Abstract
Keywords
D
To investigate the function of this glycoconjugate-rich BL, we have applied a recently developed experimental model to locally administer tunicamycin (TM), an antibiotic that interferes with glycosylation. The system consists of drilling a “window” in the alveolar bone overlying the apex of the rat incisor and connecting to it an osmotic minipump (Vu et al. 1999). This model allows local infusion with relatively large concentrations of biological or chemical agents, over a defined period of time, without disturbing the whole-animal biology.
TM inhibits the first step in the lipid carrier-dependent protein glycosylation (dolichol pathway), specifically preventing N-linked glycosylation (Takatsuki et al. 1975; Elbein 1988). It has been reported to affect morphogenesis and cell differentiation in various tissues and, notably, the organization of basement membranes (Hart and Lennarz 1978; Ekblom et al. 1979; Yang and Hilfer 1982; Giraud and Franc 1989; Kanai et al. 1991; Webster et al. 1993). TM has been largely applied in vitro because it is highly toxic in vivo, causing major alterations of the nervous system (González et al. 1981; Jago et al. 1983; Finnie and O'Shea 1988; Leaver et al. 1988; Pow and Morris 1992). To our knowledge, only three studies have been reported in the tooth organ. This inhibitor of N-glycosylation alters basement membrane formation in tooth buds and blocks odontoblast differentiation in vitro (Thesleff and Pratt 1980a,b). In vivo, it causes an accumulation of glycogen in some cells of the enamel organ and in young odontoblasts (Chardin et al. 1989).
In this study, the effect of TM on enamel maturation was examined at the light and electron microscopic levels. Lectin–gold cytochemistry was performed to reveal and map the distribution of glycoconjugates. Immunodetection of enamel proteins (EPs) and ALB was carried out to verify whether N-linked sugar depletion in the BL structural components affects the content and distribution of endogenous and exogenous proteins in the enamel layer.
Materials and Methods
Surgical Procedure
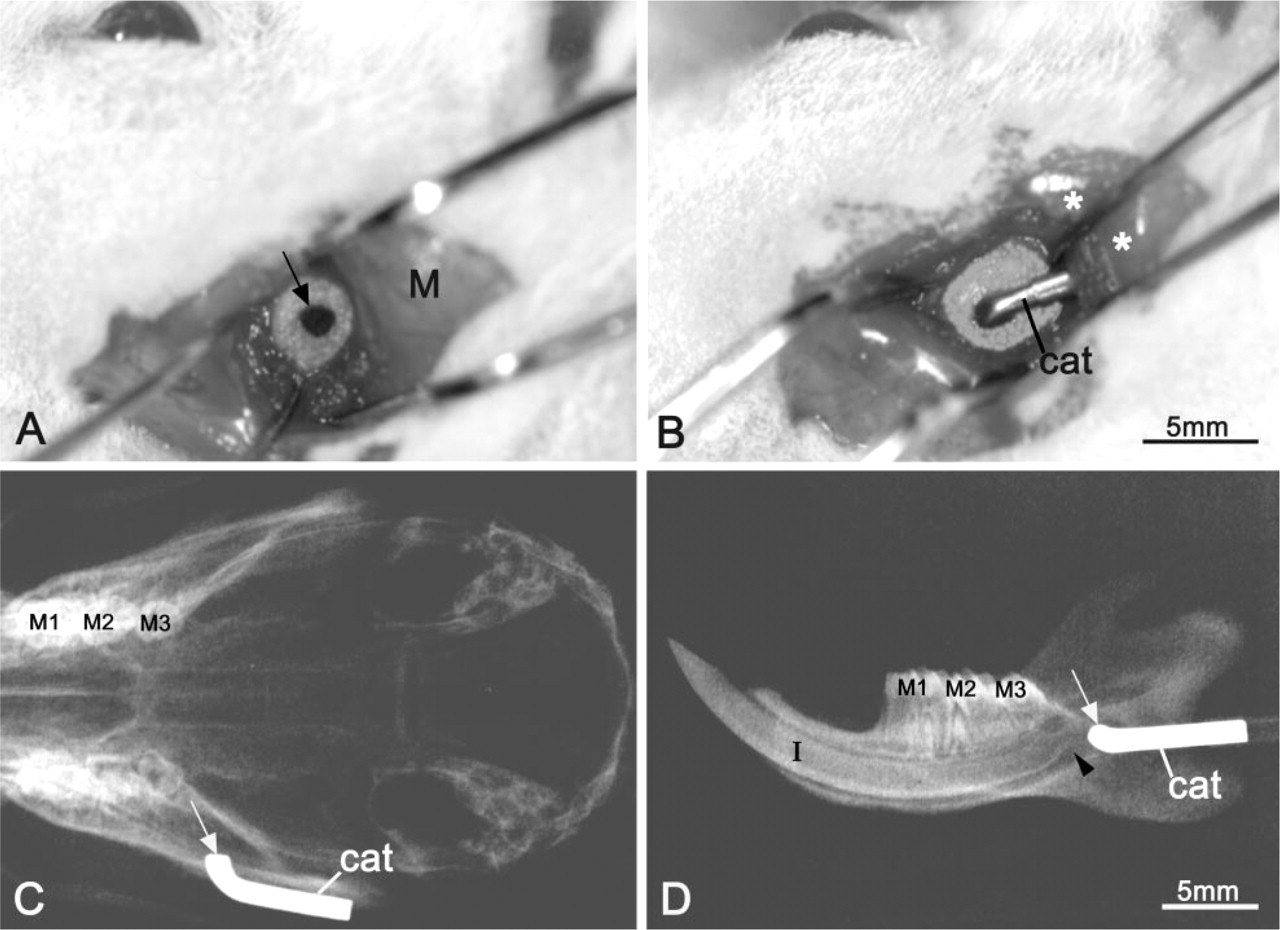
Male Wistar rats weighing 100 ± 10 g (Charles River Canada; St-Constant, QC, Canada) were anesthetized with a 0.27-ml IP injection of a 1:1:2 mixture of Hypnorm (fentanyl citrate and fluanisone; Janssen Pharmaceutica, Beerse, Belgium), Versed (midazolam; Hoffmann-LaRoche, Mississauga, ON, Canada), and distilled water. An incision was made through the skin to access the muscle layer, according to an imaginary line joining the auditory meatus and the lip commissure. After separation of the fibers of the masseter muscle and elevation of the periosteum, the bony surface of the ramus was exposed. A slow-speed dental drill equipped with a carbide round burr, size 0.14 (Brassler; Montreal, QC, Canada), was used to create a hole through the alveolar bone under manual saline irrigation (Figure 1A). The hole was drilled approximately 2 mm anterior to the posterior border of the ramus and slightly superior to the bony elevation overlying the apical end of the incisor. Another incision through the skin in the neck area was made to accomodate a 7-day Alzet osmotic minipump (model 2001D; Alza Corporation, Palo Alto, CA). The pump was tunneled into a subcutaneous pouch on the back of the animal and connected to the bony hole using a vinyl tubing and a metal catheter (Figure 1B). The tubing was passed underneath the masseter muscle and through the neck area. Tissue adhesive Indermil (distributed by Sherwood Davis & Geck; St Louis, MO) and bone cement (Zimmer; Warsaw, IN) were used to help immobilize the metal catheter against the bone surface and maintain its tip in the hole. The animals were then sutured and the surgical site was cleaned and disinfected with 70% ethanol. Some rats were administered Temgesic (buprenorphine hydrochloride; Reckitt & Colman, Hull, UK) as an analgesic to control postsurgical pain. Five days after surgery, X-ray radiographs of the rat mandibles were taken to verify that the catheter was well in place (Figures 1C and 1D). Animal procedures and experimental protocols described above were in accordance with guidelines of the Comité de déontologie de l'expérimentation sur les animaux of Université de Montréal.
Preparation and Administration of TM
Tunicamycin (Sigma Chemical; St Louis, MO) was first dissolved in 0.01 M sodium hydroxide, pH 9, and then diluted to a final concentration of 10 μg/ml with 0.1 M PBS, pH 7.2 (Pow and Morris 1992). The solution was freshly prepared before use. Six rats were each implanted with an osmotic minipump filled with ∼230 μl of TM solution. The minipumps were connected to a vinyl tubing, also filled with the drug, and soaked in sterile saline at 37C for 1–3 hr before placement, as described above. Controls consisted of three rats implanted with the same minipumps filled with physiological saline (Sigma).
Tissue Processing
On Day 8 after placement of the minipumps, the animals were anesthetized with an IP injection of 0.25 ml of 20% chloral hydrate (Sigma) and sacrificed by intravascular perfusion through the left ventricle. The vasculature was prerinsed with lactated Ringers'solution (Abbott Laboratories; Montreal, QC, Canada) for about 30 sec, followed by perfusion with a fixative solution consisting of 1% glutaraldehyde in 0.1 M sodium phosphate (PB), pH 7.2, for 20 min. Both hemimandibles were dissected out and immersed in the fixative overnight at 4C. They were then washed in 0.1 M PB, pH 7.2, and decalcified either in 4.13% disodium ethylene tetra-acetic acid (EDTA) for 21 days at 4C (Warshawsky and Moore 1967) or in a solution consisting of hexahydrate aluminum chloride, chloridric acid (10 N), 88% formic acid (Plank and Rychlo 1952), diluted 1:8 with distilled water, for 3 days at 4C. Segments of incisors containing the early and mid-maturation stage were prepared using a molar reference line (Smith and Nanci 1989). Each segment was split in half along its length using a double-edged razor blade (Smith 1974) and washed for 2 hr in 0.1 M PB. All hemisegments were then dehydrated in graded alcohols and embedded in LR White resin (London Resin; Berkshire, UK). The contralateral hemimandible, liver, duodenum, kidney, and parotid gland were similarly processed for embedding in LR White resin.
(
Each tooth segment was oriented for sectioning along its longitudinal axis. One-μm-thick sections were cut with glass knives on a Reichert Jung Ultracut E ultramicrotome and stained with toluidine blue. Thin sections of selected areas were then prepared with a diamond knife and mounted on 200-mesh nickel grids having a carbon-coated Formvar film. The sections were then processed for postembedding lectin-gold cytochemistry for detection of glycoconjugates (reviewed in Benhamou 1989), and for colloidal gold immunocytochemistry (reviewed in Bendayan 1995) for detection of EPs and ALB. All grids were stained with 4% aqueous uranyl acetate and lead citrate for examination in a JEOL JEM-1200EX-II transmission electron microscope operated at 60 kV.
Lectin Cytochemistry
Helix pomatia agglutinin (HPA), specific for N-acetyl-
Immunocytochemistry
For immunolabeling of the two main classes of EPs, amelogenin and non-amelogenin (see Fincham et al. 1999), sections were blocked by floating for 15 min on a drop of 0.01 M PBS containing 1% ovalbumin (Oval; Sigma). Briefly, for the amelogenin class, sections were transferred either on a drop of a chicken egg yolk antibody (Gassmann et al. 1990) raised against 24-kD rat amelogenin (AMELy) (Chen et al. 1995; Nanci et al. 1996b), diluted 1:150 for 3 hr, or against mouse recombinant 179 amelogenin isoform (M179y) (Orsini et al. in press) diluted 1:100 for 3 hr. They were washed with PBS, refloated on PBS–Oval, and then incubated for 1 hr with a rabbit anti-chicken IgG antibody (diluted 1:2000) (Cappel Research Products; Scarborough, ON, Canada). Finally, grids were rewashed with PBS, blocked with PBS–Oval, and incubated with protein A–gold complex for 30 min. For ameloblastin (AMBN, a non-amelogenin), sections were incubated for 1 hr with anti-rat recombinant AMBN antibody diluted 1:20, as recently described (Nanci et al. 1998), followed by protein A–gold complex for 30 min. For immunodetection of ALB, sections were incubated for 1 hr with rabbit anti-rat albumin (Cappel) diluted 1:80, followed by protein A–gold for 30 min. After immunolabeling, the grids were extensively washed with PBS, followed by distilled water. Controls consisted of incubations with the secondary antibody and protein A–gold or with protein A–gold alone. All incubations were carried out at RT.
Results
Macroscopic Observations and Light Microscopy
Appropriate positioning of the bony window was confirmed on X-rays and by visual inspection at time of dissection (Figures 1C and 1D). Continous exposure of the dental organ to TM for 7 days affected tooth eruption such that the treated incisor was about 0.5–1 mm shorter than the contralateral one (Figure 2B). None of the rats implanted with saline minipumps showed a notable difference in length between the two incisors (Figure 2A). In some cases, a slight erosion was noted surrounding the hole, probably due to the combined inflammatory effects of the adhesives used.
Histologically, in early to mid-maturation, there was no significant alteration of the enamel organ (Figure 3A). However, in some regions, groups of odontoblasts appeared to have degenerated (Figure 3B) or to have lost their secretory polarity (Figure 3C). In the latter case, the predentin layer was thickened (Figure 3C). Dentin consistently exhibited a differential metachromatic staining pattern at about half its thickness (Figures 3A and 3B). None of the above cell and matrix alterations was observed in control rats. Duodenum, kidney, liver, and parotid cells of treated and control animals showed no significant structural alterations.
Ultrastructural Observations
The BL in incisors from hemimandibles infused with saline (Figures 4A and 5A) and from contralateral teeth from both saline- and TM-infused rats showed structural characteristics similar to those previously described in the incisors of normal animals (Nanci et al. 1993). However, in TM-treated incisors, both its integrity and structure were affected. In some regions the BL was absent or difficult to distinguish (Figures 4B, 4C, and 6B), whereas in others it increased substantially in thickness (Figures 6A and 8). Where enamel is almost EDTA-soluble (Warshawsky and Smith 1974), the BL often ramified into the enamel layer (Figure 5B) and showed an irregular surface (Figure 11C).
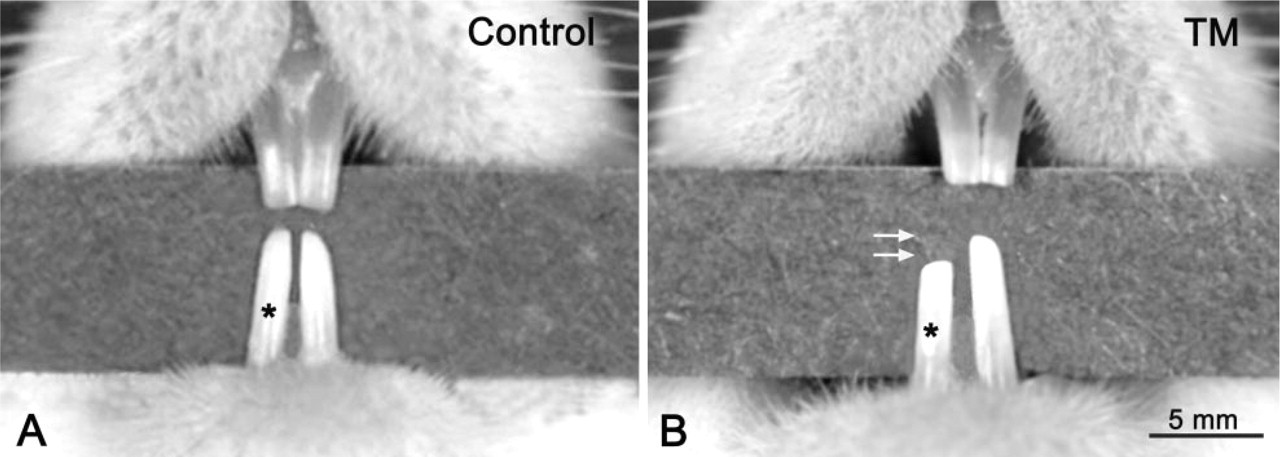
Micrographs showing the incisors in a rat infused for 7 days with (
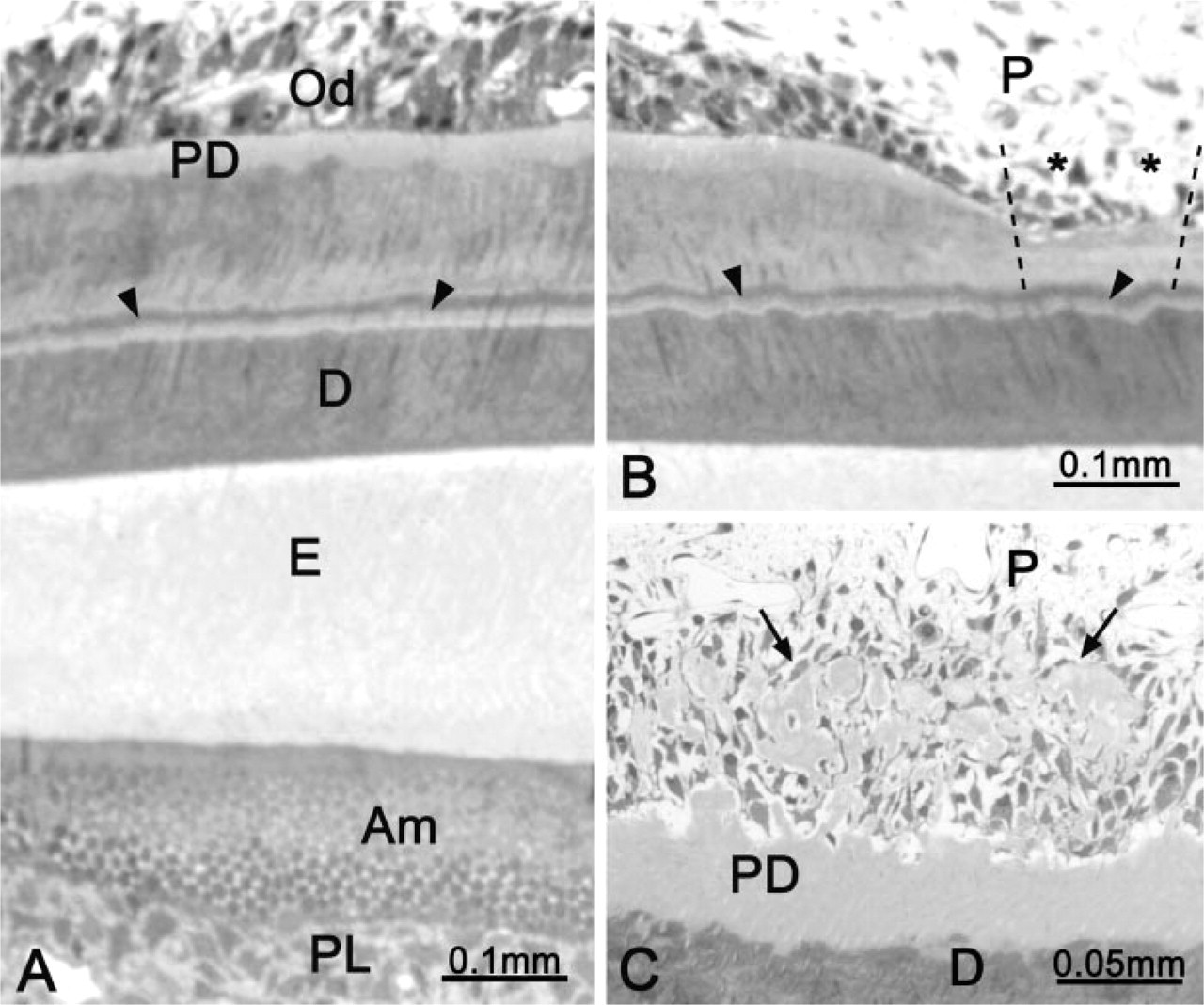
Light micrographs illustrating typical histological alterations observed after tunicamycin administration. (
Lectin Cytochemistry
In control rats, the BL showed uniform and intense labeling with HPA (Figures 4A and 5A). At the start of the maturation stage, the labeling over the BL in TM-treated rats appeared weaker than in controls, and was sometimes interrupted (Figure 4B), but for the most part the labeling appeared to have the same density as in controls. In some cases, the labeling normally associated with the BL appeared to diffuse into the overlying enamel matrix (compare Figures 5C and 5A). In regions at which the BL was enlarged, HPA binding sites were found throughout the structure, sometimes showing a concentration at the interface with enamel (Figure 6A).
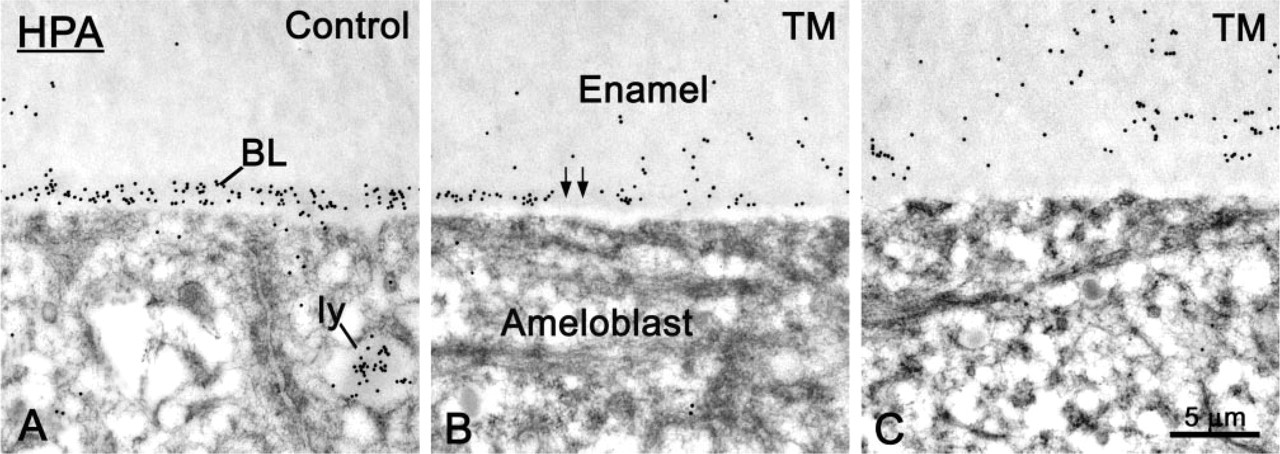
Cytochemical preparations with Helix pomatia agglutinin (HPA) for the detection of N-acetyl-D-galactosamine sugar residues. (
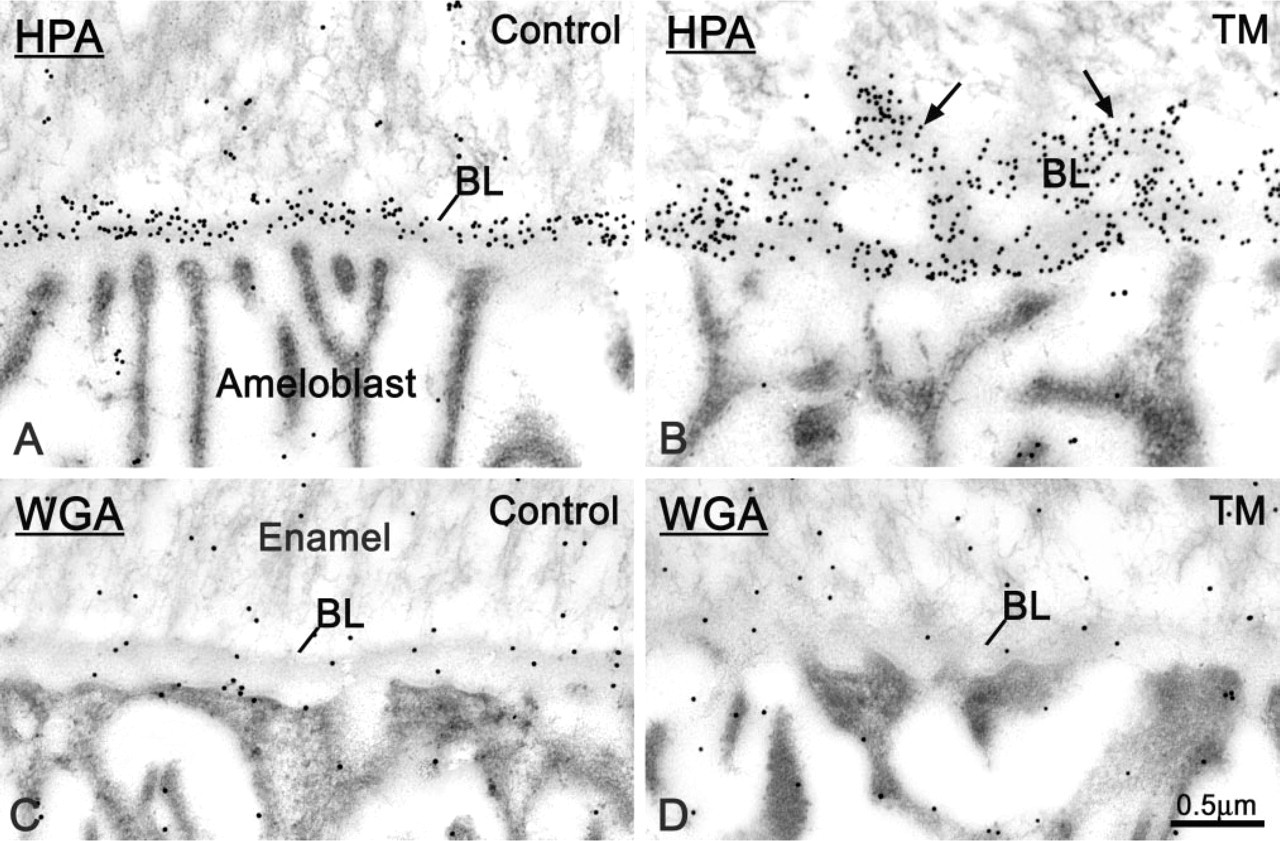
Micrographs from sections of control and tunicamycin (TM)-treated rats incubated with (
In both control and TM-treated rats, labeling with WGA over the BL was weak and was difficult to distinguish from that over the adjacent enamel (Figures 5C and 5D). In TM-treated teeth, regions of altered BL did not show any accumulation of gold particles near the cell surface (Figure 6B). Qualitative estimation suggested that the overall WGA labeling over the BL and enamel was somewhat weaker in TM-treated than in saline-infused animals (Figures 5C and 5D).
Immunolocalization of Enamel Proteins and Albumin
All three anti-EP antibodies showed slightly weaker immunoreactivity over enamel from TM-treated incisors compared to saline-infused teeth.
Both anti-amelogenin antibodies used labeled the enamel matrix. However, they yielded different distribution patterns. Anti-AMELy showed an intense reaction throughout the enamel layer (Figure 7A), whereas labeling with anti-AMEL M179y was more intense near the enamel surface (Figure 7B). In TM-treated incisors, in regions at which the BL appeared widened, immunoreactivity for amelogenin started farther away from the cell surface (Figure 8). In general, labeling for AMBN over enamel was very weak and in treated rats was almost absent in the region where rods are visible (Warshawsky and Smith 1974) (Figure 7C). With all three antibodies to EP, there was little or no labeling over the BL in both control and treated rats (Figures 7A and 8A).
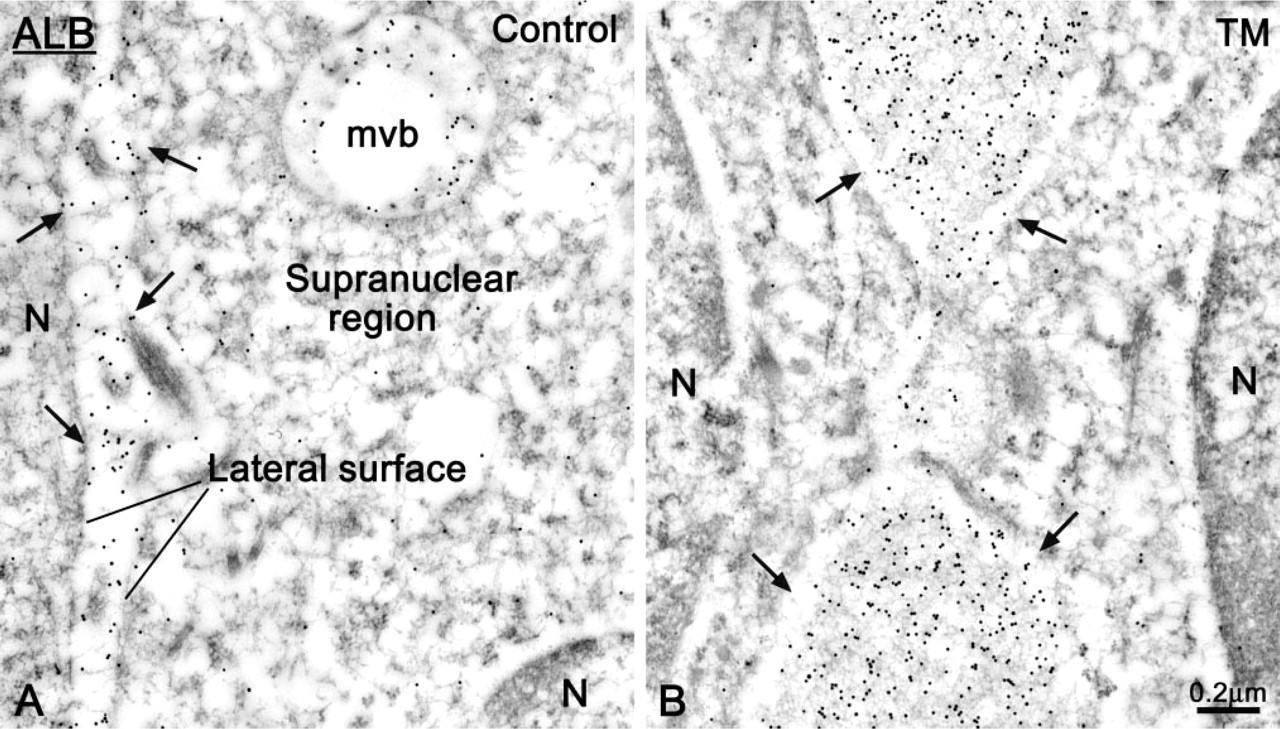
In both control and TM-treated rats, ALB was immunodetected in the intercellular space along the basolateral surface of ameloblasts. However, in treated animals, accumulation of ALB between cells was more common and abundant (compare Figures 9B and 9A, and Figures 10B and 10A). There were very few or no gold particles over enamel in control rats, but there was weak and conspicuous labeling at the dentino–enamel junction (Figures 11A and 11B). The number of gold particles over maturing enamel was significantly higher in TM-treated incisors, including at the dentino–enamel junction (Figures 8B, 11C and 11D). Labeling for ALB over enamel was patchy and more intense around regions where the BL was absent or significantly altered.
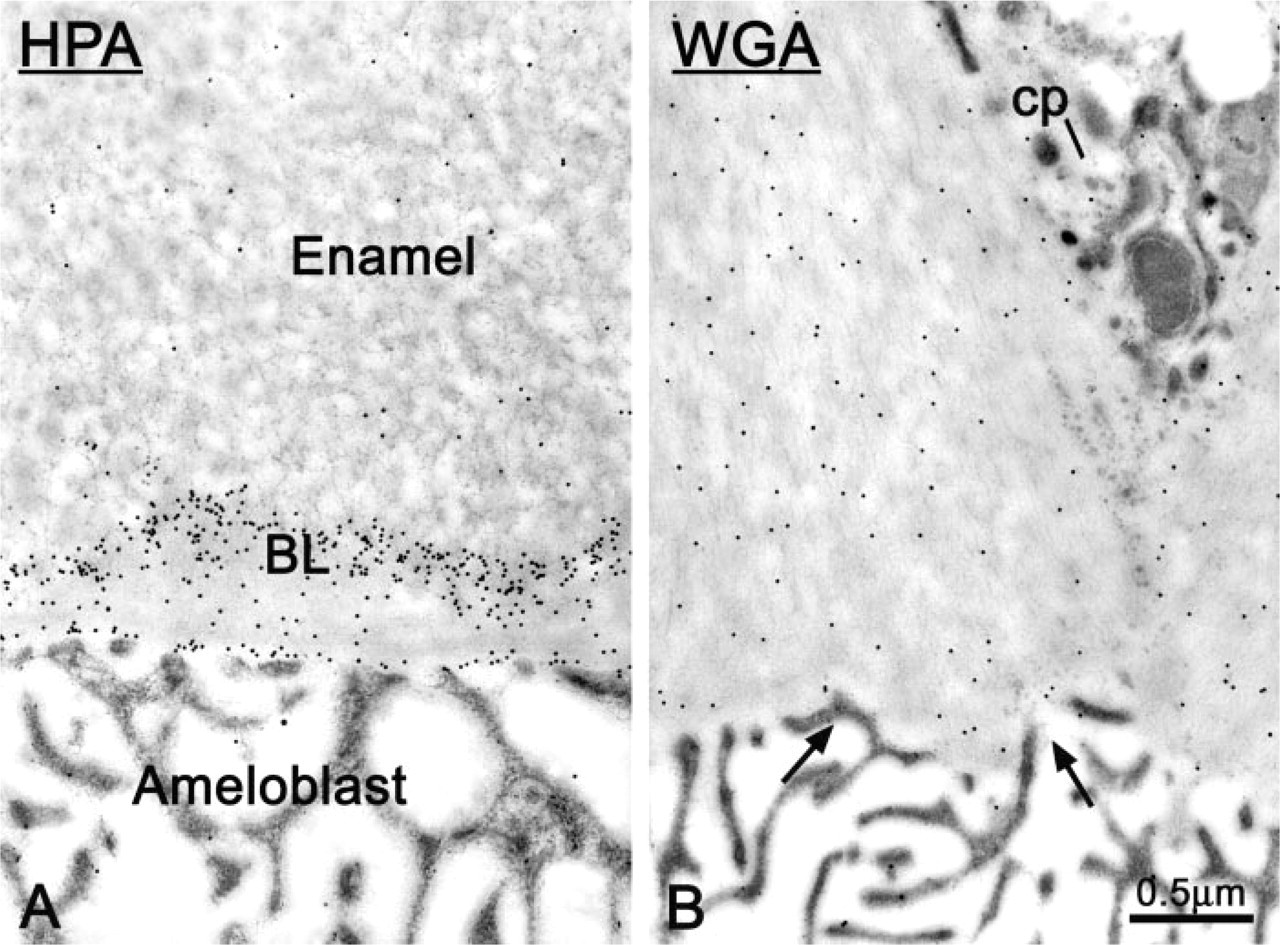
The maturation stage basal lamina (BL) is regionally affected during TM administration. In some regions (
Control Incubations
Labeling was significantly reduced when sections were incubated with HPA or WGA in the presence of their respective competing saccharides, or when they were incubated with anti-HPA or anti-WGA only, followed by protein A–gold. In the case of incubations for EPs and ALB, controls exhibited abolition of the selective labeling observed and the presence of few, randomly distributed gold particles throughout the tissue sections.
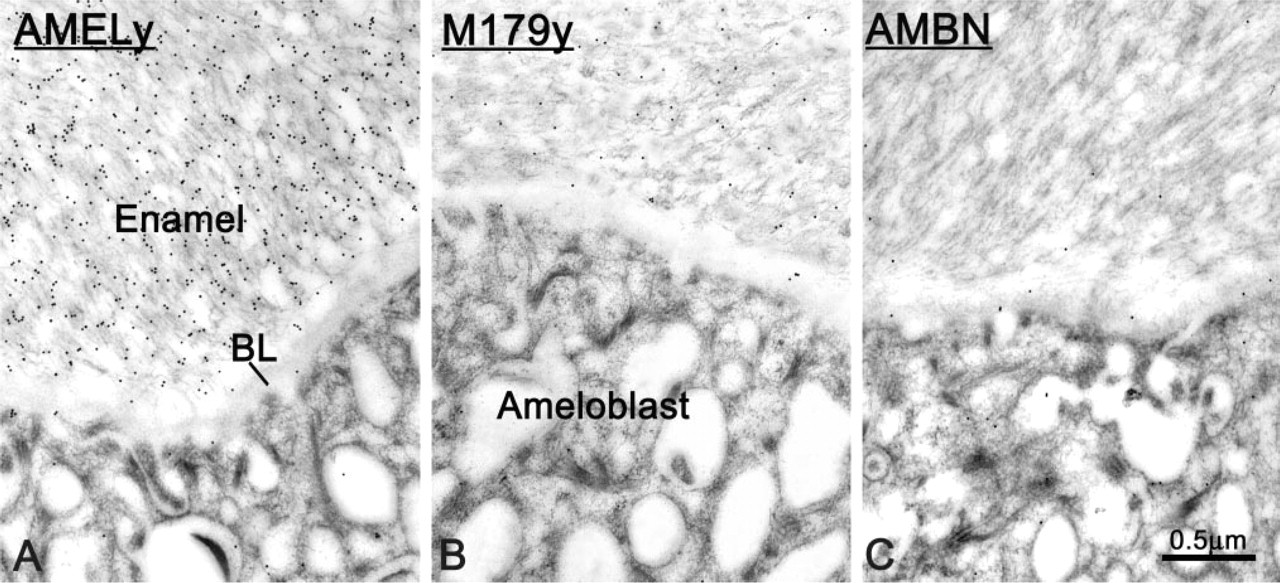
Immunocytochemical preparations from TM-treated rats incubated with antibodies raised against (
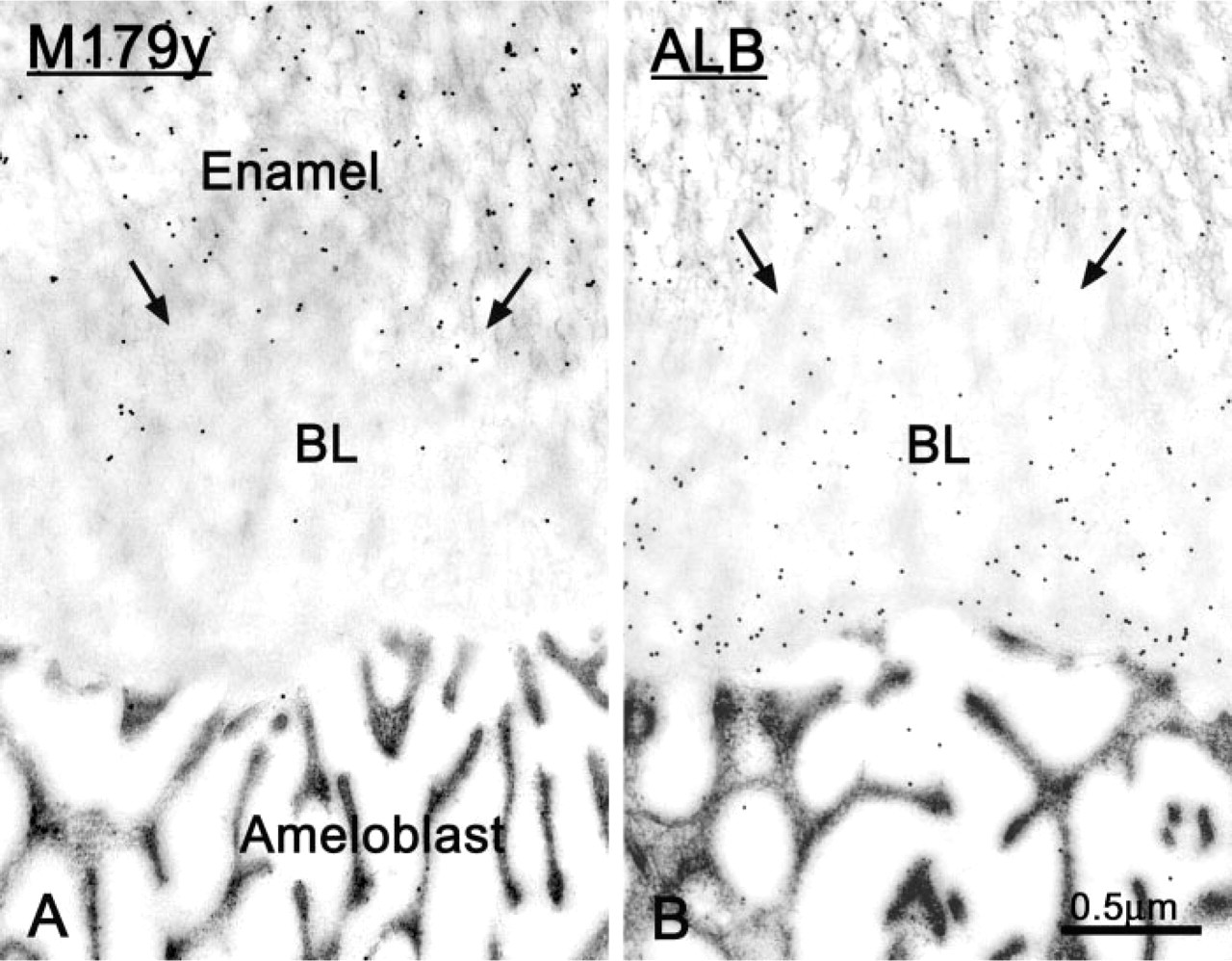
Immunocytochemical preparations from TM-treated rats in a region of the incisor at which the maturation stage basal lamina (BL) is thickened. (
(
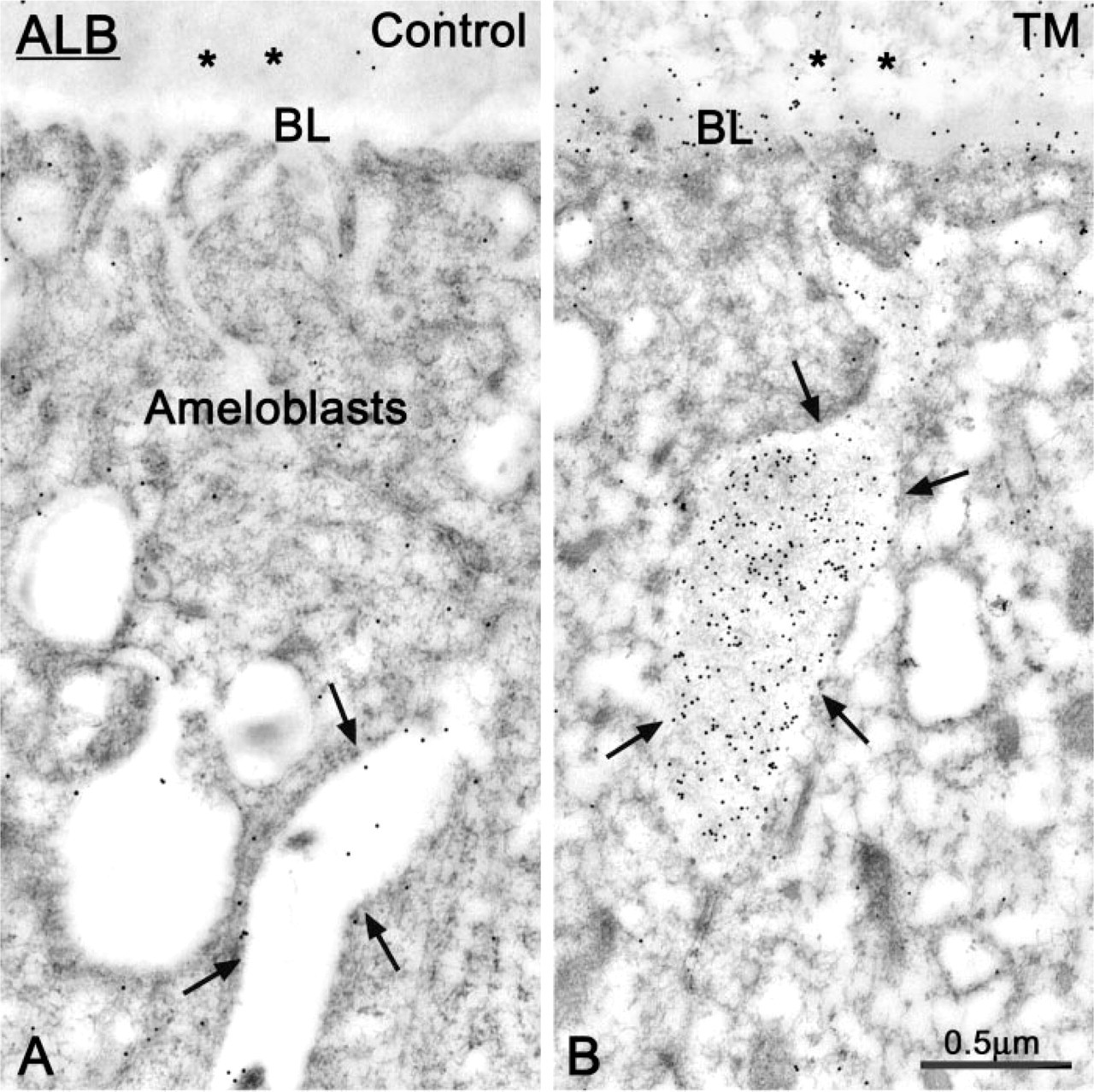
Electron micrographs from sections immunolabeled for albumin (ALB), showing the apical portion of ameloblasts. (
Discussion
This study demonstrates that it is possible to achieve a localized administration of TM in the rat hemimandible without systemic side effects. Infusion of this antibiotic through a bony window using an osmotic minipump over a 7-day period causes alterations in both the structure and composition of the BL interposed between ameloblasts and maturing enamel.
Noncollagenous matrix proteins in mineralized tissues, such as bone sialoprotein (BSP) and osteopontin (OPN), are highly glycosylated (reviewed in Butler 1989; Midura and Hascall 1996). Indeed, a large portion of the molecular weight of these two proteins is accounted for by N- and O-linked carbohydrates. On the other hand, enamel matrix proteins are distinctively less glycosylated. Sugar residues are mainly present on non-amelogenins and are predominately N-linked in the case of enamelin (Fukae et al. 1996; Hu et al. 1998) and a 65-kD sulfated enamel protein (Smith et al. 1995), and O-linked in the case of ameloblastin (Cerny et al. 1996; Krebsbach et al. 1996; Hu et al. 1997; Uchida et al. 1997; Nanci and Smith 2000). However, a highly glycosylated BL forms at the interface between ameloblasts and enamel at the start of the maturation stage of amelogenesis (Nanci et al. 1993). It is rich in glycoconjugates comprising N-acetyl-
In the present study, lectin–gold cytochemistry was applied to examine compositional alterations of the BL induced by inhibition of N-glycosylation. Under the influence of TM, the BL becomes irregular and/or is replaced by an interfacial region poor in EPs but rich in glycoconjugates. The presence of a large number of HPA binding sites throughout this region suggests that it represents a widened BL. The density of HPA binding sites over the BL did not significantly differ in treated and control animals, indicating that this lectin recognizes glycoconjugates whose concentration is not affected by TM. On the other hand, the density of labeling of WGA appeared to decrease over both the BL and the enamel in TM-treated incisors. This apparent reduction is consistent with the expectation that TM should prevent the incorporation of N-linked sugars, such as GlcNac.
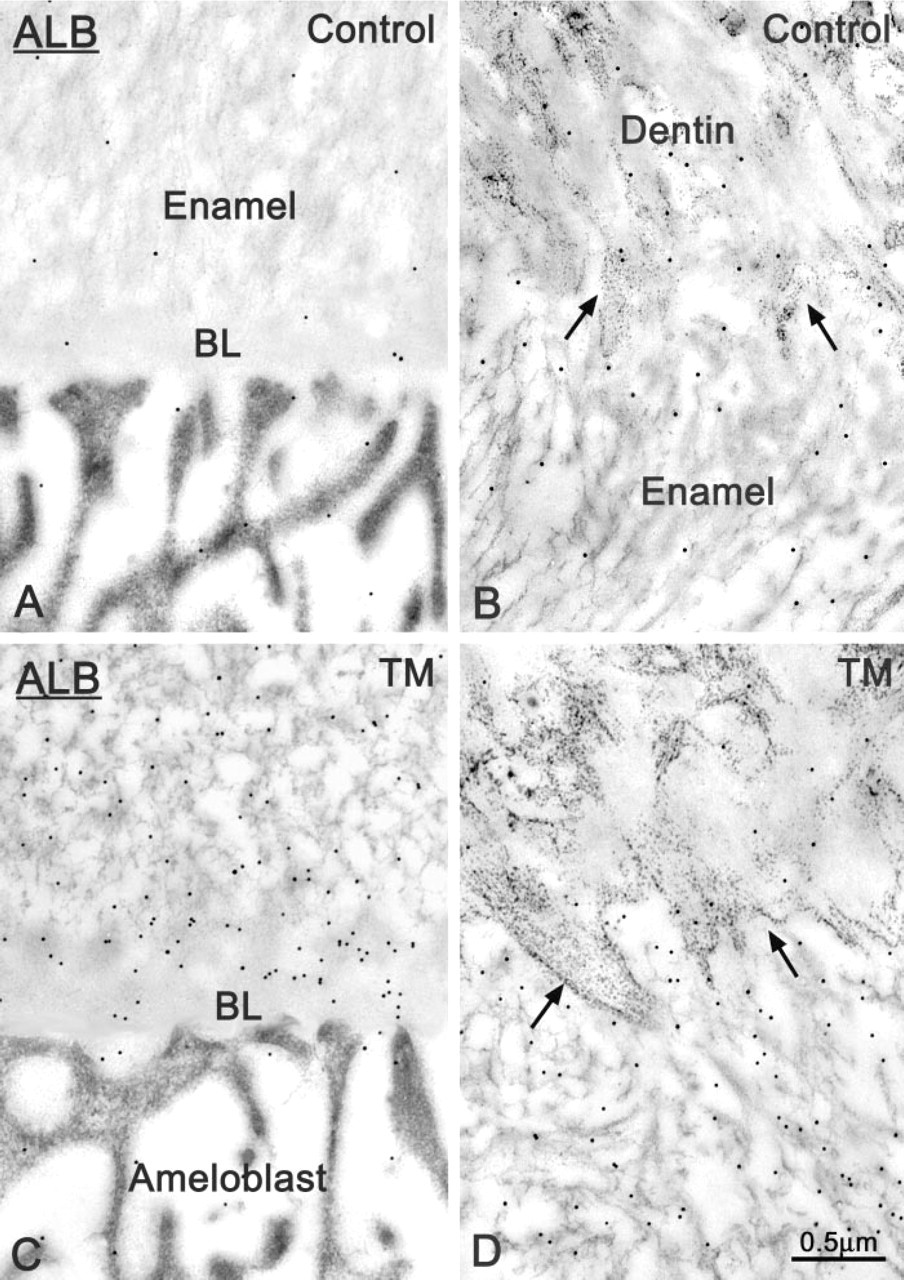
Comparative immunocytochemical preparations illustrating (
A number of enzymes are involved in the processing and degradation of EPs (reviewed in Smith 1998). TM is likely to have had an effect on the activity of enamel proteinases, because enzymes are generally glycosylated. This should have resulted in the delayed removal of EPs and a corresponding increase in density of labeling at certain times during enamel maturation. One possible explanation for why this was not observed is that the enamel being examined, and its constituting matrix proteins and enzymes, were in part formed before administration of the drug. Whereas EPs are generally not believed to serve any major structural function, the BL forms a dense mesh-work which may be, in part, dependent on carbohydrate side chains for its integrity. Protein conformation might be altered in the absence of glycosylation (Olden et al 1978; Elbein 1988), such that N-glycosylated BL components interact less efficiently, leading to a more diffuse structure or its absence. In the case of EPs, only some non-amelogenins are N-glycosylated and these represent a minor component of enamel. They are processed very soon after their secretion and their primary role is in the secretory stage, when crystals actively elongate (Nanci et al. 1998). Any alteration of the few non-amelogenins still secreted during the maturation stage is likely to have a minor impact on enamel formation. Alternatively, sugar residues on EPs may have no significant role in their function. Although no major modification of maturing enamel structure was observed, defective dentin matrix production and mineralization were noted, suggesting that N-glycosylated matrix components play an important role in its formation.
The immunodetection of large amounts of ALB in the enamel matrix of TM-treated rats indicates that there has been an abnormal influx of this plasma protein after its administration. Ameloblasts do not synthesize and secrete ALB (Couwenhoven et al. 1989; Fincham et al. 1999), and circulating ALB has limited access to the enamel layer in normal rats (McKee et al. 1986; Robinson et al. 1994; Chen et al. 1995; Nanci et al. 1996a,b), as well as in saline-infused control rats. Therefore, it is likely that the alteration of the BL may, at least in part, be responsible for its presence in enamel (Nanci et al. 2000). Similarly to proteoglycans in the kidney glomerular basement membrane (Kanwar and Farquhar 1979), glycoconjugates may confer on the BL charge-selective properties, which would likely be affected by the inhibition of N-glycosylation.
In conclusion, the rat hemimandible bony window model is well suited for studying locally the effect of drugs such as TM. This antibiotic affects both the structure and composition of the BL interposed between maturation stage ameloblasts and enamel. This structure has been proposed to have adhesive and/or filtering functions. No significant detachment of the enamel organ was observed after 7 days of TM administration, but there was an abnormal presence of ALB in maturing enamel. Although not excluding a potential adhesive role, the data therefore lend support to the proposal that this BL is a specialized structure exhibiting filtering functions.
Footnotes
Acknowledgments
Supported by a grant from the Canadian Institute of Health Research.
We thank M. Fortin for general technical assistance and Dr H. Verreault for helping during the surgical procedures.
