Abstract
We studied the specific expression patterns and distributions of α1 and β1 integrin subunits, the major cell adhesion receptors in smooth muscle, in developing smooth muscle cells from 16-, 18-, and 20-day embryonic gizzards and from 1- and 7-day post hatch chick gizzards by SDS-PAGE, immunoblotting, and immunoelectron microscopy. Antibodies raised against α1 and β1 integrins isolated from avian gizzards were used as probes. Gels and blots showed that the amount of α1 and β1 integrins increased as age increased, with major increases at 1 and 7 days post hatch. Image analysis of immunoelectron micrographs demonstrated that statistically significant labeling increases occurred between embryonic Days 16 and 18, between embryonic Day 20 and 1 day post hatch, and between 1 day and 7 days post hatch. Immunolabeling with both anti-α1 and anti-β1 integrin was prominent at membrane-associated dense plaques (MADPs) and at filament anchoring regions at cell ends. This indicates that α1 and β1 integrin expression coincides temporally with the intracellular proliferation and reorientation of myofilaments. The similarity in distribution patterns of α1 and β1 integrins during development suggests that the two integrin subunits are synchronously expressed during development and do not appear sequentially.
I
The predominant integrins in chick gizzard smooth muscle cells are the 165-kD α1 (Syfrig et al. 1991) and the 130-kD β1 (Kelly et al. 1987) subunits. The α1-integrin subunit is both a primary receptor for collagen IV (Syfrig et al. 1991; Gardner et al. 1996) and a receptor for laminin in chick smooth muscle cells (Duband et al. 1992) and in PC12 cells (Colognato–Pyke et al. 1995). Expression patterns of α1-integrin in several types of developing avian embryonic tissues, including skeletal muscles, visceral, and vascular smooth muscles, have been investigated by using immunoblots and fluorescence microscopy (Duband et al. 1992). In developing skeletal muscle cells, α1-integrin disappeared when the cells became contractile (Duband et al. 1992), and α7-integrin appeared after the beginning of terminal differentiation (George–Weinstein et al. 1993). In visceral and vascular smooth muscle cells, however, integrin α1 was present early in differentiation and continued to be expressed in adult smooth muscle cells (Duband et al. 1992). The expression of α1-integrin in muscle was frequently correlated with expression of laminin and collagen IV. The ECM is evidently involved in the control of integrin expression. Vascular smooth muscle cells in the medial layer of normal arteries interact with collagen IV and express α1β1 integrins, but cultured cells from medial explants express only α2β1-integrins, which are needed for cell migration on collagen I substrates (Skinner et al. 1994). Fibroblasts from mutant mice without the α1-integrin gene could not spread or migrate on collagen IV substrates but could on collagen I substrates (Gardner et al. 1996). The essential receptor for collagen IV, therefore, is α1-integrin.
During embryogenesis and neonatal development, smooth muscle cells undergo remarkable phenotypic changes, including reorganization of the ECM (Risau and Lemmon 1988), changes in proportions of non-muscle and smooth muscle isoforms of cytoskeletal and contractile proteins (Glukhova et al. 1990), and changes in the intermediate filament protein desmin (Chou et al. 1994). Although β1-integrin functions primarily as a cell surface receptor for ECM molecules such as collagen, laminin, fibronectin, and vitronectin, the ligand binding specificity may vary among cell types (Elices and Hemler 1989). Immunofluorescence has demonstrated that β1-integrin is present in guinea pig T. coli smooth muscle membrane-associated dense plaques (MADPs) (North et al. 1993). MADPs are characterized by a smooth extracellular surface and an electron-dense plaque at the cytoplasmic face of the plasmalemma. Several proteins, including α-actinin, vinculin, talin, paxillin, tensin, zyxin, filamin, and tyrosine kinases, have been localized or have been proposed to be located at or associated with MADPs (for review see Small 1995). Therefore, MADPs are believed to be the smooth muscle equivalents of focal adhesions or focal contacts, where cell-cell and cell-matrix adhesions and interactions with cytoskeletal proteins are mediated. In addition, MADPs may be more complex than focal adhesions because MADPs also serve as anchors for intermediate filaments (Small 1995).
The objectives of this study were to investigate the expression patterns of both α1- and β1-integrins in embryonic and neonatal smooth muscle cells to determine if the α1- and β1-subunits were sequentially or synchronously expressed, to determine when the amounts of α1- and β1-integrins increased during development, and to localize both α1- and β1-integrins in these cells by using high-resolution immunogold labeling to determine when the subunits could be localized in relation to other assembling components in these cells.
Materials and Methods
Purification of Integrin α1- and β1-subunits
Turkey gizzard α1- and β1-integrins were prepared from stripped membrane fractions by the method of Kelly et al. (1987), with the following modifications. The pH 9.0 extraction step was done overnight at 4C instead of 30 min at 37C; 1 mM MgCl2 was added to the Triton X-100 extraction solution; and a HiTrap SP column (Pharmacia LKB Biotechnology; Piscataway, NJ) was used to purify β1-integrin from β1-integrin-enriched fractions rather than mono-S and mono-Q columns.
Antibodies
Electroeluted α1-integrin (0.2–0.3 mg) was dialyzed against PBS (2.6 mM KCl, 137 mM NaCl, 1.5 mM KH2PO4, 15.2 mM Na2HPO4, pH 7.2) before emulsification 1:1 with Freund's complete adjuvant (initial injection) and with Freund's incomplete adjuvant (booster injections) for sc injections into a New Zealand White rabbit. Animal care and management in these studies were done in compliance with the guidelines of the Iowa State University Committee on Animal Care. The β1-integrin (0.4–0.5 mg) without any adjuvant was injected directly into a sterile wiffle ball that had previously been implanted in the thoracic region of a New Zealand White rabbit (Clemons et al. 1991). After an initial injection and two booster injections, antisera against α1- and β1-integrins were collected. The β1-integrin polyclonal antibody was affinity-purified by using immunoblots as described in Harlow and Lane (1988). A polyclonal anti-α1 -integrin generously donated by Dr. M. Paulsson (University of Cologne, Germany) and a monoclonal anti-β1-integrin from the Hybridoma Bank were only used to identify α1- and β1-integrins in Western blots of column fractions. Neither of these antibodies was satisfactory for immunoelectron microscope labeling, thus requiring the production of α1- and β1-integrin antibodies for localization.
SDS-PAGE and Immunoblotting
Smooth muscle samples were taken from gizzards of 16-, 18-, and 20-day embryos and 1- and 7-day post hatch chicks. The integrin-containing Triton X-100 extract was prepared by the same method used with adult gizzards, except that homogenization steps were three times for 10 sec each. Protein concentrations were determined by the Bio-Rad Protein Assay (Bio-Rad; Hercules, CA). To minimize protein degradation, gels were run the day after preparation of the extracts. Electrophoresis was done on 10% polyacrylamide gels (acrylamide: N,N'-methylenebisacrylamide = 77:1 w/w) by a combination of the methods of Laemmli (1970) and Kelly et al. (1987). The same amount of protein (13 μg) from each sample was loaded on gels. After electrophoresis, proteins were transferred to a polyvinylidene fluoride (PVDF) membrane (Immobilon-P; Millipore, Bedford, MA) (Towbin et al. 1979). Silver-enhanced immunogold staining with anti-α1-integrin (1:10,000) and affinity-purified anti-β1-integrin (1:200) was done by the method of Moeremans et al. (1989).
Tissue Preparation for Immunoelectron Microscopy
Smooth muscle samples for immunoelectron microscopy were taken from gizzards of 16-, 18-, and 20-day embryos and from 1- and 7-day post hatch chicks. Tissues were prepared by a modification of the method of Stromer and Bendayan (1988,1990). Tissues from embryos could not be clamped because of the small size of the gizzards, but gizzard strips from post hatch samples were restrained at rest length in modified Lambert–Chalazion clamps. Samples were fixed with 2% paraformaldehyde, 0.05% glutaraldehyde in a modified Kreb's Ringer buffer, pH 7.1–7.3, for 2.5 hr at 2C. After fixation, the samples were rinsed and immersed in 0.15 M glycine for 1 hr to block free aldehyde groups. Dehydration in graded methanol, infiltration and embedding in Lowicryl K4M resin, and polymerization were done by the method of Bendayan (1984).
Immunoelectron Microscopy
The polyclonal antibody to avian gizzard α1-integrin was used at 1:50 dilution, and the affinity-purified polyclonal antibody to avian gizzard β1-integrin was used undiluted. The protein A-12-nm gold complex was produced by the method of Bendayan (1984). Labeling was done as described in Stromer and Bendayan (1988,1990) and in Bendayan (1984). Controls, which included substituting preimmune sera from the same rabbits used for antibody production and substituting PBS for the primary antibody, showed virtually no labeling and provided an indication of background levels. Six blocks were randomly selected from each age for thin sectioning. Sections and labeling were examined at 80 kV in a JEOL JEM-100CXII electron microscope. Images were recorded on Kodak SO-163 electron image film.
Sample Evaluation
A minimum of 25 attachment plaques were measured and colloidal gold particles associated with each plaque were counted for each animal age. Plaques were identified in randomly selected micrographs, and the number of gold particles per μm2 of plaque area was determined for both embryonic and neonatal samples. To compare labeling intensities of plaques, where plaque area could readily be determined, with labeling associated with filament anchoring sites at the ends of cells, the area of which could not be determined, we counted the number of gold particles per μm of cell surface in each of these sites in neonatal samples. The t-test was used to determine statistical significance.
Results
Purification of α1- and β1-integrins
The α1-integrin-rich fractions (no. 60 to no. 110) from the DEAE-cellulose column were chromatographed on a hydroxylapatite column to remove some high and low molecular weight contaminating proteins. SDS-PAGE analysis showed that α1-integrin was prominent in hydroxylapatite column fractions no. 45 to no. 70. The α1-integrin band was cut out of the gel and the protein was recovered by electroelution. The β1-integrin-rich fractions (no. 140 to no. 160) from the DEAE–cellulose column were chromatographed on a HiTrap SP column. Contaminating proteins were retained by the HiTrap SP column, and β1-integrin was in the flow-through.
Characterization of α1- and β1-integrin Antibodies
Immunoblotting with anti-α1-integrin showed that only the α1-integrin subunit was recognized (Figure 1, Lane 2). Immunoblotting with anti-β1-integrin heavily labeled the β1-integrin subunit but also weakly labeled some minor components (Figure 1, Lane 3). To ensure the specificity of the β1-integrin antibody, affinity-purification was done. The affinity-purified antibody recognized only the β1-integrin band in blots (Figure 1, Lane 4). The α1- and β1-integrin antibodies from outside sources identified α1- and β1-integrins, respectively, in the column fractions (results not shown).
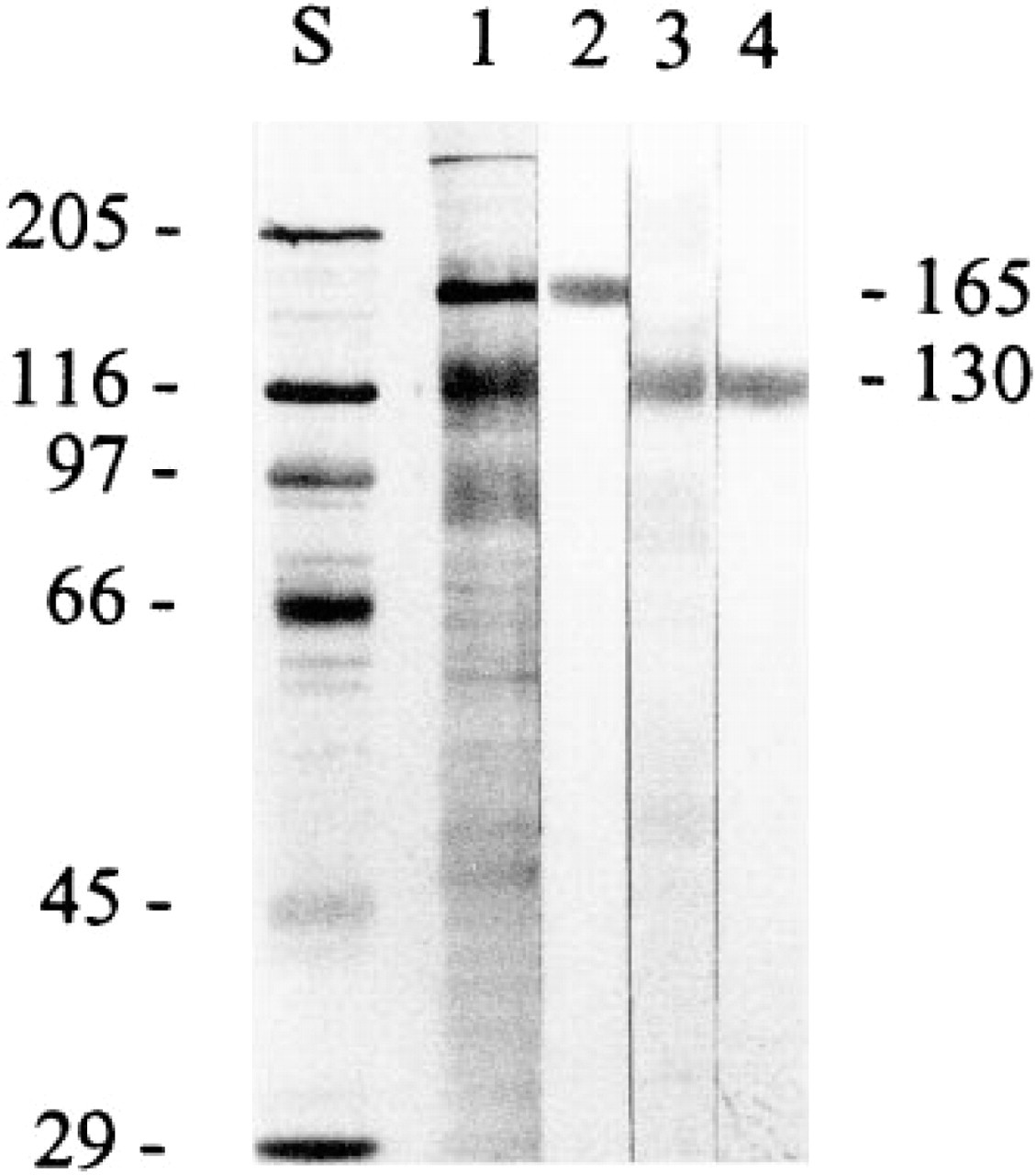
Characterization of α1- and β1-integrin antibodies. Lane S, which contains molecular weight standards, and Lanes 1–4, which are smooth muscle membrane protein extracts from adult gizzards, were transferred to a PVDF membrane. Lanes S and 1 were stained with Coomassie Brilliant Blue. Lane 2 was labeled with anti-α1-integrin. Lane 3 was labeled with anti-β1-integrin. Lane 4 was labeled with affinity-purified anti-β1-integrin. Molecular masses of avian α1- and β1-integrins are 165 kD and 130 kD, respectively.
Changes in α1- and β1-integrin Expression
Results from SDS-PAGE (Figure 2A) and from immunoblots (Figure 2B) of stripped membrane fractions demonstrated that the 165-kD α1-integrin band is already present in relatively small amounts as a percentage of total membrane proteins at embryonic Day 16 and that amounts of α1-integrin increase rapidly as age increases. The increase in the 130-kD β1-integrin band (Figure 2C) with increasing age shows a similar pattern. Both α1- and β1-integrins increase markedly after hatching (cf. Lanes 4 and 5 vs Lanes 1, 2, and 3 in Figures 2B and 2C). It is evident that, compared with the embryonic and neonatal samples in Lanes 1–5 in Figure 2, adult gizzard smooth muscle cells (Lane 6) contain more of both α1- and β1-integrin.
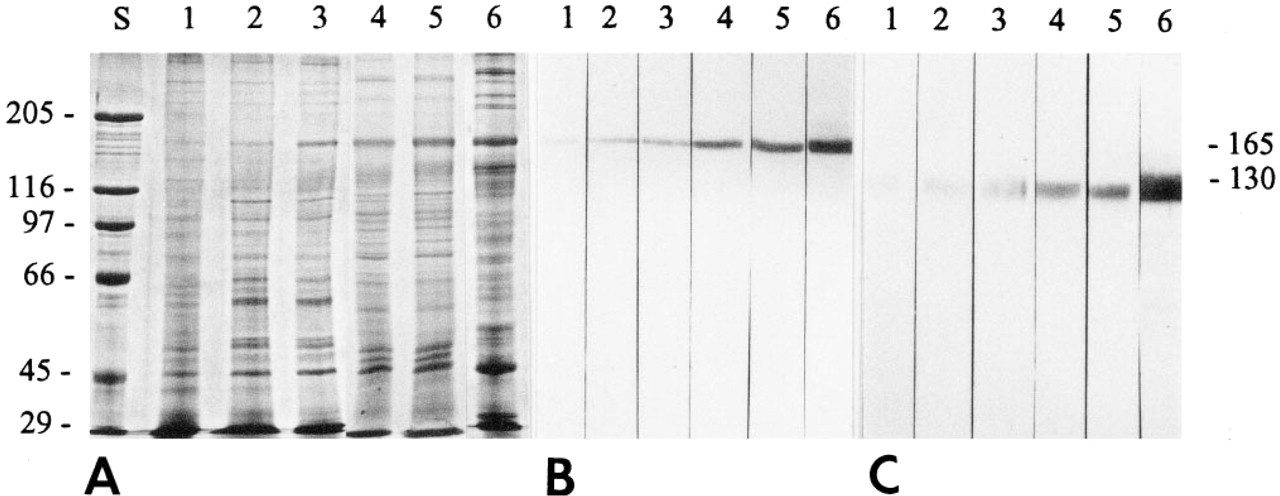
Change in the content of α1- and β1- integrins in smooth muscle membrane extracts from 16-, 18-, and 20-day embryos and 1- and 7-day post hatch cells (Lanes 1–5, respectively). For comparison, Lane 6 contains a membrane extract prepared in the same way from adult gizzards. Lane S contains molecular weight standards. (
Immunoelectron Microscopic Labeling
Because both the density and the pattern of labeling with antibodies to α1- and β1-integrins were very similar (see Tables 1 and 2), we have chosen to include micrographs from each animal age that show typical labeling with anti-α1-integrin only. Representative micrographs from each sample are shown in Figure 3, and quantitation of the labeling obtained is shown in Tables 1 and 2.
Immunogold labeling of attachment plaques with α1-and β1-integrin antibodies
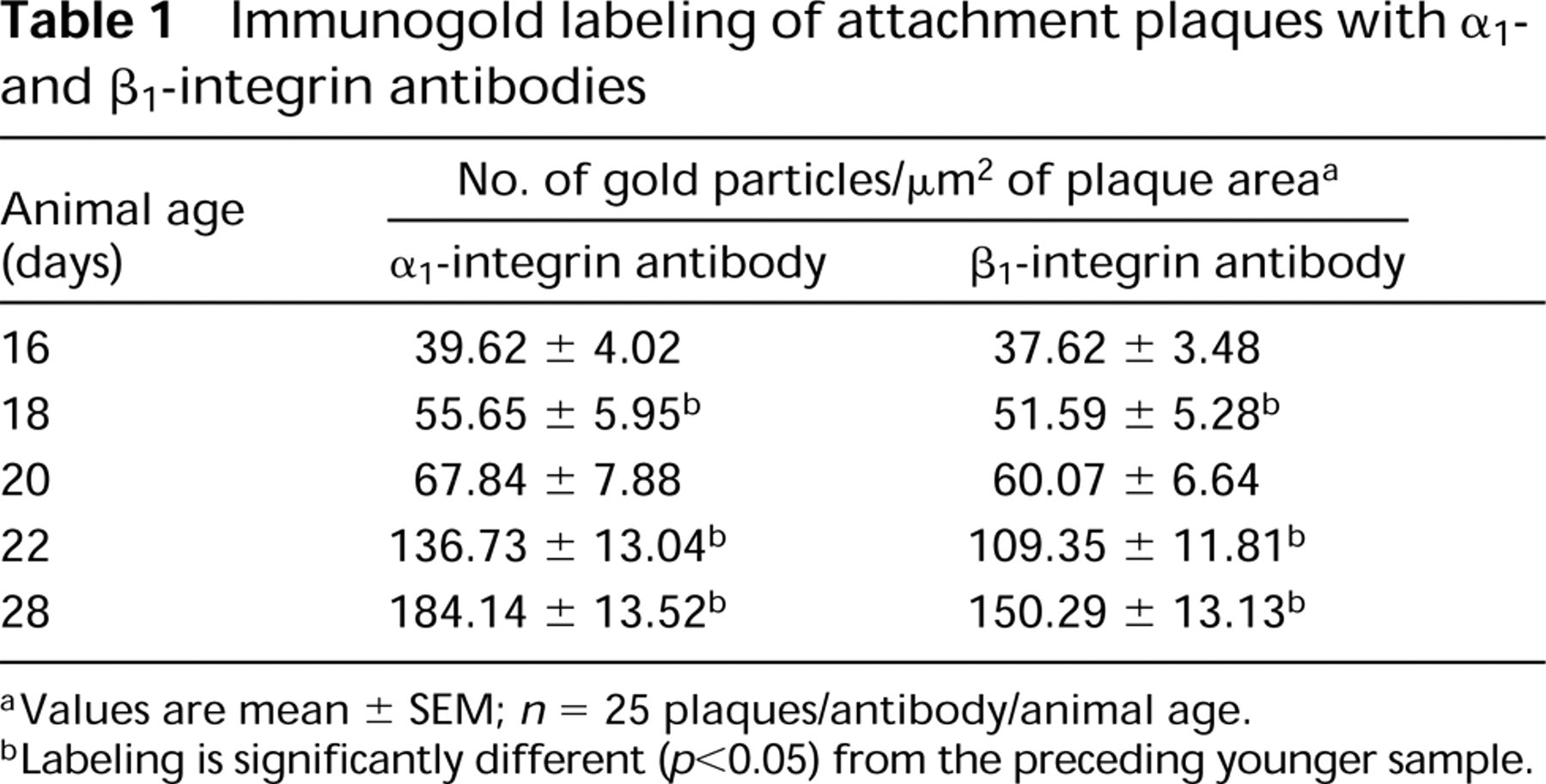
Values are mean ± SEM; n = 25 plaques/antibody/animal age.
Labeling is significantly different (p<0.05) from the preceding younger sample.
Comparison of immunogold labeling with α1- and β1-integrin antibodies at cell ends and plaques a
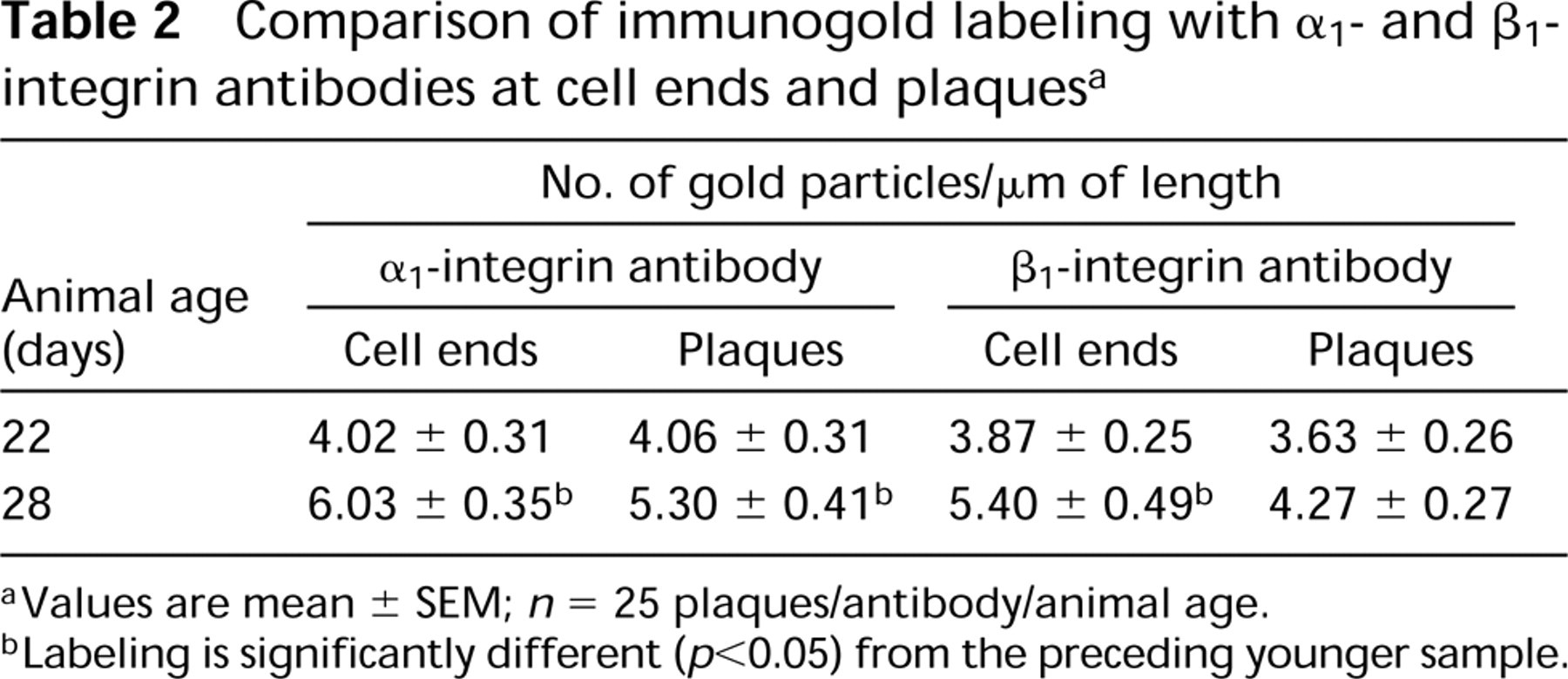
Values are mean ± SEM; n = 25 plaques/antibody/animal age.
Labeling is significantly different (p<0.05) from the preceding younger sample.
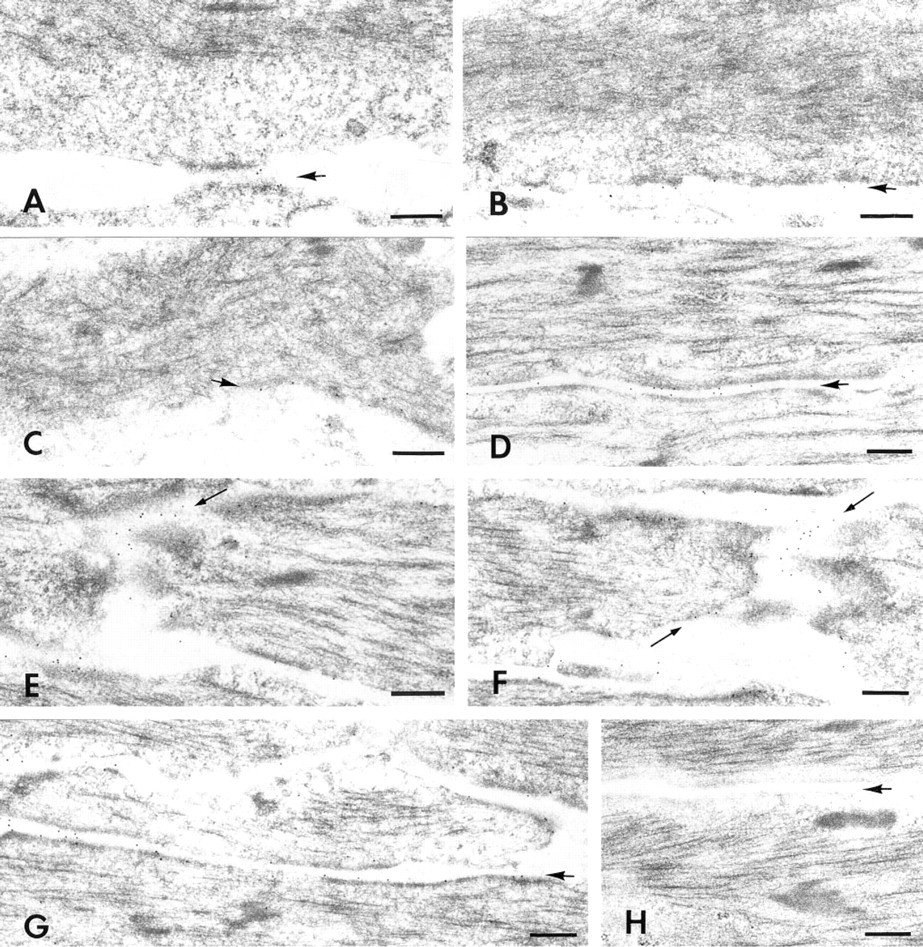
Immunogold labeling of developing smooth muscle cells with anti-α1-integrin. (
Attachment plaques were consistently labeled at all developmental stages included in this study. Sixteen-day embryonic smooth muscle cells contain both large areas of cytoplasm that have no recognizable filaments and relatively small, somewhat diffuse attachment plaques (Figure 3A). The lowest density of labeling was associated with these immature plaques. At 18 days, embryonic cells contain more filaments that are associated with plaques (Figure 3B) and labeling intensities have increased significantly over that seen at 16 days (Table 1). Plaques increase further in both size and density by 20 days of embryonic development (Figure 3C) and, although labeling density has increased (Table 1), the increase is not statistically significant. By 22 days of development (1 day post hatch), labeling associated with plaques (Figure 3D) increased significantly by 1.8 to 2 times that observed 2 days earlier. Additional significant increases in label density occurred by 28 days of development (7 days post hatch) (Table 1; Figure 3G). The overall increase in plaque labeling from Day 16 to Day 28 was 4.6 times with anti-α1-integrin and 4 times with anti-β1-integrin.
As the size of the filament compartment increased during development, filaments became oriented more nearly parallel to the longitudinal cell axis and the intracellular patches of filaments expanded towards the ends of the developing smooth muscle cell. Cell ends were a second site that was consistently labeled by anti-α1- and anti-β1-integrins. By 22 days of development (1 day post hatch), the linear boundary of the cell end was adequately developed to permit reproducible measurements of the end boundary and to compare labeling at the cell ends with labeling at the attachment plaques. In 22-day samples (Figure 3E), the number of gold particles per μm of plaque and of attachment area at the cell end was virtually identical with both antibodies (Table 2). Labeling densities per μm of plaques and cell ends in 28-day (7 days post hatch) samples were greater than in 22-day samples (Table 2; Figure 3F). Plaque labeling increased approximately 30% with the α1-integrin antibody and 20% with the β1-integrin antibody. In contrast, the cell end-labeling increased 50% with the α1-integrin antibody and 40% with the β1-integrin antibody. The increases were statistically significant at cell ends with both antibodies and at plaques with anti-α1-integrin.
Discussion
We have used SDS-PAGE, immunoblotting, immunoelectron microscopy, and image analysis to demonstrate that expression of both α1- and β1-integrin increases synchronously as age of developing smooth muscle cells increases. Our SDS-PAGE and immunoblots both show a consistent increase in both α1- and β1-integrin in gizzard smooth muscle cells from embryonic Day 16 through 7 days post hatch. This steady increase differs from the patterns observed by Belkin et al. (1990) in smooth muscle cells from developing human aorta. In the human aorta, amounts of both α1- and β1-integrins were lower at 24 weeks of gestation than at 10 weeks, increased again after birth, and reached adult levels by 1.5 years after birth. Although the relative developmental stages in the human smooth muscle studied by Belkin et al. (1990), in which decreases in integrins were observed are not directly comparable with the avian developmental stages studied herein, we found no decrease in α1- and β1-integrin expression. We also show that the timing of increased synthesis of α1- and β1-integrins coincides with the increase in intracellular filaments and the reorientation of these filaments in a direction more nearly parallel with the long axis of the cell. The low level of labeling we observed in 16-day embryonic cells coincides with the appearance of intracellular patches of myofilaments that occupy only a small central portion of the cell. Between 20 and 22 days (1 day post hatch) of development, a major increase in cell volume occupied by myofilaments is accompanied by the greatest increase in integrin content. The timing of the increase in myofilaments in these smooth muscle cells from the digestive tract is probably dictated by the need to crush and grind feed taken in by mouth after hatching. The myofilaments in smooth muscle cells consist of both contractile and cytoskeletal filaments (Small 1995). To permit cell shortening and generation of force, these filaments must be anchored. It has been suggested that both cytoplasmic β-actin filaments (North et al. 1994) and intermediate filaments (Bagby 1983) are anchored in or associated with attachment plaques. The simultaneous increase in myofilaments and in integrin content of attachment plaques is consistent with the proposal that integrins are mechanical linkages between the cytoskeleton and the extracellular matrix (Horwitz et al. 1986; for reviews see Ingber 1991; Gumbiner 1996).
The patterns of increase in labeling from 10 to 28 days of development were very similar for α1 and for β1 antibodies (Tables 1 and 2). Although it has previously been predicted that α1- and β1-integrins should be synchronously expressed during development, we know of no published reports in which this has been demonstrated. The slightly lower labeling of attachment plaques (Table 1) and cell ends (Table 2) with anti-β1-integrin is probably due to a slight loss in binding activity after affinity purification rather than a real difference in amount of α1- and β1-integrins. This interpretation is supported by Briesewitz et al. (1995), who found that, for correct integrin receptor assembly, the cytoplasmic domains of both α1- and β1-integrin are required. This suggests that α1- and β1-integrin should be present during development in similar if not identical amounts, and is consistent with our immunoblots (Figures 2B and 2C).
We also observed that, as increasing numbers of collagen fibrils became visible in the extracellular space, intensity of integrin labeling at attachment plaques and cell ends increased. Although collagen fibrils were not seen in association with all attachment plaques, we observed an increase in collagen fibrils near plaques from one to three at embryonic Days 16, 18, or 20 to six or seven collagen fibrils near plaques at 1 and 7 days post hatch. This increase coincided with the major increase in integrin labeling (Table 1). It has been demonstrated that α1β1-integrin dimers are collagen IV receptors (Syfrig et al. 1991; Eble et al. 1993; Kern et al. 1993; Gardner et al. 1996) and that the α1β1-integrin binding site is approximately 100 nm from the N-terminus of collagen IV (Vandenberg et al. 1991). The basement membrane associated with smooth muscle cells contains, among its many components, fibronectin, laminin, and collagen IV (Carey 1991). It has also been shown that interaction with collagen enhances the in vitro differentiation of vascular smooth muscle cells (Sakata et al. 1990) and influences the shape of cultured aortic smooth muscle cells (Akita et al. 1993). Therefore, the increased intensity of integrin labeling as collagen fibrils become more numerous near the developing smooth muscle cells studied herein supports a role for the integrin–extracellular matrix interaction in differentiation as described by Duplàa et al. (1997).
Footnotes
Acknowledgements
Supported in part by a grant from the American Heart Association, Iowa Affiliate.
We thank Dr M. Paulsson (University of Cologne, Germany) for providing the α1-integrin antibody. We also thank Mary Sue Mayes, Suzy Sernett, Jo Philips, and Lynn New-bold for excellent technical assistance.
This is Journal Paper J-17204 of the Iowa Agriculture and Home Economics Experiment Station, Ames, IA 50011, Projects 3349 and 2127.
