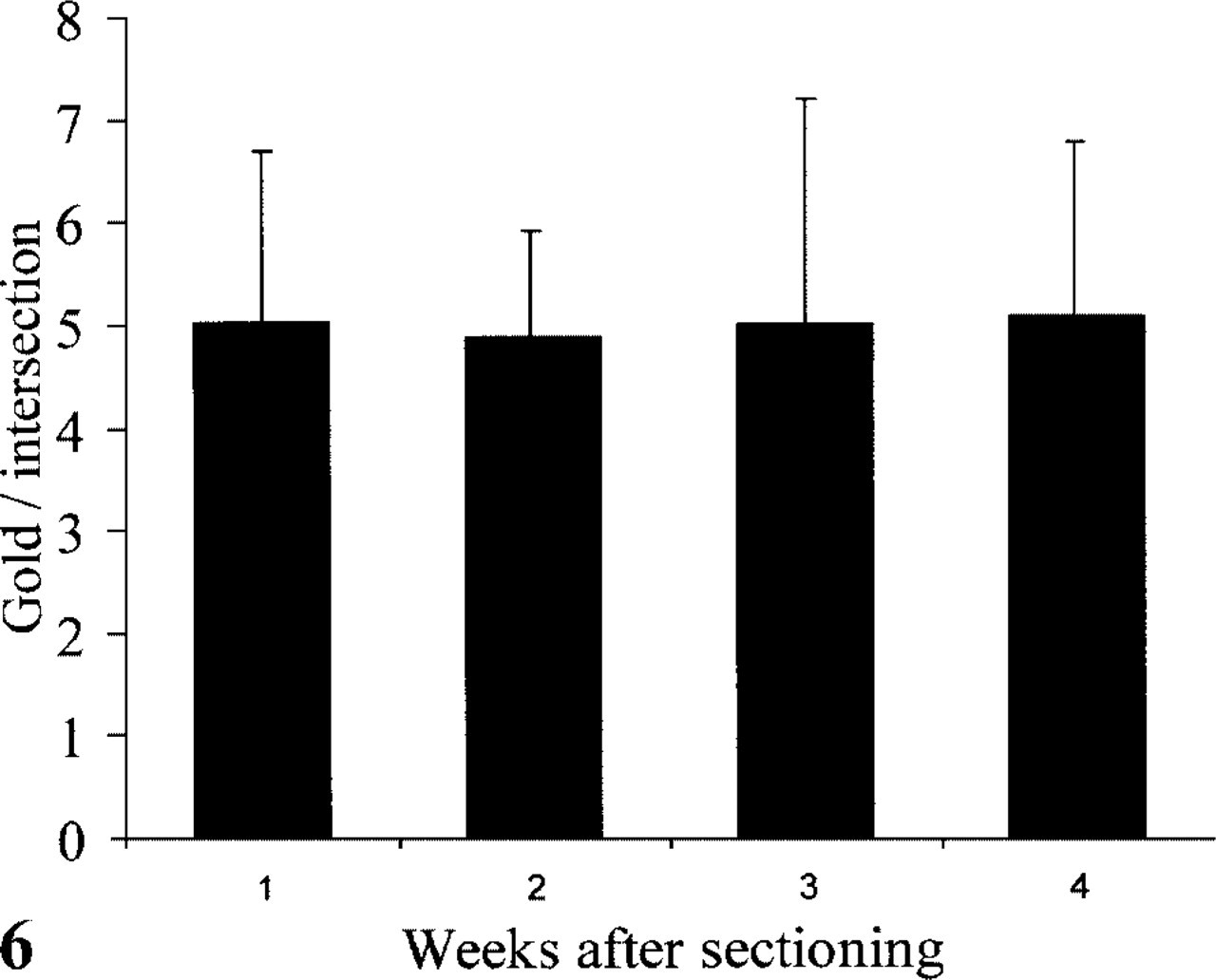
Number of gold particles/intersection on the plasma membrane of D1 cells after immunolabeling with rabbit anti-mouse invariant chain (Ii) luminal part and protein A-gold at different time points after sectioning. The sections were stored at 4C in MCS.
Next they are put on a 2% gelatin in PB plate for 1–3 hr. At first we tried to store grids on the gelatin plates, but found that the Formvar/carbon-coated copper grids turn black within a week and insoluble copper salt deposits can be observed on the grids. We have not tested nickel or golden grids, but they may well be the solution to this problem. When we started our investigations, two different pick-up solutions were used in the laboratory: 2.3 M sucrose and a 1% methylcellulose/1.15 M sucrose (MCS). Both were tested for their ability to serve as a storage solution. The ultra-structure of sucrose- and MCS-retrieved sections differed considerably, as described by Liou et al. (1996). This difference in ultrastructure did not change during storage. Sections retrieved with sucrose have a lower LD for amylase in the SGs compared to the MCS sections, even without storage. Most likely, 2.3 M sucrose extracts soluble antigens from the section, which is counteracted by the addition of methylcellulose. Changes in morphology and LD were also observed by Takizawa and Robinson (1994) when they compared the sucrose and a mixture of sucrose and gelatin as a pick-up solution. Our results support their findings that sucrose alone as a pick-up medium affects the quality of the section. The loss of antigenic determinants in 2.3 M sucrose occurs during the pick-up or the subsequent thawing of the sections, and thereafter no further reduction is observed. The stretching that normally occurs when sections are picked up with 2.3 M sucrose (Liou et al. 1996) does not contribute to the smaller LD after sucrose pick-up because the size of the SGs does not change. We cannot exclude that MCS also extracts specimen components, but a 20% better LD prompted us to use MCS as standard pickup and storage solution.
The contents of an SG are very compact, with an extremely high protein concentration (Posthuma et al. 1987; Oprins et al. 1994), which may be cross-linked by nature and/or by the fixative. A potential loss of antigens can be much greater in an organelle with a loose matrix, such as the ER. Even up to 6 months after sectioning no significant differences in the LD for amylase were measured. By qualitative evaluation, no obvious changes in the LD for albumin in liver cells were found.
Membrane-anchored proteins, such as the Ii molecules in D1 cells (Winzler et al. 1997), are less likely to diffuse out of a ultrathin cryosection. Indeed, we have not found any changes in LD after prolonged storage in MCS. We conclude that our MCS storage method allows ultrathin cryosections to be immunolabeled and processed at a long time after sectioning, irrespective of the nature of the protein or matrix in which it is contained.
Footnotes
Acknowledgements
We wish to thank Rene Scriwanek and Tom van Rijn for excellent photographical work and Drs Slot and Geuze for their helpful suggestions and critical reading of the manuscript.
