Abstract
Sepsis, which could lead to mitochondrial dysfunction and cellular energy loss, always induces acute liver injury and has a high mortality rate. Tetramethylpyrazine (TMP) is an active extract from the Chinese herb Ligusticum chuanxiong and exhibits anti-sepsis activity. In this study, a rat sepsis model was first established via cecal ligation and puncture (CLP). Then, 48 Sprague Dawley male rats were randomly divided into four groups (12 rats in each group): control group (C), sepsis group (S), TMP treatment group (T), and TMP prevention group (P). Serum aspartate aminotransferase (AST), serum alanine aminotransferase (ALT), mitochondrial aspartate aminotransferase (mAST), and adenosine triphosphate (ATP) levels and mitochondrial membrane potential (MMP) were measured and used as indicators of hepatic dysfunction severity and mitochondrial function. In addition, the activities of Na+-K+-ATPase, Mg2+-ATPase, Ca2+-ATPase, and Ca2+-Mg2+-ATPase in the mitochondrial membrane, the expression level of AQP8 and some inflammatory factors, and the level of oxidative stress were measured to explore potential mechanisms. We found that AQP8 accepts signals from inflammatory factors upon stimulation and during various infections, and low AQP8 expression levels could result in further downstream mitochondrial dysfunction. In conclusion, our data demonstrated that TMP could ameliorate hepatocellular mitochondrial dysfunction by decreasing the inflammatory response and increasing AQP8 protein expression.
Introduction
Sepsis is a clinical syndrome caused by severe infections that always trigger excessive inflammatory responses. 1 Sepsis, septic shock, and multiple organ dysfunction syndrome (MODS) represent the sequential stages of the disease process. The end stage of MODS is organ failure, which is commonly associated with poor clinical outcome and a high mortality rate. 2 During the process of sepsis, the liver plays an important role in defensive responses to scavenge bacteria and produce inflammatory mediators. In addition, sepsis-induced multiple organ injury and failure may be crucially caused by the deterioration of mitochondrial function and consequent failure of cellular energy metabolism.3–5
Mitochondria are required for normal cellular function by providing energy, utilizing approximately 98% of total body oxygen. Therefore, mitochondria play an important role in the maintenance of normal organ function, especially in skeletal muscle and the liver. However, mitochondrial structural integrity is essential for preserving normal mitochondrial function and energy metabolism. However, the exact mechanism of mitochondrial dysfunction is not fully understood.
Tetramethylpyrazine (TMP), an active extract from the Chinese herb Ligusticum chuanxiong, is widely and very safely used for the treatment of inflammatory and cardiovascular diseases in traditional Chinese medicine.6–8 TMP can act as an antioxidant to quench reactive oxygen species (ROS), inhibit lipid peroxidation, and protect enzymatic antioxidants.9,10 TMP also has anti-inflammatory and11,12 organ-protective effects and improves mitochondrial function at the cellular level. 13 However, the molecular mechanisms responsible for these effects of TMP remain elusive. Aquaporin-8 (AQP8), a member of the family of water channel of trans-membrane proteins, is located in the inner mitochondrial membrane of liver cells and is essential for mitochondrial structure because it mediates the transport of water and other metabolic substrates.14,15 AQP8 also contributes to inflammation by mediating mature IL-1β release through the induction of rapid cell swelling. 16 However, knocking down mitochondrial AQP8 can cause oxidant-induced mitochondrial dysfunction17,18 and induce necrotic cell death in human neoplastic hepatic cells. These results suggest potential therapeutic strategies against hepatoma cells. 19 In addition, tumor necrosis factor α (TNF-α) reduces the expression of AQP8 causing mitochondrial swelling in livers of septic rats. 20 Overall, data have suggested that AQP8 might play an important role in maintaining normal mitochondrial function. Our previous research also suggested that TMP could protect the structure of hepatocyte mitochondria in septic rats by increasing AQP8 expression. 13 However, whether TMP protects liver cells from inflammation by improving mitochondrial function, whether a strong correlation exists between AQP8 and the inflammatory response in the septic rat model, and whether TMP has efficacy as a treatment of sepsis remain unclear.
In this study, we sought to further investigate the effect of TMP on mitochondrial function and elucidate the potential mechanisms underlying the protective effects of TMP in sepsis-induced acute liver injury. A better understanding of mitochondrial damage and dysfunction in sepsis will potentially result in new therapies, which could reduce the progression of organ dysfunction and failure and ultimately decrease the mortality of sepsis. 21
Materials and methods
Animals and chemicals
Specific pathogen-free clean-grade adult male Sprague Dawley rats (220–260 g) were obtained from the Laboratory Animal Research Center of Anhui Medical University (China). They were fed under constant environmental conditions (temperature of 22°C–25°C, a relative humidity of 30%–70%, and continuous cycle of 12 h in light and 12 h in dark) according to the Institutional and National Guidelines for the Care and Use of Laboratory Animals. All animals received a standard rat diet and water ad libitum. The Ethics Committee of Anhui Medical University approved all animal procedures. The animal quality permit number is SCXK (WAN) 2011-02.
Injectable ligustrazine hydrochloride (each 2-mL ampoule contained active components equivalent to 40 mg of ligustrazine) was provided by Harbin MEDISAN Pharmaceutical Co., Ltd (China). The lot number was 140906D1-113. All chemicals were purchased from Invitrogen (Life Technologies, USA) unless otherwise stated.
Rat sepsis model preparation
The sepsis model was established via cecal ligation and puncture (CLP) according to the standard procedure described in a previous study. 22 The main procedures are summarized as follows: first, rats were placed on a desk and a midline laparotomy was performed after anesthetizing with 10% chloral hydrate via intraperitoneal injection. Then, the cecum was exposed and ligated with the distal ileocecal valve with a 3.0 silk suture without causing intestinal obstruction. Next, the cecum was punctured twice using a 16-gauge needle and gently compressed to extrude a small volume of cecal content. Finally, the incision was closed and the rats were subcutaneously administered 10 mL/100 g body weight of saline solution for fluid resuscitation. Sham-operated control animals underwent the same procedures (only separated cecum without ligation and puncture). Sepsis was defined by temperature variation (<36°C or >38°C) and/or decreased sensitivity to touch, diarrhea, eye hemorrhage, and piloerection. 23
Animal experimental design
In total, 48 Sprague Dawley male rats were randomly divided into four groups (12 rats in each group): control group (C), sepsis group (S), TMP treatment group (T), and TMP prevention group (P). The rats in the TMP treatment group received 60 mg/kg/day of ligustrazine through the caudal vein immediately after CLP, and those in the prevention group received 60 mg/kg/day of ligustrazine in the caudal vein for 7 days prior to CLP. The rats in the control and sepsis groups received the same volume of NaCl (0.15 mol/L) immediately after CLP. In this phase, the onset time of sepsis and survival time after the CLP operation were observed and recorded for the rats from the different treatment groups. In the next phase, an additional 48 rats were selected and divided into the four model groups. At 18 h after the rat models were generated, the rats were anesthetized with 10% chloral hydrate through intraperitoneal injection and then euthanized. The 3-mm blood samples were collected from the abdominal aorta of euthanatized rats. The liver was also quickly extracted and stored in liquid nitrogen for further detection.
Isolation of mitochondria from rat liver
Liver specimens were quickly excised and washed with 0.9% normal saline solution (ice-cold). The liver was minced and homogenized into particles using a DIAX900 electric homogenizer (Heidolph, Germany) in a separation medium (consisting of 220-mM mannitol, 70-mM sucrose, 20-mM Tris–HCl (pH 7.4), 1-mM ethylenediaminetetraacetic acid, and 5-mM egtazic acid). The homogenate was centrifuged at 1000g for 10 min at 4°C, Then, the supernatant was centrifuged at 17,000g for 15 min at 4°C after the pellet was discarded. Next, the resultant supernatant was centrifuged at 17,000g for 15 min at 4°C, and the remaining pellets contained the mitochondria, which were washed twice. Then, mitochondria pellets were resuspended by adding 1.5 mL of isolation medium, and the samples centrifuged again at 12,000g for 10 min at 4°C. Finally, mitochondria were resuspended in 100 μL of mitochondria freezing medium after discarding the supernatant and immediately frozen in liquid nitrogen to measure mitochondrial function. Mitochondrial protein concentrations were determined by the bicinchoninic acid assay method. Purified mitochondria freezing medium (GMS12198) was provided by Shanghai Jiemei Gene Pharmaceutical Ltd, and BCA protein assay kits were provided by Biyuntian Biotechnology Ltd.
Ultrastructural morphological observation of mitochondria
Liver tissue was immersed in 2.5% glutaraldehyde in phosphate buffer (pH = 7.4) for 2 h. After washing in phosphate buffer for 20 min, samples were immersed again in 1% osmium tetroxide in phosphate buffer for 2 h. Next, all tissues were dehydrated with graded alcohol and embedded with embedding medium. Then, appropriately sized sections were prepared in Reichert-Jung Ultracut-E microtome (Reichert-Jung, Austria) and stained with 4% uranylacetate and 0.5% lead citrate, separately. Finally, electron micrographs were obtained using a JEOL JEM-1230 transmission electron microscope (JEOL Ltd, Japan).
Serum preparation and liver enzyme determination
Whole blood was collected from the abdominal aorta and placed at room temperature for 1 h to clot. Serum was separated by centrifugation at 3000g for 10 min and then stored at −80°C until use. To evaluate liver function, alanine aminotransferase (ALT), aspartate aminotransferase (AST), and mitochondrial aspartate aminotransferase (mAST) isoenzyme levels in serum samples were measured with an AU5800 automatic biochemical analyzer (Beckman Coulter, USA).
Assay of adenosine triphosphate content in liver cells
After mixture with 1 mL of adenosine triphosphate (ATP) lysate, 100 mg of rat liver portions were homogenized into particles using a DIAX900 electric homogenizer. The homogenate was centrifuged at 12,000g for 10 min at 4°C. Then, the supernatant was prepared for assessment of the ATP content in liver cells after discarding the precipitate. An equal amount of test solution was added to 100 μL of supernatant. The relative light units (RLU) value was immediately measured using a BioTek Synergy 4 multifunctional microplate reader (BioTek, USA), and the ATP content in the sample was calculated according to the standard curve of ATP concentration. The ATP detection kit (S0026) was provided by Biyuntian Biotechnology Ltd. The liver cell ATP content was measured using the ATP determination kit (Biyuntian Biotechnology Ltd, China) according to the manufacturer’s protocol.
Mitochondrial membrane potential measurements
The mitochondrial membrane potential (MMP) was determined using the mitochondrial dye JC-1. 24 JC-1 exhibits potential-dependent accumulation in mitochondria indicated by a fluorescence emission shift from green (530 nm) to red (590 nm). 25 Accordingly, mitochondrial depolarization is indicated by a decrease in the red/green fluorescence intensity ratio. MMP was measured using the JC-1 MMP assay kit (Biyuntian Biotechnology Ltd, China) according to the manufacturer’s protocol. Briefly, the mitochondria sample was diluted to 1 mg/mL mitochondria suspension with mitochondria dilution buffer at 4°C. Then, 0.1 mL of the mitochondria sample (mitochondrial density > 1 × 109) was added to 0.9 mL of JC-1 working solution (10 mg/mL), and MMP was detected using a BD FACS Verse Flow Cytometer (Becton Dickinson, USA). To detected the JC-1 monomer, the excitation light was set to 490 nm, and the emitted light was set to 530 nm. To measure the JC-1 polymer, the fluorescence ratio of 590 to 525 nm was used for quantitative analysis. 26
Measurement of ATP enzyme activity in mitochondrial membranes
Diluted mitochondrial samples were resuspended and sonicated at 4°C using an ultrasonic cleaning device. Mitochondrial membrane ATP enzymes activity was measured by colorimetry according to the instructions of the ATP enzyme assay kit (Nanjing Jiancheng Bioengineering Institute, China).
AQP8 expression levels as detected by reverse-transcription quantitative polymerase chain reaction
Total ribonucleic acid (RNA) was prepared from fresh tissue samples using the Trizol kit according to the manufacturer’s protocol (Invitrogen, USA). Briefly,1 mg of total RNA was used for the reverse-transcription reaction. The primers used for reverse transcription quantitative polymerase chain reaction (RT-qPCR) are listed in Table 1. Quantitative polymerase chain reaction (qPCR) was performed in a Real-Time PCR 7500 System (Applied Biosystems, USA) using SYBR Green I (TaKaRa). qPCR products were also analyzed on a 1.5% agarose gel containing ethidium bromide substitutes.
Primers of AQP8 and β-actin for qPCR.

AQP8: aquaporin-8; qPCR: quantitative polymerase chain reaction.
AQP8 expression levels as detected by western blotting
Isolated mitochondria protein samples (120 μg) prepared as described above were heated to 90°C and electrophoresed via 12% SDS-PAGE (Beyotime, China) with a low molecular weight protein ladder (Fermentas, Canada). Then, the resolved proteins were transferred electrophoretically onto polyvinylidene difluoride membranes that were blocked in 5% (weight/volume) bovine serum albumin (BSA) for 1 h. Next, the membranes were incubated with rabbit anti-rat AQP8 polyclonal antibody (Abcam Inc., USA) at a 1:500 dilution at 4°C for 12 h. The secondary antibody was goat anti-rabbit horseradish peroxidase-conjugated immunoglobulin G (IgG) (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd, China), and it was used at a dilution of 1:1000. Ultrasensitive enhanced chemiluminescent reagent (Beijing Full product speed biotech companies, China) was used for sample exposure and development. Image-Pro Plus software was employed for protein expression analysis and calculation of the relative AQP8 expression levels (AQP8 relative expression level = AQP8 band density/COX-IV band density × 100%).
Statistical analysis
All data were presented as mean ± standard deviation (SD) with a minimum of three independent experiments, and the differences between groups were evaluated using SPSS version 18.0 (USA). The optical density of the AQP8 band and the relative AQP8 messenger RNA (mRNA) levels among the four groups were compared by analysis of variance (ANOVA). Significance was determined using Kaplan–Meier survival analysis, Student’s t test, and one-way ANOVA (Tukey test). Pearson correlation analysis and simple linear regression analysis were also performed using SPSS version 18.0 (SPSS, USA) and Microsoft Office Excel 2007 (USA). Statistical significance was set at P < 0.05. Extreme statistical significance was set at P < 0.01.
Results
Survival time of different rat models
In total, 48 rats were equally divided into four groups: control group (C), sepsis group (S), treatment group (T), and prevention group (P). All rats were observed for no more than 48 h. Therefore, the survival time of the rats in control group was 48 h. However, in other groups, the survival time was defined by the time from the end of the operation to death. The survival time was 15.34 ± 2.35 h in the sepsis group, 23.20 ± 2.49 h in the treatment group, and 24.55 ± 1.86 h in the prevention group. Kaplan–Meier survival analysis revealed that the rats in the sepsis group died earlier than those in the treatment group and prevention group (P < 0.0001) (Figure 1).
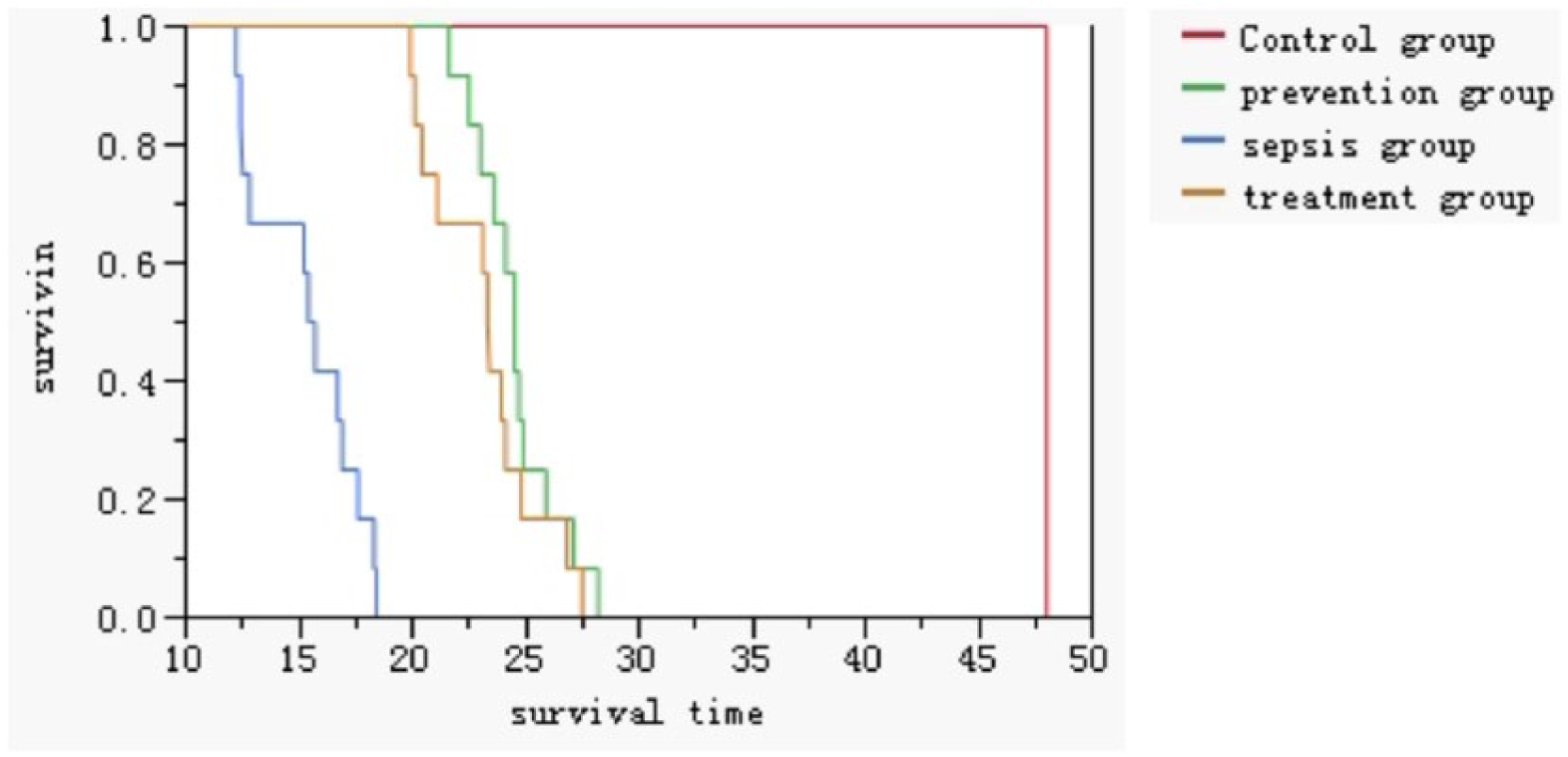
Survival time and Kaplan–Meier analyses among different groups of rats.
Hepatic character of septic rat model
After CLP treatment, numerous instances of rough, dilated, and ruptured endoplasmic reticulum were observed in rat hepatocytes via electron microscopy. The number of mitochondria was significantly reduced, and mitochondrial swelling occurred. Vacuolization and reduced cytoskeletal features were also noted. In addition, irregular nuclear-shaped protrusions were formed in the nuclear membrane, resulting in nuclear pyknosis (Figure 2(a)). The severity of hepatic dysfunction and mitochondria function was measured based on the serum ALT levels, serum AST levels, mAST levels, ATP content, and mitochondrial morphology in hepatocytes. Serum analysis revealed that ALT, AST, and mAST levels in the sepsis group were extremely significantly increased compared with the control group (P < 0.001) (Figure 2(b)). Furthermore, the ATP content in the sepsis group was extremely significantly decreased (P < 0.001) (Figure 2(c)). These results revealed that a sepsis model of liver injury was established successfully in Sprague Dawley rats.
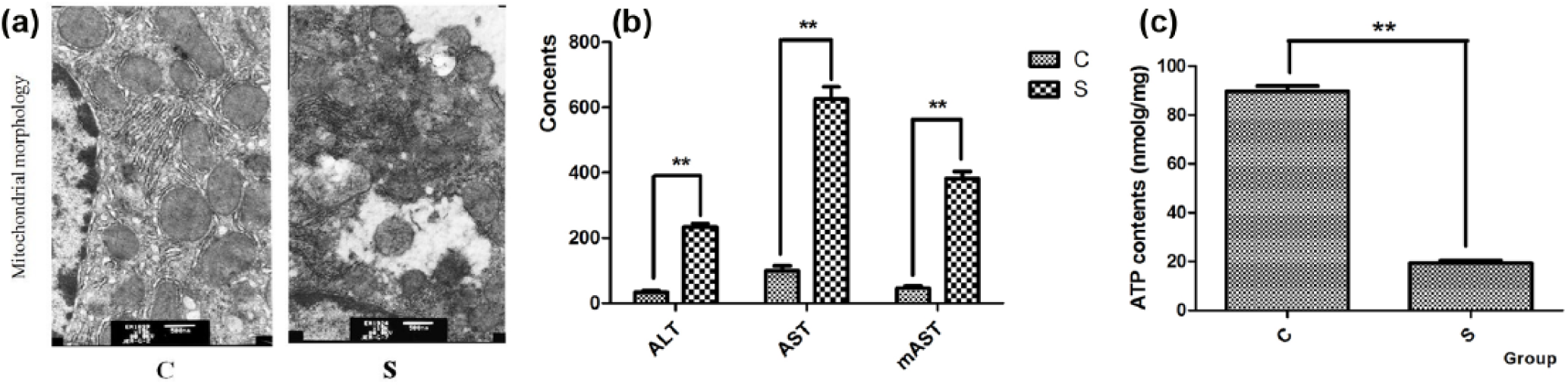
Related liver characteristics in a rat septic model generated via CLP: (a) mitochondrial morphology of hepatocytes in the control and sepsis group (magnification is 15,000 ×), (b) ALT, AST, and mAST levels in group C and group S, and (c) ATP content of hepatocytes in the control and sepsis groups.
Effect of TMP on liver mitochondria in septic rats
After TMP treatment, the mitochondria were slightly swollen with only individual vacuolization and no vacuoles. In addition, the endoplasmic reticulums of liver cells was slightly expanded and the nuclear deformity improved. In the treatment group, liver mitochondria were round and oval with almost no swelling and no vacuolization. The ridge exhibited a concentric, dense, well-defined, and vertical arrangement. Nuclear membrane integrity was noted, and the chromatin was distributed uniformly (Figure 3(a)). ALT, AST, and mAST levels were significantly increased at 18 h after the CLP operation (Figures 2(b) and 3(b)–(d)). These increases were also markedly reduced by the TMP treatment and TMP prevention (Figure 3(b)–(d)). Compared with the control group, the ATP content was extremely reduced in the sepsis group (P < 0.001). TMP treatment and TMP prevention could also improve the sepsis-induced reduced in ATP content in the rat model (Figure 3(e)). These results demonstrated that TMP prevention and treatment reversed sepsis-induced liver injury.

Effects of TMP on liver mitochondria in septic rats: (a) mitochondria morphology of hepatocytes in the TMP treatment group and TMP prevention group. Bar chart of (b) ALT, (c) AST, (d) mAST levels in serum from the control group (C), sepsis group (S), TMP treatment group (T), and TMP prevention group (P), and (e) ATP content in hepatocytes from the four groups (C, S, T, and P).
Effect of TMP on MMP and ATP enzyme activities in septic rats
The MMP in the four groups was measured using JC-1 staining. The ratio of red to green fluorescence intensity was used as an indicator of MMP. Stronger red fluorescence indicated that the MMP was increased. From Figure 4(a) and (b), the MMP in the sepsis group was lower than that in the control group. Moreover, TMP treatment and prevention could ameliorate this effect (Figure 4(a) and (b)).

Effects of TMP on mitochondrial membrane potential (MMP) and ATP enzyme activities in septic rats: (a) MMP was assess by flow cytometry in the four groups (C, S, T, and P), (b) MMP results are presented as the ratio of red and green fluorescence intensity, and (c) Na+-K+-ATPase, Mg2+-ATPase, Ca2+-ATPase, and Ca2+-Mg2+-ATPase levels in mitochondria in groups C, S, T, and P.
Some ATPases, such as Na+-K+-ATPase, are located in the inner mitochondrial membrane and are ion pumps that are important for mitochondrial structure and function. Compared with the control group, mitochondrial Na+-K+-ATPase, Mg2+-ATPase, Ca2+-ATPase, and Ca2+-Mg2+-ATPase activities were extremely significantly decreased (P < 0.01) in the sepsis group (Figure 4(c)). Compared with the sepsis group, mitochondrial Na+-K+-ATPase, Mg2+-ATPase, Ca2+-ATPase, and Ca2+-Mg2+-ATPase activities were extremely significantly increased in the treatment group and the prevention group (Figure 4(c)).
Effect of TMP on AQP8 and related inflammatory factors in septical rats
RT-qPCR and immunoblotting were performed to examine AQP8 expression at the mRNA and protein levels in the compared samples. Antibodies against β-actin and COX-IV were used as controls to normalize equal loading in the qPCR and immunoblotting experiments, respectively. As shown in Figure 5(a), compared with the control group, AQP8 mRNA levels were significantly reduced in the other three groups (P < 0.01), Compared with the septic group, AQP8 mRNA levels were significantly increased in the TMP treatment and prevention groups (P < 0.01). AQP8 protein levels were extremely significantly different (P < 0.01) between any two groups with the exception of the treatment and prevention groups (Figure 5(b)) in which a significant difference was noted (P < 0.05).

Quantification of the levels AQP8 and related inflammatory factors among groups C, S, T, and P: (a) AQP8 mRNA expression in the four groups, (b) AQP8 protein expression in the four groups. Western blotting results are present at the top of the figure. An anti-COX-IV antibody (15 kDa) was used as a control for equal protein loading. Densitometric analysis of AQP8 protein expression levels is presented at the bottom of the figure (data are expressed in arbitrary units as mean ± SE from 12 independent mitochondrial preparations per experimental condition). Bar chart of (c) IL-6, (d) TNF-α, and (e) HGMB1 levels in the four groups. Bar chart of (f) IL-1β and (g) NO levels in the four groups.
IL-6, IL-1β, TNF-α, HGMB1, and NO levels in the three experimental groups were also increased compared with the control group. However, TMP reduced the release of inflammatory factors and alleviated the excessive inflammatory response and stress (Figure 5(c)–(g)).
Correlation analysis among cytokines, AQP8 proteins, ATP content, and liver function
As shown in Figure 6, serum AST, serum ALT, and mAST levels decreased significantly with the increase in mitochondrial AQP8 proteins levels (Figure 6(a)–(c)). These results demonstrated significant positive correlations between AQP8 protein levels and the levels of markers of liver function. Furthermore, significant positive correlations were noted between liver cell ATPases levels and AQP8 proteins expression levels (Figure 6(d)) and between liver cell ATP levels and AQP8 protein expression levels (Figure 6(e)). Moreover, with respect to the inflammatory response, AQP8 levels also exhibited good correlations with NO (Figure 7(a)) and TNF-α levels (Figure 7(b)); in contrast, poor correlations were observed between AQP8 levels and IL-1β, IL-6, and HGMB1 levels.
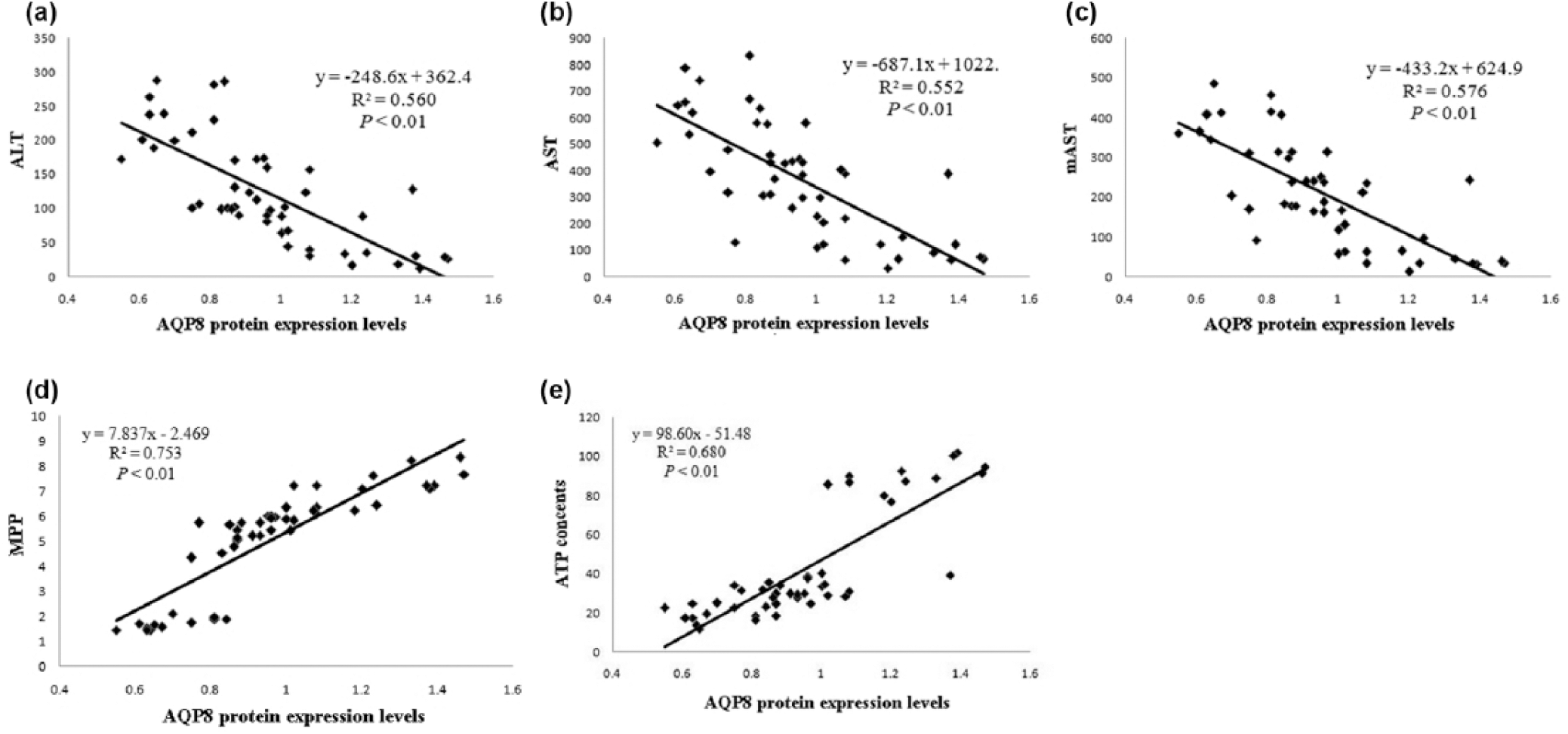
Correlation analysis among liver functions parameters (ALT, AST, and mAST levels and ATP content), MMP and AQP8 protein levels in the sepsis model: (a) correlation analysis between ALT levels and AQP8 protein expression levels, (b) correlation analysis between AST levels and AQP8 protein expression levels, (c) correlation analysis between mAST levels and AQP8 protein expression levels, (d) correlation analysis between MMP and AQP8 protein expression levels, and (e) correlation analysis between ATP levels and AQP8 protein expression levels.

AQP8 expression correlates with the levels of various inflammatory factors in the sepsis model: (a) linear correlation analysis between AQP8 protein expression levels and NO levels and (b) linear correlation analysis between AQP8 protein expression levels and TNF-α levels.
Discussion
A growing body of evidence from experimental studies and clinical data has demonstrated that excessive pro-inflammatory mediators and oxidative stress are implicated in various pathological conditions such as sepsis.27–29 During sepsis, the liver plays an important role in defensive responses to scavenge bacteria and produce inflammatory mediators. 30 Recent studies have revealed liver dysfunction as an early event in sepsis 31 and inflammation may induce a massive loss of hepatocytes and exacerbate the severity of various hepatic conditions associated with irreversible liver damage, fibrosis, and carcinogenesis 32 The liver is known to play a central role in metabolism of substances and energy and the host innate immune system. The liver is thought to be a major organ responsible for the initiation of sepsis. However, mitochondria play a central role in the intracellular events associated with inflammation and sepsis. 33 Reducing intramitochondrial ATP content in sepsis is a fundamental aspect contributing to organ dysfunctions. Given the strong association of altered liver functions with mitochondrial function, protection and improvements in mitochondrion function may tend to reduce MODS and sepsis.
In this study, CLP was performed to create a sepsis rat model that closely resembles the progression and characteristics of human sepsis. Our results demonstrated that serum ALT, serum AST, and mAST levels were significantly increased at 18 h after CLP. ATP content and MMP were significantly decreased at 18 h after CLP (Figures 2 and 4(b)). Mitochondrial morphology was also significantly altered (Figure 2(a)). These data suggested that a rat sepsis model was successfully established via CLP and obvious signs of liver and mitochondrial dysfunction were noted in septic rats. These results were consistent with those of a previous study. 29 However, the survival time results demonstrated that TMP could delay the onset time of sepsis in rats and increase the survival time after CLP operation in septic rats (Figure 1).
In addition to its vasodilatory and antiplatelet activities, TMP has recently been found to exhibit hepatic protection for multiple liver injuries and to function as an antioxidant, an anti-inflammatory agent, and a suppressor of apoptosis.10,34 In this study, the effect of TMP on liver function was further assessed to investigate the mechanism of Chinese medicine in the cure and prevention of sepsis. In Figure 3, we observed that hepatic mitochondrial morphology (Figure 3(a)) and serum ALT (Figure 3(b)), serum AST (Figure 3(c)), mAST (Figure 3(d)), and ATP (Figure 3(e)) levels were significantly improved in the TMP treatment and prevention groups. In addition, the levels of four related ATPases in the mitochondrial membrane (Na+-K+-ATPase, Mg2+-ATPase, Ca2+-ATPase, and Ca2+-Mg2+-ATPase) were significantly increased in the TMP treatment group and prevention groups compared with the sepsis group. In addition, as an index of ATP synthesis, the MMP was sensitive to toxic insults and was used to measure both cell viability and mitochondrial function. Reduced MMP represents a decreased ability of mitochondria to synthesize ATP. In this article, mitochondrial depolarization was indicated by a decrease in the red-to-green fluorescence intensity ratio using the JC-1 reagent. In Figure 4(a) and (b), MMP was increased in the treatment group and the prevention group compared with the sepsis group. These data demonstrate that TMP could reverse the damages in septic rats and was effective in treating sepsis.
To explore the molecular mechanism of TMP, the levels of some key inflammatory factors and mitochondrial proteins were further assessed. Five factors (IL-1β, IL-6, HGMB1, TNF-α, and NO), which were classically represent the inflammatory pathway and have been used frequently in previous studies, 35 were analyzed. Mitochondrial protein homeostasis can be perturbed, causing excessive ROS transport through the electron transport chain. This action directly damages protein folding and structure. Protein carbonyls formation could occur either through protein oxidative cleavage or direct oxidation of several amino acid residues. Therefore, we assessed mitochondrial AQP8 levels in the mitochondrial inner membrane and five factors in blood samples. Furthermore, some key ATPases were also detected. Moreover, linear correlation analysis was performed between related indexes and factors. In our study, the CLP operation significantly reduced mitochondrial AQP8 expression at the mRNA (Figure 5(a)) and protein (Figure 5(b)) levels. TMP treatment and pre-treatment alleviated the decline in AQP8 expression. However, the following descending order of expression levels for IL-1β, IL-6, HGMB1, TNF-α, and NO were observed in groups S, T, P, and C (Figure 5(c)–(g)). Na+-K+-ATPase, Mg2+-ATPase, Ca2+-ATPase, and Ca2+-Mg2+-ATPase expression improved after TMP treatment compared with sepsis alone. These phenomena demonstrate that the CLP operation caused a serious inflammatory response. Further linear correlation analysis revealed strong correlations of AQP8 expression with NO and TNF-α levels (Figure 7(a) and (b)). Similar relations between AQP8 and TNF-α were demonstrated in previous studies.36,37 We also identified the strong correlations of AQP8 expression with ATP content and the levels of various ATPases (Figure 6(a)–(e)). These data suggest that AQP8 plays a central role in the hepatoprotective effects of TMP in a sepsis model. In our opinion, AQP8 could accept signals from inflammatory factors upon stimulation and in response to various infections, and low AQP8 expression levels could result in further downstream mitochondrial dysfunction. Therefore, based on the results of this study, we suggest that TMP can ameliorate hepatocellular mitochondrial dysfunction by reducing the inflammatory response and increasing AQP8 protein expression.
Of note, a significant reduction in mitochondrial AQP8 mRNA level was detected in the septic rats after CLP in this study. However, interesting and controversial previous observations revealed increased AQP8 mRNA expression and reduced AQP8 protein expression after the CLP operation and lipopolysaccharide (LPS)-induced sepsis.13,20,38 One possible explanation for the observation of reduced AQP8 protein levels and increased AQP8 mRNA levels in previous studies is a common compensatory mechanism in septic hepatic mitochondria.20,39 These phenomena perhaps reflected a method-specific and cell-specific response, and little is known about the complete pattern of changes at the transcriptional level that occur during the development of acute sepsis. Additional studies will be required to assess the role of aquaporin (AQP) expression in human liver and to understand the transcriptional regulation of human and rat AQP in hepatocytes under pathophysiological conditions.
Based on previous reports and the results of this study, we postulated a model of the effect of TMP on liver mitochondrion in septic rats, as described in Figure 8. Injury factors, such as various infections and CLP operation, could induce an uncontrolled inflammatory response that leads to mitochondrial damage and liver dysfunction. We have highlighted the important role of AQP8 in this process, and we present novel findings that TMP can ameliorate the damage induced by sepsis through protecting and improving liver mitochondrial functions in rats. Such effects may be partially mediated by up-regulating the expression of AQP8 and inducing reduction in the inflammatory response.
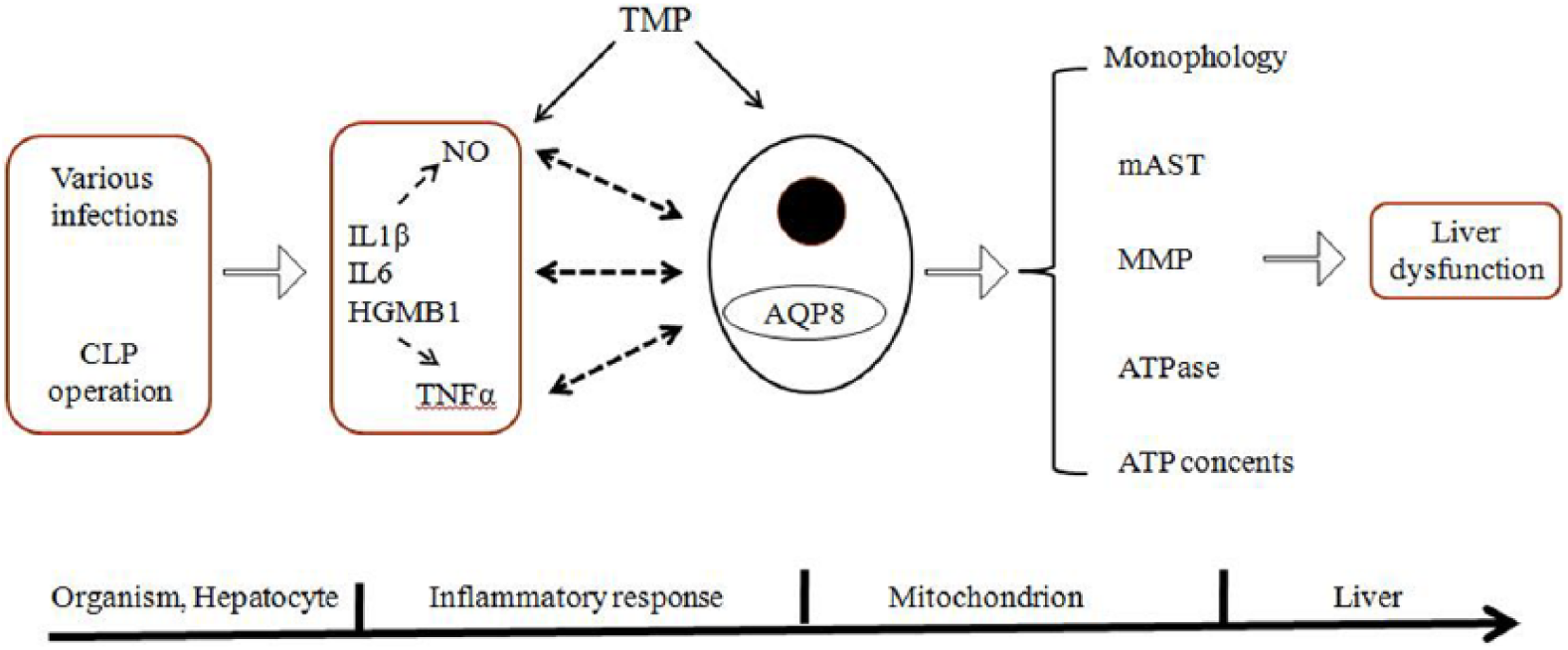
Postulated model of the effect of TMP on liver mitochondria in the rat sepsis model.
Our data suggest that TMP may serve as a starting point in the search for novel antioxidants for the treatment of sepsis and associated complications and TMP may be beneficial to sepsis patients in the future.
Footnotes
Acknowledgements
All authors equally contributed to the design, data acquisition, and manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the funds for International Science & Technology Cooperation Program of Anhui Province (No. 1403062022; Hefei, Anhui, China).
