Abstract
In previous studies, collagen XI mRNA has been detected in colon cancer, but its location in human colon tissue has not been determined. The heterotrimeric collagen XI consists of three a chains. While it is known that collagen XI plays a regulatory role in collagen fibril formation, its function in the colon is unknown. The characterization of normal human colon tissue will allow a better understanding of the variance of collagen XI in abnormal tissues. Grossly normal and malignant human colon tissue was obtained from pathology archives. Immunohistochemical staining with a 58K Golgi marker and α1(XI) and α2(XI) antisera was used to specifically locate their presence in normal colon tissue. A comparative bright field microscopic analysis showed the presence of collagen XI in human colon. The juxtanuclear, dot-like collagen XI staining in the Golgi apparatus of goblet cells in normal tissue paralleled the staining of the 58K Golgi marker. Ultra light microscopy verified these results. Staining was also confirmed in malignant colon tissue. This study is the first to show that collagen XI is present in the Golgi apparatus of normal human colon goblet cells and localizes collagen XI in both normal and malignant tissue. Although the function of collagen XI in the colon is unknown, our immunohistochemical characterization provides the foundation for future immunohistopathology studies of the colon.
C
Accessing the major triple helix of collagen XI is not possible without disrupting the fibrils, but the NTD is exposed on the fibril surface. The NTD remains on the surface for an extended period of time after biosynthesis (Thom and Morris 1991; Keene et al. 1995). Preliminary data have indicated that the proteolytic removal rate of the α1(XI) Npp is dictated by the splice forms present in the Vr (Medeck et al. 2003). This function requires the Vr to be on the fibril surface (Gregory et al. 2000). The fibril surface location of the NTD facilitates immunohistochemical localization.
The presence of collagen XI has been studied in both cartilaginous and non-cartilaginous tissue, such as human chondrocytes (Burgeson and Hollister 1979; Swoboda et al. 1989; Lui et al. 1995), skeletal muscle (Lui et al. 1995), placenta (Bernard et al. 1988; Yoshioka and Ramirez 1990; Oxford et al. 1995), lung (Lui et al. 1995; Chong et al. 2006), and brain (Lui et al. 1995), and collagen XI mRNA has been shown to be upregulated in colorectal cancer (Fischer et al. 2001a, b). Fischer et al. (2001a) concluded through northern blotting and RT-PCR that collagen (α1)XI was not present in the adult human colon, and Sandberg et al. (1993) concluded through Southern blotting that α2(XI) is not present in the fetal colon. Although both of these support the hypothesis that collagen XI is not produced in the colon, it is possible that collagen XI was either below the limit of detection by the experimental technique or is posttranslationally relocated within the cells of the colon. Detailed structural localization of the components of the collagen (α1)XI Vr in normal and malignant colon tissue using immunohistochemical staining has not been published to date. To evaluate the upregulation of collagen XI observed in cases of colorectal cancer, it is advantageous to document the presence in both normal and malignant tissue.
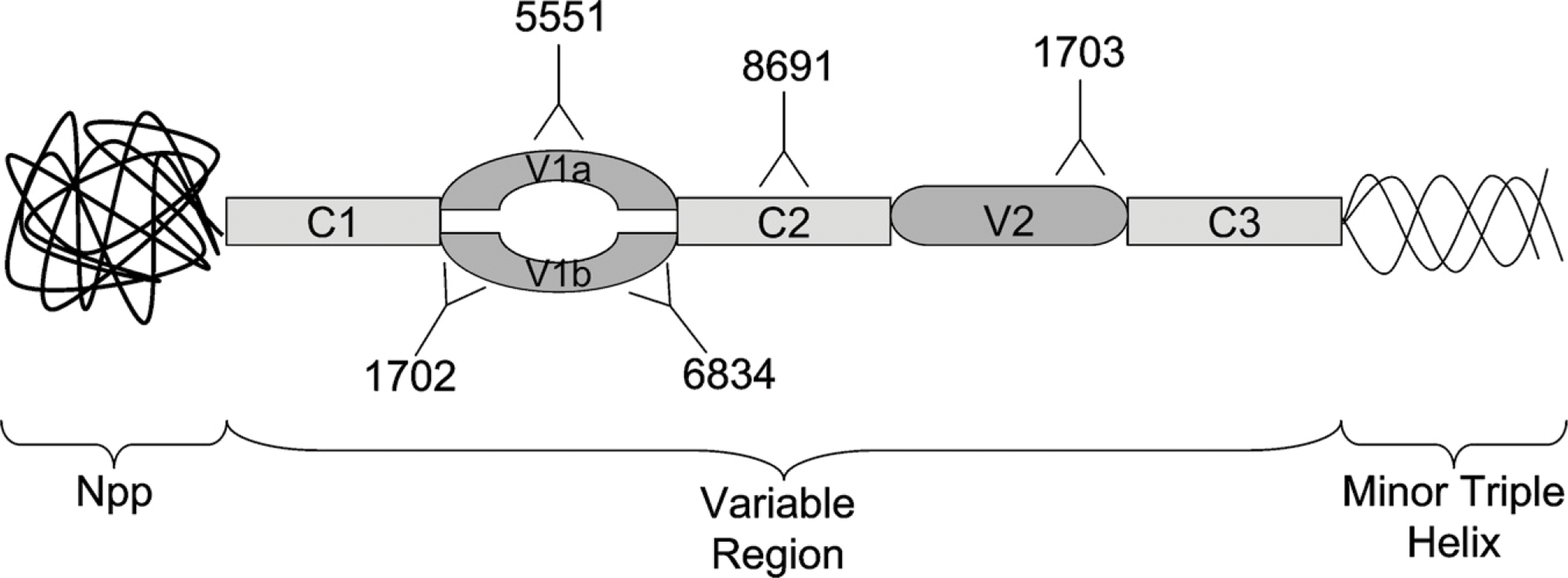
Schematic diagram of collagen (α1)XI amino terminal domain. Approximate locations of target sequences are indicated for each of the antisera used for this study. The V1a region is recognized by antiserum 5551, the V1b region is recognized by antisera 1702 and 6834, the C2 region is recognized by antiserum 8691, and the V2 region is recognized by antiserum 1703.
Materials and Methods
Antisera
Using specific α1(XI) chain antisera allowed a circumspect observation of collagen XI presence. Antibodies were raised against a 20-aa synthetic peptide designed from the amino portion of V1b (exon p6b), a 20-aa synthetic peptide from the carboxyl portion of V1b (exon p6b), and a 39-aa synthetic peptide from V1a (exon p6a) of the collagen α1(XI) chain as described previously (Oxford et al. 1994; Keene et al. 1995, Davies et al. 1998). All antisera were initially characterized by ELISA to verify that the antibodies recognized the target peptides and to determine titer, that they did not recognize similar but unrelated peptides, and that they had the capacity to recognize the target protein by immunoblot and immunofluorescence against tissues and cells from several species including human, bovine, horse, chicken, mouse, and rat. Other antibodies raised against the 14-aa bovine sequence of the collagen α2(XI) chain (Oxford et al. 2004), a 29-aa synthetic peptide from C2 (exon p7) (Oxford et al. 1994; Morris et al. 2000), and a 20-aa human synthetic peptide from the carboxyl portion of V2 (exon p8) (Davies et al. 1998) were characterized by ELISA, immunoblot, and immunofluorescence staining on cells and tissue sections previously as indicated above. A 58K Golgi protein monoclonal antibody, formiminotransferase cyclodeaminase, (6284; Abcam, Cambridge, MA), was used for a side-by-side comparison with the collagen XI antibodies.
The percent of sequence identity of peptide antigen and human target was determined using BLAST 2 sequences (http://www.ncbi.nlm.nih.gov/blast/bl2seq/wblast2.cgi), where the known sequence of each antigen was compared with the human collagen XI α1 or α2 chains. In addition, each antigen sequence was used (http://www.ncbi.nlm.nih.gov/BLAST) to identify any proteins that could potentially introduce artifactual staining results.
Colon Tissue
Grossly normal and malignant human colon tissue, removed of identity in accordance with institutional review board guidelines, was obtained from pathology archives (Mercy Medical Center; Nampa, ID). Tissue was fixed with formalin containing zinc, paraffinized, cut to 1 μm thickness, and heat fixed onto glass slides, as previously described (Fyffe et al. 1999).
Immunohistochemical Staining
The colon tissue slides were deparaffinized through incubation in a VWR incubator (model 1320; Sheldon Manufacturing, Cornelius, OR) for 25 min at 65C. Deparaffinization and rehydration was carried out through sequential washes in xylene (twice at 5 min), absolute ethanol (twice at 3 min), 95% ethanol (twice at 3 min), and distilled water for 5 min. For the remainder of the procedure, the tissues were maintained in a humidified chamber and not allowed to dry out. A Pap Pen (DakoCytomation; Dako North America, Carpinteria, CA) was used to ensure that the solution remained on the slide.
Hyaluronidase (H3506; Sigma-Aldrich Corporation, St. Louis, MO) was diluted to 0.01 mg/ml with Trisbuffered saline (TBS), pH 7.5 (0.2 M Tris, 0.17 M NaCl); 200 μl was placed on each slide to incubate in a Slide Moat (Boekel Scientific; Feasterville, PA) at 25C for 45 min. Subsequent 25C incubations also used this slide moat. Slides were rinsed with TBS and distilled water and subsequently placed into 95–99C target retrieval solution, pH 7.5, (DakoCytomation; Dako North America) for 40 min. The slides were allowed to cool in the target retrieval solution for 20 min and were rinsed with fresh Wash Buffer, a TBS saline solution containing 0.05% Tween 20 (DakoCytomation; Dako North America). To quench endogenous peroxidases, the slides were incubated with 200 μl Dual Endogenous Enzyme Block for Autostainer (DakoCytomation; Dako North America) for 25 min at 25C, rinsed with fresh Wash Buffer, and submerged in Wash Buffer three to five times.
Each primary antibody was diluted with antibody diluent (DakoCytomation; Dako North America) to a dilution optimized by serial dilutions (1:300 for carboxyl V2; 1:400 for V1a and C2; 1:450 for amino and carboxyl V1b, and α2). The tissue was incubated in 200 μl primary antibody for 1 hr at 25C and rinsed as was done subsequent to enzyme block incubation. A positive control of placental tissue (Bernard et al. 1988; Yoshioka and Ramirez 1990) and a negative control of only secondary antibody coincided with this process. EnVision+ Dual Link System Peroxidase (DakoCytomation; Dako North America), an anti-rabbit and anti-mouse secondary antibody, was applied, 200 μl/slide, and incubated at 25C for 30 min. The slides were rinsed with Wash Buffer. Liquid DAB+ Substrate Chromagen System for Autostainer (DakoCytomation; Dako North America) was added, 200 μl/slide, to each slide. After a 20-min incubation at 25C, the slides were rinsed with distilled water. Each slide was placed in hematoxylin (Hematoxylin 7211; Richard-Allan Scientific, Kalamazoo, MI) for 2 min, rinsed thoroughly with distilled water, and placed in distilled water for 5 min.
The tissue was dehydrated through graded ethanol-concentration steps and xylene. Glass coverslips were glued (Pro-Texx Mounting Medium; Baxter Diagnostics, Deerfield, IL) over tissue to enhance preservation of the tissue. Ultra light microscopy was carried out using a Leica DM500 microscope (Leica Microsystems; Bannockburn, IL) with a 1.6-cm resolving lens, ×10 optical, ×1.25 magnifier, and a ×100 (numerical aperture, 1.46) objective. This allows for × 2000 magnification.
Results
Characterization of α1(XI) and α2(XI) Antisera
The antibody specificity has been previously established (Keene et al. 1995; Oxford et al. 1995; Davies et al. 1998). BLAST results indicated that peptide antigen sequences were highly conserved between rat and human and between bovine and human for the sequences of interest (Figure 2).
Each antigen sequence was compared with the human genome through BLAST searches to find any proteins that have similarities to collagen XI. Mitogenactivated protein kinase (MAPK) (accession number BAD92330) has a similar sequence to the p6a antigen (EYDYEY). Titin (accession number Q8WZ42), a muscular fiber protein, has multiple short sequences in common with the antigen to the carboxyl end of p8 (GPGVP). Mib (accession number BAF03565), a newly discovered brain membrane protein, also has a similar sequence to p8 (GPGVPA). The potential targets identified by BLAST searches are less likely targets than the intended collagen α1(XI) targets of the polyclonal antisera used in this study.
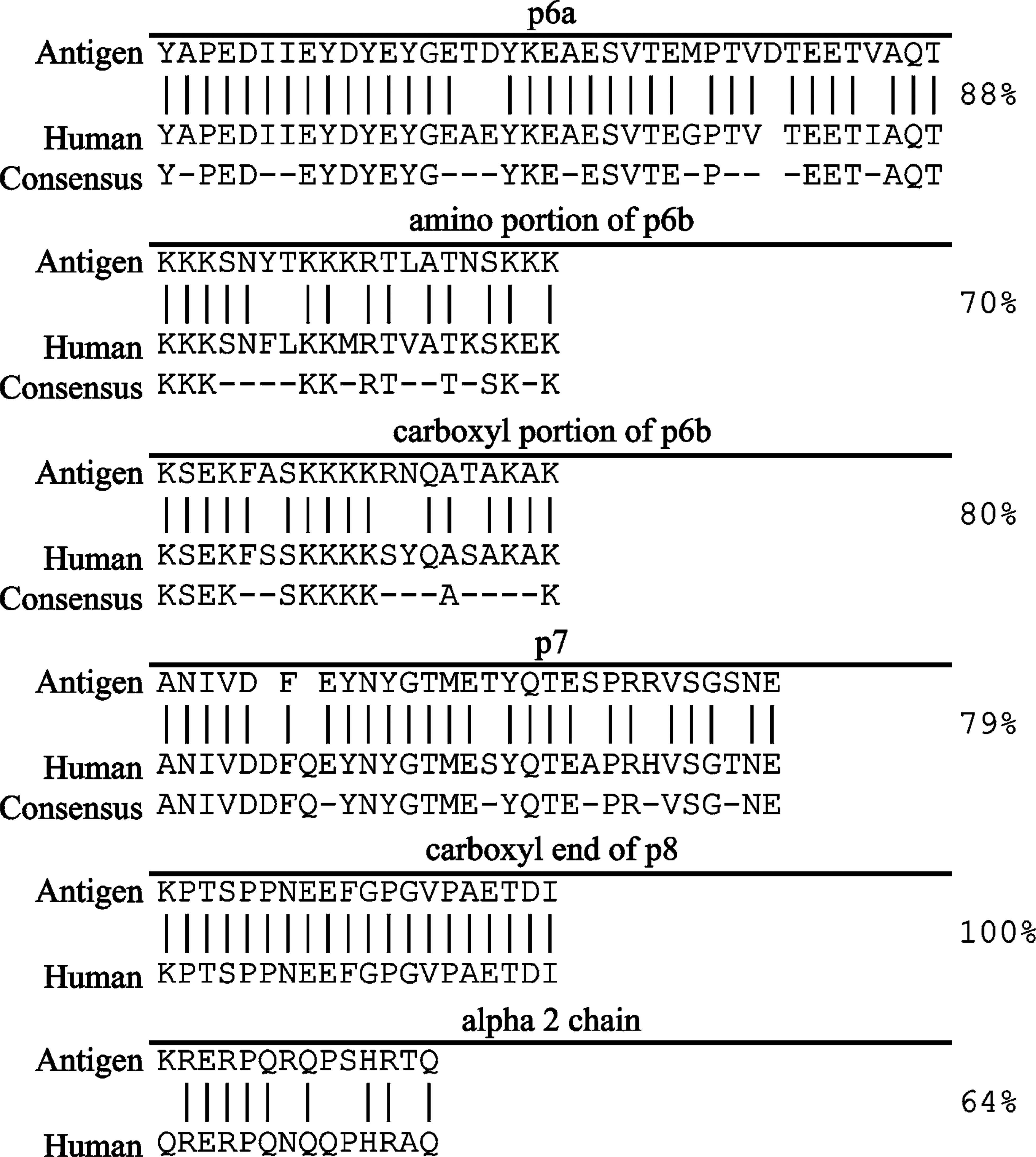
Comparison of collagen XI antigen sequences to humans. Vertical lines indicate identical residues. Dots represent dissimilarities in sequences, although aa have similar properties. Spaces in sequence denote gaps introduced for alignment of the sequence. Dashes in conserved sequence indicate nonconserved residues. The published consensus sequence between bovine, human, mouse, chicken, and rat, when available, is presented below each sequence (Medeck et al. 2003). Fraction and percent identity included missing aa as a deviation.
Collagen XI Staining in the Golgi Apparatus of Colonic Crypts
Presence of the collagen XI α1 and α2 in normal and malignant colon tissue was visualized using antibodies against the α1(XI) splice forms and the α2(XI) α chain (Figures 3 and 4). From the staining and analysis, it was evident that there was specific staining present in normal colon tissue. Each of the collagen XI antibodies yielded intense juxtanuclear, dot-like staining within the colonic crypts epithelial cells, accompanied by diffuse, light cytoplasmic staining and a lack of stromal staining (Figure 3). Furthermore, malignant colon tissue showed the presence of α1(XI) splice forms and the α2(XI) α chain, although appearing more cytoplasmic in comparison to the juxtanuclear, dot-like staining seen in normal tissue. Malignant colon tissue and placental tissue served as positive controls for staining of each antibody. The negative control using antiserum-diluent in place of the primary antibody did not stain (Figure 3H).
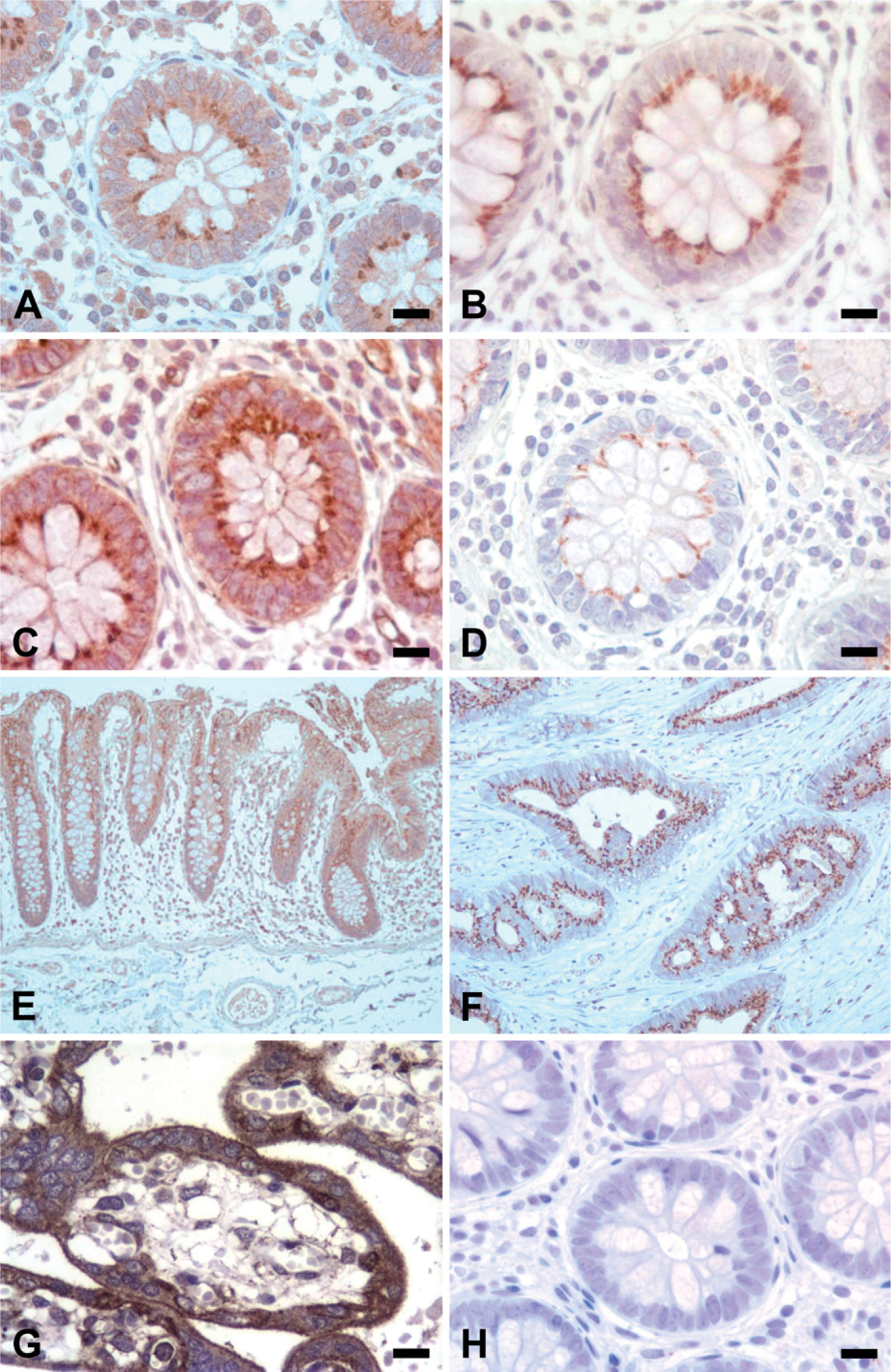
Collagen (α1)XI colonic staining. Novel antibodies, raised against each splice form of the variable region of collagen (α1)XI, were used to locate specific collagen markers in normal colon tissue. Human malignant colon tissue is shown for comparison (
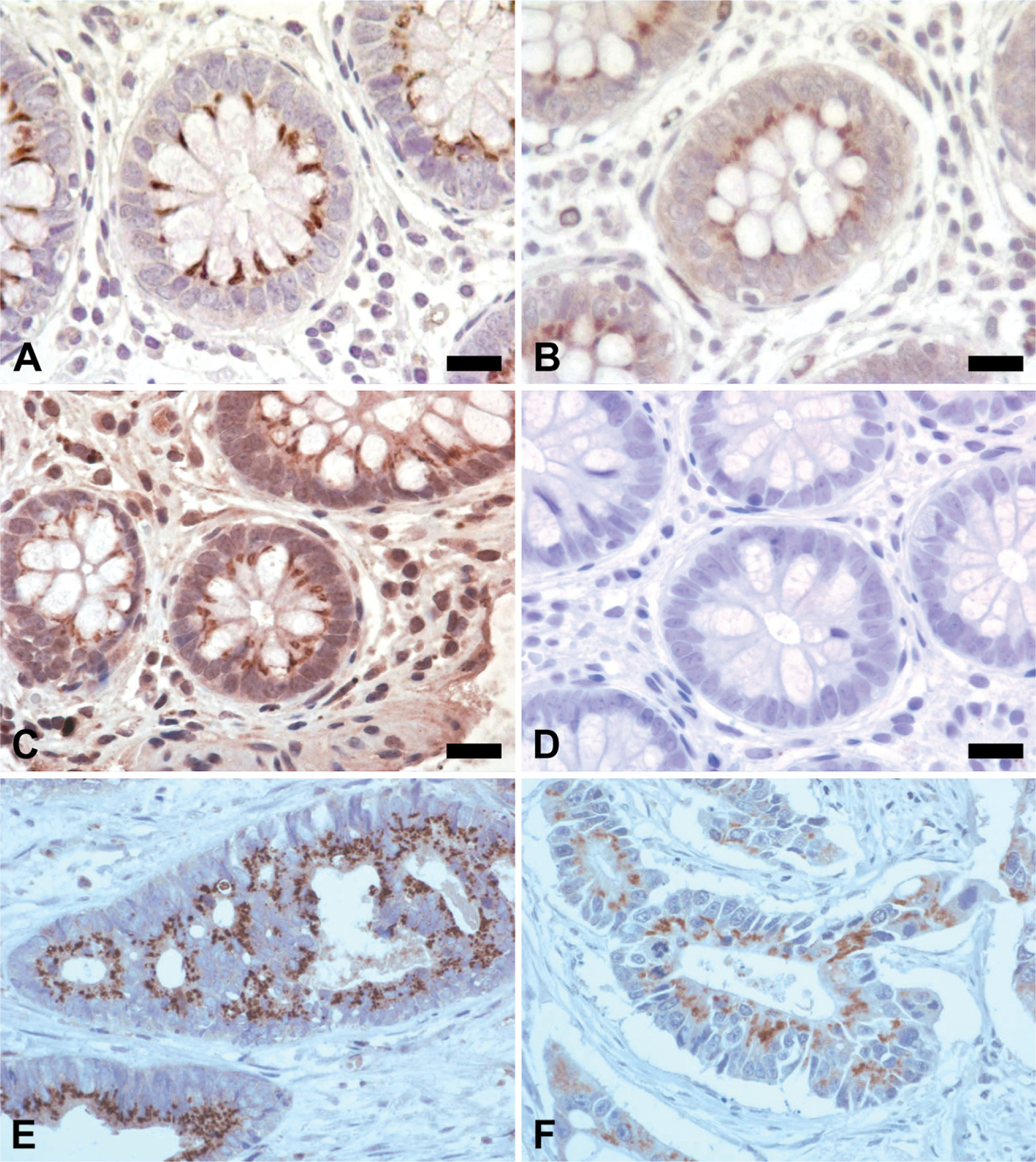
Comparison of αl(XI) and α2(XI) to the 58K Golgi marker. Bright field microscopy images show the presence of collagen XI in the Golgi apparatus of human colon tissue. Human colon cancer tissue is shown for comparison (
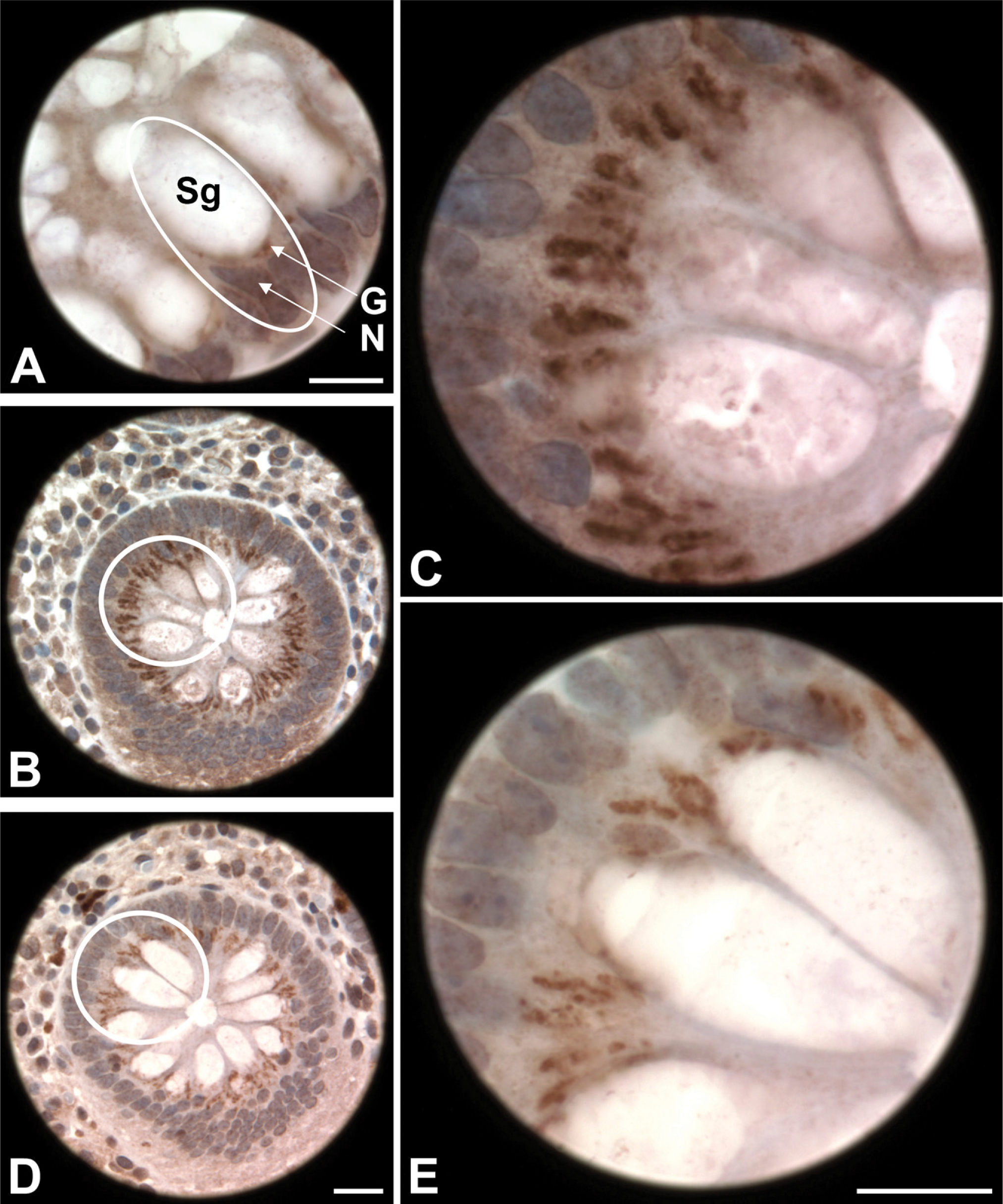
Focal examination of a normal colonic crypt. (
Juxtanuclear staining in normal colon tissue was located in the same subcellular area as the Golgi apparatus of colonic epithelial cells as confirmed by the 58K Golgi marker (Figure 4). A staining comparison of each collagen XI antibody and the 58K Golgi marker at high magnification reiterated Golgi apparatus staining (Figure 5).
Discussion
Collagen XI is a minor but essential constituent of collagen fibrils because of its well-characterized role in the regulation of fibril diameter (Morris and Bächinger 1987; Mendler et al. 1989). Its function is less well understood in non-cartilaginous tissues. The NTD of the α1(XI) chain remains attached to the rest of the molecule for a longer period of time after synthesis than the other amino propeptides of fibrillar collagens and has been localized to the surface of thin collagen fibrils (Keene et al. 1995). Because the NTD of collagen XI is exposed at the surface of collagen fibrils, antisera against various sequences within the NTD of α1(XI) and α2(XI) were used. Although best characterized for its role in cartilage, the presence of collagen type XI within colon tissue may be relevant in the onset of inflammatory bowel disease, including ulcerative colitis and Crohn's disease, and may in part explain the link between intestinal and extraintestinal manifestations common in articular joints and the eyes. Collagen XI may be considered a candidate for an autoantigen or the target of an aberrant self-recognition that contributes to an exaggerated inflammatory response by the adaptive immune system. Additionally, changes in collagen XI expression as a function of colon cancer progression may provide the opportunity for development of new diagnostic reagents.
In this study, immunostaining has been shown by bright field microscopy in normal and malignant colon tissue using collagen XI polyclonal antibodies against the splice forms of α1(XI) and the α2(XI) a chain. In normal colon tissue, juxtanuclear, dot-like staining has revealed collagen XI localized to the Golgi apparatus. A review of the literature indicated that this is the first confirmation of collagen XI staining in normal colon tissue. A Golgi marker, raised against a resident enzyme (58K, formiminotransferase cyclodeaminase), confirmed the location of Golgi apparatus in goblet cells, which mirrored the collagen XI staining locale. Ultra light microscopy also explicated the Golgi apparatus staining.
In comparing normal and malignant colon tissue staining with collagen XI antisera, there was an apparent decrease of Golgi staining and an increase of cytoplasmic staining in cancerous tissue compared with normal colon tissue. Further studies will investigate the statistical significance of these apparent differences in localization and intensity of collagen XI staining in normal and malignant tissue. It is possible that a dysregulation of protein localization within cells may accompany tumor progression and result in the changes observed.
Potential artifacts caused by cross-reactivity of the antisera were studied; however, no likely alternative targets for the antisera used in this study were identified. Titin, a cytoskeletal protein, has not been found in the colon or the Golgi and is therefore an unlikely candidate for cross-reactivity with the antibody to the carboxyl end of V2. Mib, a novel cerebral membrane protein, first found in senile plaques, was discovered by Satoh et al. (2006) and was shown to be present in colon among other tissues. Through colocalization with markers for endoplasmic reticulum (ER), ER–Golgi intermediate compartment, and
Collagen XI mRNA upregulation has been noted in the mucosa stromal cells of both familial adenomatosis polyposis and sporadic colorectal cancer, but the baseline expression and localization has not been established. Through northern blot and RT-PCR, using primers within the C1 and V1a regions and within the C-terminal domain respectively, collagen XI was not detected or perhaps was below the level of detection in normal tissue (Fischer et al. 2001a, b). The methods of Fischer et al. (2001a, b) and our study are dissimilar in that northern blot and RT-PCR detect collagen XI mRNA in the colon, whereas immunohistochemistry as carried out in our study detects collagen XI protein. Our study documents the presence of collagen XI protein in normal and malignant colon tissue using six polyclonal antisera to various locations to the exposed NTD of both the α1(XI) and α2(XI) chains.
The method and function of collagen XI's presence in the Golgi of the colonic crypt merits further study. Thyberg et al. (1979) suggested that the Golgi complex participated in the transfer of collagen to vesicles of the rat aorta. Conversely, collagen XI could play a role in mucus production and/or excretion. If endocytosis does occur, it might be beneficial to compare this process with disease-causing retrograde endocytosis of cholera toxin (Lencer and Tsai 2003; Feng et al. 2004) and Shiga toxin (Falguières et al. 2001), which both enter cells of the colon through the Golgi but are further transported into the ER. Collagen XI may be synthesized and localized to the Golgi and not further secreted or secreted normally and endocytosed back to the interior of the cell. Further studies using these antibodies will be carried out to document the transformation of the staining pattern during various stages in the development and progression of colon adenocarcinoma.
Footnotes
Acknowledgements
This study was supported by National Institutes of Health Grants P-20RR-016454 from the IDeA Network of Biomedical Research Excellence (INBRE) Program of the National Center for Research Resources, AR-47985 (to JTO), and AR-48672 (to JTO), the Lori and Duane Stueckle professorship, and funding from the M.J. Murdock Foundation.
The authors thank Dorthyann Isaakson, Karen Halsted, Angela Modin, Raquel Brown, and Janee Mestrovich for technical support and Dr. Tom Donndelinger for light microscopy.
