Abstract
Laminin-5 (Ln-5) is a heterotrimeric basement membrane (BM) molecule (α3β3γ2). It is a principal protein constituent of the anchoring filaments, which connect the BM with the hemidesmosomes of the basal keratinocytes and possess a crucial function in keratinocyte adhesion. Confocal immunofluorescence imaging is introduced for a quantitative evaluation of the Ln-5 content in the BM of oral squamous epithelium. The BM of normal oral mucosa was used as a reference (100%) for comparative analysis and showed a nearly uniform Ln-5 immunofluorescence intensity (99–100%). In all hyperplastic lesions of oral mucosa, the Ln-5 immunofluorescence intensity was increased (107–141%). The increased Ln-5 content in the BM of hyperplastic lesions suggests an increased keratinocyte-BM adhesion, possibly resulting in a higher stability of the oral mucosa. In contrast, in the oral squamous cell carcinoma (OSCC) invasive front, the remaining BM segments were characterized by a decrease in Ln-5 immunofluorescence intensity (35–74%). A stronger decrease of Ln-5-linked kerationocyte-BM adhesion correlates with a higher tumor grade. Because in central areas of carcinoma BM segments with a normal Ln-5 content could be demonstrated, the fundamental Ln-5 diminution in BM segments of the invasive front should be considered as an invasion-associated phenomenon.
A
In contrast to the well-documented structural irregularities and the qualitative differences in the protein content of the BM, only little is known concerning quantitative variations of the BM protein content. Owing to the signal amplification of enzyme-based immunohistochemical techniques and the projection of all immunohistochemical signals of a BM protein yielded from a usual 4-μm-thick immunohistochemical slide in one level, only few variations of the signal intensity occur and a nearly uniform BM staining is seen independent of possible quantitative variations of the protein within the BM. In consequence, for carcinomas a “black-and-white picture” of the BM is accepted in general, because segments with immunohistochemically normal BM exist adjacent to segments with a complete loss of BM proteins (breaks).
Confocal laser scanning microscopy (CLSM) allows the excitation of a fluorochrome and the collection of the emitted light in a defined narrow level within the slide. Quantitative evaluation of the emitted light yields information about the number of primary antibody-binding proteins in the defined level of the BM.
Among the BM constituents, special attention has been devoted to laminin-5 (Ln-5; α3β3γ2), a member of the growing laminin molecule family. Ln-5 has been identified in the BM of squamous epithelium, where it is localized in the anchoring filaments. These filaments connect the hemidesmosomes of the cell with the basal lamina (Rousselle et al. 1991). Mutations in the genes of Ln-5 cause a loss of the basal keratinocyte–BM adhesion, resulting in epidermolysis junctionalis bullosa Herlitz (Matsui et al. 1998).
Whereas the structural irregularities of the BM and qualitative variations of the BM proteins are well documented in association with several disease processes, only little is known about quantitative variations. Therefore, we have introduced a procedure for quantitative confocal immunofluorescence imaging of BM proteins.
Because Ln-5 plays a crucial role in the epithelial cell–BM adhesion and may be modified in reactive or tumor lesions, we have analyzed the keratinocyte–BM adhesion via a comparison of the Ln-5 content in the BM of normal, hyperplastic, and carcinomatous oral mucosa.
Materials and Methods
Tissue Material
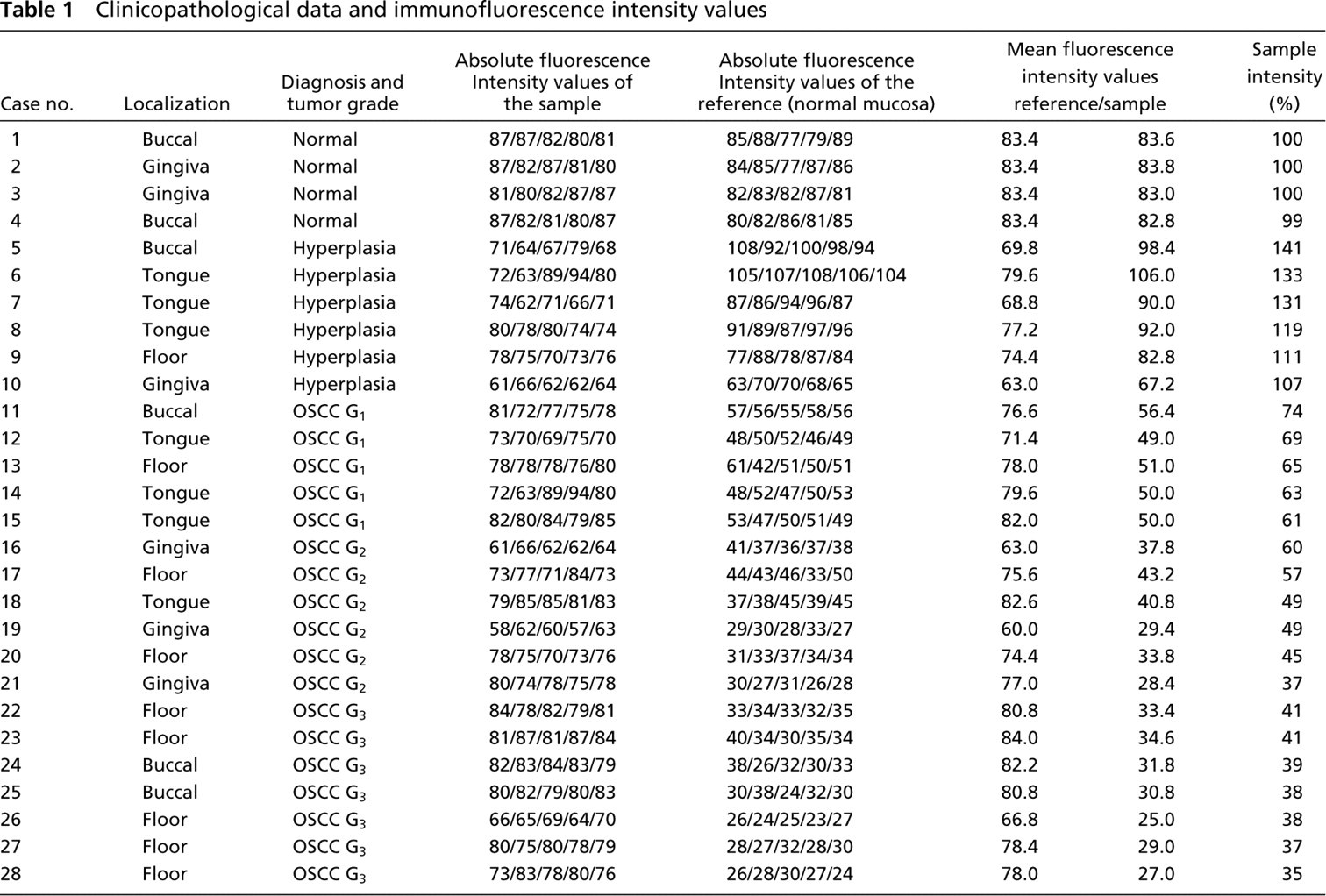
The study included five samples of normal buccal and gingival mucosa, six cases of hyperplasia of oral mucosa, and 18 cases of oral squamous cell carcinoma (OSCC). Samples of 4 mm2, including the epithelial–stromal interface or the deep invasive margins of OSCC, were shock-frozen in isopropanol, cooled by liquid nitrogen, and stored at −75C. The diagnosis and tumor grade were confirmed in the cryostat sections and in the corresponding paraffin-embedded tissue according to the revised WHO classification and Bryne's malignancy grading (Bryne et al. 1992; Pindborg et al. 1997).
Immunofluorescence Labeling
Four-μm-thick cryostat sections of the respective frozen tissue samples were fixed in ice-cooled acetone for 15 min and subsequently subjected to immunofluorescence labeling. The immunofluorescence labeling for collagen Type IV (CIV22, 1:50; Odermatt et al. 1984) was used to indentify the BM region in serial sections. The laminin chain immunolabelings (Ln-α3 chain: BM165, 1:5000; Rousselle et al. 1991; Ln-β3 chain: 6F12, 1:50; Marinkovich et al. 1992; Ln-γ2 chain: GB3, 1:10; Hsi and Yeh 1986; Ln-γ2 chain: D4B5; Mizushima et al. 1998) were interpreted behind the background of the collagen Type IV-based BM identification. The Ln-γ2 chain occurs exclusively in Ln-5. In contrast to the clone D4B5, which recognizes only the Ln-γ2 chain alone or in combination with the Ln-β3 chain and/or Ln-α3 chain, the monclonal antibody GB3 recognizes the Ln-γ2 chain only in the heterotrimeric molecule. Therefore, GB3 was used for quantitative analysis of the assembled Ln-5 molecule (Matsui et al. 1995).
The primary antibodies were diluted in a background-reducing medium (Dako; Glostrup, Denmark). The incubation with the primary antibody was carried out overnight at 4C. After being washed with a Tris buffer, the sections were incubated with secondary goat anti-mouse FITC-conjugated antibodies (Dianova; Hamburg, Germany) diluted 1:50 in a RPMI cell culture medium (BioWhittaker Europa; Verviers, Belgium) for 45 min at room temperature (RT). After rinsing, the slides were mounted with Vectashield (Vector Laboratories; Burlingame, CA) to prevent bleaching (Florijn et al. 1995). As a negative control, the primary antibody was replaced by a non-immune serum.
Laser Scanning Microscopy
The fluorescence labeling was analyzed by CLSM (LSM 510, ApoChromat ×40; Zeiss, Jena, Germany) immediately after the fluorescence labeling procedure. The 488-nm line of the argon laser (LASOS; Jena, Germany) was used to excite the FITC. For the quantitative Ln-5 evaluation, the laser was set to 25% power output and 2% excitation. With these settings no bleaching was observed during the measurement period. The FITC-mediated emission was detected using the LSM-510 configuration “FITC narrow band” (HST 488, filter BP 505–550; Zeiss). The pinhole was set to 90 μm (1 airy unit) and the amplification gain was set to 1. To provide an optimal dynamic range for the measurement, the amplification offset and the detector gain were adjusted for each slide. The nonspecific fluorescence of the background, calculated from areas without Ln-5 deposits, was determined as the threshold value.
To evaluate the Ln-5 content within the areas of BM breaks in the OSCC, the CLSM was set at maximal sensitivity.
Measurement Procedure
For the quantitative analysis, two tissue sections were mounted on one slide: a sample of normal oral mucosa as a reference and a sample of non-normal mucosa, by which the immunofluorescence intensity should signal a variation of Ln-5, on the other. To identify the histopathology before scanning, both sample and reference were visually evaluated using differential interference contrast (DIC).
Using constant settings, five representative high-power Fields (HPFs) of the non-normal sample and five HPFs of the reference mucosa were scanned with 512 × 512 pixels. The images were recorded with 8 bits per pixel, resulting in 255 intensity levels. Using the “area” function of the LSM-510 software, the intensity of Ln-5 immunofluorescence above the threshold was measured (mean intensity per pixel). Areas with artifacts or necrosis were excluded. A periodic (non-continuous) LSM analysis of the same slides or switching the laser illumination off and on was avoided to prevent variations in the laser intensity.
For a comparative evaluation, the immunofluorescence intensity of the sample was given as a percent of the immunofluorescence intensity yielded from the reference (normal oral mucosa) mounted on the same slide.
Statistical Analysis
The significance of the deviations of the mean fluorescence intensity as measured from normal, hyperplastic, and carcinomatous mucosa as well as within G1, G2, and G3 oral carcinomas was performed using the Exact Mann–Whitney test (SPSS, Version 10; SPSS, Chicago, IL).
Results
Ln-5 in Normal Mucosa, Hyperplastic Mucosa, and OSCC Using Conventional Immunofluorescence Microscopy
With monoclonal antibodies to the α3-, β3-, and γ2-chains of Ln-5 in normal adult squamous epithelium, a narrow and sharply contoured immunolabeling was demonstrated in agreement with collagen Type IV immunolabeling at the epithelial–stromal interface (BM region) (Figure 1A). In hyperplastic lesions of oral mucosa, the Ln-5 immunostaining was retained in the BM region. Compared to the Ln-5 immunostaining of the normal oral epithelium, the BM showed irregularities that included thickening and a blurred outline. No BM breaks were observed (Figure 1B). Only a few small Ln-5 spots were seen beneath the BM within the stromal compartment. In the OSCC, fundamental irregularities of Ln-5 immunolabeling occurred. Three distinct patterns could be distinguished: a cytoplasmic Ln-5 immunolabeling within the invading carcinoma cell complexes; many defects in Ln-5 immunolabeling of the BM; and spots of Ln-5 within the stromal compartment adjacent to invading OSCC cell complexes. In contrast to the Ln-γ2-chain antibody D4B5 and the Ln-β3-chain antibody, the Ln-γ2 antibody GB3 revealed, if at all, only sparse and weak immunolabeling outside the collagen Type IV-marked BM region. The Ln-α3-chain was also detected in pre-existing vessels (Figure 1C–1F).
Ln-5 Content in the BM of Normal Oral Mucosa
Under the following conditions [all compared tissue samples on the same slide, a standardized immunolabeling procedure (monoclonal antibody GB3), laser and filter settings kept constant, and a continuous (unbroken) laser scanning in the microscopic evaluation] the immunofluorescent analysis of the Ln-5 content in the BM of normal oral mucosa showed only minimal variations (Figure 2A and 2B). The analysis showed a nearly uniform Ln-5 immunofluorescence intensity (99–100%; Table 1) in the BM of the normal oral mucosa, indicating a uniform Ln-5 content and distribution independent of the localization. The mounting of two different tissue samples on one slide enables the comparative and reproducible analysis of the Ln-5 immunofluorescence intensities.
Ln-5 Content in BM of Hyperplastic Lesions of Oral Mucosa
An increase of Ln-5 immunofluorescence in the BM of all hyperplastic lesions of oral mucosa (monoclonal antibody GB3) could be detected (Table 1; Figure 3). Compared to normal oral mucosa, the fluorescence intensity was raised to minimal 107% and maximal 141% and proved to be significant (p = 0.01).
Ln-5 Content in BM of OSCC
The detection of the extent of BM breaks is dependent on the intensity of the laser light illumination used. Despite the use of high-powered laser illumination, a complete focal loss of Ln-5 (all antibodies against Ln-5 chains) or collagen Type IV immunofluorescence from the BM was demonstrated.
There was an Ln-5 immunofluorescence gradient within the tumor, visible with all antibodies applied to chains of Ln-5. The Ln-5 content of the remaining/newly forming BM of the invasive front was lower than in the more central areas of the carcinoma. The center of the OSCC contained BM segments with Ln-5 immunofluorescence intensities comparable to those of normal oral mucosa. Moreover, the number and extent of the BM breaks were lower here than in the invasive front (Figure 2B and 2C).
In all the carcinoma samples, the Ln-5 content of the retained BM in the invasive front was statistically significantly reduced (p < 0.001) compared to the normal oral mucosa (35–74%). With a higher OSCC malignancy grade, a diminution of the Ln-5 content occurred within the retained/reestablished BMs of the invasive front (Table 1; Figure 3). This difference was particularly pronounced if the malignancy grading of OSCC invasive front according to Bryne and co-workers (1992) is applied (G1-G2 p = 0.04; G2-G3 p = 0.035; G1-G2/3 p < 0.001; Table 1).
Discussion
Pyke and co-workers (1994) described an increased Ln-γ2-chain synthesis in budding tumor cell complexes in the invasive front of various carcinomas and suggested an invasion via Ln-5. This finding was confirmed for OSCC by Kainulainen and co-workers (1997). Concerning the distribution of Ln-5 chains in OSCC, non-small-cell lung cancer, and in gastric, colon, and pancreatic adenocarcinoma, three distinct invasion-associated patterns were revealed: a retention of Ln-5-chains in the cytoplasm of invading carcinoma cell complexes; BM segments with retained Ln-5 immunostaining adjacent to segments with a complete loss of Ln-5 in the BM; and a deposition of Ln-5-chains within the stroma beneath the invading carcinoma cell complexes (Kosmehl et al. 1996, 1999; Tani et al. 1997; Thorup et al. 1997; Sordat et al. 1998; Maatta et al. 1999).
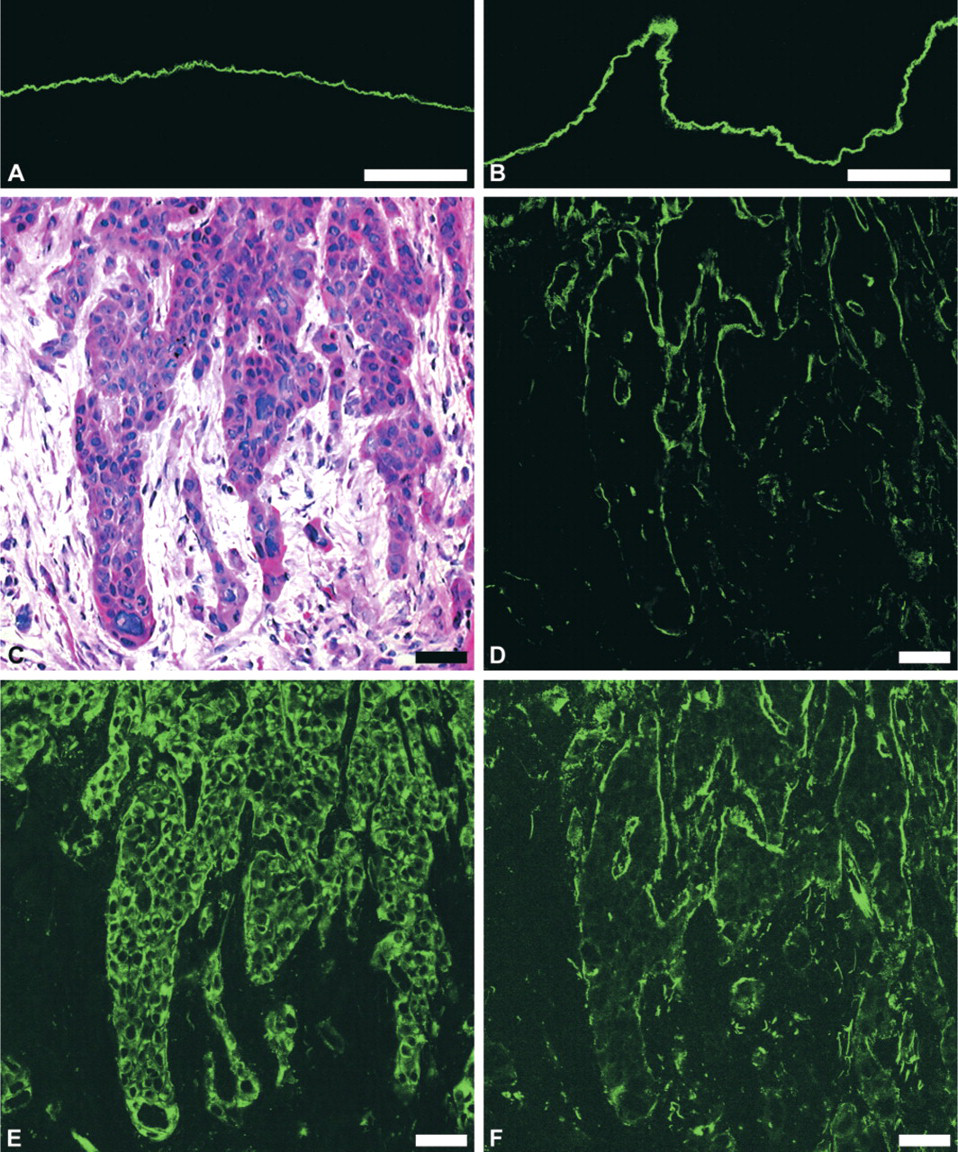
Demonstration of Ln-5 in normal, hyperplastic, and carcinomatous oral mucosa by CLSM. In normal oral mucosa a uniform Ln-5 immunofluorescence labeling of the BM is demonstrated (
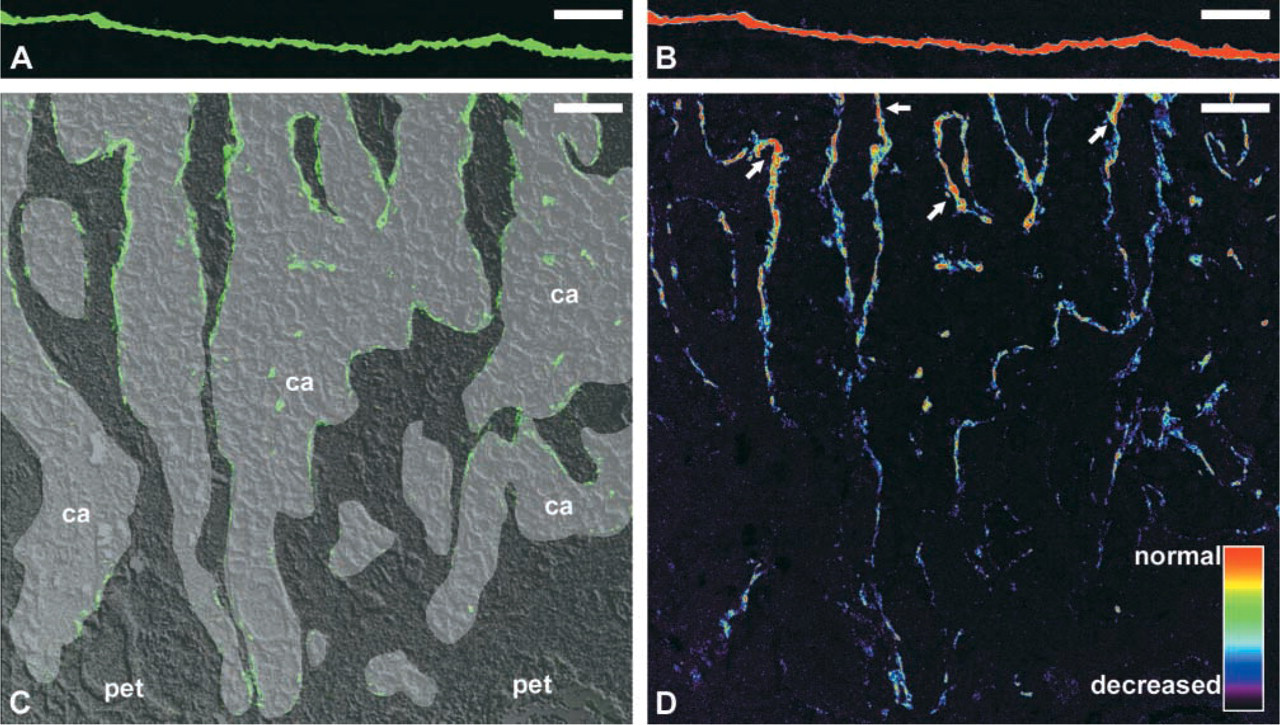
Demonstration of the Ln-5-γ2-chain immunofluorescence intensity in the BM of normal oral epithelium (
Prostate carcinoma and invasive duct or lobular carcinoma of the breast are characterized by an almost complete or total loss of Ln-5 in the BM without stromal Ln-5 deposition (Yeh et al. 1986; Hao et al. 1996; Henning et al. 1999). Therefore, it has recently been proposed that Ln-5 plays different roles in the invasion of different carcinomas (Henning et al. 1999).
Comparison of the immunolabelings of the monoclonal antibodies D4B5 (recognizing the Ln-γ2-chain alone or in combination with other chains of Ln-5) and GB3 (recognizing the Ln-γ2-chain exclusively in combination with other chains of Ln-5) reveals that, outside of the BM, the γ2-chain is most frequently not associated with the other chains of Ln-5 in OSCC. The functional properties of complete Ln-5, single Ln-γ2-chain, or Ln-5 fragments may be different. It is assumed that a complete Ln-5 is associated with keratinocyte adhesion, whereas Ln-5 fragments promote migration (Koshikawa et al. 2000; Sordat et al. 2000; Supurna and Stack 2000; Tsubota et al. 2000). Therefore, the GB3 antibody represents an ideal tool to monitor the Ln-5-mediated BM–keratinocyte adhesion in a carcinoma invasive front.
Because of the almost exclusive immunolabeling of GB3 in the BM region, the GB3 immunofluorescence signals can be considered as BM-specific. Antibodies with an immunolabeling pattern not restricted to the BM require a predefinition of the BM region for determination of measurement area.
The quantitative analysis of the Ln-5 content in the retained or re-established BM segments of an OSCC invasive front, using CLSM, reveals a clear decrease of Ln-5 in congruence to prostate and breast carcinoma. Our results reveal a reduced carcinoma cell–BM adhesion mediated by Ln-5 as a common step in the process of cancer cell invasion, obviously independent of carcinoma type. The diminution of Ln-5-mediated cell adhesion is in line with previous observations and parallels the loss of hemidesmosomal integrins. Whereas within the whole OSCC the number of integrin α6β4-bearing carcinoma cells is increased, in the invasive front a diminution of this hemidesmosomal lamininbinding integrin is found in association with a loss of Ln immunostaining of the BM (Downer et al. 1993; Kosmehl et al. 1995). Moreover, in prostate and breast carcinoma, a loss of hemidesmosomes connecting the cell with the Ln-5-containing anchoring filaments of the BM was demonstrated (Bergstraesser et al. 1995; Nagle et al. 1995). Carcinoma invasion embraces simultaneous BM degradation as well as formation of new BM. The reduced Ln-5 content in the BM segments of invasive front can be explained by a loss of integrins essential for assembly of Ln-5 into the BM or by proteolytic activities in the context of the invasion. The ability of carcinomas to form BMs is evidenced by the occurrence of BMs in metastasis (Hida et al. 1994).
Clinicopathological data and immunofluorescence intensity values
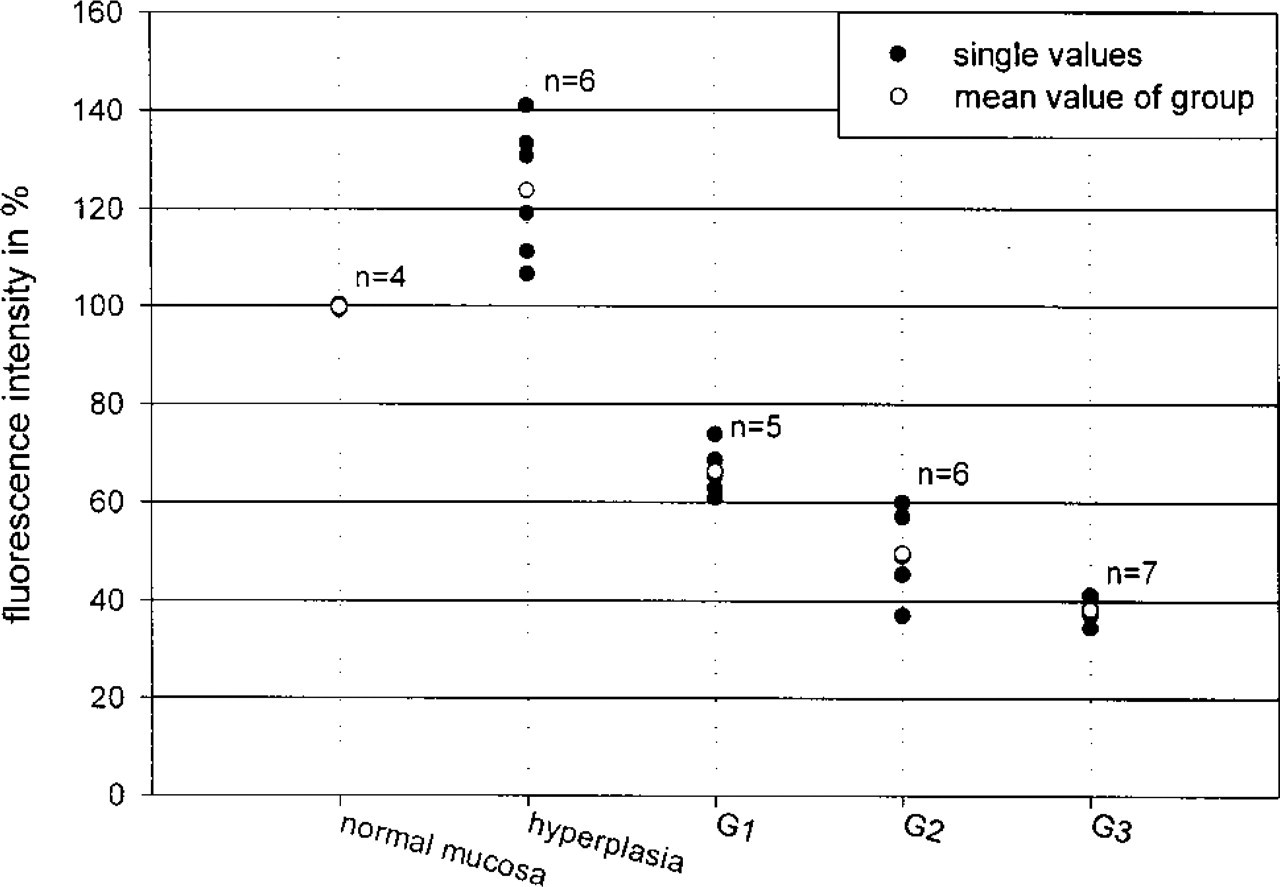
Variation of the Ln-5 immunofluorescence intensity in the BM of normal and hyperplastic oral epithelium as well as in the OSCC invasive front in relation to malignancy grade (Bryne et al. 1992).
To our knowledge, this is the first study for the quantitative analysis of BM proteins using laser scanning microscopy. As yet there have been only a few attempts to quantify BM proteins histologically by immunogold particle counting using an electron microscope (Posthuma et al. 1986; Chevalier et al. 1995; Moriya et al. 2001). The quantitative evaluation of immunolabeled proteins and other fluorescent substances in situ represents an already established application of laser scanning microscopy (Brown et al. 1993; Huntsman et al. 1999; Verschure et al. 1997). The CLSM presents images that are almost free of out-of-focus signals and dramatically reduces interference from out-of-focus structures by allowing optical sections of a specimen with adjustment of plane of focus (Hashimoto et al. 1999). Nevertheless, the combination of quantitative immunofluorescence labeling and confocal microscopy needs special attention regarding methodological problems. First, there is a quantitative but not a stoichiometric relation between bound antibodies, fluorescence intensity, and absolute protein amount. Second, the measured intensity may be reduced by some excitation and emission light absorption from tissue more superficial than the plane of focus. Relative measurements between identically treated samples eliminate the use of strict quantification in the amounts of a protein and still enable one to assess whether biological conditions result in a measurable relative change in the amounts of a protein (Rastogi et al. 1997). This has been our rationale for reporting only relative fluorescent intensity differences among our samples.
The number of all BM defects correlates with the invasive and metastatic potential of OSCC (Harada et al. 1994). As recently confirmed in a tumor model, the loss of Ln-5 and hemidesmosomes correlates with invasive behavior (Tomakidi et al. 1999). By comparative quantitative analysis of the retained BM segments in OSCC, we have shown that a decrease of Ln-5 corresponds to an increased malignancy grade. Moreover, we have also demonstrated an intratumor gradient regarding the Ln-5 content in the BM of the tumor complexes. The lowest Ln-5 content was found in the BM of tumor complexes at the deep invasive margins of OSCC. In contrast to the periphery, in the central area of carcinoma the BM of tumor complexes showed Ln-5 immunofluorescence intensity reaching normal levels. The finding that the most reduced Ln-5-mediated cell–BM adhesion occurs in the invasive OSCC margins concurs with the suggestion that the invasive front of carcinomas represents an important area for tumor prognosis. Furthermore, our findings support the application of the histopathological malignancy grading of the invasive front for OSCC (Bryne et al. 1992, 1998).
Surprisingly, a significant difference was demonstrated in the Ln-5 of BM between hyperplastic lesions of oral mucosa and OSCC. In contrast to OSCC, the Ln-5 immunofluorescence in hyperplasia was increased compared to that in normal oral mucosa. Hyperplastic lesions of oral squamous epithelium represent an adaptive response to irritation. The increased Ln-5 content in hyperplasia may be considered a contribution to a higher stability of the oral mucosa by increased keratinocyte–BM adhesion. As in OSCC, the hyperplasia of oral mucosa can include an increased mitotic activity and an increase of basal-respective parabasal cells. Therefore, the decrease of Ln-5 in the retained BM segments in OSCC may represent an invasion- but not a differentiation-associated phenomenon.
In summary, the analysis of the keratinocyte–BM adhesion distinguishes benign oral squamous epithelium from invasive malignant oral squamous epithelium by the level of Ln-5 content.
Footnotes
Acknowledgements
Supported by Grant ThMWK no. 973214.
We thank Dr Patricia Rousselle (Institut de Biologie et Chimie de Protéines, Université Lyon I, France) for kindly providing the monoclonal antibodies BM165 and 6F12 and for encouragement and critical advice in the ECM studies. We thank Ms Christiane Rudolph for skillful technical assistance.
