Abstract
In the retina, zinc is believed to be a modulator of synaptic transmission and a constituent of metalloenzymes. To determine whether the intracellular localization of zinc correlates with function, we examined the localization of endogenous zinc in the rat retina using the silver amplification method. By light microscopy, reaction products were detected in the pigment epithelial cells (PE), the inner segment of photoreceptors (IS), the outer nuclear layer (ONL) and the inner nuclear layer (INL), the outer plexiform layer (OPL) and the inner plexiform layer (IPL), and the ganglion cell layer (GC). The heaviest accumulation of precipitate was observed in PE and IS, whereas only a little precipitate was found in GC. When the intracellular zinc was chelated with diethyldithiocarbamate, a small amount of precipitate was observed only in ONL. By electron microscopy, zinc was associated with three compartments. In OPL and IPL, zinc was associated with neural processes, while in PE, IS, INL, and GC it was associated with the Golgi apparatus. In ONL, zinc was associated with the nucleus. Zinc in the neural processes is believed to act as a modulator of synaptic transmission, and zinc associated with the Golgi apparatus is assumed to catalyze metalloenzyme reactions.
Keywords
I
Because zinc modulates the activity of the amino acid receptors (Wu et al. 1993; Dong and Werblin 1994; Qian et al. 1996, 1997; Kaneda et al. 1997, 2000; Han and Wu 1999; Han and Yang 1999), zinc is believed to be an endogenous modulator of synaptic transmission in the retina. However, zinc can also regulate the metabolic function of pigment epithelial cells. Pigment epithelial cells show active uptake of zinc (Newsome et al. 1992), and protein synthesis is reduced when the supply of zinc to cultured pigment epithelial cells is insufficient (Tate et al. 1995). These functions of zinc are supported by the histochemical demonstration of zinc in both the synaptic regions (Wu et al. 1993; Qian et al. 1997; Ugarte and Osborne 1998; Kaneda et al. 2000) and the pigment epithelial cells (Ugarte and Osborne 1998). However, these histochemical studies used only light microscopy, and the intracellular localization of zinc in the retina has not been clarified at the ultrastructural level.
In this study we examined the precise intracellular localization of zinc in the rat retina with the silver amplification method by both light microscopy and electron microscopy. We found that zinc was localized in specific compartments: the neural processes of the outer plexiform layer and the inner plexiform layer, the Golgi apparatus of the pigment epithelial cells, the inner segment of photoreceptors, the cells of the inner nuclear layer and the ganglion cells, and the nucleus of photoreceptors. The presence of zinc in these specific compartments is believed to reflect the fact that zinc is involved in multiple functions in the retina. A portion of the present study has been published in abstract form (Akagi et al. 1999).
Materials and Methods
Fixation of Animals
Research protocols were approved by the Animal Care and Use Committee of RIKEN. For the silver amplification, we followed the methods described in detail by Danscher (1981). Briefly, 12–16-week-old Wistar rats were anesthetized by IP injection of pentobarbital (Nembutal, 20 mg/kg body weight). Animals were perfused with 0.1 M phosphate buffer (200 ml/kg body weight) containing 0.1% sodium sulfide. Then the perfusate was switched to 0.1 M phosphate buffer containing 0.1% sodium sulfide and 3% glutaraldehyde for 15 min. After fixation, both eyes were enucleated, opened at the equator, and the retina was detached from the sclera. To obtain better fixation, the retina was kept in 0.1 M phosphate buffer containing 0.1% sodium sulfide and 3% glutaraldehyde for 60 min. All fixations were carried out at room temperature. When it was necessary to chelate the zinc in the retina, rats were pretreated by IP injection of diethyldithiocarbamate (DEDTC) (1000 mg/kg body weight) for 60 min before sacrifice (Danscher 1996). Nembutal was purchased from Abbott Laboratories (North Chicago, IL) and DEDTC from Sigma (St Louis, MO).
Silver Amplification
Fixed retina was embedded in 3% agar and sectioned at 100 μm thickness with a microslicer (DTK-1500; Dosaka EM, Kyoto, Japan). Sections were washed thoroughly with distilled water before the silver amplification. The IntenSE M silver Enhancement kit (Amersham International; Little Chalfont, Bucks, UK) was used to intensify zinc signals (Danscher et al. 1987; De Biasi and Bendotti 1998). A one-to-one cocktail of the IntenSE M kit solution and 33% gum arabic solution was used as a reagent. Because the intensification procedure of zinc signals in a reagent is a chemical reaction, the increase in size of silver precipitates proceeded with time. When the reaction period in a reagent was within 45–120 min, there was no difference in the distribution pattern of silver precipitates except for the size. In the present study, a reaction period of 60–75 min in the dark at 26C gave an optimal size of silver precipitates for observation. The intensification of zinc signals was halted by a through wash in distilled water. To reduce the contamination of metals as much as possible, all staining procedures were carried out in new plastic ware.
After the silver amplification, retinas were postfixed with 1% osmium tetroxide in 0.1 M phosphate buffer for 1 hr at 4C, dehydrated with a graded ethanol series, and flat-embedded in epoxy resin (Araldyte CY212; TAAB, Aldermaston, UK). Semithin 2-μm-thick sections were cut for light microscopic observation. For electron microscopic observation, the retina was sectioned at 80-nm thickness with an ultramicrotome (Ultracut-UCT; Leica, Heidelberg, Germany). Ultrathin sections were stained with uranyl acetate and lead citrate.
Light and Electron Microscopic Observation
To observe the distribution of silver precipitates under the microscope (Nikon, ECLIPS E800; Tokyo, Japan), no counterstaining was carried out. Photomicrographic data recorded on color reversal film (Fujichrome 100, Fuji Photofilm, Tokyo, Japan) were scanned and digitized through a film scanner (LS-4500; Nikon). Digitized images processed by commercially available software (Photoshop; Adobe, San Jose, CA) were printed on a color printer (Pictrography 3000; Fuji Photofilm).
Electron micrographs recorded on imaging plates through an LEO 912 electron microscope (LEO Electron Microscopy; Oberkochen, Germany) were scanned and digitized by an FDL 5000 imaging system (Fuji Photofilm). Digitized images were processed by the computer program “Image Gauge” and printed out on a Pictrography 3000 color printer.
All experiments were carried out on at least three different animals.
Results
Light Microscopic Observation
Figure 1A shows the distribution of silver precipitates corresponding to zinc in retinal layers developed by the IntenSE M silver Enhancement kit. In the pigment epithelial cells and the inner segment of photoreceptors, precipitates accumulated heavily and were observed as a black band. No precipitate was observed between the pigment epithelial cells and the inner segment of photoreceptors. In the outer plexiform layer and the inner plexiform layer, precipitate was diffusely distributed. There was substantially less precipitate in the outer nuclear layer and the inner nuclear layer than in the outer plexiform layer and the inner plexiform layer. In the inner nuclear layer, most zinc was distributed near the marginal regions of the layer. Some precipitate was also observed in the ganglion cell layer.
In the silver amplification method, the perfusion of sodium sulfide is necessary to precipitate free zinc and zinc weakly bound to protein as sulfide compounds in the tissue (Frederickson 1989). Such a sulfide compound can become a core of silver precipitates and finally the distribution of zinc is detected. However, the formation of sulfide compounds is not specific to zinc and can occur with other metals (Danscher 1981), indicating that the silver precipitates can be sulfide compounds of other metals. To rule out this possibility, we used DEDTC pretreatment to chelate zinc (Danscher 1981, 1996). In our study, when animals were treated with DEDTC before the perfusion of sodium sulfide, precipitate disappeared in most layers, as has been reported in previous papers (Danscher 1981, 1996). We occasionally observed the association of precipitate with the nucleus of photoreceptors after pretreatment, but at a much lower level than in untreated retina (Figure 1B). Therefore, we concluded that the sulfide complex detected by the silver amplification methods accurately reflected the free or weakly protein-bound zinc in the rat retina.
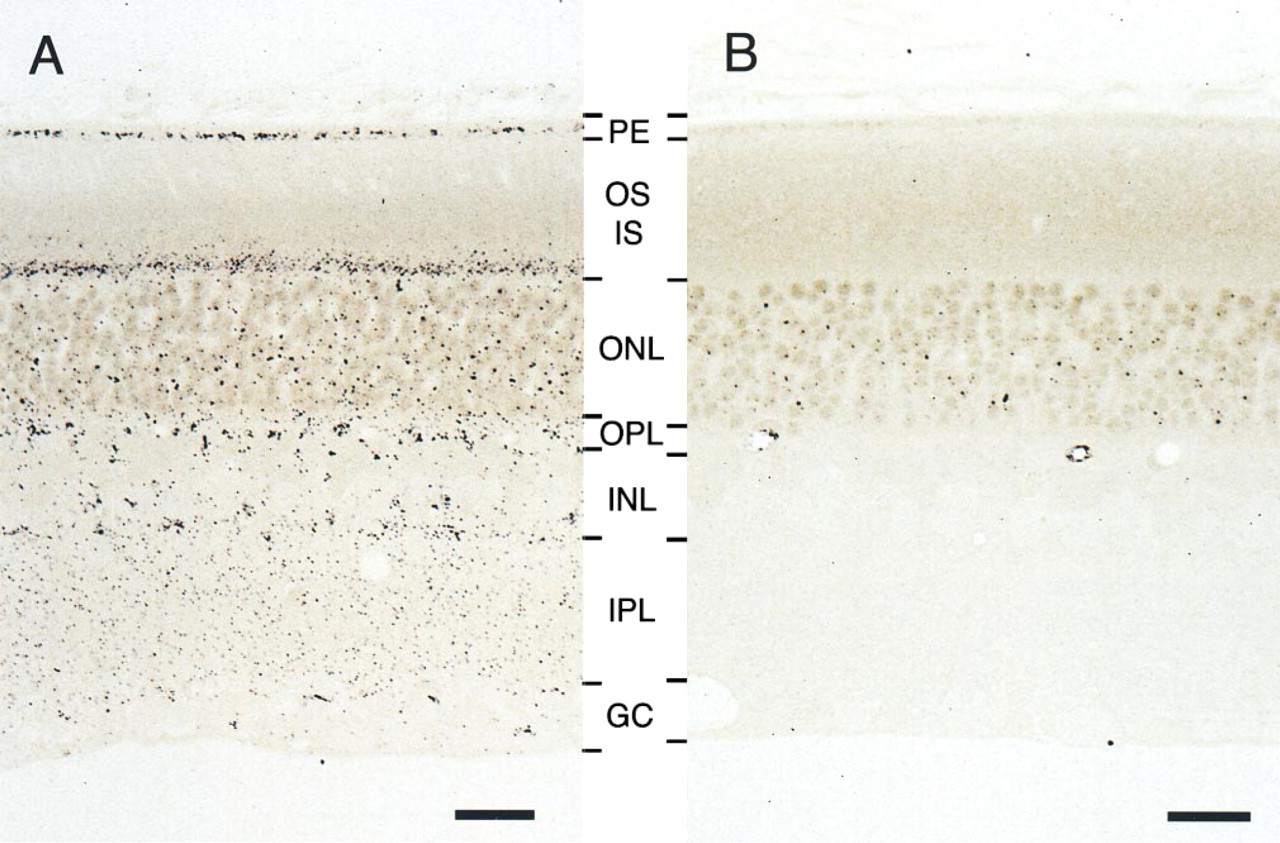
Photomicrographs showing the distribution of zinc in the rat retina. Zinc was detected as silver grains developed by the IntenSE M silver Enhancement kit. (
Electron Microscopic Observation
Pigment Epithelial Cells. Silver grains corresponding to zinc were distributed in the cytoplasm of the pigment epithelial cells (Figure 2A). These silver grains were frequently associated with the Golgi apparatus. Silver grains were usually localized on one side of the lamellae (Figure 2B). Because the lamellae appeared to be straight, we could not identify whether silver grains were associated with the cis side or the trans side of the Golgi apparatus. There were very few silver grains not associated with the Golgi apparatus.
Outer and Inner Segments of Photoreceptors. Silver grains were localized to the inner segment (Figure 2C), where the grains had accumulated in the myoid region. Most grains were associated with only one side of the Golgi apparatus (Figure 2D). No grains were found in the outer segment.
Outer Nuclear Layer. Many silver grains were associated with the nucleus of photoreceptors (Figure 3A). These nuclei were polygonal and had uniform density. Some of the grains were found in the internuclear space, but most of them were found in the processes of the Müller cells (Figure 3B). A few precipitates were observed in both the nucleus and the processes, even after DEDTC pretreatment.
Outer Plexiform Layer. On the scleral side, silver grains were found in the electron-lucent neural processes containing vesicles (Figure 3C). These electron-lucent neural processes had contact with electron-dense neural processes (Figure 3D), and an invagination of the electron-lucent processes into the electron-dense neural processes was also observed (Figure 3C). Because the electron-dense terminals were located near the scleral side and had a single synaptic ribbon (Rodieck 1973; Dowling 1987) and the cones of the rat retina are 1% photoreceptors (Szél and Röhlich 1992), these electron-dense terminals were identified as rod spherules. Silver grains could not be found in the rod spherules. Neural processes containing a few silver grains were occasionally observed on the vitreal side (Figure 3C). This neural process had no contact with the rod spherules.
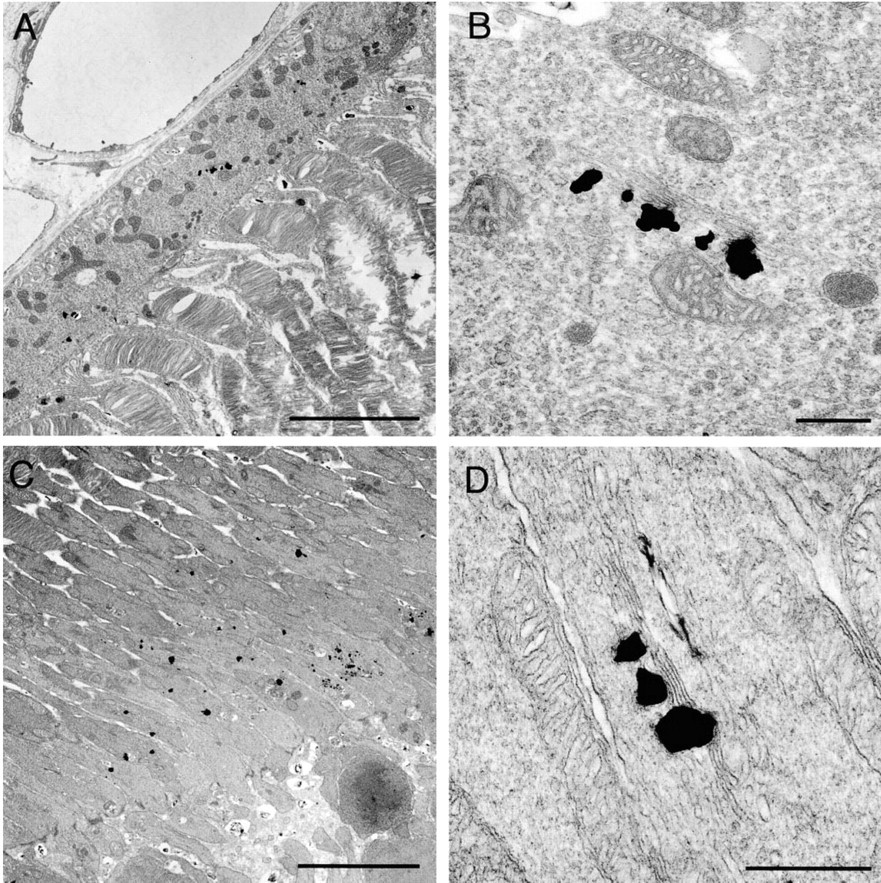
Electron micrographs showing the distribution of zinc in the pigment epithelial cell (
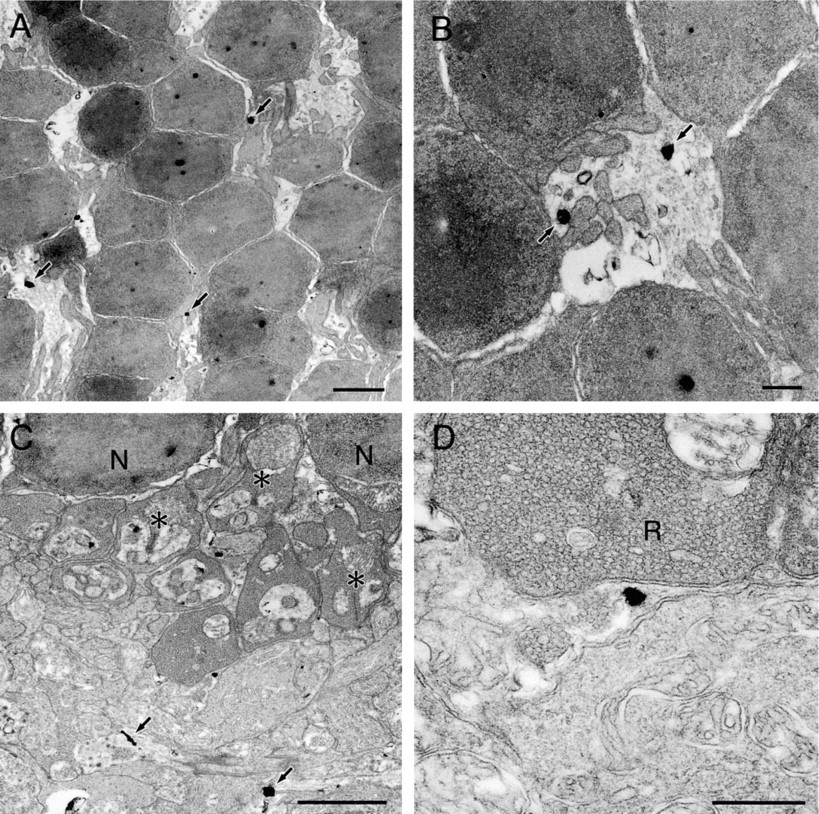
Inner Nuclear Layer. There are different types of cells in this layer. Cell bodies of the horizontal cells and the bipolar cells are located on the scleral side, whereas those of the amacrine cells are located on the vitreal side and often have an indented nucleus. Cell bodies of the Müller cells are also localized together with them, and often show a fusiform shape (Rodieck 1973). The majority of silver grains were observed in the cytoplasm of the following two types of cells. The first type was located on the scleral side and had a round light nucleus (Figure 4A). Most silver grains were associated with one side of the Golgi apparatus (Figure 4B). The second type was located on the vitreal side and often had a light nucleus with a notch (Figure 4C), presumably amacrine cells. In these cells, some silver grains were associated with the Golgi apparatus (Figure 4D), where the grains were localized to one side. Some silver grains were also observed in other types of cells with nuclei of various morphologies. It will be interesting in future experiments to see if the presence of zinc is a characteristic feature of specific cell types.
Electron micrograph showing the distribution of zinc in the outer nuclear layer (
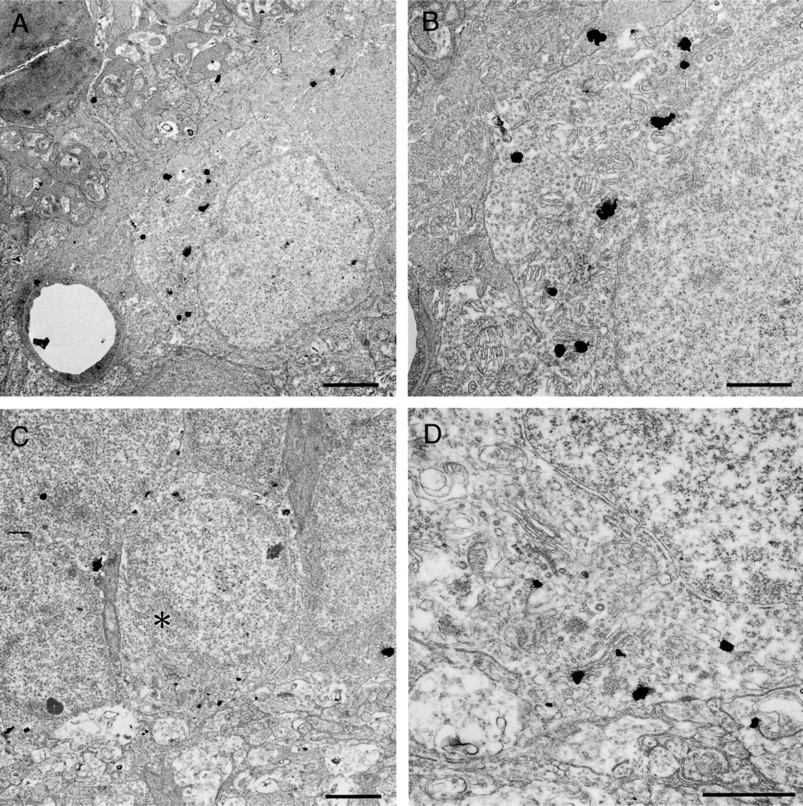
Inner Plexiform Layer. Silver grains were frequently found in the neural processes (Figure 5A). They contacted both the electron-lucent (Figure 5B) and electron-dense processes (Figure 5C). In Figure 5B, the processes contain small clear vesicles and occasionally cored vesicles, and contact electron-lucent neural processes with sparse vesicles. This contact is believed to be a non-synaptic contact. In Figure 5C, the electronlucent processes contain small clear vesicles and contact electron-dense neural processes with rich vesicles. This contact is believed to be a synaptic contact. In the present study, the ratio of two types of contacts was not analyzed quantitatively. The presence of synaptic ribbons was used to identify terminals of bipolar cells, where we could not find any silver grains (Figures 5A and 5C).
Electron micrographs showing the distribution of zinc in the inner nuclear layer. (
Ganglion Cell Layer. A few ganglion cells had silver grains in the cytoplasm. Asymmetrical distribution of the grains was obvious when they were found near the Golgi apparatus (Figure 5D).
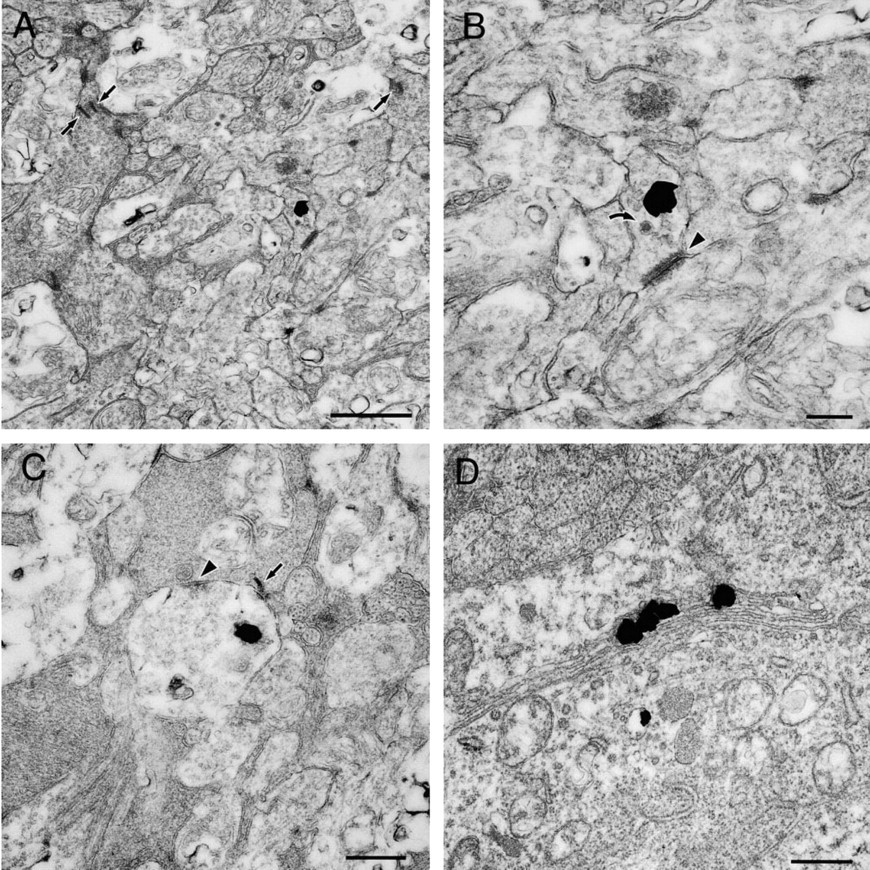
Electron micrographs showing the distribution of zinc in the inner plexiform layer (
Discussion
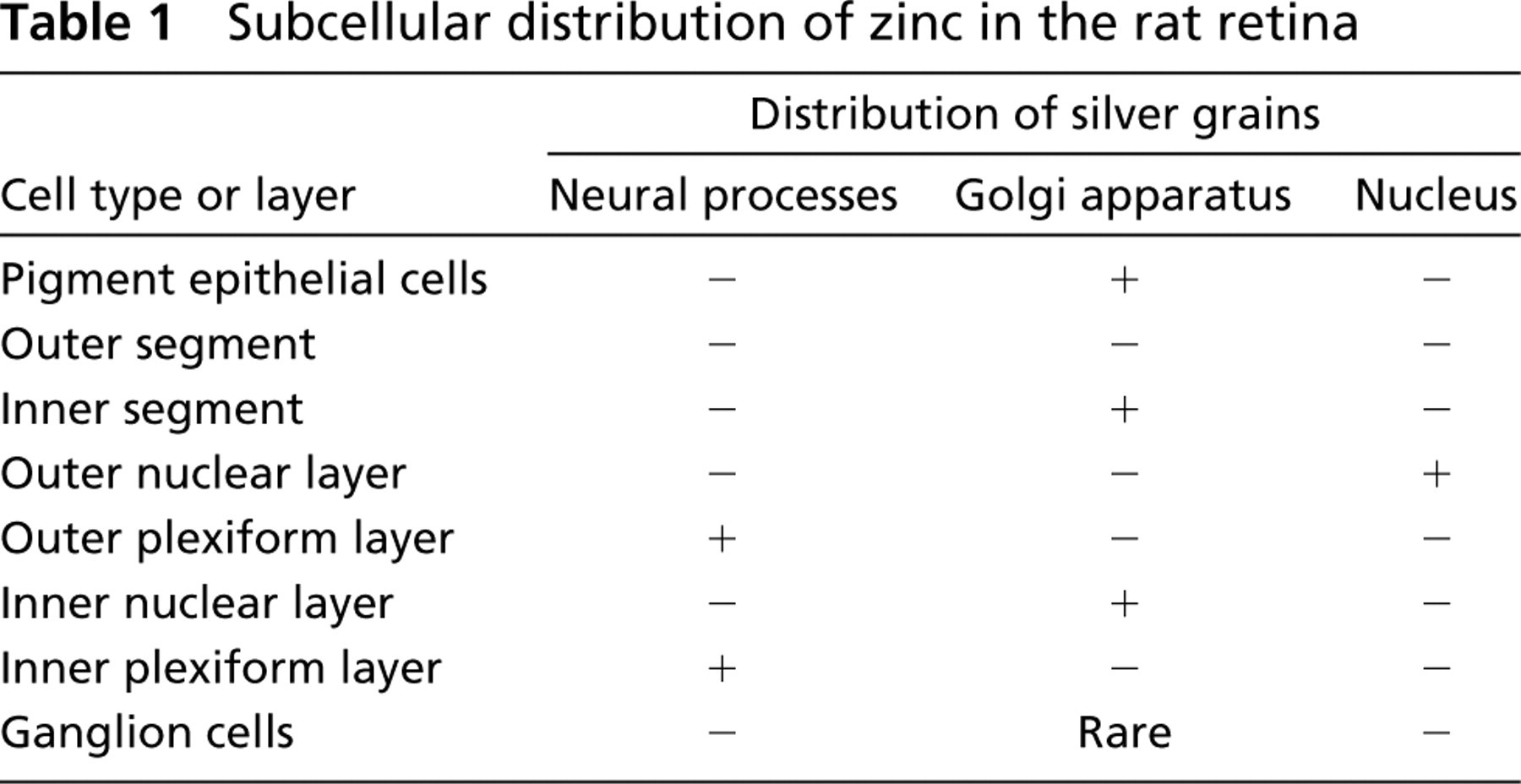
In this study we employed silver amplification methods to localize zinc in the rat retina at the ultrastructural level. We found that zinc was localized in three distinct subcellular compartments, each of which was specific for each layer. Table 1 summarizes the subcellular localization of silver grains in the rat retina. These compartments were not detectable by light microscopic observation.
Zinc Stained by the Silver Amplification Method
The silver amplification method is a useful means of detecting free zinc and zinc weakly bound to protein because the soluble zinc is precipitated in the tissue as sulfide compounds (Frederickson 1989). Although the formation of a sulfide complex is not specific to zinc and can occur with other metals, zinc–sulfide complexes in the tissue are specifically identified by the DEDTC test (Danscher 1981, 1996). In our study, we observed that the formation of zinc–sulfide complexes was blocked by pretreatment with DEDTC, as has been reported in previous papers (Danscher 1981, 1996). These findings demonstrate that the silver precipitates accurately reflected the distribution of the free or weakly protein-bound zinc in the rat retina.
We observed that reaction products were widely distributed throughout the retina except for the outer segment of photoreceptors. A similar distribution of reaction products has been reported using the neo-Timm method in the rat retina (Ugarte and Osborne 1998). It has been reported that the IntenSE M silver Enhancement kit can detect the same component of zinc detected by the conventional neo-Timm method (Danscher et al. 1987; De Biasi and Bendotti 1998). However, reaction products by the neo-Timm method were mainly limited in the outer nuclear layer and the outer plexiform layer in the cold blooded animals (tiger salamander, Wu et al. 1993; skate, Qian et al. 1997).
Subcellular distribution of zinc in the rat retina
Zinc in the Neural Processes
In the present study, we found endogenous zinc in the neural processes of the outer plexiform layer and the inner plexiform layer. We observed many synaptic vesicles in these processes. In the hippocampus, zinc in the synaptic terminals (Pérez-Clausell and Danscher 1985, 1986) is released from mossy fibers to the synaptic cleft, where the concentration of zinc is ~300 μM (Assaf and Chung 1984; Frederickson 1989). Because micromolar zinc inhibits the activity of NMDA and GABA receptors in cultured hippocampal neurons (Westbrook and Mayer 1987), zinc is believed to be an intrinsic modulator of synaptic transmission. A number of studies have described the modulatory actions of micromolar zinc on amino acid receptors in the retina (Wu et al. 1993; Dong and Werblin 1994; Qian et al. 1996, 1997; Kaneda et al. 1997, 2000; Han and Wu 1999; Han and Yang 1999). Although the release of endogenous zinc from synaptic regions of the retina was not examined, our findings strongly support the idea that zinc in the vertebrate retina can also work as an endogenous modulator of synaptic transmission.
In the central nervous system, neural processes containing zinc are glutamatergic (Frederickson 1989; Frederickson and Danscher 1990). We identified rod spherules in the outer plexiform layer on the basis of previous electron microscopic findings (Rodieck 1973; Dowling 1987). In our study, silver grains were not observed in rod spherules but were found in the neural processes contacting the rod spherules. It has been reported that photoreceptors receive feedback from horizontal cells (Baylor et al. 1971; Piccolino and Gerschenfeld 1980; Wu 1991, 1992). Because this feedback is mediated by the GABAergic system (Tachibana and Kaneko 1984; Kaneko and Tachibana 1986; Wu 1991, 1992), it is likely that zinc can co-localize with GABA in synaptic terminals of the horizontal cells.
Zinc in the Golgi Apparatus
We observed that zinc was associated with one side of the Golgi apparatus in pigment epithelial cells. Melanin is the main source of zinc in pigment epithelial cells (Samuelson et al. 1993). The formation of melanin requires activation of tyrosinase, which is distributed in the endoplasmic reticulum and the Golgi apparatus in pigment epithelial cells (Schraermeyer and Stieve 1994). Although tyrosinase itself is a copper-containing metalloenzyme (Hearing and Tsukamoto 1991), the zinc-containing metalloenzyme is also a member of the tyrosinase family in the melanogenic biosynthetic pathway (Solano et al. 1994). In addition, the presence of zinc in the active site of enzymes has been proposed (Solano et al. 1994). In the Golgi apparatus, enzymes tend to show one-sided distribution (Dunphy and Rothman 1985; Farquhar 1985); enzymes in the cis side are different from those in the trans side. Therefore, it is likely that zinc forms the catalytic site of metalloenzymes in the pigment epithelial cells. Consistent with this, zinc is important for the maintenance of metabolic status in pigment epithelial cells (Leure-Dupree and Bridges 1982; Tate et al. 1995; Miceli et al. 1999; Schraermeyer and Heimann 1999). We also found that zinc was associated with one side of the Golgi apparatus in the inner segment of photoreceptors, the inner nuclear layer, and the ganglion cell layer. Therefore, it would be interesting to see if the one-sided association of zinc with the Golgi apparatus represents the presence of zinc-containing metalloenzymes in these regions.
Zinc in the Outer Nuclear Layer
In the outer nuclear layer, silver precipitate was associated with the nucleus and some silver precipitate was insensitive to DEDTC pretreatment. Therefore, it is likely that free zinc or zinc weakly bound to protein exists in the nucleus. In the nucleus, however, zinc has been found at the structural site of some transcription factors (Coleman 1992). Such tightly protein-bound zinc would react to neither sodium sulfide nor DEDTC (Frederickson 1989). However, if the affinity of protein-bound zinc to sodium sulfide or DEDTC depends on the three-dimensional structures of those proteins, some protein-bound zinc may react with sodium sulfide or DEDTC. The possible staining of zinc tightly bound to protein should be studied in future experiments. The presence of zinc in the outer nuclear layer has also been reported in previous papers (Wu et al. 1993; Qian et al. 1997; Ugarte and Osborne 1998; Kaneda et al. 2000).
The silver amplification method is a useful means of detecting zinc in the central nervous system (Frederickson 1989). In this study we found that zinc has a broad subcellular localization in the rat retina at the ultrastructural level. This specific subcellular localization may reflect the multiple functions of zinc in the rat retina. In neural processes zinc modulates synaptic transmission, while in the Golgi apparatus zinc is an important regulator of intracellular metabolism.
Footnotes
Acknowledgments
Supported by a BSI grant (No. 57911) from RIKEN and a grant-in-aid to M. Kaneda (No. 09680820) from the Ministry of Education, Science and Culture.
We thank Prof A. Kaneko for critical reading of our manuscript.
