Abstract
One of the characteristics of pancreatic cancer is its tendency to invade neural tissue. We hypothesized that the affinity of cancer cells for nerve tissue is related to the presence of growth factors in neural tissue and their receptors in cancer cells. Sections of pancreatic cancer and normal pancreatic tissue were examined by immunohistochemistry for the expression of the neurotrophins NGF, BDNF, NT-3, NT-4, and their receptors TrkA, TrkB, and TrkC, as well as the low-affinity receptor, p75NTR. TrkA expression was found in duct, islet, and cancer cells; TrkB was found in the α-cells of the islet only. The anti-pan-Trk antibody (TrkB3), which is presumed to recognize all three receptors, immunoreacted with duct and acinar cells in normal tissue and with cancer cells. The staining with TrkC was similar to that of TrkA. The low-affinity receptor p75NTR was expressed in the neural tissue and in scattered duct cells of the normal tissue only. Duct and acinar cells, as well as neural tissue and cancer cells, showed weak to strong immunoreactivity with NGF. NT-3 expression was noted in capillary endothelia and erythrocytes. NT-4 showed specific staining for ductule cells. The expression and distribution of neurotrophins and their receptors suggest their role in the potential of pancreatic cancer cells for neural invasion.
P
The grave prognosis of the disease is related to its invasive and metastatic nature. Perineural invasion is one of the most common features of pancreatic cancer. Up to 90% of patients show the involvement of intrapancreatic nerves and 69% of extrapancreatic nerves (Nagakawa et al. 1992a,b,1993; Nakao et al. 1996; Takahashi et al. 1997). There appears to be no relationship between the size of the cancer and its potential for perineural invasion, because even microscopic cancers show this tendency (Kimura et al. 1998). Reasons for the high affinity of PC cells for neural tissue are obscure. It has been suggested that this process is related to an inherent ability of tumor cells to penetrate the perineural sheet (Bockman et al. 1994). This possibility is unlikely because, unlike the nerves, lymphatic and vascular structures are affected in the late stages of the disease. Consequently, it is possible that some substances produced by nerves act as chemotactic factors. Nerve growth factor (NGF) might be one of these factors. To investigate this possibility, we examined the expression of nerve growth factors (NGF, BDNF, NT-3, NT-4) and their receptors (TrkA, B, C, and p75NTR) in pancreatic cancer cells and in pancreatic neural tissue.
NGF and its relatives, brain-derived neurotrophic factor (BDNF), NT-3, and NT-4, are a family of polypeptides that promote the outgrowth and differentiation of peripheral and central neurons. Thus far, two classes of corresponding receptors have been identified. The high-affinity tyrosine kinase receptors TrkA, TrkB, and TrkC (kD ≈10−11 to 10−10 M) and the low-affinity receptor p75NTR (kD ≈10−9 to 10−8 M). All three tyrosine kinase receptors are transmembrane molecules that bind NGF, BDNF, NT-3, and NT-4 with a different affinity. The low-affinity receptor (p75NTR), with an approximate weight of 75 kD, is a transmembrane glycoprotein and a member of the TNF receptor superfamily. It can promote cell survival or induce apoptosis, at least in neural tissue, whether or not TrkA, TrkB, or TrkC is co-expressed (Rabizadeh et al. 1993). NGF is preferentially bound to TrkA. BDNF and NT-4 bind to the TrkB receptor. NT-3 is the preferential ligand of the TrkC receptor (Lamballe et al. 1991; Weiner 1995; Friedman and Greene 1999).
The purpose of the present study was to examine whether these growth factors and their receptors play a role in the affinity of pancreatic cancer cells for neural tissue.
Materials and Methods
Nineteen surgically removed pancreatic cancer specimens were examined. The cancers were classified according to the International Histological Classification of Tumors published by the World Health Organization (WHO) (Kloeppel et al. 1996). Sixteen cancers were duct adenocarcinomas, two were cystadenocarcinomas, and one was of anaplastic type. Six of the 16 duct adenocarcinomas were well differentiated and 10 were moderately to poorly differentiated. Five of these specimens had a portion of tumor-free pancreatic tissue. Twelve pancreas specimens from multi-organ donors were used as control.
Antibodies
The following primary antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Polyclonal rabbit TrkA and goat TrkA were against an epitope mapping adjacent to the carboxy terminus of the human TrkA protein. The polyclonal rabbit anti-TrkB (794) recognizes an epitope corresponding to amino acids 798–812 of mouse TrkB. Polyclonal rabbit anti-TrkC immunoglobulin (798) recognizes an epitope corresponding to amino acids 798–812 of porcine TrkC and NGFR p75NTR goat polyclonal antibody against a peptide mapping at the carboxy terminus of p75NTR receptor of human origin. NGF (h-20) and BDNF (n-20) are rabbit polyclonal antibodies were against a peptide mapping at the amino terminus of the mature form of NGF and BDNF of human origin. The NT-3 (n-20) rabbit polyclonal antibody recognizes a peptide mapping at the carboxy terminus of the mature form of NT-3 of human origin. The NT-4 (n-20) rabbit polyclonal antibody recognizes a peptide mapping to an internal domain of the mature form of NT-4 of human origin. Pan Trk (TrkB3) is monoclonal mouse antibody that reacts with a highly conserved carboxy terminus of the precursor form of TrkA gp140 of human origin. The anti-TrkB3 “pan Trk” is reactive with TrkA, TrkB, and TrkC epitopes. For details of the specificities of anti-receptor antibodies see Hoehner et al. (1995). The optimal antibody dilution (1:400) was determined by serial dilution and was applied to all sections.
Immunohistochemical Examinations
All specimens were fixed in 10% buffered formalin and processed for histology by conventional methods. Serial sections 4 μm thick were prepared from paraffin blocks. After deparaffinization the slides were submerged in methanol containing 0.3% hydrogen peroxide for 30 min at room temperature (RT) to inhibit endogenous peroxidase activity. Antigen retrieval was performed by incubating the sections with 0.05% saponin in deionized water at RT for 30 min. Sections were washed with Tris-buffered saline (TBS 0.1 M, pH 7.4). According to the origin of the primary antibody, the slides were incubated with normal goat or donkey (p75NTR, goat TrkA) serum for 60 min at RT. The sections were washed with TBS buffer and incubated with the primary antibody at 4C overnight. Biotinylated secondary antibody (Multilink; BioGenex, San Ramon, CA) was used against rabbit or mouse primary antibodies. Biotin-conjugated anti-goat IgG (Santa Cruz Biotech) was used with the p75NTR or TrkA goat antibody. Detection of the antibody complex was preformed by the streptavidin–peroxidase reaction kit (Kirkegaard & Perry Laboratories; Gaithersburg, MD). Multilabeling technique was also used as reported (Pour et al. 1994).
Hyaluronidase (Sigma; St Louis, MO) 1 mg/ml in 100 mmol/liter sodium acetate and 0.85% NaCl or saponin (Sigma) 0.05% in deionized water was used for antigen retrieval for anti-TrkA, anti-TrkB, and anti-TrkC antibodies. After preincubation with Tris-buffered saline containing 0.1% (v/v) Triton X-100 for 15 min, the sections were incubated at 37C for 30 min. Formalin-fixed, paraffin-embedded sections of human prostate cancer served as control for the immunostaining of TrkA as reported (Pflug et al. 1995; Shibayama and Koizumi 1996).
Results
Table 1 summarizes the expression of the growth factors and their receptors in normal and malignant pancreatic cells.
The immunoreactivity of anti-TrkA differed depending on the pretreatment with saponin or hyaluronidase. The latter treatment resulted in staining of the tunica muscularis of blood vessels, the neural tissue, duct and ductule cells, scattered islet cells, and cancer cells. Duct, ductule, and cancer cells showed moderate to strong granular and membranous staining. Acinar cells were unstained. Pretreatment of the same sections with saponin showed weak staining of smooth muscle tissue of blood vessels only.
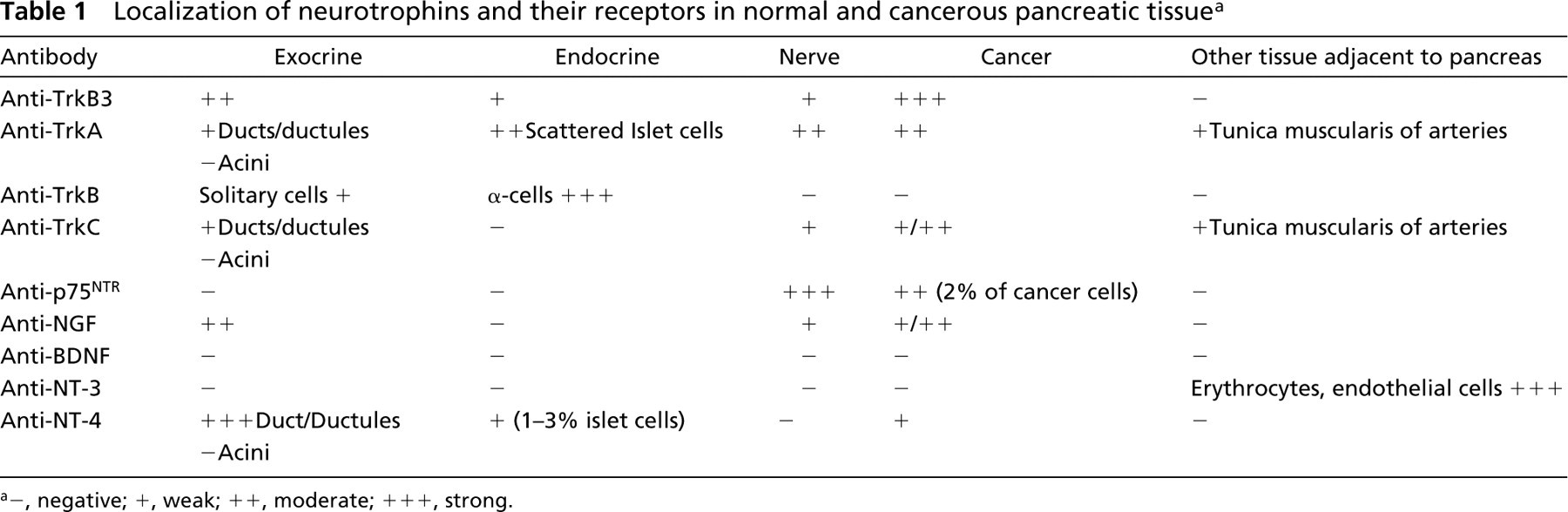
The pattern of staining with anti-TrkB did not differ between sections pretreated with saponin or hyaluronidase. Cancer cells did not express TrkB in any specimen. Strong cytoplasmic staining of up to 20% of islet cells within each islet was detected with this antibody in both normal and cancer tissues. Double immunostaining with the TrkB antibody and islet hormone antibodies showed strict co-localization of TrkB in α-cells (Figure 1). The scattered extra-insular endocrine cells between the exocrine cells were also stained. Neural tissues were not stained.
Localization of neurotrophins and their receptors in normal and cancerous pancreatic tissuea
–, negative; +, weak; ++, moderate; + + +, strong.
Similar to the findings with anti-TrkA, the immunoreactivity of anti-TrkC differed according to the method of pretreatment. Although the slides pretreated with saponin did not show staining in any cells, in the specimens pretreated with hyaluronidase the anti-TrkC antibody showed strong membranous staining of ducts and ductules in both normal and cancerous tissues. Acinar and endocrine cells were not stained. Although cancer cells, in general, revealed a fine membranous staining, in well-differentiated tumors a few cells also showed very strong cytoplasmic staining. Neural and smooth muscle tissues of intra-and peripancreatic blood vessels showed similar but weaker staining than with the anti-TrkA antibody.
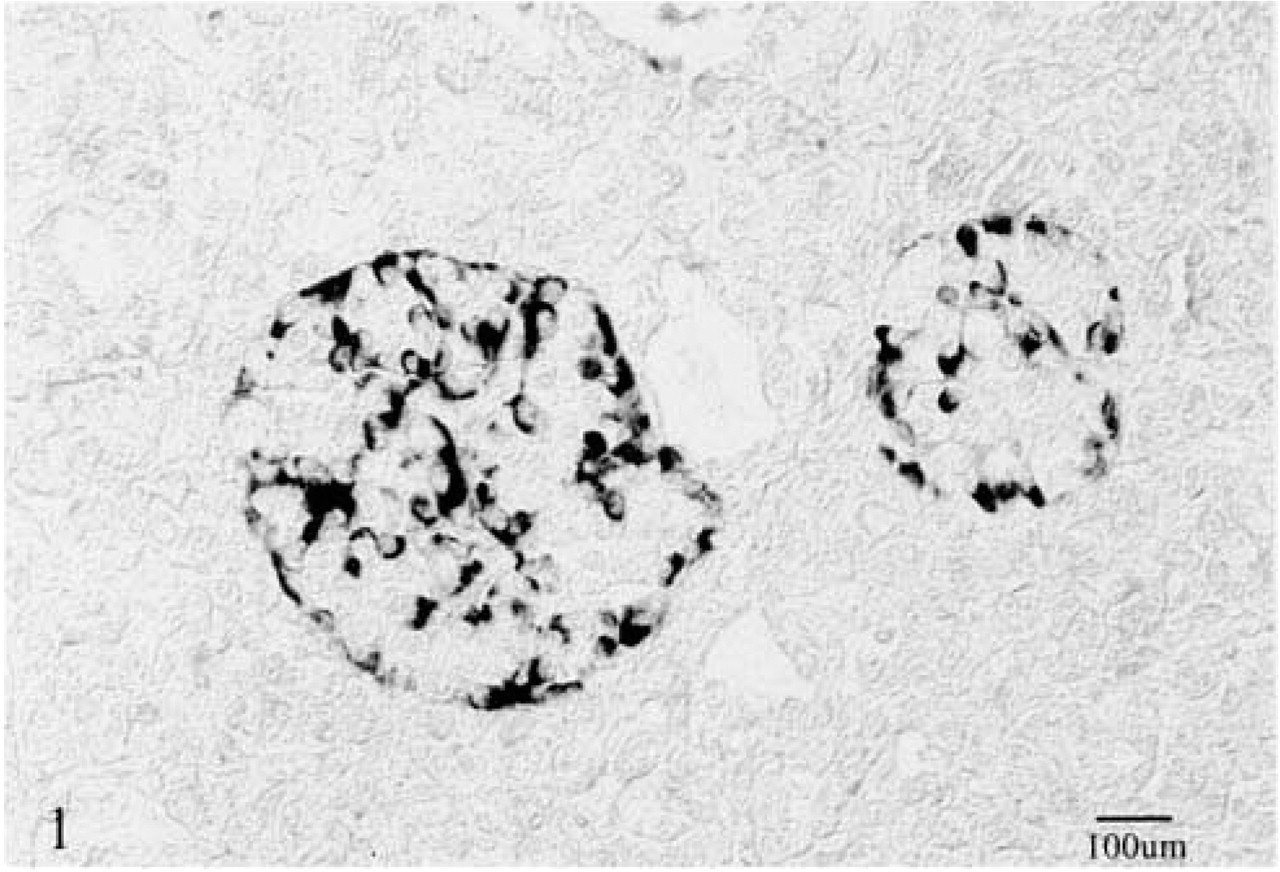
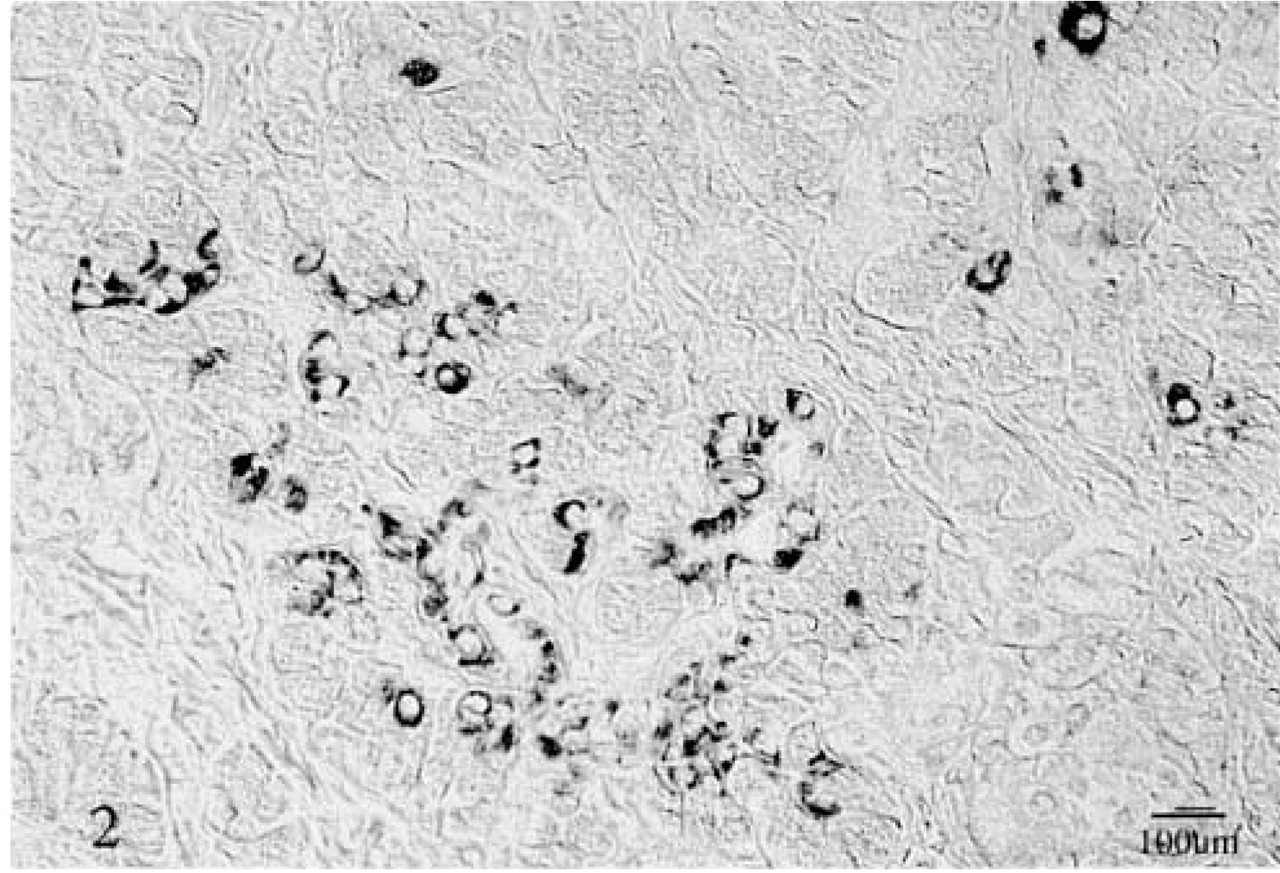
In normal tissue and in the tumor-free pancreatic tissues of five pancreatic cancer specimens, the pan Trk (TrkB3) antibody showed a cytoplasmic granular staining of duct and acinar cells. Scattered polygonal cells, grouped in small clusters, also showed strong cytoplasmic granular staining in the normal tissue and in tumor-free tissue adjacent to the cancer (Figure 2). Cancer cells presented a granular cytoplasmic staining, with stronger reactivity in the supranuclear regions. This finding was consistent throughout the tumors with various grades of differentiation (Figure 3). No staining was found in the neural structures. There were no differences in the staining patterns and intensity between the specimens pretreated with saponin and those pretreated with hyaluronidase.
The low-affinity receptor p75NTR was expressed in nerve fibers, perineurium, and ganglia in both normal and cancer tissues. In all specimens the perineurium showed the strongest staining (Figure 4). Ductules and ducts showed strong membranous staining only in atrophic areas close to the cancer cells and only in specimens pretreated with saponin. Pancreatic cancer cells did not show any immunoreactivity with the antibody regardless of the pretreatment.
Acinar, duct, ductule, and centroacinar cells, particularly in atrophic pancreatic areas, showed diffuse cytoplasmic staining with the anti-NGF antibody. Neural fibers were also stained. Strong nuclear staining of a few Schwann cells was noted in neural tissue. Cancer cells showed weak and diffuse cytoplasmic staining in glandular or anaplastic areas but not in cystic areas of the two cystadenocarcinoma specimens (Figure 5). Nuclear staining of a few cancer cells was also present within the malignant glands.
NT-3 antibody showed immunoreactivity with erythrocytes and capillary endothelia in both normal and cancerous tissues. No immunoreactivity was found in any pancreatic cells, cancer cells, or nerves. NT-4 antibody, on the other hand, showed strong cytoplasmic staining of duct and ductule cells (Figure 6) and heterogeneous staining of cancer cells. Although acinar cells were negative for NT-4, about 1–3% of islets showed weak granular staining. No significant differences in the immunoreactivity of anti-NGF, anti-NT-3, and NT-4 antibodies were found between the tissues pretreated with saponin and those treated with hyaluronidase.
Immunoreactivity with the anti-BDNF antibody was not detected in any tissues, regardless of the method of pretreatment.
Discussion
Perineural invasion is one of the most common characteristics of pancreatic cancer and appears to occur more often than in cancer of the prostate, biliary tree, or other gastrointestinal tumors. Characteristically, cancer cells appear to migrate along the nerve fibers for a long distance to reach the celiac plexus, where they apparently provide a nidus for cancer recurrence after surgery (Nakao et al. 1996). The reasons for the prolivity of pancreatic cancer for neural invasion are not understood. Cancer cells also invade islets, but this could be due to the lack of a protective mantle of the islets of Langerhans. The great affinity of PC cells to neural tissue, even at the very early stages of cancer development (Kimura et al. 1998), indicates a particular attraction of cancer cells to the neural tissue (Matsuda and Nimura 1983; Nagakawa et al. 1989).
Expression of TrkB in islet cells. The strong staining of the cells is in sharp contrast to the surrounding unstained cells. Multilabeling immunohistochemistry demonstrated. TrkB cells are expressed exclusively in the α-cells. ABC method.
Clusters of small polygonal cells stained with anti-pan Trk (TrkB3) in normal pancreatic tissue. Some of these cells were α-cells. Lack of staining in the surrounding acinar cells. ABC method.
Immunoreactivity of the pan Trk (TrkB3) antibody with cancer cells. The reactive product is granular cytoplasmic. In many cylindrical cancer cells the staining was localized around the nuclei, leaving a clear area in the luminal cytoplasmic portion (upper middle and right). ABC method.
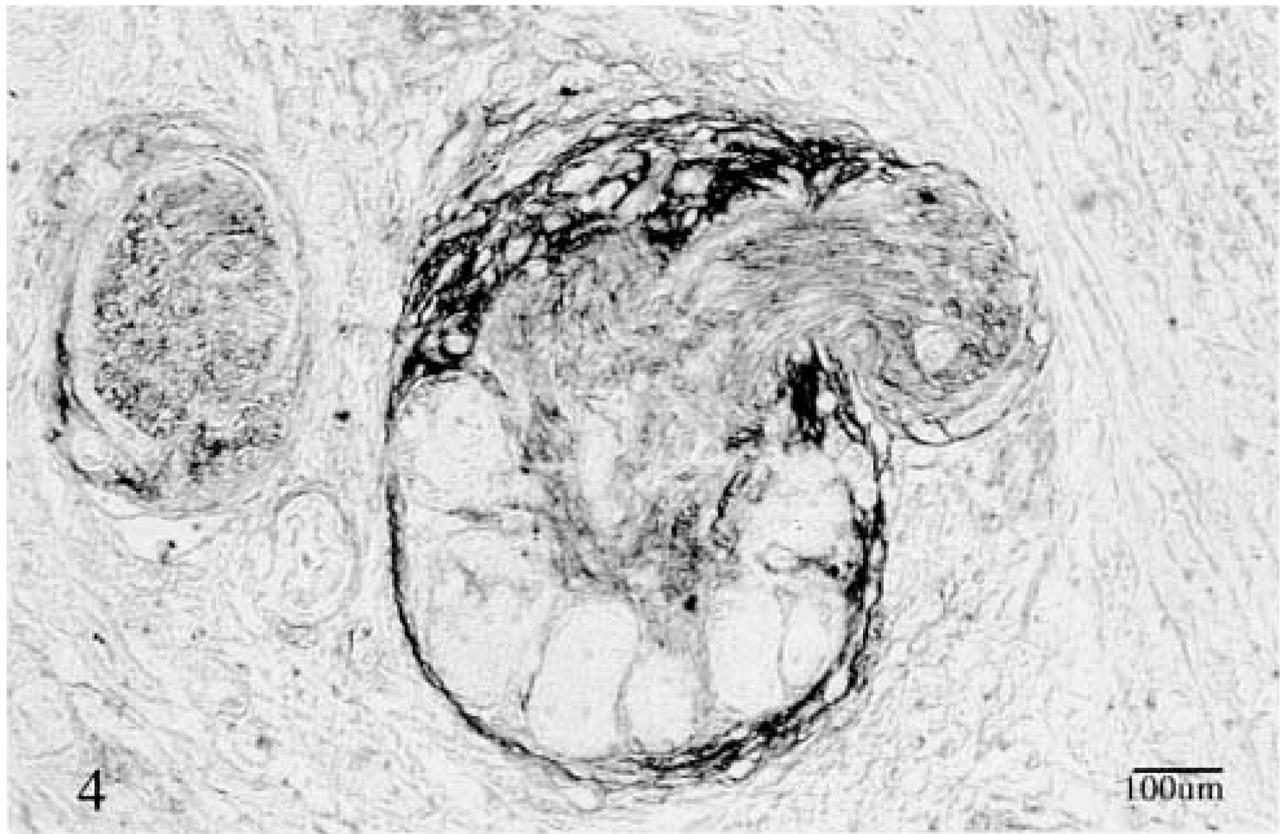
Expression of the low-affinity receptor p75NTR in a swollen nerve embedded in scarred tissue. Note the stronger staining of the perineurium and the weaker staining of the nerve fibers. Ganglia cells were not stained. ABC method.
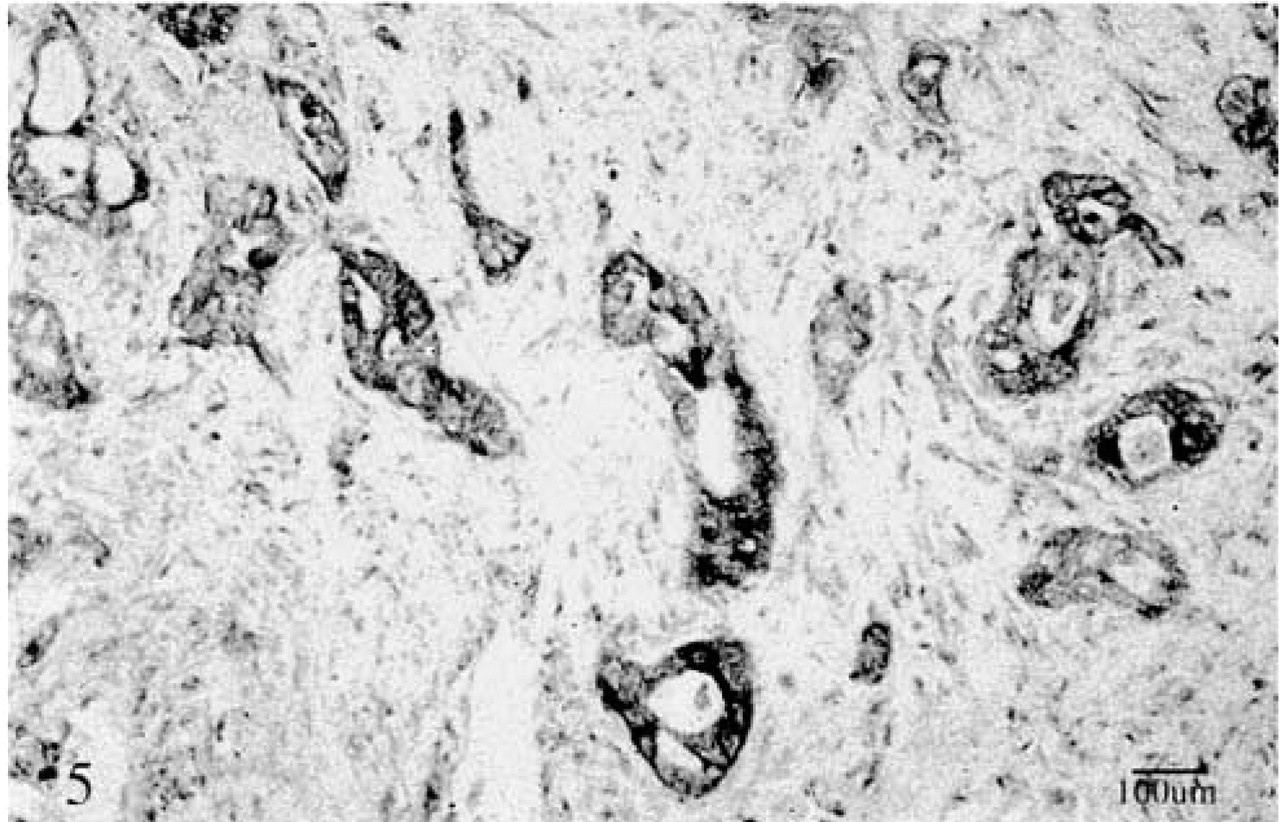
A poorly differentiated cancer showing fairly strong staining of cells with the anti-NGF antibody. In the weakly stained cells, the immunoreactive product was diffuse and cytoplasmic. ABC method.
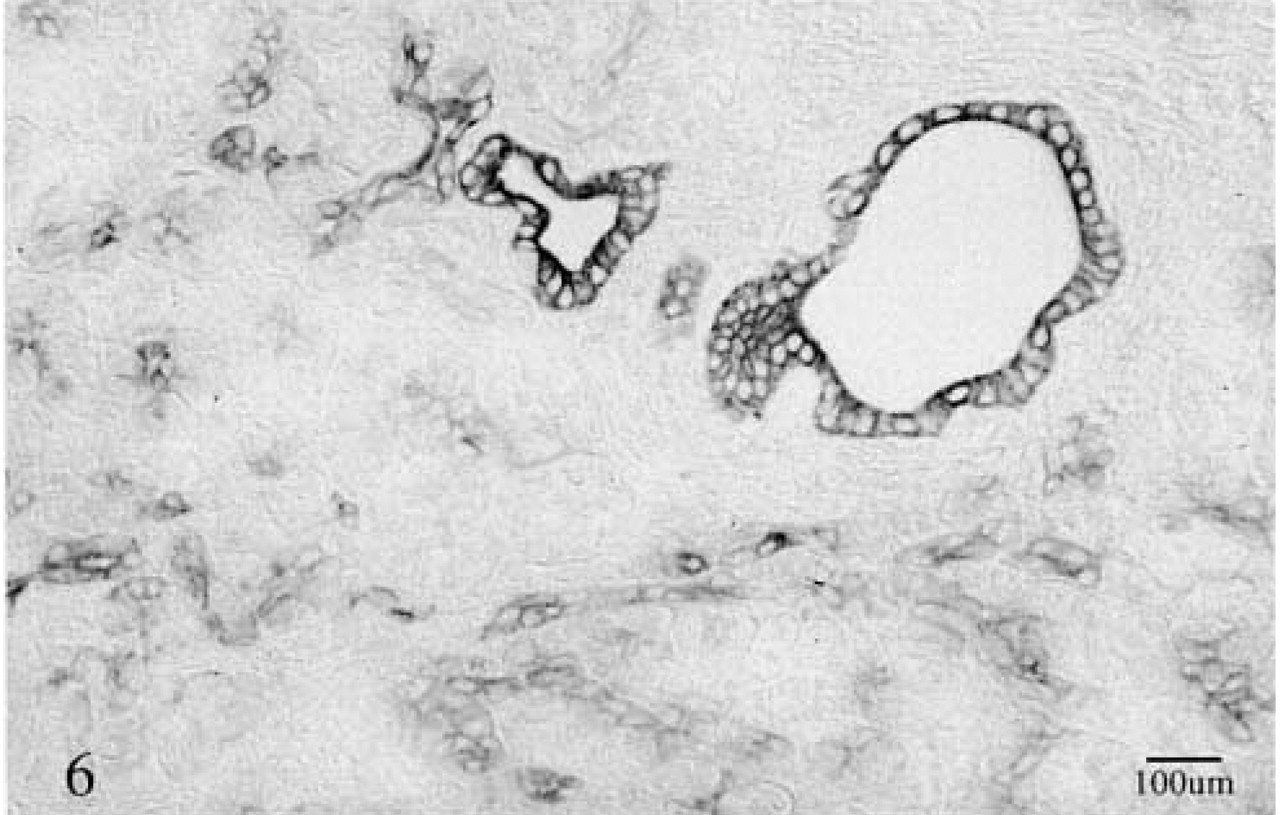
Strong membrane staining of duct cells and weaker staining of ductule cells with the anti-NT-4 antibody. ABC method. Centroacinar cells seen in the mid-portion of some acini (middle left) are also stained. ABC method.
It is suggested that the abundant expression of EGFR by cancer cells and the overexpression of TGF-α in nerves might constitute a reason for the affinity of cancer cells for nerves (Bockman et al. 1994). It could be argued, however, that TGF-α and EGFR are also produced in a variety of cancer cells, with or without the potential for perineural invasion. The easy access of cancer cells to neural tissue through certain segments of the perineurium (Bockman et al. 1994) has also been suggested but similarly appears unlikely because cancer cells do not show the same tendency to invade vascular or lymphatic channels. It is possible that specific growth factors produced by neural tissue act as chemotactic factors. In that case, the tumor cells should express receptors for a particular growth factor that is expressed in neural tissue.
Our study could, in part, confirm the reported immunoreactivity of pancreatic cancer cells with TrkA and TrkC (Miknyoczki et al. 1999). Contrary to the findings of Miknyoczki, we could not demonstrate the expression of TrkB in cancer cells. Moreover, in contrast to a previous study (Miknyoczki et al. 1999), we found strong immunostaining of cancer cells with the anti-NGF antibody.
Remarkably, only in hyaluronidase-pretreated slides the immunoreactivity of the pan Trk (TrkB3) antibody which, according to the manufacturer, recognizes all three Trk epitopes, corresponded to the immunoreactivity of TrkA, TrkB, and TrkC. This finding suggests that the epitope is masked by a complex carbohydrate structure in formalin-fixed tissues. This also may explain, in part, the differences in the reported cellular expression of these growth factors and their receptors.
The striking selective expression of TrkA and TrkB by α-cells, also noted by Shibayama and Koizumi (1996), clearly indicates differences in growth factor production by individual islet cells. Differences in the number of immunoreactive cells in individual islets could well be related to the number of immunoreactive cells (α-cells) present in a given section of the islets.
The strong cytoplasmic expression of NT-4 in duct cells in all specimens is noteworthy. It appears that expression of this growth factor represents a duct cell marker. In that case, the presence of a few cells within the islets could be considered to represent the tendency of some islet cells towards duct cell differentiation as has been shown to invariably occur in vitro (Lucas–Clerc et al. 1993; Kerr–Conte et al. 1996; Yuan et al. 1996; Schmied et al. 2000a,b). NT-4, NT-3, and BDNF are, thus far, known ligands of the TrkB receptor (Klein et al. 1991; Klein 1994; Maness et al. 1994). Our findings suggest an intrapancreatic paracrine signaling pathway between ductule cells secreting NT-4 and α-cells expressing the TrkB receptor.
In summary, strong expression of pan Trk (TrkB3), TrkA and TrkC on cancer cells and NGF in neural tissue may be, at least in part, the reason for the affinity of cancer cells for neural tissue. However, the existence of other mechanisms, which are of great clinical importance, awaits further studies.
Footnotes
Acknowledgements
Supported by the National Institute of Health, National Cancer Institute Grant, 5ROICA60479 and Spore Grant P50CA72712, the National Cancer Institute Laboratory Cancer Research Center Support Grant CA367127, and the American Cancer Special Institutional Grant.
Alexis Ulrich is a recipient of a scholarship from the Deutsche Forschungsgemeinschaft, Germany.
