Abstract
Laminins (Ln), together with Type IV collagen and nidogen-1, form the structural integrity of the basement membranes (BM). In this study we used immunohistochemistry to show the distribution of laminin chains α1, α3, α5, β1, β2, β3, γ1, γ2, as well as Type IV collagen, in various types of carcinomas and in normal tissues. Except for diffuse gastric carcinomas and infiltrative breast carcinomas, the malignant epithelial tumor clusters were surrounded by quite a continuous BM in most tumors. These BMs comprised most abundantly Ln α5, β1, and γ1 chains. Conversely, the Ln α1 chain, a component of laminins-1 and -3, showed the most restricted distribution in BMs of both normal tissues and malignancies, being moderately present in carcinomas of thyroid gland and ovary and in intraductal carcinomas of breast. In other types of carcinomas, immunoreactivity for Ln α1 chain was found more randomly and was practically negative in carcinomas of tongue, stomach, and colon. These findings were comparable to those observed by in situ hybridization, which showed that carcinomas of thyroid gland and intraductal carcinomas of breast constitutively expressed Ln α1 mRNA and that the epithelial tumor cells were the main producers of it. The results suggest that epithelial malignancies, except for infiltrative breast and diffuse gastric carcinomas, produce more notable amounts of BM macromolecules in their growth substratum than has previously been anticipated. Corroborating their widespread distribution in normal epithelial tissues, the chains of Lns-5 and -10 are the most abundant Ln molecules in the corresponding carcinomas.
Basement membranes (BMs) form a ubiquitous and highly specialized network in the extracellular matrix (ECM). As important structural barriers, they additionally possess many functional properties and take part in cell adhesion, migration, molecular ultrainfiltration, and signaling, and are synthesized and deposited, together with adjacent epithelial and stromal cells, in a highly organized manner. The laminins (Lns) are an expanding group of large BM glycoproteins that are associated with Type IV collagen through nidogen-1. In addition, various proteoglycans (PGs) are associated with BMs (Colognato and Yurchenco 2000). The Ln molecule consists of a large molecular weight α chain to which lighter β and γ chains are joined to generate a heterotrimeric laminin molecule, either cruciform or T-shaped. We currently know of 11 Ln chains in the human, α1–5, β1–3, and γ1–3, forming 14 Ln heterotrimers, and others are likely to exist (Burgeson et al. 1994; Miner et al. 1997; Koch et al. 1999; Libby et al. 2000).
A linear BM normally separates the epithelium from the adjacent mesenchyme and surrounds the nerves, vessels, and some individual cell types such as adipocytes, decidual and muscle cells, and Schwann cells. The original observation of Barsky et al. (1983), who showed discontinuities to be present in the BMs of malignant tumors but not in those of their benign counterparts, was followed by several confirmatory reports (Cam et al. 1984; Dahler et al. 1987). The BM is the first structural barrier that carcinoma cells must penetrate to pass from one compartment to another. The fragmentation or total absence of BM structures seen in malignant tumors is therefore believed to be partially due to active proteolytic degradation by tumor cells (Chambers and Matrisian 1997; Murphy and Gavrilovic 1999). Decreased synthesis of BM macromolecules, along with their altered assembly into BMs in tumor tissues, may also be involved in modulation of the invasive potential of malignant cells.
Antisera against Ln isolated from an Engelbreth–Holm–Swarm (EHS) tumor have been widely used to detect the immunohistochemical distribution of Ln in human tissues. Such antisera have usually been considered to reveal the Ln-1 molecule in immunostaining, but they also detects Ln γ1 chain complexed with other α chains than α1, which explains why they stain all BMs (Aumailley and Smyth 1998; Erickson and Couchman 2000). Later, the monoclonal antibody (MAb) 4C7, which was raised before there was any knowledge of the existence of Ln heterotrimers other than Ln-1, was also believed to detect the Ln α1 chain, and antisera against EHS Ln and MAb 4C7 have therefore been used for many morphological studies (Engvall et al. 1986, 1990; Hewitt et al. 1997; Virtanen et al. 1997). However, gene expression studies aimed at specific detection of mRNA for the human Ln α1 chain have shown a rather restricted distribution for the Ln α1 chain (Nissinen et al. 1991; Vuolteenaho et al. 1994). This discrepancy was resolved when the 4C7 antibody was demonstrated to actually detect the Ln α5 chain (Tiger et al. 1997; Church and Aplin 1998; Kikkawa et al. 1998). There are antibodies that detect the Ln α1 chain and, in line with the mRNA expression results, they show that the Ln α1 chain in both mouse and rat tissues is present only in certain epithelial BMs (Klein et al. 1988; Ekblom et al. 1990; Falk et al. 1999). This has been shown to be the distribution pattern also in human tissues in which Ln α1 chain is expressed by certain epithelia and the muscle–tendon junction (Pedrosa–Domellöf et al. 2000; Virtanen et al. 2000).
Because the Ln α1 chain expression in human tumor BMs has not been clearly presented due to the fact that many previous investigations have been made using the MAb 4C7 we collected a series of tumor samples to clarify systematically the role of Ln α1 chain in malignancies by immunohistochemistry and to compare its distribution to other Ln chains. In situ hybridization (ISH) was also used to confirm Ln α1 chain mRNA expression in corresponding tumors. Here we show that only certain malignancies contain the Ln α1 chain in their BMs and that epithelial cells express its mRNA. In general, moreover, malignant tumors display a similar Ln chain composition to that in their normal tissues.
Materials and Methods
Materials
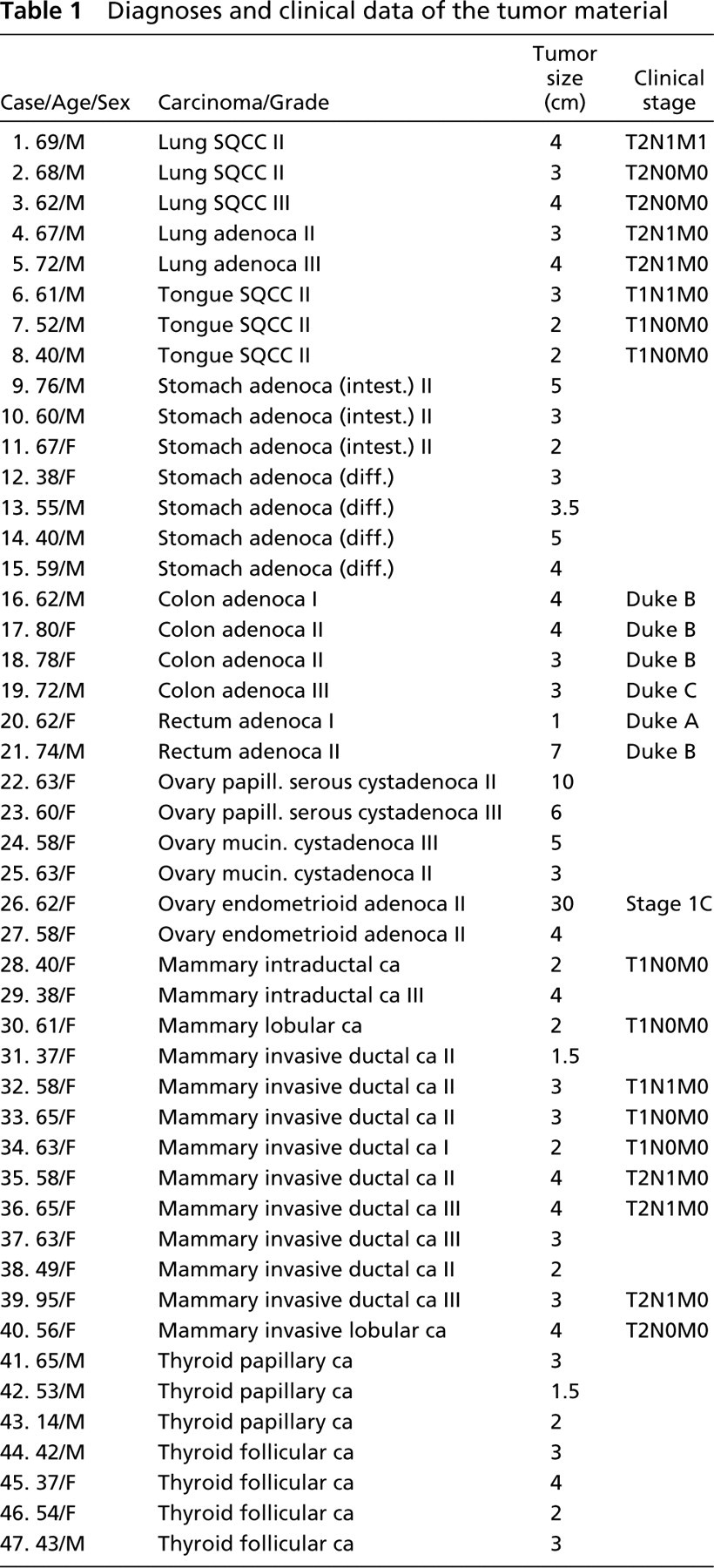
The material used here was collected from the files of the Department of Pathology, Oulu University Central Hospital, for the years 1995–1999. The diagnoses had been based on light microscopy of routine hematoxylin–eosin-stained sections and had been classified and graded according to the WHO classifications. The material comprised 47 malignant tumors: five lung carcinomas [three squamous cell carcinomas (SQCCs) and two adenocarcinomas], three SQCCs of the tongue, seven gastric adenocarcinomas (three intestinal-type and four diffuse-type), six colorectal adenocarcinomas, six ovarian adenocarcinomas (two mucinous and two serous cystadenocarcinomas and two endometrioid adenocarcinomas), 13 breast carcinomas (10 ductal carcinomas comprising invasive and intraductal carcinomas and three lobular carcinomas), and seven carcinomas of the thyroid gland (four follicular and three papillary adenocarcinomas). The samples for immunohistochemical staining had been frozen immediately and kept in liquid nitrogen at –80C until used. The corresponding paraffin-embedded sections were used for ISH experiments. The material and clinical data are presented in Table 1.
Antibodies
The MAbs used in this study were as follows: MAb against the Ln α1 chain (clone 161 EB7) (Virtanen et al. 2000); MAb detecting Ln β1 (PLB1) (Virtanen et al. 1997); MAb against Ln α3 (clone BM-2) (Rousselle et al. 1991); MAb for Ln β3 (clone 6F12) (Marinkovich et al. 1992); MAb detecting Ln α5 chain (Engvall et al. 1986; for its specificity see Tiger et al. 1997; Church and Aplin 1998; Kikkawa et al. 1998) purchased from Chemicon (Temecula, CA); and MAb against Ln β2 chain (clone C4) (Hunter et al. 1989) purchased from Developmental Studies Hybridoma Bank (Iowa, City, IA). The polyclonal antibody (PAb) for the Ln γ2 chain (Pyke et al. 1995) was a kind gift from Professor Karl Tryggvason (Karolinska Institute; Stockholm, Sweden) and the PAb detecting the 7S domain of Type IV collagen (Risteli et al. 1980) was kindly provided by Professor Juha Risteli (University of Oulu, Finland). A new MAb against the Ln γ1 chain (113BC7) was raised and is characterized here.
Production and Characterization of MAb 113BC7
The new MAb against the Ln γ1 chain (113BC7) was raised using a pepsin digest of human placental Ln (Wewer et al. 1983). Balb/c mice were immunized with ~50 μg of the antigen in Freund's complete adjuvant, followed by two immunizations with Freund's incomplete adjuvant. The spleens of the immunized mice were then collected and suspended cells were fused with X63-Ag8–653 myeloma cells by standard techniques (Köhler and Milstein 1976).
Diagnoses and clinical data of the tumor material
To demonstrate the specificity of the new MAb, hybridomas were screened by immunoprecipitation, SDS-PAGE, and immunoreactivity on selected human tissues, and positive hybridomas were cloned manually under the microscope. JAR human choriocarcinoma cells (American Type Culture Collection; Manassas, VA) were then radioactively labeled with [35S]-methionine (Amersham Pharmacia Biotech; Uppsala, Sweden) and the labeled culture medium was subjected to reduction and alkylation to dissociate the laminin-1 and laminin-10 trimers secreted by this cell line (Tani et al. 1999). The supernatant was then subjected to immunoprecipitation using the hybridoma supernatant. For this purpose GammaBind-Sepharose (Amersham Pharmacia Biotech) was pretreated with the hybridoma supernatant by standard techniques. The bands were then mixed with the culture medium and after the washes the binding was detected by SDS-PAGE (Laemmli 1970) using 5% gels under non-reducing conditions. After electrophoresis the gels were subjected to fluorography. As seen in Figure 1, MAb 113BC7 precipitated two high-Mr α chains (α1 and α5; Figure 1, Lane 1) as well as Mr 220,000 and Mr 200,000 β1 and γ1 chains. On the other hand, when the reduced alkylated culture supernatant was precipitated with MAb 113BC7 under non-reducing conditions, only Mr 200,000 Ln γ1 chain was seen (Figure 1, Lane 2). For further domain specificity see Geberhiwot et al. (2000).
Immunohistochemistry
For immunohistochemistry, 4-μm frozen sections were airdried for 15 min, followed by fixation in cold acetone for 10 min and then air-drying, and were incubated with fetal calf serum (FCS) (Hyclone Laboratories; Logan, UT) 1:5 in PBS for 20 min to block nonspecific binding of IgG. The sections were then incubated with the primary antibody at 4C for 2–20 hr, followed by biotinylated anti-mouse or anti-rabbit IgG secondary antibody (DAKO; Glostrup, Denmark) for 30 min and avidin–peroxidase complex for 30 min. The color was developed with diaminobenzidine tetrahydrochloride–H2O2 (DAB) (Sigma; St Louis, MO) in Tris buffer, pH 7.4. Finally, the sections were lightly counterstained with hematoxylin. As a negative control, the sections were treated as described above but PBS was used instead of the primary antibody.
Indirect Immunofluorescence
Indirect immunofluorescence stainings were performed using the MAb against Ln β2 chain. Unfixed frozen tumor samples of 4 μm were first washed twice in PBS for 15 min and then incubated with FCS (Hyclone Laboratoires) for 20 min. The primary antibody was applied overnight, followed by incubation with fluorescein isothiocyanate (FITC) coupled with rabbit anti-mouse secondary conjugate (DAKO) for 30 min, and finally the sections were washed in PBS for 30 min and mounted. In the control stainings, PBS was used instead of the primary antibody. An Olympus microscope, model BHT, together with a fluorescence epi-illuminator, was used for analysis and photography of the stainings. The microscope was equipped with a mercury lamp (HBO 100 W), a dichroic mirror B (DM-500) with a built-in barrier filter (O-515), and an excitation filter (EY 455) for FITC fluorescence.
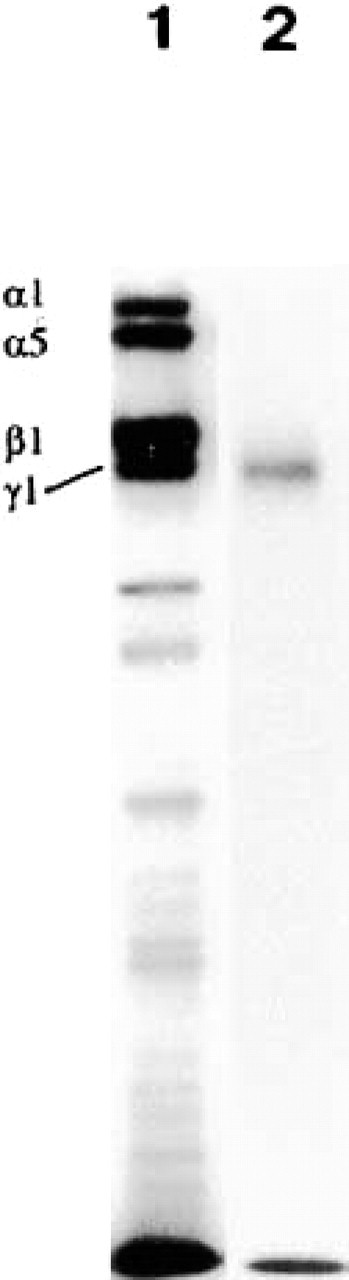
The MAb 113BC7 against Ln γ1 chain precipitated two high-Mr α chains (α1 and α5, Lane 1) as well as Mr 220,000 and Mr 200,000 β1 and γ1 chains, respectively. When the reduced, alkylated Ln culture supernatant was precipitated with the MAb 113BC7 under non-reducing conditions only the Mr 200,000 Ln γ1 chain was seen (Lane 2).
Riboprobes
The riboprobes used for ISH were as follows: a 348 Sal I-Not I fragment (bases 3100–3448) for the Ln α1 chain (Nissinen et al. 1991); a 906 Pst I fragment of clone HL2 (bases 920–1826) for the Ln β1 chain (Pikkarainen et al. 1987); an 845-bp Pst I-EcoR I fragment of clone L15 (bases 2995–3840) for the Ln γ2 chain (Kallunki et al. 1992); and a 916 BamH I-Hind III fragment of clone HT21 (bases 528–1444) for the α1(IV) chain (Pihlajaniemi et al. 1985). All the cDNAs were ligated into the M13 polylinker site of pSP64 and pSP65 vectors (Promega; Madison, WI) except for Ln α1, for which the pBluescript II SK vector (Stratagene; La Jolla, CA) was used. All probes were tested by Northern hybridization (not shown).
In Situ Hybridization
Paraffin sections were deparaffinized by incubation in xylene followed by dehydration. The sections were then treated with 0.2 M HCl at room temperature (RT) for 20 min and washed in diethylpyrocarbonate-treated water (DEPC-H2O) for 5 min. For proteolysis, the sections were incubated with proteinase K (1 mg/ml) (Roche Diagnostics; Indianapolis, IN) for 30 min at 37C and the reaction stopped with 0.2% glycine in PBS. After that, the sections were washed twice in PBS for 30 sec, fixed in 4% paraformaldehyde in PBS for 20 min, and washed again in PBS. Acetylation was performed in 0.25–0.5% acetic anhydride in 0.1 M triethanolamine for 10 min, after which the sections were washed in 1 × PBS, dehydrated, and air-dried for 1–2 hr at RT. The sections were then treated for 2 hr with the prehybridization mixture (10 mM dithiothreitol (DTT; Sigma), 10 mM Tris-HCl, 10 mM NaPO4, 5 mM dinatrium-diamin-tetra-acetic acid (EDTA), 0.3 M NaCl, 1 mg/ml yeast tRNA, deionized formamide 50%, and dextran sulfate 10% (w/v), 0.02% (w/v) Ficoll (Amersham Pharmacia Biotech), 0.02% (w/v) polyvinylpyrrolidone, and 0.02 mg/ml bovine serum albumin (BSA) and washed in 1 × PBS and dehydrated. In the hybridization step the probes were first denatured by boiling for 1 min and placed on ice. Approximately 3 × 106 cpm of the [35S]-labeled antisense or sense probe in 40 μl prehybridization buffer was applied to each section and the hybridization was carried out at 50C overnight. The posthybridization washes were performed as follows: twice at 50C for 1 hr in prehybridization mixture except for dextran sulfate and tRNA, 15 min in TE (0.5 M NaCl in 10 mM Tris-HCl, 1 mM EDTA) at 37C, 30-min incubation in 0.5 M NaCl in TE containing 40 μg/ml RNase A (Sigma) at 37C, washing for 15 min in 0.5 M NaCl in TE at 37C, 15 min twice in 2 × standard saline citrate (SSC), and 15 min twice in 1 × SSC, both at 50C. The sections were dehydrated in ethanol containing 300 mM ammonium acetate and air-dried at RT for 1 hr. In autoradiography, the slides were dipped into NTB-2 film emulsion (Kodak; Rochester, NY) and then placed in light-tight boxes for 10–14 days. They were developed in GBX developer (Kodak), fixed in Agefix (Kodak), and counterstained with hematoxylin and eosin.
Labeled probes in sense orientation were always used as negative controls for each case in every experiment.
Evaluation of the Results
The immunohistochemical and immunofluorescence staining reactions were analyzed by two observers. Adjacent normal structures were analyzed if present.
Immunohistochemical staining intensity was assessed by taking into account the intensity of staining in the BMs and the latitude of the staining reaction, and was scored as negative (–) when the staining reaction was indistinguishable from the background, weak (+) when the reaction was slightly increased or expression was observed only in short strips of BMs, moderate (++) when a clear reaction was observed, and strong (+++) when the reaction was abundant and observed generally in all BMs.
Results
Semiquantitative results regarding the presence of Ln and Type IV collagen in the tumor BMs and normal structures are presented in Tables 2and 3, respectively.
Laminin Chains in Carcinomas
Lung Carcinomas. All the Ln chains α3, α5, β1 (Figure 2B), β2 (Figure 2D), β3, γ1 (Figure 2E), and γ2 (Figure 2C) in SQCCs of the lung were moderately or strongly present in the BMs around tumor islands. The expression pattern for the various Lns was in most cases linear, but discontinuities were seen in some invasive tumor islands. There was no significant difference in the BM staining reaction between large tumor clusters and smaller invasive islands. Ln α1 was linearly present in the BMs of occasional tumor islands (Figure 2A), although it was generally absent from tumor BMs. Weak to moderate expression was found for the β2 chain in tumor BMs (Figure 2D). In one case, Ln γ2 had an intracellular staining pattern in some tumor cells situated in the peripheral parts of tumor clusters (Figure 2C). In some areas there was a diffuse immunoreactivity for laminin chains α3 and β3 in the stroma around tumor islands (not shown).
In the two adenocarcinomas of the lung, all the Ln chains, except for the α1 chain, were present in the tumor BMs. The staining reaction was mostly fragmented and contained clear disruptions. The Ln γ2 chain was also detected intracellularly in tumor cells in close contact with the BM and in the tumor stroma.
Presence of laminin (Ln) chains and type IV collagen (IVc) in tumor basement membranes
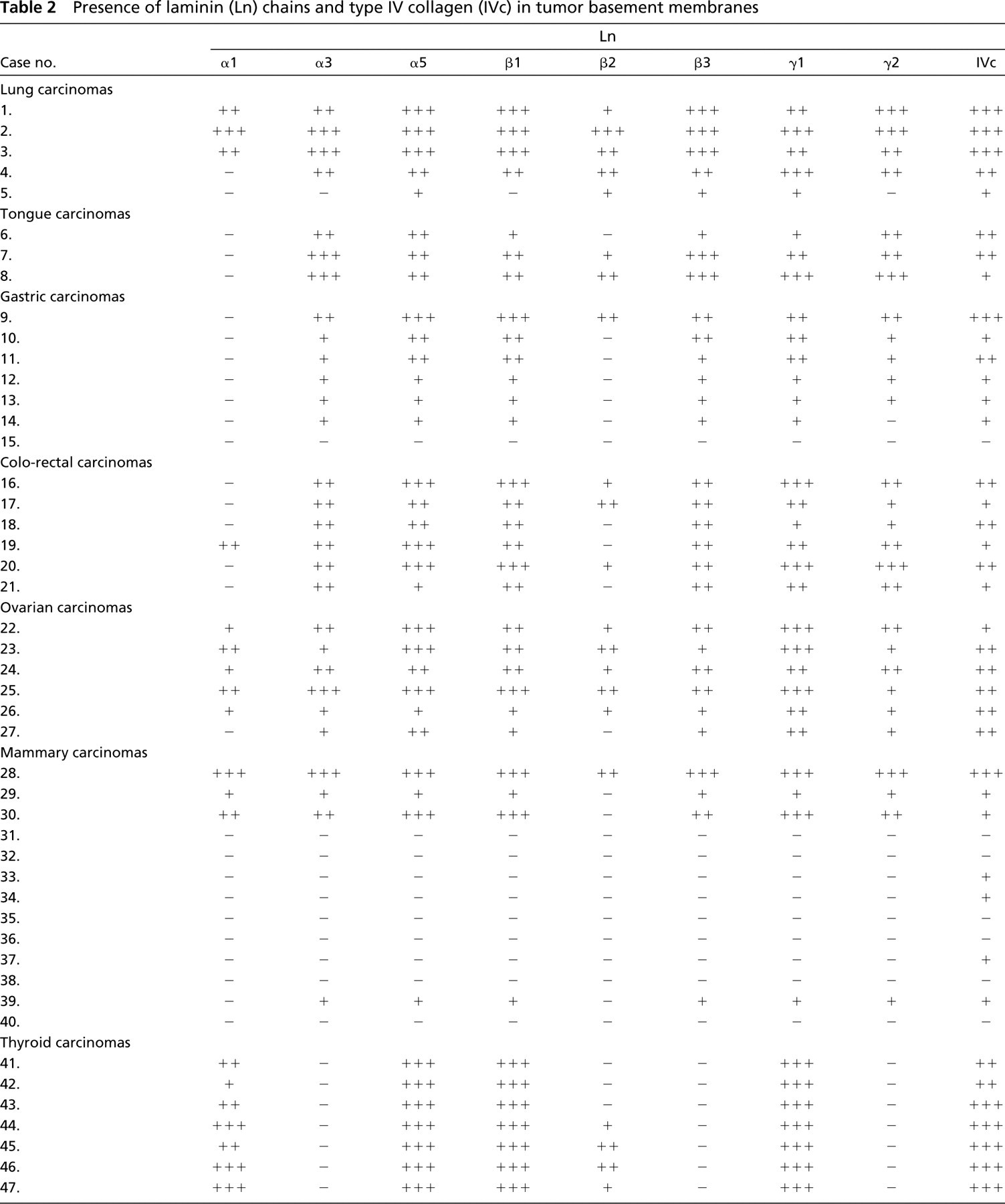
Type IV collagen was present in abundance in all the lung carcinomas and generally showed a linear staining pattern, but there were discontinuities in some invasive areas. The small and larger tumor islands expressed Type IV collagen in a similar manner. In some areas the immunoreactivity for Type IV collagen was seen to be thicker in the tumor BMs than in those of normal bronchi or peribronchial glands.
Presence of laminin (Ln) chains and type IV collagen (IVc) in basement membranes of normal structures a
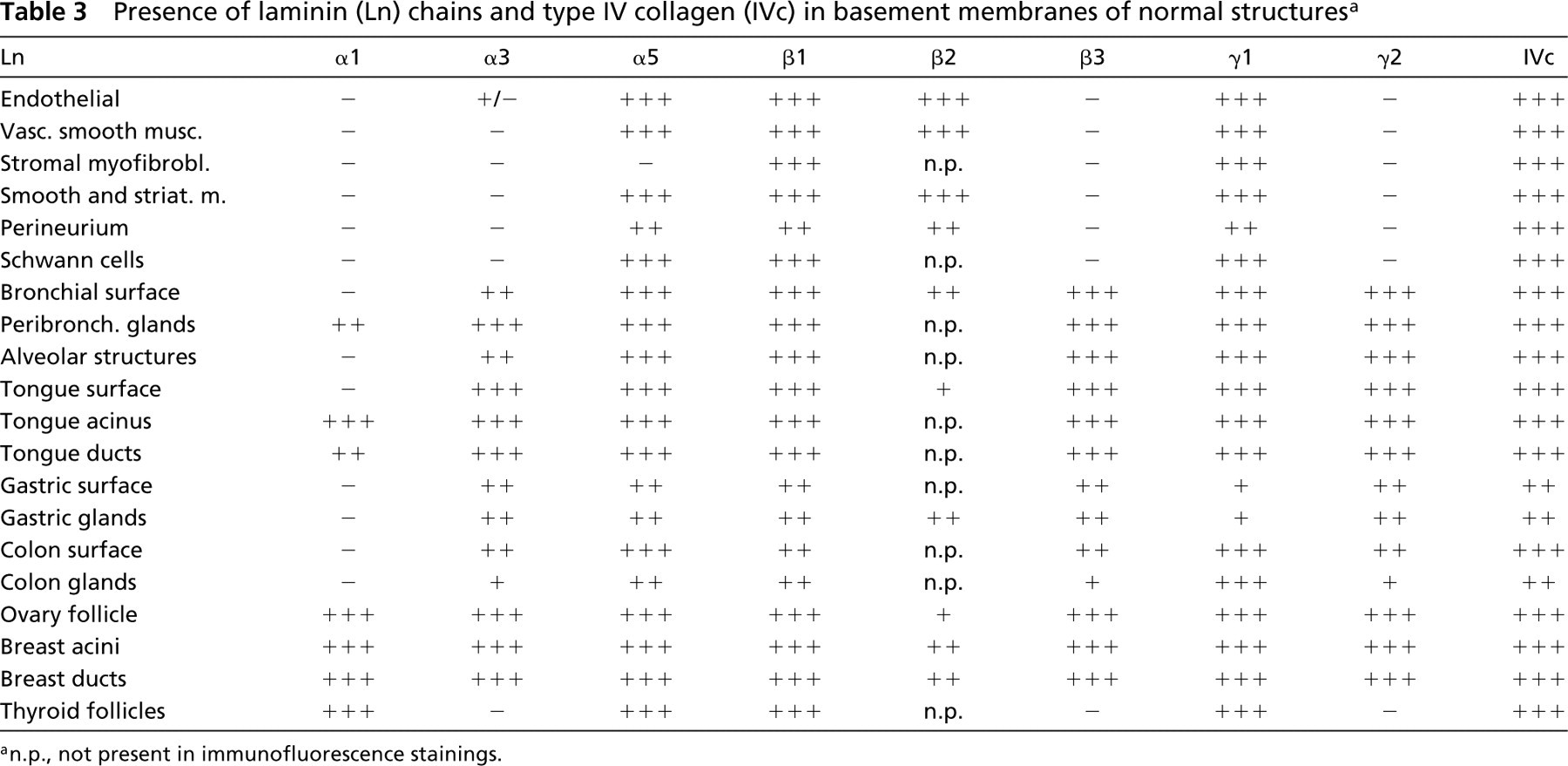
an.p., not present in immunofluorescence stainings.
Tongue Carcinomas. All the Ln chains, except for α1, were present in the tumor BMs of SQCCs of the tongue (Figures 3A, 3B, and 3D). In areas of obvious invasion, however, the immunoreactivities were clearly weaker or totally absent, showing a disrupted pattern. Larger tumor islands showed linear reactivity of their BMs. In Case 6, in which the growth pattern was more diffuse than in the other specimens of tongue carcinoma, the BM reaction was clearly reduced and was generally lacking in the invasive areas. These areas were also characterized by intracytoplasmic staining of carcinoma cells for Ln chains α5, α3 (Figure 3A), β1, and γ2. The intracytoplasmic staining reaction for Ln γ2 seemed to be more pronounced in the paraffin-embedded sections, and the diffusely scattered tumor cells in invasive areas exclusively expressed intracytoplasmic Ln γ2 (Figure 3D).
Type IV collagen showed a comparable result to that seen in the Ln stainings, being poorly expressed or totally lacking in the smaller invasive tumor islands, although the larger clusters were surrounded by quite linear, well-formed BMs (Figure 3C).
Gastric Carcinomas. The intestinal-type gastric carcinomas were totally negative for the Ln α1 chain, whereas the other Ln chains were seen to be present in the tumor BMs. The most intense expression was observed for the α5 (Figure 4A), β1, and γ2 (Figure 4B) chains, which showed a fairly continuous staining pattern. Other chains were more limited in their expression, but when present they showed quite linear staining of the tumor BMs. The Ln α3 chain differed from the others by showing a more fragmented staining pattern. Type IV collagen was present in about 80% of the tumor glands, but especially in areas showing active invasion through the muscularis mucosa, the tumor clusters were characterized by highly discontinuous or totally absent immunoreactivity for Type IV collagen.
There were diffuse-type gastric carcinomas in which there was no extracellular BM material that was positive for either for Lns or Type IV collagen. In these cases there was, however, intracellular positivity for Lns α3 (Figure 4C), β2, and γ2 (Figure 4D) in some diffusely scattered individual carcinoma cells. Other diffuse carcinoma samples had very weak and fragmented but visible BM structures for all the other Ln chains except for Ln α1 in areas forming trabecules and small clusters within the mucosal part of the tumor, but the more diffuse infiltrative parts did not contain any BM material.
Colorectal Carcinomas. The Ln α1 chain was present in the tumor BMs in only one case (Case 19), and was totally lacking in the remaining five cases. Ln β2 was seen in 3/6 tumors. When present, both of these Ln chains were present around almost 70% of the neoplastic glands. Ln chains other than α1 and β2 were moderately to strongly present in the BMs in all cases. The α5, β1 (Figure 5A) and γ1 chains were linearly expressed around tumor clusters, whereas the α3, β3 (Figure 5B) and γ2 chains had in some areas a more disrupted staining pattern, showing fragmentations, especially in the invasive areas. The Ln γ2 chain showed intracytoplasmic positivity in tumor cells close to the tumor stroma in all the samples. Type IV collagen was weakly to moderately expressed by tumor BMs, which variably showed disruptions and fragmentations.
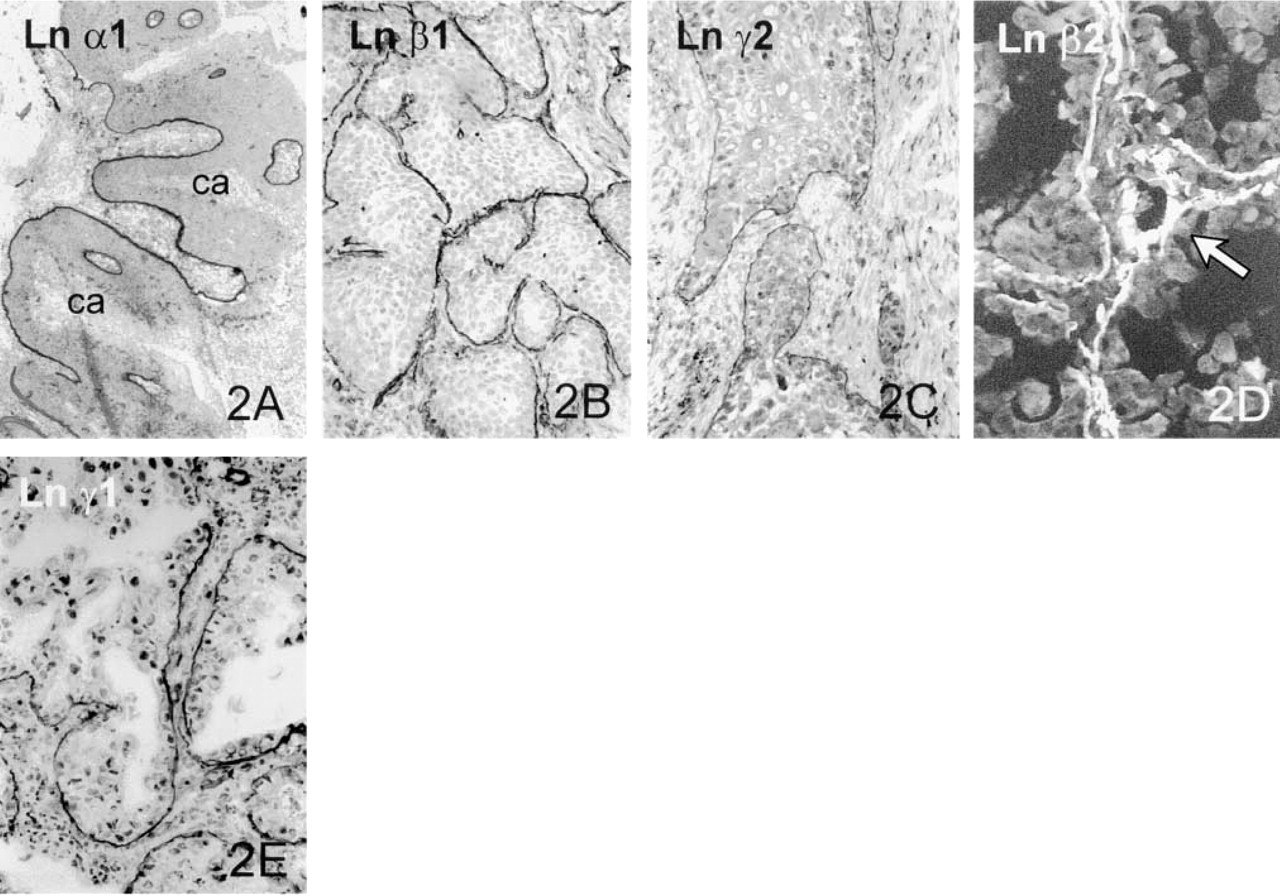
Lung carcinoma. Linear BMs are seen around carcinoma islands (ca) of the grade II SQCC using Ln α1 (
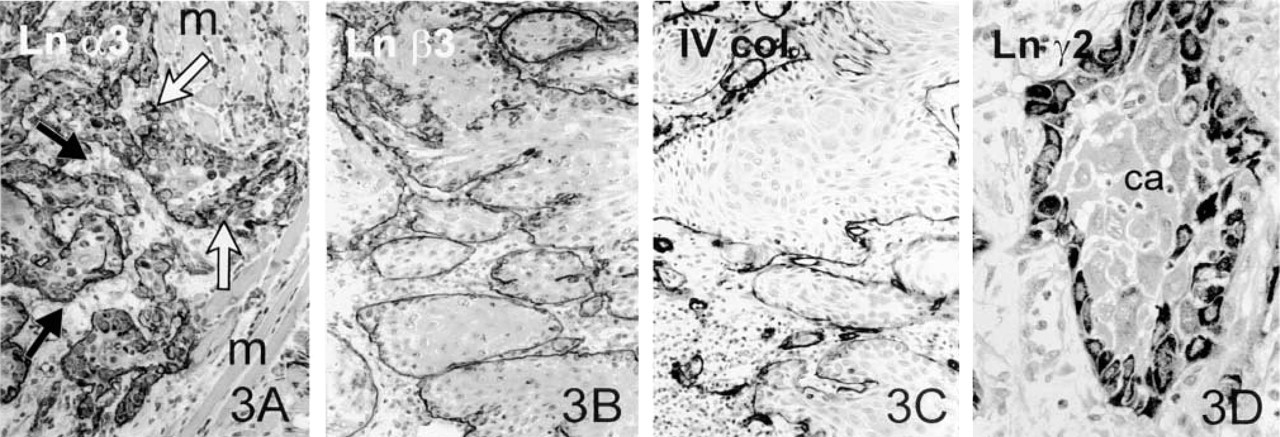
Tongue carcinoma. Immunohistochemical staining for the Ln α3 chain in the grade II SQCC of tongue (
Ovarian Carcinomas. In general, all the ovarian carcinomas expressed all the Ln chains, except for one case which lacked Ln α1. In the serous papillary cystadenocarcinomas, α1, α3, β2, β3 (Figure 6A) and γ2 chains were present in about 50% of the neoplastic structures, while α5, β1, and γ1 (Figure 6B) chains were seen in over 80% of the tumor BMs. Only the Ln α5 chain antibody stained the tumor BMs fairly linearly (not shown), whereas the other Ln chains had focal disruptions in their staining pattern. The Ln γ2 chain was also seen intracytoplasmically in tumor cells located opposite the tumor stroma.
All Lns were present in the tumor BMs of the mucinous cystadenocarcinomas. In this tumor type, Ln α1 was seen only in about 30% of the neoplastic glands and the other Ln chains in 50–70%. The most continuous staining pattern was seen for the Ln α5 chain, although all chains showed disruptions in their BM stainings to various degrees, and there were areas of active invasion in which only small fragments of BM positivity could be seen.
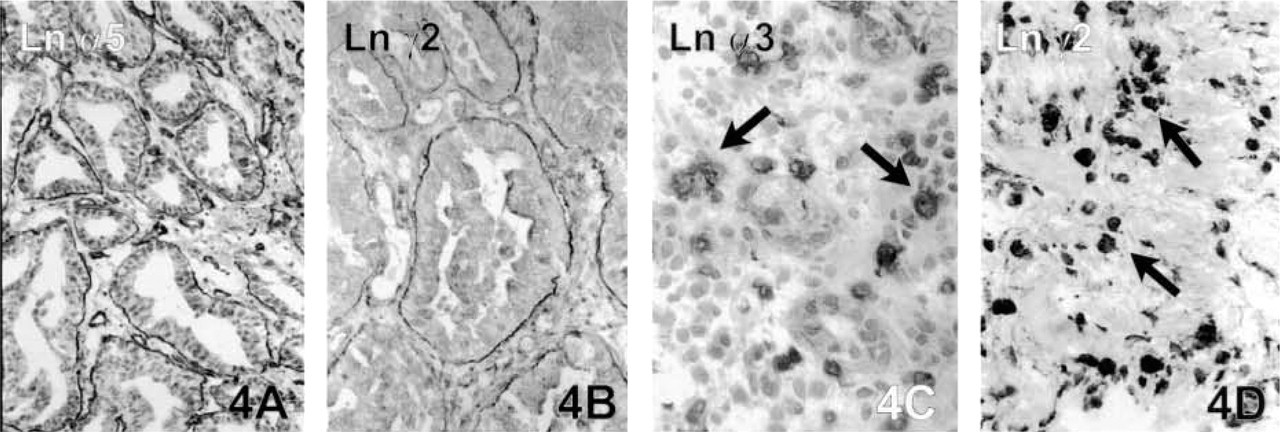
Gastric adenocarcinoma. Linear BMs with Ln α5 (
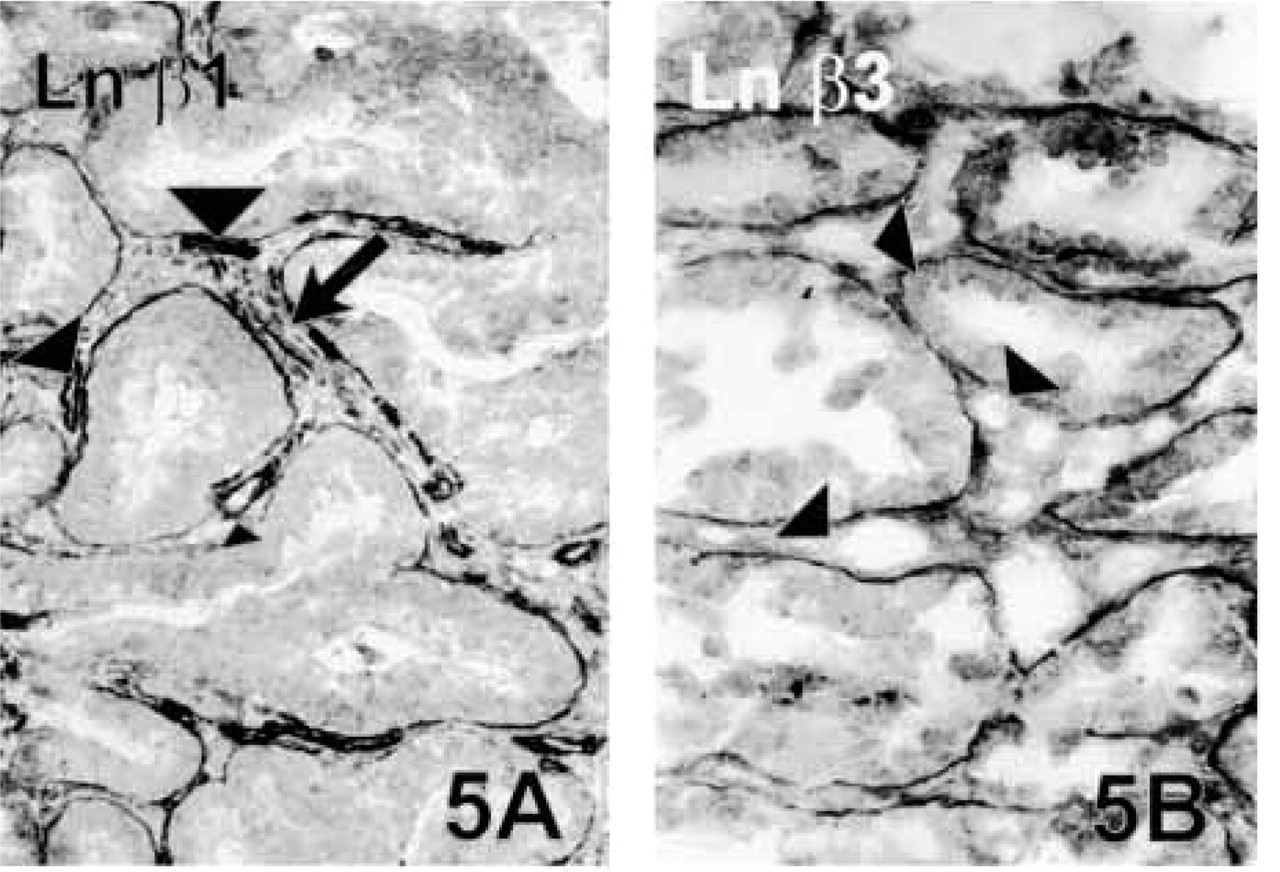
Colon adenocarcinoma. Strong BM reaction is seen using antibodies against Ln β1 (
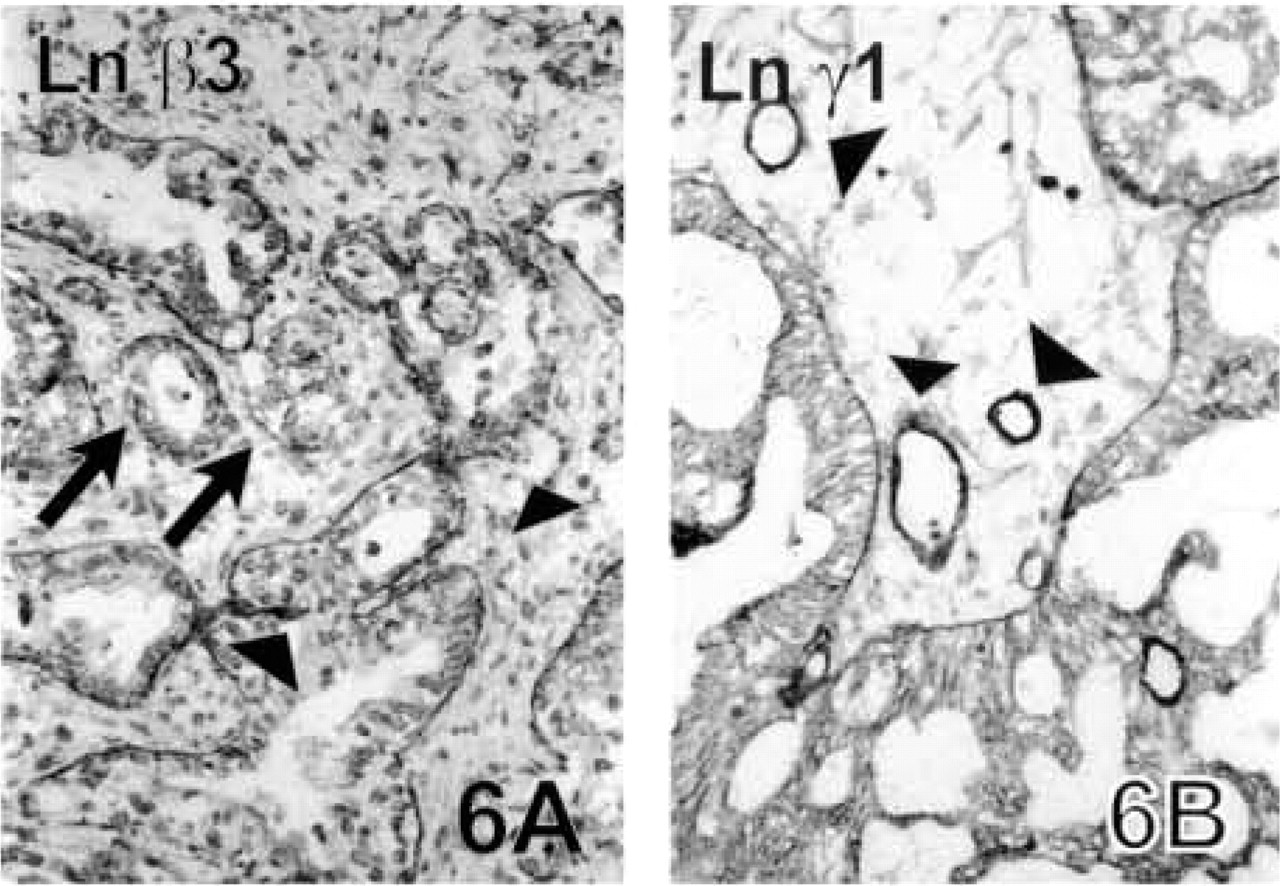
Ovarian carcinoma. In the grade III mucinous cystadenocarcinoma of ovary, BM staining reaction with Ln β3 chain antibody is linear in large carcinoma islands but can be detected only as short strips around smaller tumor clusters (arrows). The endothelial BMs are negative (arrowheads) (
Breast carcinoma. Linear BMs are seen around carcinoma clusters (ca) of the intraductal carcinoma with the Ln α1 chain antibody (
Thyroid carcinoma. BMs are clearly visible in the Ln α1 chain staining in the papillary thyroid adenocarcinoma (
Type IV collagen was present in about 50% of the neoplastic glands in both types of ovarian carcinomas, and the BM structures in these glands were more or less attenuated thin membranes.
The results for the two endometroid adenocarcinomas of the ovary were comparable to those seen in the mucinous cystadenocarcinomas, but one of the tumors lacked Ln α1 chain staining in its BMs.
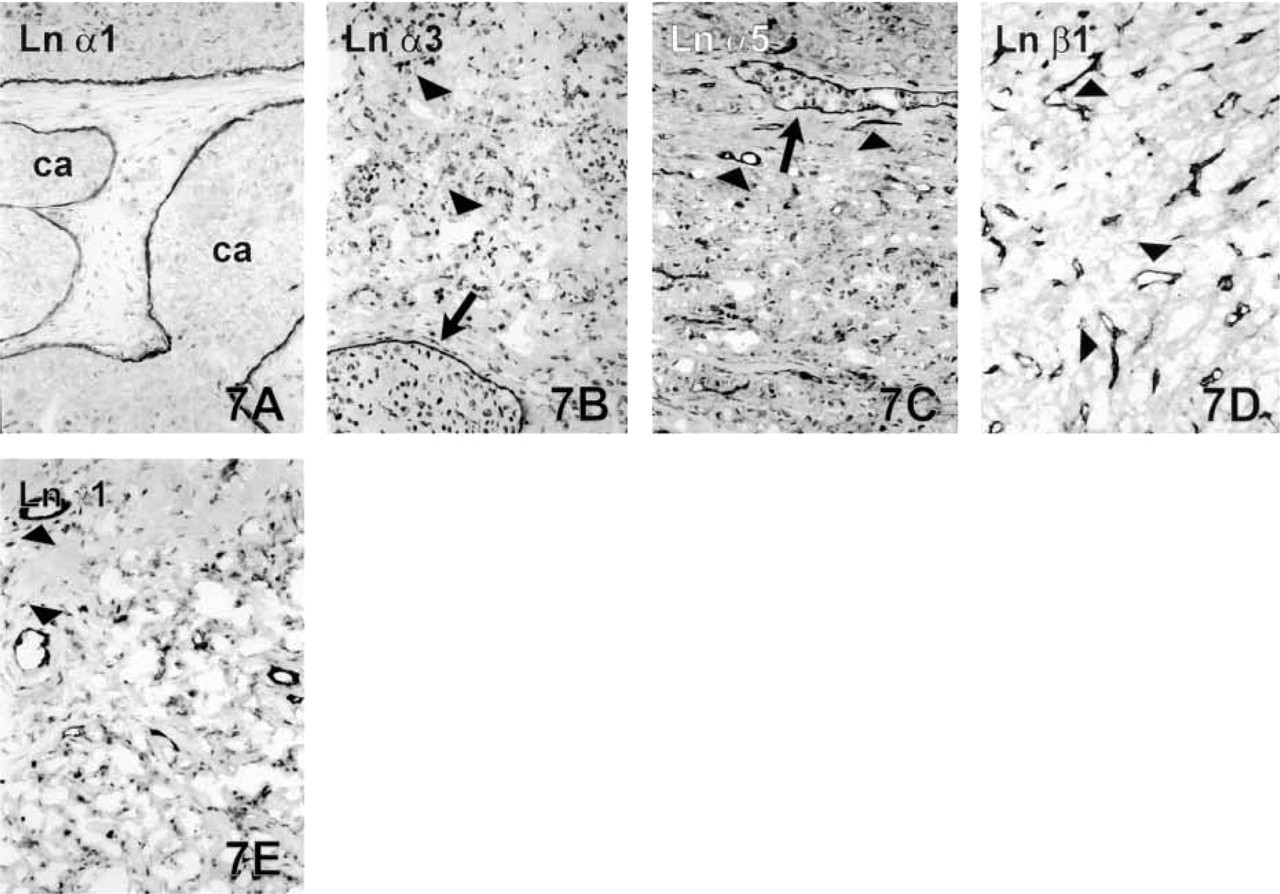
Breast Carcinomas. The pre-existing ductal structures of the intraductal carcinomas strongly expressed all the Ln chains and Type IV collagen (Figure 7A). BMs in this tumor type were always linear and well-formed, without any disruptions. The opposite result was seen with the invasive ductal and lobular carcinomas, which generally lacked immunoreactivity for the Ln chains, and Type IV collagen was detected only occasionally in short strips around neoplastic clusters (Figures 7B-7E). Ln γ2 chain was present intracellularly in invasive tumor cells in seven cases, however, and Ln α5 and α3 chains in two cases (not shown).
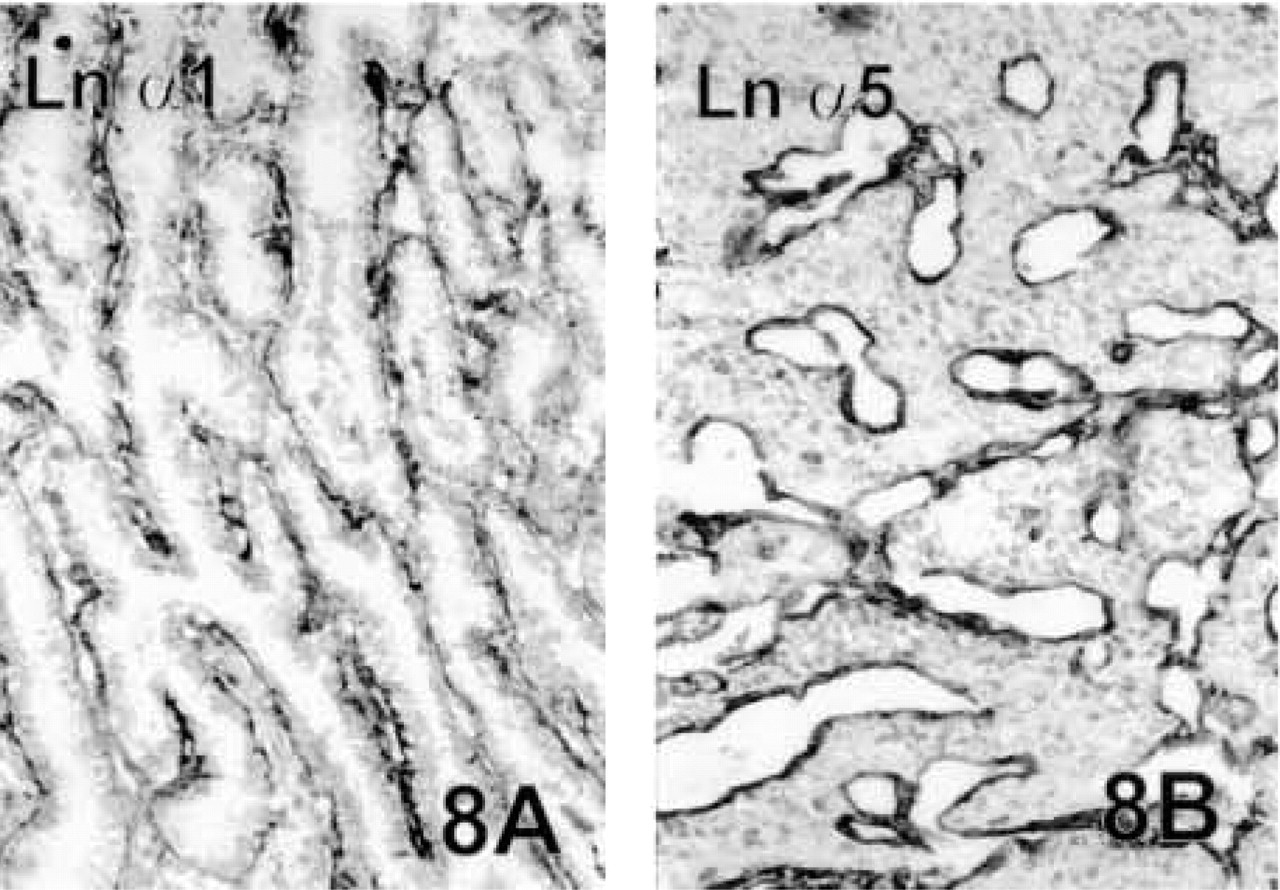
Carcinomas of the Thyroid Gland. Ln α1 (Figure 8A), α5 (Figure 8B), β1, β2, and γ1 chains were strongly expressed in the BMs of neoplastic follicular and papillary structures of the thyroid gland. Large tumor follicles showed quite linear BMs, but BMs of the smaller ones in the peripheral invasive areas had a clearly reduced staining pattern. Ln α3, β3, and γ2 chains forming the laminin-5 heterotrimer were either seen in occasional faint strips around the tumor clusters or else the result was unconvincing.
Type IV collagen was abundantly present in the BMs around neoplastic follicles, although a reduced staining pattern was seen in the follicles of the invasive front.
In Situ Hybridization Findings
ISH was carried out to determine which cells are responsible for the production of mRNAs for the Ln α1, β1, and γ2 chains and the collagen α1(IV) chain in tumor tissues, to investigate the intensity of expression, and to show the tissue distribution of their mRNA transcripts. One feature common to all the mRNAs was intensified expression in the tumor area compared with the adjacent normal tissue.
The level of synthesis of mRNA for the Ln α1 chain was generally low or at most moderate in the carcinomas. For this reason, samples of early human 8–10 gw placentae and deciduae from legal abortions were hybridized simultaneously as a positive control. The human placentae gave clear signals and positive grains were abundantly localized to the double-layered trophoblastic epithelium (Figure 9G). The corresponding antigen was localized to the BMs of trophoblastic epithelium (result not shown).
The strongest expression of Ln α1 chain mRNAs in the carcinomas was seen in the SQCCs of the lung (Figure 9A) and the intraductal breast carcinomas (Figure 9B), where the neoplastic epithelial cells contained positive grains. Invasive cells of the infiltrative breast carcinomas showed only occasional labeling. Occasional labeled carcinoma cells were also seen in ovarian and thyroid carcinomas, but the SQCCs of the tongue, stomach, and colorectal adenocarcinomas were negative or unconvincing in their Ln α1 chain mRNA expression.
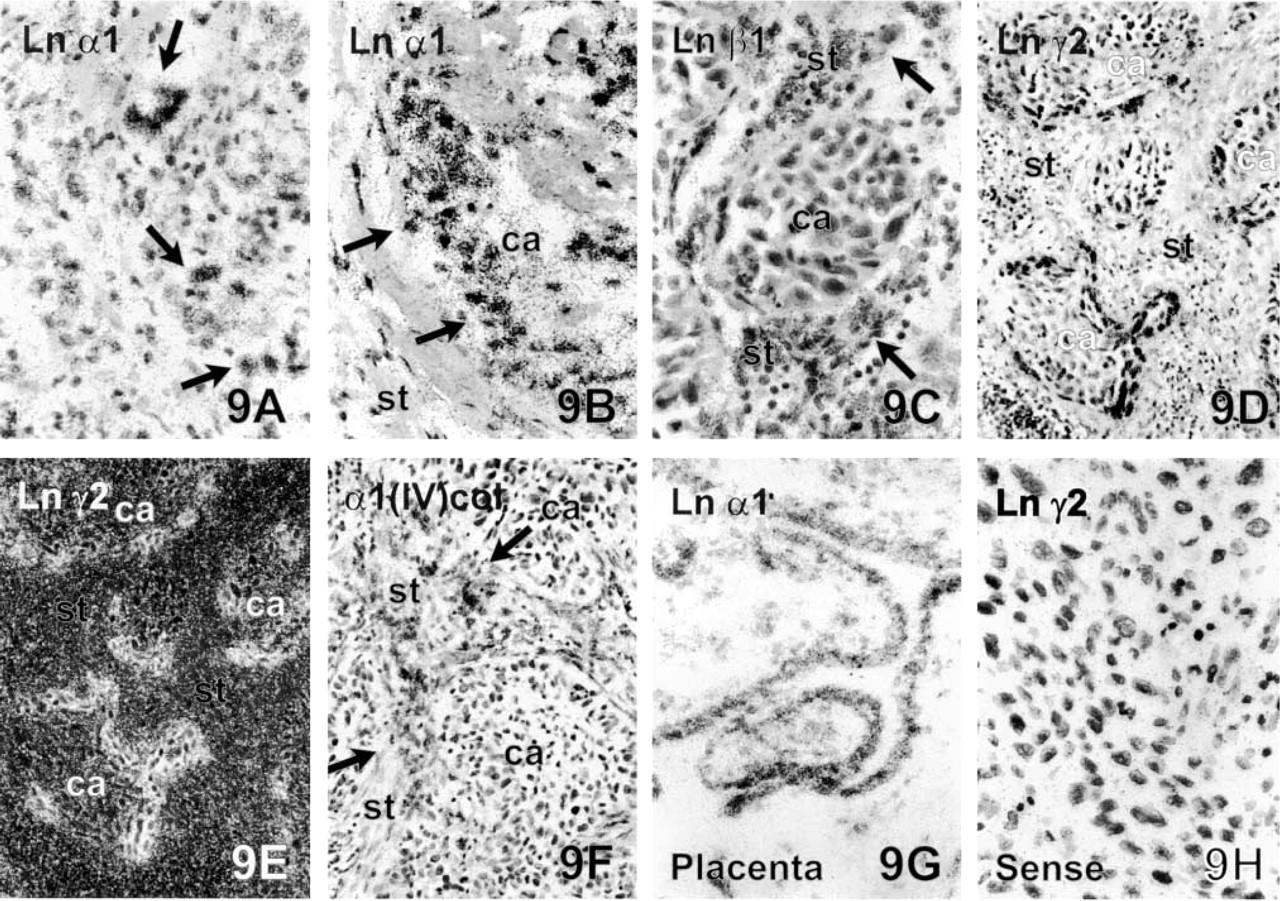
ISH using antisense mRNA probes. mRNA synthesis of the Ln α1 chain can be localized to the neoplastic epithelial cells (arrows) in the grade II SQCC of lung (
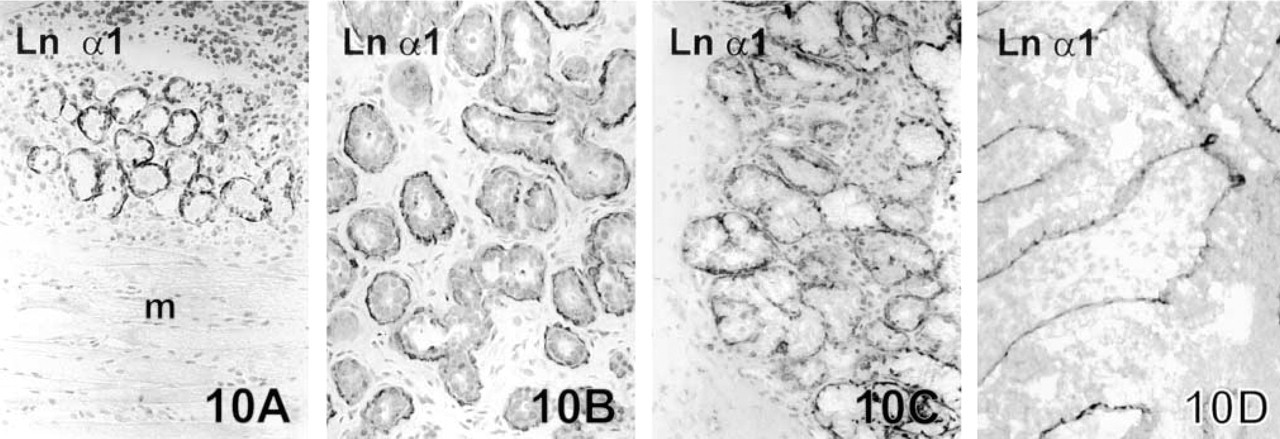
The Ln α1 chain in the BMs of normal tissue structures. The BMs of salivary glands of tongue (
Ln γ2 chain mRNA was produced solely by carcinoma cells. The most intense expression was observed in the SQCCs of the lung (Figures 9D and 9E) and tongue, followed by the colorectal and intestinal type of gastric adenocarcinoma. mRNA production was seen especially in the cancer cells of the tumor-stromal interphase, but scattered invasive cancer cells in the tumor stroma and individual smaller tumor islands showed focal but strong expression. The central parts of larger tumor clusters contained only low amounts of transcripts or were negative. Intense synthesis was also seen in cells adjacent to necrotic areas of the SQCCs (not shown), while very low synthesis was observed in the invasive breast and ovarian carcinomas and the diffuse type of gastric adenocarcinoma, where occasional labeling was observed in carcinoma cells. The thyroid carcinomas remained negative for Ln 2 chain mRNA in ISH.
mRNAs for the Ln β1 chain and the collagen α1(IV) chain were mainly synthesized by the stromal cells in all the tumors. Tumors expressed low levels of Ln β1 chain mRNA in their carcinoma cells, but only occasional mRNA transcripts for the collagen α1(IV) chain were present. The most intense expression of both transcripts was observed in endothelial cells and in fibroblasts around tumor clusters, collagen α1(IV) chain mRNA being more abundant in the endothelial cells (Figures 9C and 9F). Tumors accompanied by excessive fibrous desmoplasia, as in colorectal adenocarcinomas, had especially strong expression of mRNA for the Ln β1 chain and the collagen α1(IV) chain in stromal fibroblasts. In infiltrative breast tumors, carcinoma cells did not contain trascripts either for Ln β1 or collagen α1(IV) chain, but stromal fibroblasts and vascular endothelium were strongly labeled.
Laminin Chains in Normal Tissue Structures
The Ln chains generally showed differential expression patterns in the various BMs. Expression of Type IV collagen was stable and it was abundantly present in the BMs of all the normal structures, which were always surrounded by a continuous BM layer. Of the Ln chains, α5, β1, and γ1 were the most widespread, whereas the α1 chain was the most restricted in its expression in the normal tissues, as also in the tumors. Ln α1, α3, β3, and γ2 chains were generally the ones present in the epithelial BMs, whereas Ln α5, β1, β2, and γ1 were also found in other BMs, such as the vascular endothelium, neural structures, and stromal myofibroblasts.
BMs of normal glands of the tongue (Figure 10A), glands of the breast (Figure 10B), peribronchial glands (Figure 10C), and follicles of fertile ovary (Figure 10D) expressed Type IV collagen and all the Ln chains. An undulating immunoreactivity for Ln α1 chain was sometimes seen beneath myoepithelial cells of the peripheral mammary ducts. The staining pattern was otherwise similar in glands of the gastric mucosa, except that the α3, β3, and γ2 chains were present only in BMs of the gastric surface and the foveolar parts of glands, whereas α5, β1, β2, and γ1 were also present in BMs around the more deeply situated glandular parts. There was no immunoreactivity for the Ln α1 chain in gastric or colon glands. Normal follicular BMs of the thyroid gland expressed the Ln chains α1, α5, β1, γ1, and Type IV collagen. These structures lacked the Ln chains of α3, β3, and γ2.
Ln chains α5, β1, β2, and γ1 and Type IV collagen were all abundantly present in basal laminae of blood vessels. In some cases Ln α3 was also weakly seen in the endothelial BMs, but they were generally negative for this chain (not shown). The staining reaction for the α1, β3, and γ2 chains was negative in all cases. The BMs of the smooth and striated muscle cells expressed Ln α5, β1, β2, and γ1 chains and Type IV collagen in abundance. Fibroblasts in areas rich in stromal tissue contained intracytoplasmic Ln β1 and γ1 chains and Type IV collagen in some areas.
Perineural BMs and BMs around Schwann cells in neural structures expressed α5, β1, and γ1 chains of Ln and Type IV collagen. Immunoreactivity for other Ln chains was negative.
Negative Controls
PBS was applied instead of primary antibody to generate negative controls. These samples never contained specific staining in their BMs or at other locations (result not shown). In ISH the mRNA probes in sense orientation were simultaneously applied to separate sections and hybridized as controls. They did not show any specific labeling, but only the background signal (Figure 9H)
Discussion
BMs are known to mediate many essential properties that are important to cell adhesion, migration, and proliferation (Aumailley and Smyth 1998; Colognato and Yurchenco 2000). They are also known to protect cells from apoptosis (Mooney et al. 1999). In malignant transformation, they form the first sructural barrier that the malignant cells must penetrate to achieve an invasive phenotype, and it has long been a generally accepted fact that BMs are variably fragmented or even totally lacking in epithelial malignancies (Barsky et al. 1983; Hewitt et al. 1997; Hagedorn et al. 1998; Henning et al. 1999). The present results, however, show that human carcinomas are generally capable of synthesizing substantial amounts of Ln chains and Type IV collagen in their BMs. It is also noteworthy that BMs in carcinomas contained almost all Ln chains investigated, suggesting that they bear more than one Ln heterotrimer in their BMs. Furthermore, in many cases the BMs around the tumor clusters showed quite a linear immunoreactivity, suggesting that the capacity to deposit BM macromolecules is not totally lost during tumor progression but is rather an important event that provides a growth substratum for the infiltrative malignant cells, at least in some types of malignant tumor. These findings are in line with previous reports using Ln chain-specific antibodies (Tani et al. 1996, 1997; Sordat et al. 1998; Määttä et al. 1999).
Although the BMs around tumor clusters in this material were generally well formed, there were some differences and variations in the immunoreactivities between the tumor types and between cases of the same tumor type. The infiltrative ductal and lobular breast carcinomas and diffuse gastric carcinomas differed most strikingly from other carcinoma types by their almost total lack of extracellular immunoreactivity for Ln chains or Type IV collagen. Moreover, ISH showed that carcinoma cells of infiltrative breast and diffuse gastric carcinomas synthesized only low amounts of Ln α1, β1, and γ2 chains and collagen α1(IV) chain mRNA. This speaks for the fact that these two tumor types do not appear to produce significant amounts of BM material around their malignant cell clusters and trabecules, indicating that invasive growth in these tumors is not associated with a substratum that contains BM macromolecules. The low synthesis level seen in ISH also suggests that an altered BM macromolecule synthesis and assembly, rather than an increased matrix proteolysis, leads to the lack of BMs around tumor clusters in these tumor types. Our knowledge of the functional role of BMs in carcinomas is still limited, but on the basis of the present study and of previous reports, malignant tumors can be divided at least into two groups, one of which comprises tumors that exhibit quite well-formed BMs around their tumor clusters, e.g., carcinomas of tongue, lung, thyroid gland, colon, intestinal-type gastric carcinoma, and intraductal carcinomas of breast (Hewitt et al. 1997; Pyke et al. 1995; Sordat et al. 1998; Tani et al. 1996; Määttä et al. 1999). The other group includes carcinomas that do not appear to produce significant amounts of BM material, e.g., invasive ductal and lobular breast carcinomas and diffuse-type gastric carcinomas (Barsky et al. 1983; Tani et al. 1996; Hewitt et al. 1997; Henning et al. 1999). There are also tumors which cannot be clearly classified into the enclosed groups, e.g., carcinomas of ovary and larynx (Cam et al. 1984; Hagedorn et al. 1998). Although the amount of BM material appears to be dependent on the tumor type, common to all tumors investigated here was that the BMs were less well formed in the tumor clusters situated in the areas of obvious active invasion.
All the three chains of laminin-5 (α3β3γ2) were most abundant in the SQCCs of lung and tongue, where linear BMs were seen to encircle the tumor clusters. The invasive areas in these tumors contained intracytoplasmic positivity for the Ln γ2 chain. These staining reactions were less well expressed in the various types of adenocarcinoma. Laminin-5, which was originally found in hemidesmosomes of the skin, seems to be preferentially expressed by SQCCs, and its expression relative to the adjacent normal tissues had even increased along with tumor progression. Previously we have noted similarly that the Ln γ2 chain can be detected from the BMs and in the tumor stroma in different types of the lung carcinoma and pancreatic adenocarcinomas (Soini et al. 1996; Määttä et al. 1999). This is in line with a recent observation by Koshikawa et al. (1999) that a monomeric Ln γ2 chain can be secreted into the ECM space in addition to trimeric laminin-5 molecules. Ln γ2 chain of laminin-5 has been shown to be a substratum for both matrix metalloproteinase 2 (MMP-2) and membrane-type matrix metalloproteinase 1 (MT1-MMP), and the processed form of the γ2 chain was shown to trigger a marked cellular migration over laminin-5 surfaces (Giannelli et al. 1997; Koshikawa et al. 2000).
The current available immunohistochemical data on normal tissues indicate that laminin-10 is the most widely distributed heterotrimer and Ln-1 a more restricted one (Klein et al. 1988; Ekblom et al. 1990; Miner et al. 1997; Falk et al. 1999; Tani et al. 1999; Pedrosa–Domellöf et al. 2000; Virtanen et al. 2000). Our results showed the Ln α1 chain to be present in some individual lesions of SQCCs of the lung, adenocarcinomas of the ovary and thyroid gland, and one colon adenocarcinoma. It was exclusively present in pre-existing ductal structures of intraductal breast carcinomas but not in invasive breast carcinomas. In other locations its expression was occasional, and it was entirely absent from carcinomas of the tongue and intestine and from diffuse gastric carcinomas. In normal tissue structures, the Ln α1 chain present in the BMs of some glandular types and also around the follicles of the thyroid gland, but it was not detected in muscle, fibroblasts, or vascular or neural structures. It is also noteworthy that when the Ln α1 chain was present in BM of the normal tissues, it was also found in tumors originated from this tissue type. This distribution pattern indicates a high epithelial specificity for the Ln α1 chain, in line with the results of Virtanen et al. (2000). The findings further suggest that expression of the α1 chain, present in laminin-1 (α1β1γ1) and -3 (α1β2γ1), is restricted and can be detected only in certain carcinomas. Conversely, the Ln α5 chain, present in laminin-10 (α5β1γ1) and laminin-11 (α5β2γ1), was found to be a component of almost all tumor BMs. Because the β2 chain was detected only occasionally in epithelial BMs, this supports the notion that laminin-10, but not laminin-11, is among the most widespread Ln heterotrimers, not only in normal tissues but also in malignancies.
ISH was carried out to show which cells are responsible for the production of mRNAs and to investigate their tissue localization. The results indicated that the Ln β1 chain and the collagen α1(IV) chain were preferentially expressed by tumor stromal cells. The Ln β1 chain was synthesized especially by stromal fibroblasts, whereas the collagen α1(IV) chain was more notably produced by vascular endothelium. Abundant stromal expression for the Ln β1 chain supports the immunohistochemistry that revealed the corresponding antigen to be present around stromal myofibroblasts and in the BMs of vascular endothelium. It is also notable that these same stromal cells actively synthesize matrix-degrading enzymes (Autio–Harmainen et al. 1993). Ln γ2 chain mRNA was produced solely by neoplastic epithelium, especially in carcinoma cells that were in close contact with the tumor stroma and in individual infiltrative carcinoma cells, although invasive breast carcinomas expressed only very low amounts of mRNA for the Ln γ2 chain. Ln α1 mRNA synthesis was localized solely in the epithelial tumor cells, which is concordant with the immunohistochemical findings and with previous ISH and Northern hybridization results (Ekblom et al. 1990; Nissinen et al. 1991; Vuolteenaho et al. 1994; Tani et al. 1999; Virtanen et al. 2000). There are many in vitro experiments showing Ln chain mRNA synthesis in carcinoma cell lines (Wewer et al. 1994; Tani et al. 1999). The lack of tumor–stromal interaction may modulate the expression pattern in such experiments and the results may not be relevant in vivo, because BM macromolecules are known to be synthesized and assembled by synergistic involvement of both epithelial and adjacent stromal cells (Perreault et al. 1998). One feature common to all the mRNAs investigated here was that their synthesis in carcinoma tissues was intensified compared with that in the adjacent normal tissue. There are several reports indicating that the upregulation of mRNA synthesis of BM macromolecules is also connected with tumor dedifferentiation (Nerlich et al. 1998; Pfohler et al. 1998), but the synthesis of mRNA for the Ln α3 and β3 has been shown to be downregulated in invasive breast carcinomas (Martin et al. 1998), a finding that is corroborated by the low level of Ln γ2 chain mRNAs seen in this study and by the negative immunostaining result for all Ln-5 chains in invasive breast carcinomas.
In conclusion, we showed in this study that Ln chains are generally well preserved, with few exceptions, in BMs of human carcinomas. Ln α1 and β2 were present only in certain epithelial BMs, whereas α5, β1, and γ1 chains of Ln were more ubiquitous in expression. The results show that Ln-5 and Ln-10 are the most abundant Ln heterotrimers in carcinomas and normal tissues. Except for the infiltrating breast and diffuse gastric carcinomas, human carcinomas seem to actively synthesize BM macromolecules to their growth substratum and bear quite well-formed BMs around tumor clusters, which suggests their important connection to malignant spreading.
Footnotes
Acknowledgements
Supported by the Cancer Foundation of Finland, Helsinki, and by the Cancer Foundation of Northern Finland, Oulu.
We wish to thank Professor Karl Tryggvason (Karolinska Institute; Stockholm, Sweden) for the Ln α1, β1, γ2, and α1(IV) chain cDNAs and Ln γ2 chain antibody and Professor Juha Risteli (University of Oulu, Finland) for Type IV collagen antibody. The antibody to β2 Ln chain developed by Dr Dale Hunter and Dr Joshua Sanes was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the University of Iowa (Department of Biological Sciences; Iowa City, IA). We also thank Ms Heli Auno, Ms Annikki Huhtela, Mr Tapio Leinonen, and Mr Hannu Wäänänen for excellent technical assistance.
