Abstract
Although mineral deposits have long been described to be a prominent feature of atherosclerosis, the mechanisms of arterial calcification are not well understood. However, accumulation of the non-collagenous matrix bone-associated proteins, osteopontin, osteocalcin, and osteonectin, has been demonstrated in atheromatous plaques. The aim of this study was to evaluate the role of these proteins in arterial calcification and, more precisely, during the initiation of this process. A model of rapid aortic calcification was developed in rabbits by an oversized balloon angioplasty. Calcification was followed using von Kossa staining and osteopontin, osteocalcin, and osteonectin were identified using immunohistochemistry. The aortic injury was rapidly followed by calcified deposits that appeared in the media as soon as 2 days after injury and then accumulated in zipper-like structures. Osteonectin was not detected in calcified deposits at any time after injury. In contrast, osteopontin and osteocalcin were detected in 8- and 14-day calcified structures, respectively, but not in the very early 2-day mineral deposits. These results suggest that these matrix proteins, osteopontin, osteocalcin, and osteonectin, are not involved in the initiation step of the aortic calcification process and that the former two might play a role in the regulation of arterial calcification.
A
A large number of recent reports on the mechanisms of connective tissue mineralization and bone remodeling underline the potential role of three non-collage-nous matrix proteins, osteopontin (OPN), osteonectin (ON), and osteocalcin (OC) (Holland et al. 1987; Price et al. 1987; Chen et al. 1993; Takano-Yamamoto et al. 1994).
OPN is an RGD-containing acidic phosphoprotein first isolated in bone (Oldberg et al. 1986). It is over-expressed by arterial smooth muscle cells (SMCs) according to their phenotype (Giachelli et al. 1991; Shanahan et al. 1993) and their position in the cell cycle (Gadeau et al. 1991). In vitro, OPN demonstrates adhesive and chemotactic activities for SMCs (Liaw et al. 1994) and its overexpression is closely associated with proliferation of this cell type (Gadeau et al. 1993; Ashizawa et al. 1996). In vascular tissue, OPN is strongly detected in the intima during neointimal formation induced by balloon injury (Giachelli et al. 1993) in association with areas of intense proliferation (Gadeau et al. 1994). OPN has also been characterized in human atherosclerotic plaques, in which it was synthesized by macrophages, SMCs, and endothelial cells (O'Brien et al. 1994), and it has been found to accumulate in calcified areas (Giachelli et al. 1993; Hirota et al. 1993; Fitzpatrick et al. 1994; Shanahan et al. 1994). OPN contains a hydroxyapatite binding site and a potential EF-hand calcium binding site, suggesting that this protein plays a role in calcium metabolism and potentially in the calcification process. Hunter et al. (1996) demonstrated that OPN is a potent inhibitor of hydroxyapatite formation, suggesting that OPN could have an inhibitory action on the calcification process. However, such a role has not been clearly demonstrated in the vessel wall.
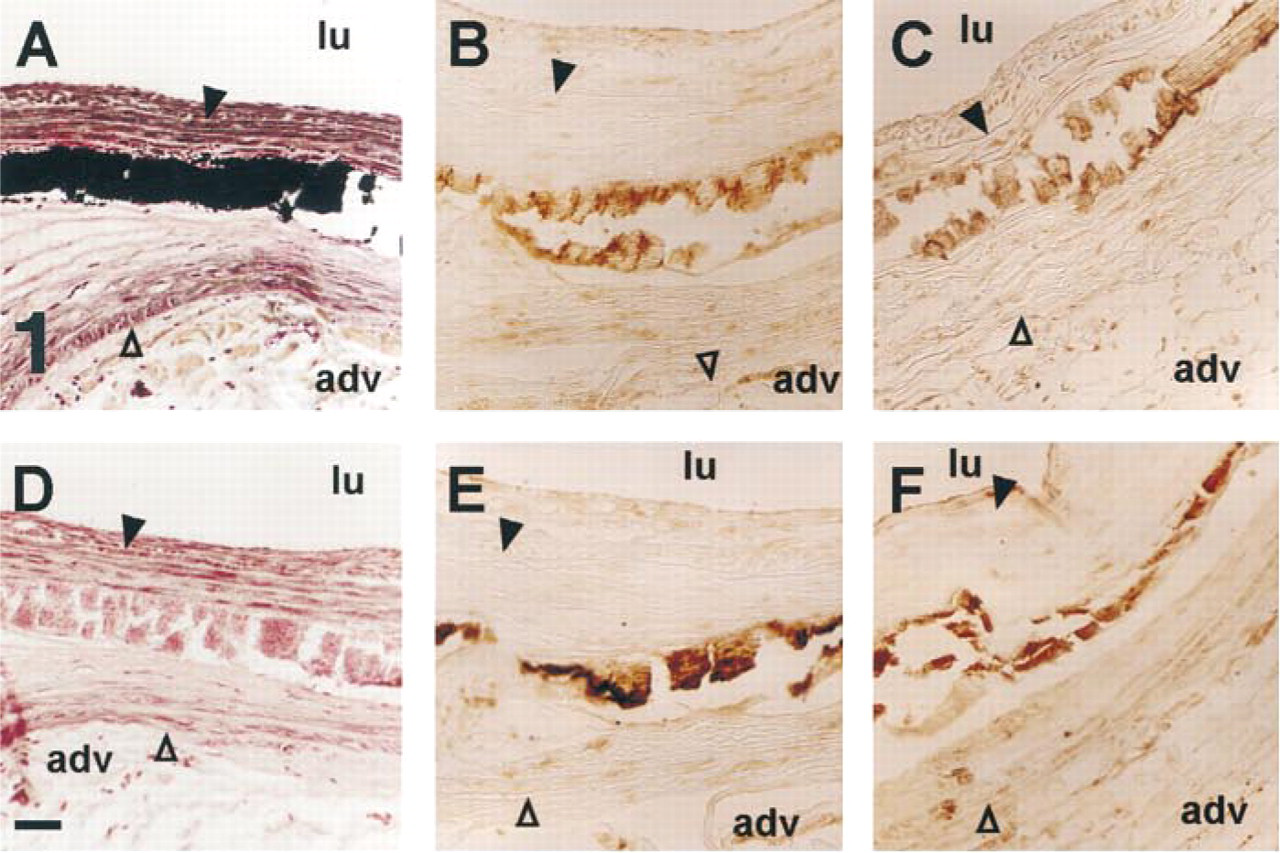
Osteopontin and osteocalcin detection in rabbit medial calcinosis. Calcium deposits of cholesterol-fed rabbit aorta detected by von Kossa staining in the middle of the media (
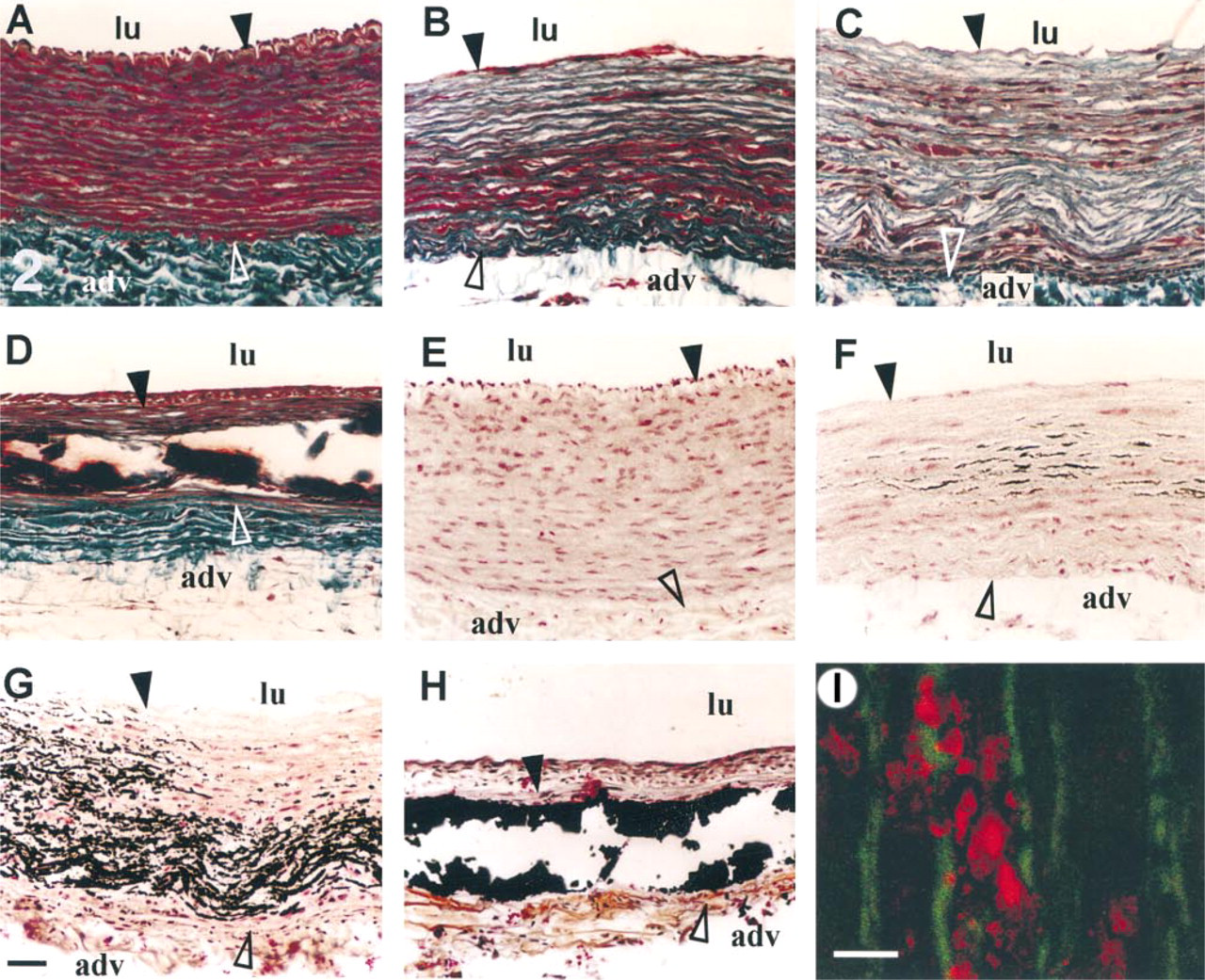
Morphology and kinetics of calcification of rabbit aorta after oversized balloon injury. Rabbit aortas were harvested before injury (
OC is a vitamin K-dependent matrix protein that contains γ-carboxyglutamic acid. In vitro, this protein strongly inhibits calcium salt precipitation (van de Loo et al. 1987), shows a strong affinity for hydroxyapatite, but inhibits crystal growth by delaying nucleation (Hunter et al. 1996). Although its role is still unclear, it has been demonstrated that OC normally limits bone formation (Ducy et al. 1996). In humans, OC has been demonstrated in calcified atherosclerotic plaques and calcified aortic valves (Levy et al. 1983).
ON, also called SPARC or BM40, is a secreted calcium binding protein involved in bone development (Mundlos et al. 1992) that demonstrates affinity for hydroxyapatite and collagen (Fujisawa et al. 1996; Sasaki et al. 1997). ON is detected in cultured SMCs (Severson et al. 1995) and is strongly expressed in young but not in healthy adult vessel walls. However, a few ON-expressing cells were scattered throughout the atherosclerosic lesions in humans (Hirota et al. 1993). Moreover, ON was found in association with large calcifications in human atherosclerotic plaques (Bini et al. 1999). ON also demonstrates a counter-adhesive effect on vitronectin-induced adhesion (Rosenblatt et al. 1997) and is involved in lowering the adhesiveness of tumor cells, thus increasing their invasive properties (Ledda et al. 1997). ON is also able to inhibit proliferation of endothelial cells and fibroblasts (Funk and Sage 1991; Pichler et al. 1996). We studied the distribution of OPN, ON, and OC during the early development of calcification in the rabbit model of injured aorta. Our results show that none of these proteins is detected in early calcifications but that OPN and OC are present in late calcium deposits, suggesting that these proteins might be involved in the control of calcification rather than in its genesis.
Materials and Methods
Tissue Collection and Staining Method
Animal studies were performed in accordance with French ministry of agriculture guidelines. Aortic tissues were obtained from male Fauve de Bourgogne rabbits. Animals were divided into two groups. In the first group, 6 month-old rabbits (n = 7) were fed for 6 months with an 0.3% cholesterol-enriched diet and sacrificed. In the second group, 3 month-old rabbits (n = 15) were subjected to aortic balloon injury (1.2–1.5 atm, three times) with a 3F angioplasty balloon (Baxter; Irvine, CA) after anesthesia by IM administration of 20 mg/kg ketamine (Imalgene) (Merial; Lyon, France) and 2 mg/kg xylazin (Rompum) (Bayer Pharma; Puteaux, France). Three rabbits were sacrificed after a defined duration of recovery (2, 4, 8, 14, or 30 days). Aortas were quickly dissected from the descending aorta to the iliac branch and eight pieces (0.5 cm) were removed at the same level in each aorta. As a control, a piece of healthy aorta was removed in all rabbits, just under the aortic arch, and was checked for calcification. These controls were always free of calcified deposits. Moreover, a healthy rabbit aorta was harvested under the same conditions and used as control. Aortas were fixed in ice-cold 4% paraformaldehyde for at least 3 hr. Specimens were rinsed in PBS, then embedded in paraffin. Seven-μm-thick sections were spread over APES (3-amino-propyltriethoxy-silane; Sigma-Aldrich, Saint-Quentin Fallavier, France)-coated slides.
Von Kossa-stained sections counterstained with Kernechtrot (Nuclear Fast Red) and Masson's trichrome were made according to classical methods (Gabe 1968). Quantitation of medial and calcified areas was performed on aortic cross-sections using a color video image analysis system (Quant'Image; INSERM U441, Bordeaux, France).
Calcium deposits were also specifically stained with Alizarin Red S and the sections were examined with a confocal microscope (Nikon PCM 2000) as previously described (Bernacca et al. 1994).
Immunohistochemistry
Serial sections were used for immunocytochemistry. SMCs were detected using an SM α-actin monoclonal antibody from Sigma-Aldrich, macrophages using RAM 11 monoclonal antibody from Dako (Trappes, France), OPN using MPIIIB10, and ON using AON-1 monoclonal antibodies purchased from DHSB, (Iowa City, IA) and OC using OCG2 from Takara (Biowhittaker Europe; Verviers, Belgium). Bound primary antibodies were detected using biotinylated anti-mouse secondary antibodies and the streptavidin–biotinylated horseradish peroxidase complex (Amersham Pharmacia Biotech; Orsay, France). The final complex was visualized by treatment with PBS containing 0.5 mg/ml diaminobenzidine and 0.03% H2O2 (Sigma-Aldrich). Antibody dilution and washes were carried out in PBS buffer containing 0.2% Tween-20 (Sigma-Aldrich) and 0.5% bovine serum albumin (ICN Biomedicals; Orsay, France). Controls were performed without primary antibodies and with nonspecific IgG1 antibody. To verify that antibodies did not bind nonspecifically to the calcium deposits, control sections with large calcifications were decalcified by 3% citric acid (Prolabo; Bordeaux, France) treatment for 1 hr before immunolabeling (Kockx et al. 1996).
Results
OPN, OC, and ON Accumulation in Rabbit Atherosclerotic Plaques
Aortic calcification of cholesterol-enriched diet-fed rabbit occurred frequently (Rokita et al. 1991). These calcifications were found in atherosclerotic plaques or in the center part of the media (Figure 1A). OC and OPN, but not ON, were immunodetected in these calcified deposits (Figures 1B and 1C).
Because hydroxyapatite has a high binding capacity for many proteins, we verified whether OPN and OC immunodetection found in calcified deposits was really due to the presence of OPN and OC antigens. Calcium phosphate was removed by a 1-hr citric acid treatment. Completion of calcium removal was controlled by the absence of black stain deposit after von Kossa staining (Figure 1D). However, a residual material, detected by Kernechtrot counterstaining (a nuclear red and cytoplasmic pink stain) remained in place of the calcium deposit. After this treatment, OC and OPN were still detected in the same areas with a nearly equal staining intensity to that in untreated sections (Figures 1E vs 1B and 1F vs 1C), demonstrating that the antibodies were not nonspecifically adsorbed on hydroxyapatite.
When primary antibody was omitted or replaced by nonspecific IgG1, the second antibody or streptavidin–peroxidase complex did not bind to calcifications (not shown).
Early Events in the Mineral Deposits
Early events in the mineral deposits were monitored in the rabbit aorta balloon injury model. The aortic injury was performed at 1.2–1.5 atm by moving a 3F angioplasty balloon from the thoracic to the abdominal aorta. Aortas were harvested either before injury or 2, 4, 8, 14, or 30 days after injury, and serial sections were made. Immunostaining with SM α-actin and RAM 11 antibodies demonstrated that only SMCs were detected in the injured tissue. At Day 30, some macrophages were observed in association with the calcification (not shown).
Masson's trichrome staining showed extensive structural tissue reorganization of the media in comparison to healthy aorta (Figure 2A). Two days and 4 days after injury, the interlaminae spaces mainly located in the inner half of the media were devoid of cells, as demonstrated by the absence of red cell staining (Figures 2B and 2C). Sections of 8, 14, and 30 days were torn, suggesting the presence of “hard” components, such as calcified materials, in the media (Figure 2D shows a 14-day picture). Indeed, von Kossa's staining of serial sections showed that the arterial wound induced medial calcification. Although calcifications were never observed in healthy tissue (Figure 2E), 2 days after injury calcium deposits were found in the aorta of two of the three rabbits (Figure 2F). These deposits were not homogeneously distributed in the media but appeared only in cell-free areas. High magnification showed that calcified deposits were first present between the elastic laminae, mainly in association with remaining cellular materials stained by Kernechtrot (not shown). Confocal microscopic examination after Alizarin Red S treatment confirmed that the first calcified deposits (red fluorescence) were not closely associated with the elastic laminae (green autofluorescence) (Figure 2I). On Day 4, more extended calcifications were also detected in the media of two rabbits (Figure 2G), and deposits began to fill the empty areas in place of the cells between the elastic laminae. From Day 8, the elastic laminae were totally embedded in the calcified structure (not shown) and became fragmented into small pieces, and large calcium deposits demonstrating a dense, zipper-like morphology were present on Day 14 (Figure 2H). Therefore, calcium deposits rapidly increased between Days 2 and 4 after injury from 2.5–20% of the total media surface, reaching more than 30% 1 month later (Figure 3).
Successive sections of injured aortas were investigated for OPN, OC, and ON protein location. OPN was not detected in healthy media (Figure 4A) but it appeared very rapidly 2 days after injury (Figure 4B). OPN was detected only in the remaining cells of injured media. In the same way, 4 days after injury, OPN was essentially detected in SMCs of the middle and internal part of the media (Figure 4C). At that time, OPN accumulation was not associated with very early calcified deposits. Then, from Day 8, OPN was detected in the neointima and also in the media, where it was clearly associated with calcified areas (Figure 4D).
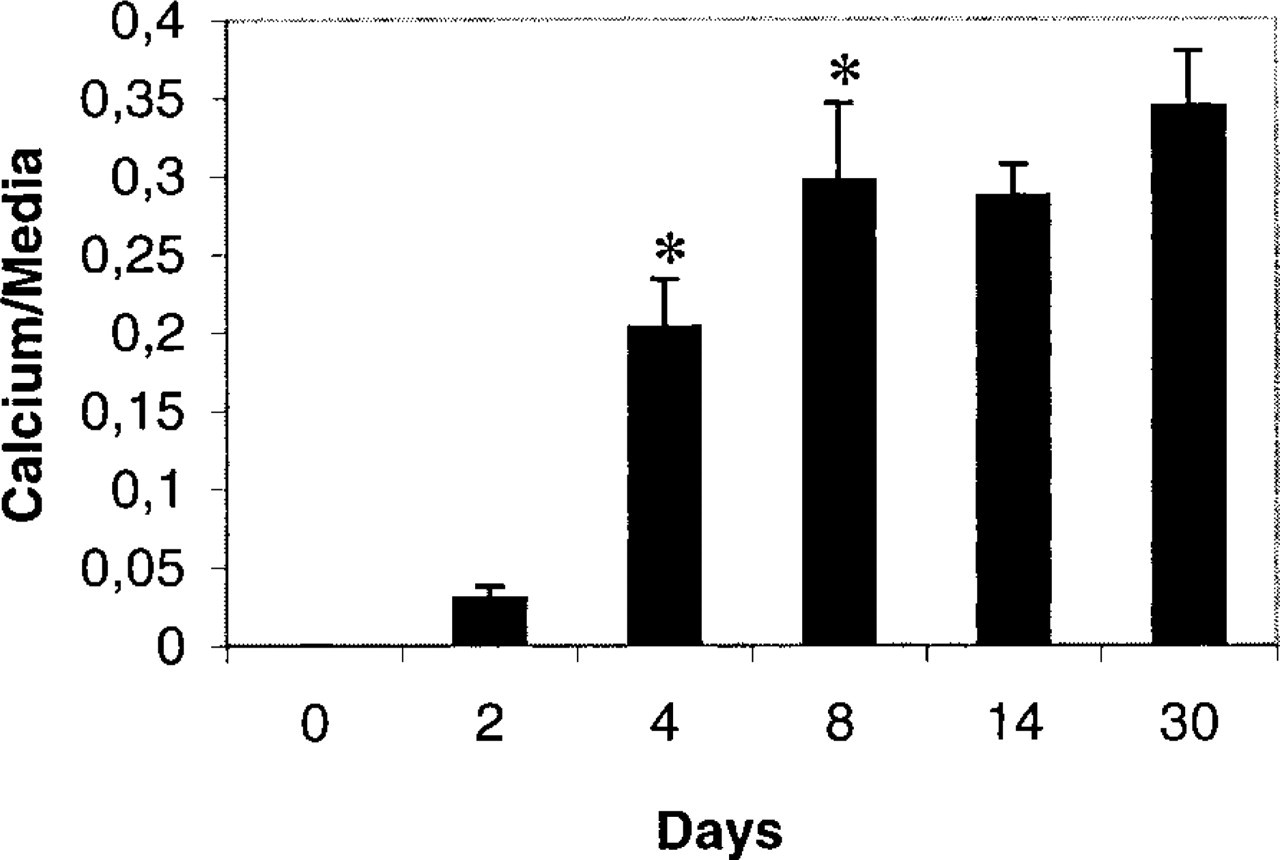
Quantitative analysis of calcium deposit areas in rabbit aorta media after balloon injury. The ratio of calcium deposit areas to total medial area at the level of the calcified zones of the vessel was determined at different days after balloon injury. Four sections of calcified rabbit aortas (two for Days 2 and 4 and three for Days 8 and 30) were stained by the von Kossa stain and calcium deposit and total medial areas were evaluated using video color image analysis. Analysis of variance demonstrated differences between values on Days 2, 4, 8, 14, and 30 (ANOVA p<4 × 10–13). Ratios between Days 2 and 4 and between Days 4 and 8 were significantly increased (∗) as determined by the Student's t-test (p < 0.005).
In healthy rabbit and before the eighth day after injury, OC was not detected in the media (Figures 5A–5C). Then, from Day 14 after injury, OC accumulated in association with calcium deposits (Figure 5D) and began to be detected in SMCs. In healthy tissue, ON was detected only in endothelial cells and adventitia (Figure 6A), but 14 days after injury this protein was found in the intima, in the cellular part of the media, and all around but not inside calcifications (Figure 6B).
Discussion
To understand the role of OPN, OC, and ON accumulation in calcified areas and their involvement in the initiation of this process, we developed a model of rapid aortic calcification that enabled us to follow non-collagenous bone proteins throughout the development of aortic calcification. In this model, severe aortic injury was induced by balloon angioplasty.
Surprisingly, calcified deposits appeared very rapidly, only 2–4 days after injury, in areas of the injured aortic media where cells had been killed. This rapid phenomenon, detected only in dead-cell areas, suggests that the early initiation of calcification is linked to SMC death, unveiling necrotic areas which can function as nidus for this process. During balloon injury a large number of cells were killed by crushing and by apoptosis (Perlman et al. 1997). However, 2 days after injury, this apoptosis phenomenon was quite completed and only very few TUNEL-positive cells could be detected (personal data). During initiation, calcified deposits were first observed in place of cells between elastic laminae but not in close association with them (demonstrated by confocal microscopy) as previously described by Bobryshev et al. (1995), who hypothesized a role for damaged extracellular matrix elastin or collagen in calcium deposition. However, later on when the calcium crystals increased, many contacts with the elastic laminae were observed, as shown by these authors.
Our results suggest that the early steps of the calcification process after rabbit injury could be related to cell death. In this case, dead cells might create an active nucleation of calcium crystals. The mechanisms involved in the subsequent steps are not clearly understood but could involve the bone-associated matrix proteins detected in calcified deposits, OPN, OC, matrix Gla protein (MGP) (Shanahan et al. 1994), or bone sialoprotein (BSP) (personal observations). Indeed, recent studies suggest that OPN and OC may play a regulatory role in the bone calcification process (Chen et al. 1993; Denhardt and Guo 1993). However, the role of these proteins in arterial calcification is still unclear. In vitro physicochemical studies using a cell-free model have shown that OC and OPN have no ability to induce hydroxyapatite crystal formation (Hunter et al. 1996). Our results, showing that OPN and OC proteins were not present in very early calcification stages but appeared after calcified deposits were formed, suggest that these proteins are not necessary for initiation of arterial calcification. These findings are paralleled by in vitro studies showing the absence of OPN during the formation of calcified foci by pericytes in culture (Proudfoot et al. 1998). Our results also demonstrated that calcification appeared in cell-free areas in which OPN synthesis cannot occur.
Two weeks after injury, all calcified deposits contained OPN and OC, suggesting that these proteins were adsorbed on the calcium crystals because of their strong affinity for hydroxyapatite. This observation indicates that these proteins could be involved in the regulation of calcium crystal development. Although Ducy et al. (1996) demonstrated that OC limits bone formation without impairing mineralization, it has been shown in vitro that OC can delay hydroxyapatite nucleation and that OPN is a potent inhibitor of crystal growth (Shiraga et al. 1992; Hunter et al. 1996). Therefore, the hypothesis that these proteins are involved in the limitation of arterial wall calcification should be seriously considered. This is particularly so for OPN, which inhibits calcification of vascular SMC cultures (Wada et al. 1999). Although OPN and OC were detected in atherosclerotic plaques, it is not clear why pronounced hydroxyapatite formation was not inhibited in some advanced atherosclerotic plaques. It may be that the presence of a positive regulator of bone formation (Boström et al. 1993) and crystal growth, such as BSP, which exhibits hydroxyapatite nucleation activity (Hunter et al. 1996) and can bind collagenous matrix (Nagata et al. 1991), counterbalances the inhibition.
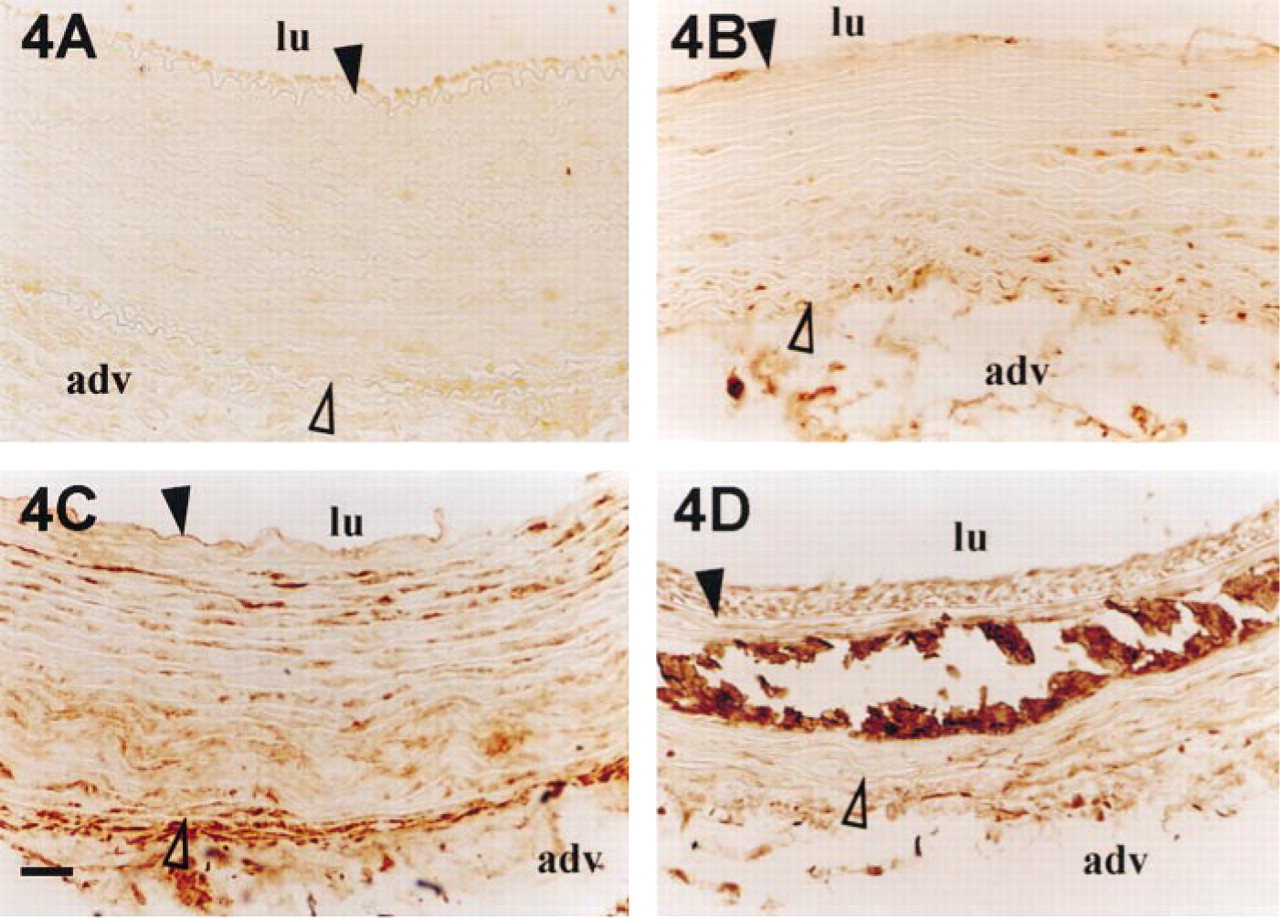
Osteopontin accumulation in rabbit aortas after balloon injury. Rabbit aortas were harvested before injury (
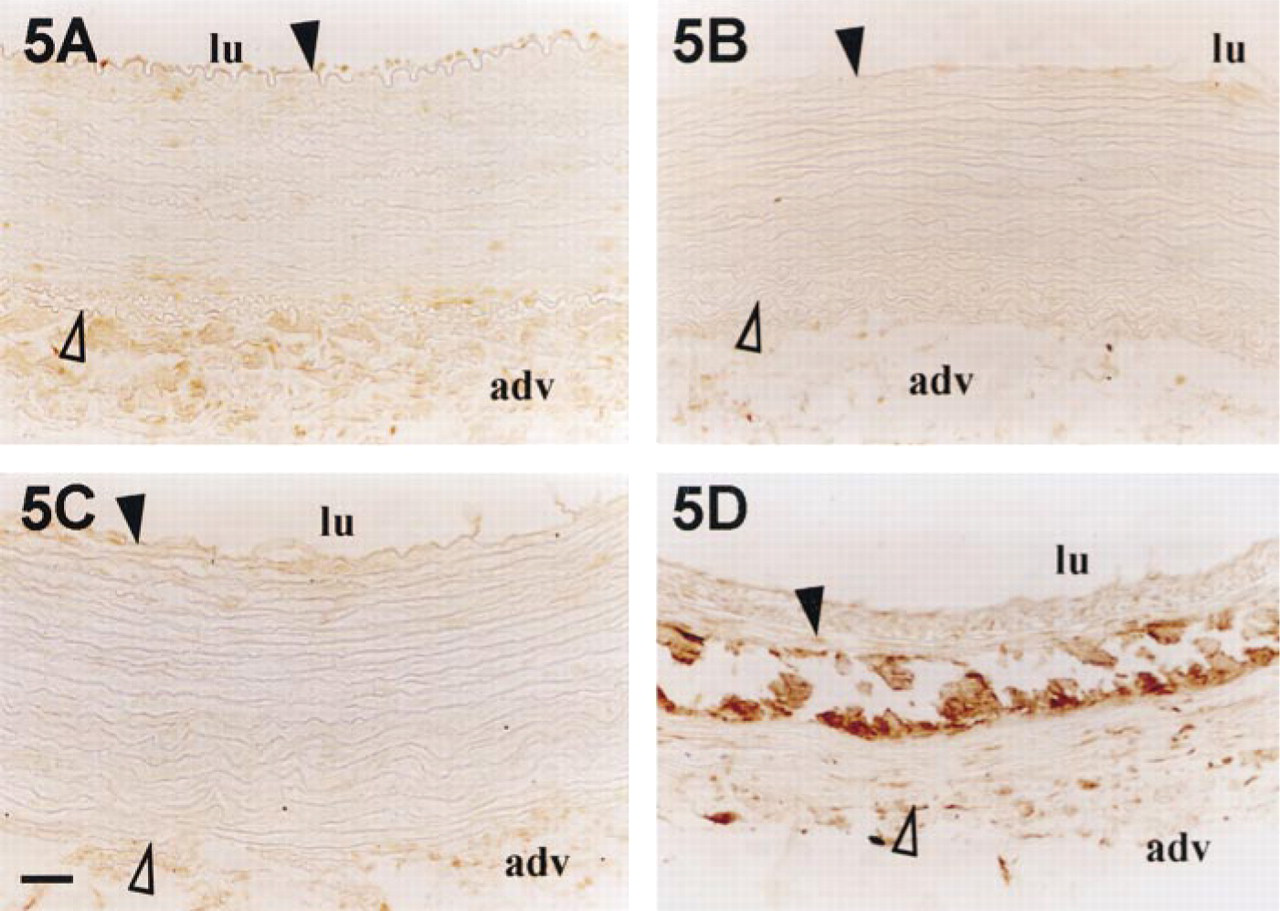
Osteocalcin accumulation in rabbit aortas after balloon injury. Rabbit aortas were harvested before injury (
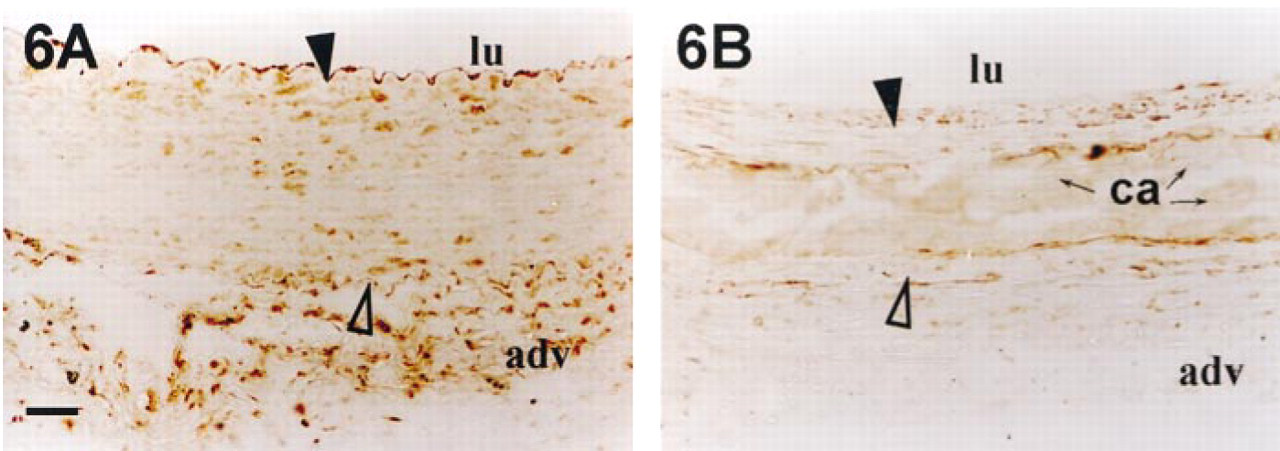
Detection of osteonectin in rabbit aortas after balloon injury. Rabbit aortas were harvested before injury (
OPN and OC could come from the surrounding cells or from plasma that has infiltrated the injured aortic media. In the calcification of human atherosclerotic plaques, it has been shown that OPN may be of macrophage or SMC origin (O'Brien et al. 1994). In the media of rabbit balloon-injured aortas, during initiation of calcification only SMCs but not macrophages were detected. OPN at an early stage and OC later on were detected in medial and intimal SMCs and also in adventitial cells when calcium deposits accumulated, demonstrating the role of these cells in the production of OPN and OC associated with calcified structures. Moreover, OPN and OC can come from plasma because it has been reported that these proteins are present in serum (Jie et al. 1995; Panda et al. 1997).
Footnotes
Acknowledgments
Supported by grants from Institut National de la Santé et de la Recherche Médicale.
We wish to thank P. Alzieu for excellent technical assistance. Monoclonal antibodies MPIIIB 10 (1) developed by M. Solursh/A. Franzen and AON-1 developed by J.D. Termine were obtained from the Developmental Studies Hybridoma Bank under the auspices of NICHD and maintained by the University of Iowa Department of Biological Sciences (Iowa City, IA).
