Abstract
We generated a new monoclonal antibody (MAb), F7.2.38, by immunizing mice with CD3∊γδ/CD3ω complexes purified from human T-cells by OKT3 MAb–Sepharose affinity chromatography. Immunoprecipitation experiments and Western blotting analysis showed that MAb F7.2.38 recognized the CD3∊ chain in CD3∊ cDNA-transfected FOX B-cells and in various T-cell lines. Using flow cytometry on permeabilized or intact cells, the epitope was found to be located in the cytoplasmic tail of the CD3∊ chain. Immunohistochemical staining on paraffin-embedded sections showed that the reactivity of MAb F7.2.38 was comparable to that of the commercially available anti-CD3∊ polyclonal antibody. Of the 52 well-characterized T-cell lymphomas, 41 were positive for F7.2.38 (79%), whereas all 37 B-cell lymphomas and 69 non-lymphoid tumors were unreactive. This new anti-CD3∊ antibody would be particularly useful for phenotyping T-cell lymphomas on routinely processed paraffin-embedded tissue sections.
Keywords
T
These considerations have led us to develop methods for the production of MAbs against all components of known human TCR/CD3 complexes (pre-TCR, TCRγδ, or TCRαβ). Such MAbs will be very useful for studies on TCR/CD3 structure and in the characterization of TCR complexes on T-lymphocytes in various diseases such as immunodeficiency, allergy, autoimmunity, and cancer. Our goal has been to produce MAbs useful in many different analytical systems, i.e., cytofluometry on living or permeabilized cells, immunohistochemistry on tissue sections prepared by different methods, immunoprecipitation in different detergents, and Western blotting analyses. Because immunization with living T-cells has been unsuccessful, we attempted to isolate CD3 complexes from NP40-T cell lysates on anti-CD3∊ MAb affinity columns. Mice were injected with the eluted material, which contained mainly CD3γ, δ, and ∊ molecules, plus an intracellular chaperone, CD3ω, implicated in CD3 complex assembly (Pettey et al. 1987; Neisig et al. 1993).
Here we present the characterization of one of the MAbs obtained using this strategy. The MAb, F7.2.38, directed against a cytoplasmic epitope in the CD3∊ chain, recognizes an epitope present in both frozen and formalin-fixed material. The reactivity of this antibody was compared with that obtained using the commercially available rabbit anti-CD3∊ antibody (Dako; Trappes, France).
Materials and Methods
Production of MAb F7.2.38
CD3 complexes were obtained by affinity chromatography of 2% NP-40 detergent (Fluka Chemie; Buchs, Switzerland)-solubilized proteins from Jurkat T-cells (250–500 × 106 cells/immunization/mouse) on a 5-ml Sepharose bead column (Amersham Pharmacia Biotech; Orsay, France) with covalently coupled MAb OKT3. Elution was performed with 1 M NH4SCN or 0.2 M glycine buffer, pH 2.8. F1 hybrid female mice (C57B6 × Balb/c) were immunized three times, once SC and twice IP, with eluted material in 250 μl of DPBS (NaCl 0.14 M, Na2HPO4 8 mM, KH2PO4 1.5 mM, KCl 2.7 mM, pH 7.2) with 250 μl of complete Freund's adjuvant (Statens Seruminstitut; Copenhagen, Denmark). Four months later the mice were immunized regularly (at least five times at 2-week intervals) with IP injections of equal doses of the CD3 complexes, still adsorbed onto the beads, without adding Freund's adjuvant. All mice produced antibodies precipitating CD3γ, CD3δ, CD3∊, and CD3ω proteins as well as molecules with higher (<30,000 Daltons) or lower (>20,000 Daltons) molecular weights (not shown). Five days after the last IP immunization, fusion of the spleen cells with the non-Ig-producing myeloma cell line X63 Ag8 653 was performed using standard techniques (Al Saati et al. 1989). When hybridoma growth could be detected, supernatants were tested by cytofluorometry for the presence of MAbs that could recognize permeabilized Jurkat cells (see below). Supernatants from positive hybridomas were tested again in immunoprecipitation experiments with [35S]-methionine/cysteine-labeled Jurkat cell lysates, as described in Geisler et al. (1990). MAb F7.2.38 (of IgG1 subclass) was selected for further detailed studies because it precipitated proteins with molecular weights similar to those of CD3γ, CD3δ, CD3∊, and CD3ω chains.
Cell lines and MAbs
Human leukemia αβ (Jurkat subclone E6.1, HBP-ALL, CEM, and MOLT4) and γδ (LYON and PEER) T-cells, human B-lymphoma Raji cells, and murine B-lymphoma FOX cells were grown in RPMI 1640 with
Mouse SP-34, OKT3, and F101.01 MAbs specific for single or CD3-associated CD3∊ components, respectively, were obtained from Dr. C. Terhorst (Boston, MA), ATCC (Rockville, MD), and Dr. T. Plesner (Copenhagen, Denmark). CD20/L26 anti-B cell MAb and the rabbit polyclonal anti-CD3∊ (A0452) were purchased from Dako.
Flow Cytometric Analyses
Cells were permeabilized using 0.1% saponin (Sigma; Saint Quentin Fallavier, France). A total of 106 cells were washed in DPBS and labeled for 30 min at 4C with F7.2.38, SP-34, and OKT3 MAbs supplemented with 0.1% saponin. After two washes in DPBS containing 0.1% saponin, cells were incubated for 30 min at 4C with a fluorescein isothiocyanate-conjugated rabbit Fab2′ anti-mouse (F0313; Dako) supplemented with 0.1% saponin. Cells were washed in DPBS with 0.1% saponin and analyzed with the Coulter Electronics Cytometer (Coulter EPICS XL; Hialeah, FL).
For surface staining analysis, exactly the same procedure was followed, but without adding saponin, and using a phycoerythrin-conjugated goat anti-mouse (IM0551; Immunotech, Marseille, France) as secondary antibody.
Transfections
Murine B-lymphoma FOX cells were transfected by electroporation. Approximately 5 × 106 cells were washed twice in culture medium without FCS, resuspended in the DNA transfection mixture [5 μg of pSRa-CD3∊del12 described by B. Alarcon (Mallabiabarrena et al. 1992), linearized by digestion with BamH1, precipitated, and resuspended in 750 μl of the cell culture medium without FCS] and then electroporated in 0.4-cm cuvettes (Ozyme; Montigny, France) at 270 V, 960 μF using a Biorad Gene Pulser apparatus (Bio-Rad; Ivry Sur Seine, France). The cells were carefully resuspended in a 25-cm2 flask containing 10 ml of culture medium. At 48 hr later, 2 mg/ml of G418 (Life Technologies) was added to the medium. When viable cells began to appear (approximately 10 days later), clonings by limiting dilutions were made in the presence of 1 mg/ml of G418. The positive clones were selected by flow cytometry using 0.1% saponin and the anti-CD3∊ MAb SP-34. In this report, all results shown were obtained with clone T116.4.3.
Western Blotting Experiments
A total of 5 × 106 FOX, T116.4.3, or Jurkat E6.1 cells were washed twice in DPBS and then lysed in 500 μl of Tris 0.5 M, pH 6.8, 2% SDS, 10% glycerol, bromophenol blue, with or without 5% β-mercaptoethanol. After three 5-sec sonications, lysates were boiled for 10 min at 100C and centrifuged for 10 min at 14000 × g, 4C. Thirty μl of supernatant was separated by SDS-PAGE (12%) according to the Laemmli (1970). Electrophoresis was followed by transfer of proteins to PVDF membranes (NEN Life Science Products; Paris, France) at 4C, 190 mA, 3 hr. At the end of the electrotransfer, membranes were colored with Ponceau's red to control the quality of the transfer and the loaded samples. Then they were incubated for 1 hr, first with blocking buffer [DPBS; 0.1% Tween-20 (Merck; Fontenay, France), 0.1% gelatin, 5% skimmed milk, and 0.5 g/liter BSA], second with blocking buffer diluted twice and containing SP-34 or F7.2.38 MAb supernatant diluted 10 times. Then membranes were washed three times for 5 min in DPBS with 0.1% Tween-20, and bound antibodies were detected with HRP-conjugated rabbit anti-mouse Ig (P0161; Dako). Peroxidase activity was revealed using an Enhanced Chemiluminescence Detection system (Amersham Pharmacia Biotech).
Immunostaining of Various Human Normal and Neoplastic Tissue Sections
F7.2.38 supernatant diluted 1:10 was used to study its reactivity on a large panel of a variety of normal and pathological human tissue samples from the tissue bank of the department of Pathology at Purpan Hospital in Toulouse, using the multi-tissue block method (Battifora 1986). Tissue samples were fixed in 10% neutral buffered formalin or Duboscq-Brasil (ethanol-based Bouin's fluid), and in a few cases in Bouin's fixative, Bouin Holland, and B5, to assess the fixative dependence of the staining intensity.
Different human tumors (n = 179) were investigated. These included: (a) 110 cases of lymphoid neoplasms [including 89 non-Hodgkin's lymphomas classified according to the REAL and the WHO classifications (Harris et al. 1994, 2000) and 21 cases of Hodgkin's disease (Table 1)]; and (b) 69 cases of non-lymphoid tumors of different categories. In addition, 142 cases of normal or non-neoplastic lymphoid (n = 28) and non-lymphoid (n = 114) tissue samples were investigated to evaluate the reactivity of this antibody in normal human organ tissue specimens.
Reactivity of MAb F7.2.38 with malignant cells of 110 cases of malignant lymphomas and Hodgkin's disease on paraffin-embedded tissue sections classified according to the REAL and WHO classifications (Harris et al. 1994, 2000)

Paraffin-embedded sections were immunostained by the streptavidin–biotin–peroxidase complex (ABC) method using Dako StrepABComplex/HRP Duet (mouse/rabbit) kit (K0492; Dako). The antigen retrieval method used heat pretreatment of sections in a 750-W microwave oven (10 min of boiling twice in citrate buffer, pH 6) (Shi et al. 1991). In some cases, we also evaluated the immunostaining on paraffin-embedded tissue sections using this antibody with two different immunostains: Techmate 500 (Dako) and Ventana 320/ES (Ventana; Strasbourg, France).
Results
Characterization of the Molecule Targeted by MAb F7.2.38
Similar Patterns of Proteins Precipitated by Known Anti-CD3∊ and F7.2.38 MAbs. MAb F7.2.38 was selected because it could (a) recognize the human leukemia T-cell line Jurkat by intracellular staining analyzed by flow cytometry and (b) immunoprecipitate proteins with molecular weights similar to those of the CD3γ, CD3δ, CD3∊, and CD3ω chains from 2% NP-40 lysates of Jurkat cells. All TCR/CD3 proteins except CD3ω are usually co-precipitated with an anti-CD3∊ MAb from a 1% digitonin T-cell lysate. However, if 2% NP-40 is used, only CD3γ, δ, ∊, and ω chains are co-precipitated. Therefore, comparable immunoprecipitations with MAb F7.2.38 of either digitonin or NP-40 lysates may indicate whether the MAb is specific for CD3γδ∊ or CD3ω molecules. The results in Figure 1 demonstrate that MAb F7.2.38, like MAb OKT3, co-precipitated three bands corresponding to CD3γδ∊ and CD3ω from NP-40 lysates and no CD3ω molecules from digitonin lysates. Consequently, MAb F7.2.38 appeared to react against the CD3γδ∊ chains rather than CD3ω. One difference between the immunoprecipitations with MAbs OKT3 and F7.2.38 was the presence of an additional band of 40 kD in the F7.2.38 immunoprecipitates (arrow). This 40,000-Dalton band was also present in immunoprecipitates with the SP-34 MAb specific for single CD3∊ chains (data not shown). The same results were obtained when we used MAb F7.2.38 with lysates from other human T-cell lines, such as the αβ HBP-ALL, CEM, and MOLT4 T-cell lines or the γδ LYON and PEER T-cell lines (data not shown). MAb F7.2.38 did not immunoprecipitate any TCR/CD3-like proteins from Raji B-cell lysates (Figure 1). These data indicate that MAb F7.2.38 reacts similarly to some other anti-CD3∊ MAbs in both detergents used.
MAb F7.2.38 Specifically Recognizes the Human CD3∊ Chain. These results led us to postulate that MAb F7.2.38 could be directed against the CD3∊ chains or against CD3γ∊ or CD3δ∊ dimers. Therefore, we transfected the murine B-cell lymphoma FOX with a cDNA encoding a truncated CD3∊ chain (Mallabiabarrena et al. 1992). The transfected cells are designated T116.4.3. We observed that MAb F7.2.38, like MAb SP-34, could stain permeabilized transfected, but not permeabilized untransfected, FOX cells (Figure 2A). MAb OKT3 did not recognize the transfectants because this MAb is specific for a “conformational” CD3∊ epitope (see Introduction). To confirm these results, a Western blotting experiment was carried out using lysates of the CD3∊-expressing FOX cells and blotting the membranes with MAb F7.2.38 or SP-34. Figure 3 shows that a ~40-kD band was revealed by MAb F7.2.38 in T116.4.3 cell lysates but not in FOX cell lysates. The proteins in this band were reduced (in the presence of β-mercaptoethanol) to a ~20-kD band, indicating that the 40-kD band represented a disulfide-linked homodimer of CD3∊ chains. In E6.1 cell lysates, MAb F7.2.38 revealed a 20-kD band and a 40-kD band. As before, the 40-kD molecules (but not the 20-kD molecules) were reduced to 20 kD. This 40-kD band appears similar to a corresponding molecule in Figure 1 (arrow). The same pattern was observed when the membranes were blotted with MAb SP-34 (data not shown). Similar results were obtained when immunoprecipitation experiments were performed with lysates of [35S]-methionine/cysteine-labeled cells and MAb SP-34 or F7.2.38. However, the results obtained with MAb F7.2.38 proved more constant than those obtained with MAb SP-34 in Western blotting analysis. In contrast, MAbs F7.2.38 and SP-34 demonstrated equivalent activities against CD3∊ molecules in [35S]-methionine/cysteine-labeled cell lysates (data not shown).
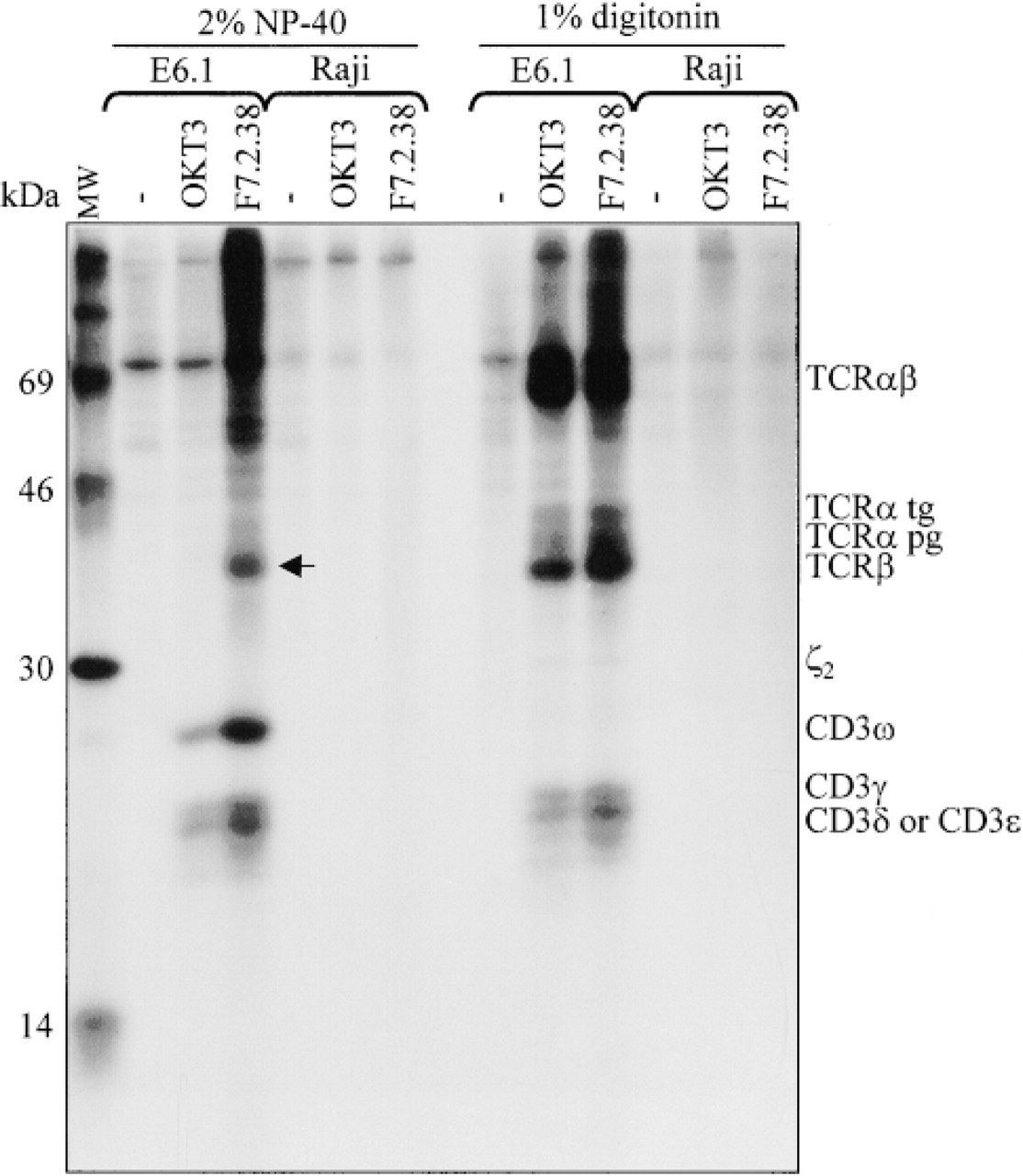
35S-Immunoprecipitates from E6.1 T-cells and Raji B-cells, prepared with MAbs OKT3 and F7.2.38 and separated under non-reducing conditions in a 12% SDS-PAGE. Using 1% digitonin or 2% NP-40 for cell lysis, very similar patterns of immunoprecipitated proteins are observed with the two MAbs, representing the CD3γ, CD3δ, and CD3∊ chains, and indicating that MAb F7.2.38 is directed against the CD3∊. As expected, the additional bands observed using 1% digitonin correspond to the ζ2 homodimer, TCRβ, TCRα partially (pg) or totally (tg) glycosylated, and the TCRαβ dimer, which have been immunoprecipated with the CD3∊ chain. Similarly, the additional band observed using 2% NP-40 corresponds to the CD3ω chain, which has been immunoprecipitated with the CD3∊ chain. Note also that using 2% NP-40, one additional band of 40 kD (probably corresponding to CD3∊2 homodimers) is observed using MAb F7.2.38 (arrow) but not with OKT3. Proteins are indicated at right and standard molecular weights are shown at left.
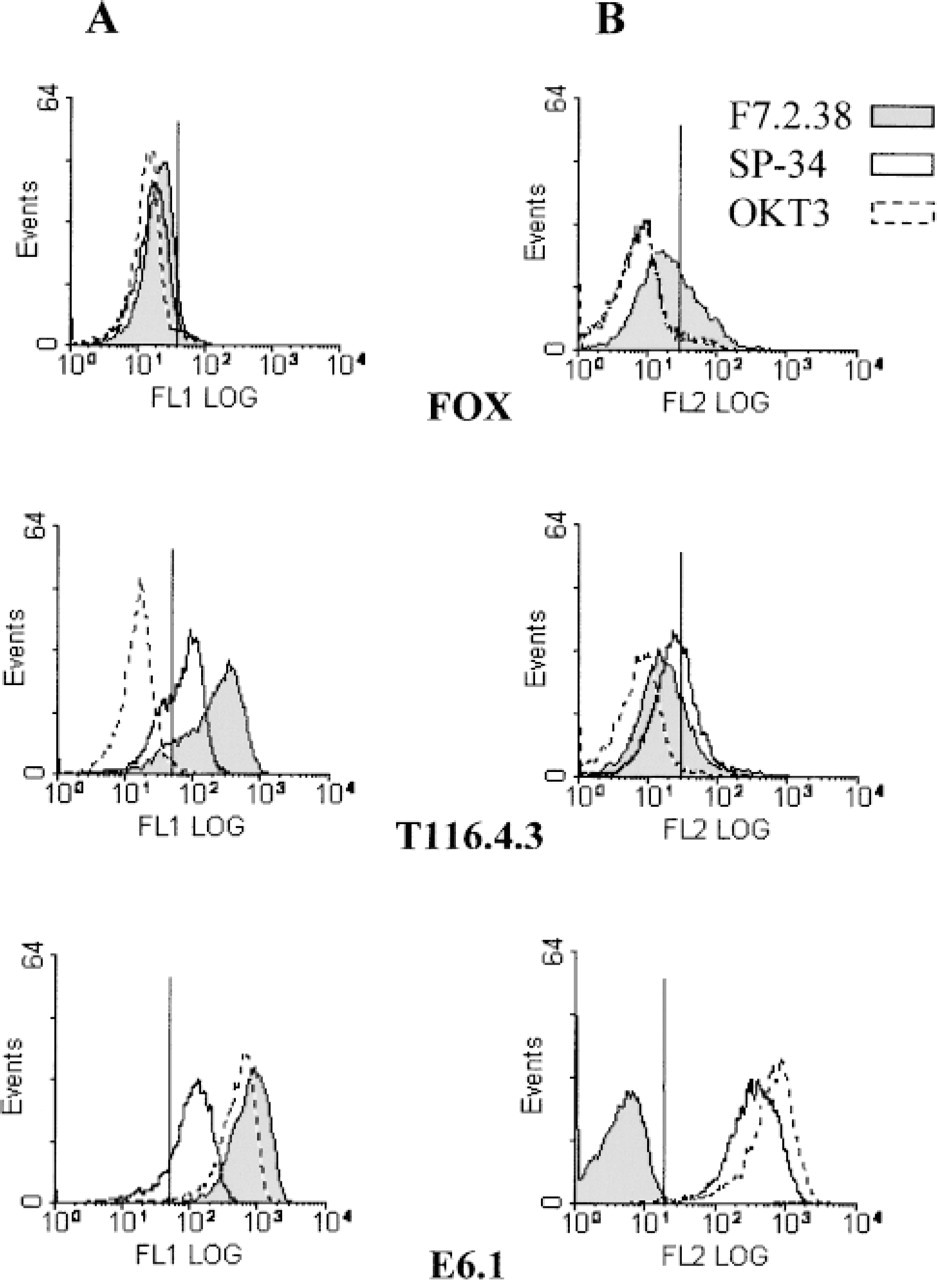
CD3∊ chain labeling by MAb F7.2.38 revealed by flow cytometric analyses. FOX cells either expressing CD3∊ chains after cDNA-transfection (T116.4.3) or negative for CD3∊ (wild-type FOX), and E6.1 T-cells were stained with SP-34, OKT3 or F7.2.38 supernatants, with (
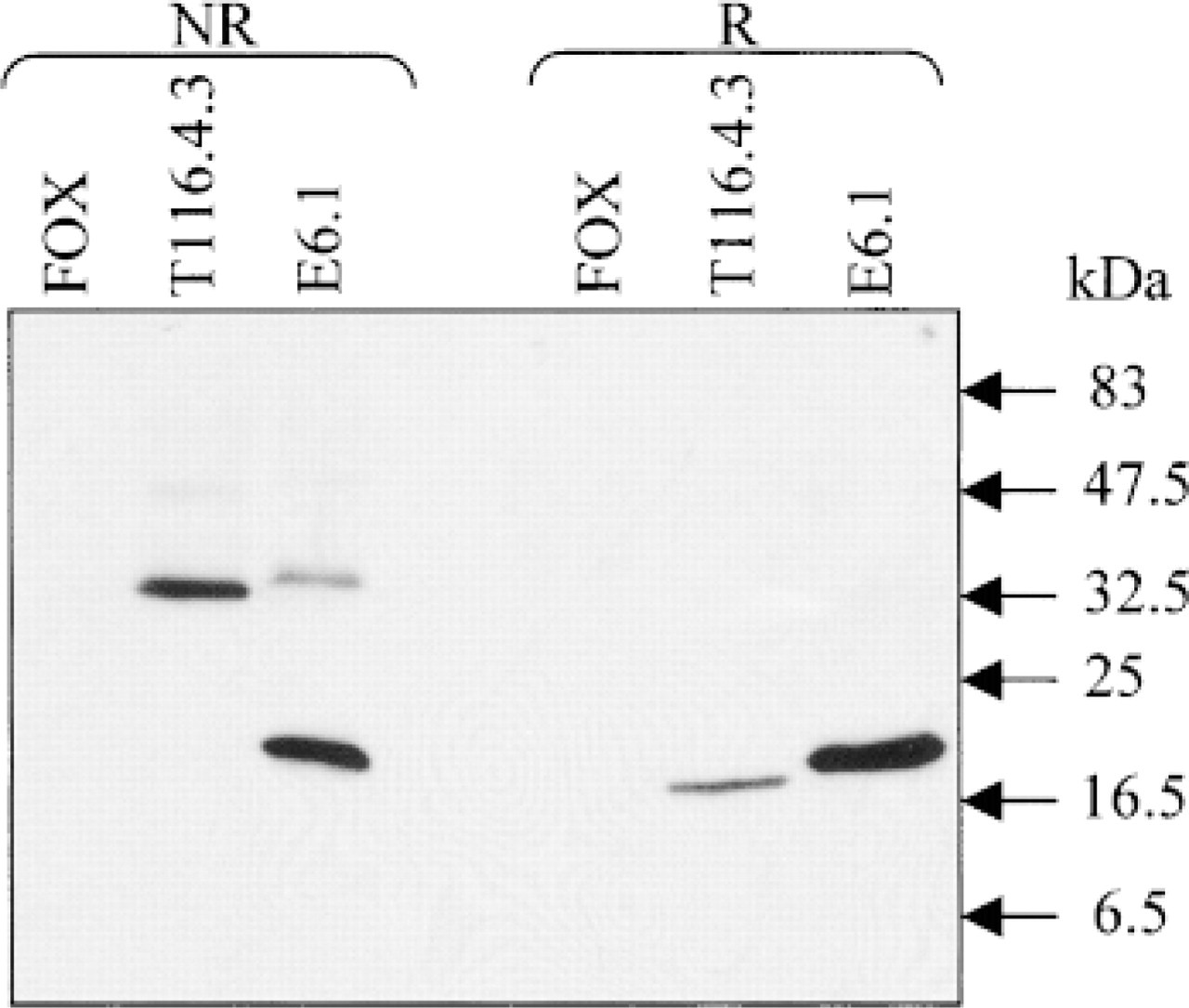
CD3∊ chain recognition by MAb F7.2.38 revealed by Western blotting. FOX cells either expressing CD3∊ chains after cDNA transfection (T116.4.3) or negative for CD3∊ (wild-type FOX) and E6.1 T-cells were lysed in non-reducing (NR) or reducing (R) conditions. Western blotting was performed with F7.2.38 or SP-34 as control (not shown) supernatants. MAb F7.2.38 reveals bands at the expected size for CD3∊ (20 kD) chains and CD3∊2 (40 kD) disulfide-linked homodimers in T116.4.3 and E6.1 lysates. The proteins in the 40-kD bands shifted to 20-kD bands under reducing conditions. The same pattern is obtained with MAb SP-34. Standard molecular weights are shown at right.
MAb F7.2.38 Does Not Recognize the CD3∊ Chain on the Surface of Cells. The cDNA used for FOX cell transfection experiments encodes the CD3∊ chain with a deleted endoplasmic reticulum retention signal sequence, which in principle should allow the molecule to be expressed at the cell surface (Mallabiabarrena et al. 1992). This deletion explains the small molecular weight difference between T116.4.3 CD3∊ chain and the E6.1 CD3∊ chain (Figure 3). However, cell surface staining was detected on T116.4.3 and E6.1 cells with MAb SP-34 but not with MAb F7.2.38 (Figure 2B). Therefore, unlike MAb SP-34, this MAb is most likely directed against the cytoplasmic region of the CD3∊ chain.
Reactivity of MAb F7.2.38 on Paraffin-embedded Tissue Sections
Normal Human Tissues. In normal lymphoid tissue, MAb F7.2.38 showed strong staining of lymphoid cells in the medulla and cortex of normal thymus (Figure 4A). As expected, only scattered B-lymphocytes in the cortex and medulla were labeled with MAb CD20/L26 (Figure 4B). In human lymph node and tonsil, interfollicular areas containing the T-lymphocytes were strongly stained, well delimiting B-cell follicles in which some small reactive T-cells were labeled (Figure 4C). This reactivity was complementary with immunostaining of B-cell zones using MAb CD20/L26 in lymph node tissue (Figure 4D). With the exception of scattered T-cells, MAb F7.2.38 was unreactive with all other normal human tissues studied (data not shown).
Immunostaining on frozen and paraffin-embedded tissue sections was comparable, and therefore only paraffin sections were used in further experiments. Almost all results obtained with MAb F7.2.38 were comparable to the reference antibody, the rabbit polyclonal anti-CD3∊ antibody.
Neoplastic Human Tissues Lymphoid Tumors. The reactivity of MAb F7.2.38 with lymphoid tumors is shown in Table 1. All B-cell lymphomas were negative for F7.2.38 (0+/37), and only small reactive T-cells were strongly positive with this antibody. On the contrary, the majority of T-cell lymphomas [41+/52 (79%)] were positive for MAb F7.2.38 (Figure 4E–4H). As expected, anaplastic large-cell lymphomas (ALCLs) showed a lower rate of immunostaining with this antibody [6+/14 (43%)] in comparison with the other types of T-cell lymphomas [35+/38 (92%)] (Table 1) (Figure 4E–4G). In this group of T-cell lymphomas, only 6/14 cases studied showed a positive staining of a variable percentage of neoplastic cells for F7.2.38. The labeling intensity was also variable from case to case and overall was lower than that obtained in the other types of T-cell lymphomas. It is noteworthy that in 2/6 positive ALCLs, only rare neoplastic cells showed weak cytoplasmic staining with MAb F7.2.38. Of note was the reactivity of MAb F7.2.38 with nasal type extranodal NK/T-cell lymphoma. In this last category, 3/5 cases studied were positive for MAb F7.2.38 (Table 1) and virtually all neoplastic cells were labeled (data not shown). Lastly, among the 21 cases of Hodgkin's disease of different categories, only two cases (9.5%) showed focal cytoplasmic staining of a variable number of Hodgkin and Reed–Sternberg cells. In the other cases, neoplastic cells were clearly negative and were often surrounded by strongly labeled small T-lymphocytes.
Non-lymphoid Tumors. The 69 non-lymphoid tumors investigated were always negative for MAb F7.2.38, which stained only small reactive T-cells (data not shown). The same results were obtained with the polyclonal anti-CD3∊ antibody.
The F7.2.38 antibody worked well on paraffin sections stained either manually, or with either of two automated immunostainers (TechMate 500 and Ventana 320/ES).
Discussion
In this article we have described a novel MAb, F7.2.38, which recognizes the human CD3∊ chain, associated or not with the TCR/CD3 complex. This MAb can be used for all biochemical and immunohistochemical methods in T-cell receptor research and in routine clinical laboratory tests. Our results show that MAb F7.2.38 recognizes both monomeric and dimeric forms of the CD3∊ chain (see Figure 3). The latter form is dominant in the CD3∊ cDNA-transfected non-T-cells compared to normal T-lymphocytes, in which all components of the TCR/CD3 complex are present. These results are in agreement with those described by others (Pessano et al. 1985; Alarcon et al. 1991; Salmeron et al. 1991; Huppa and Ploegh 1997).
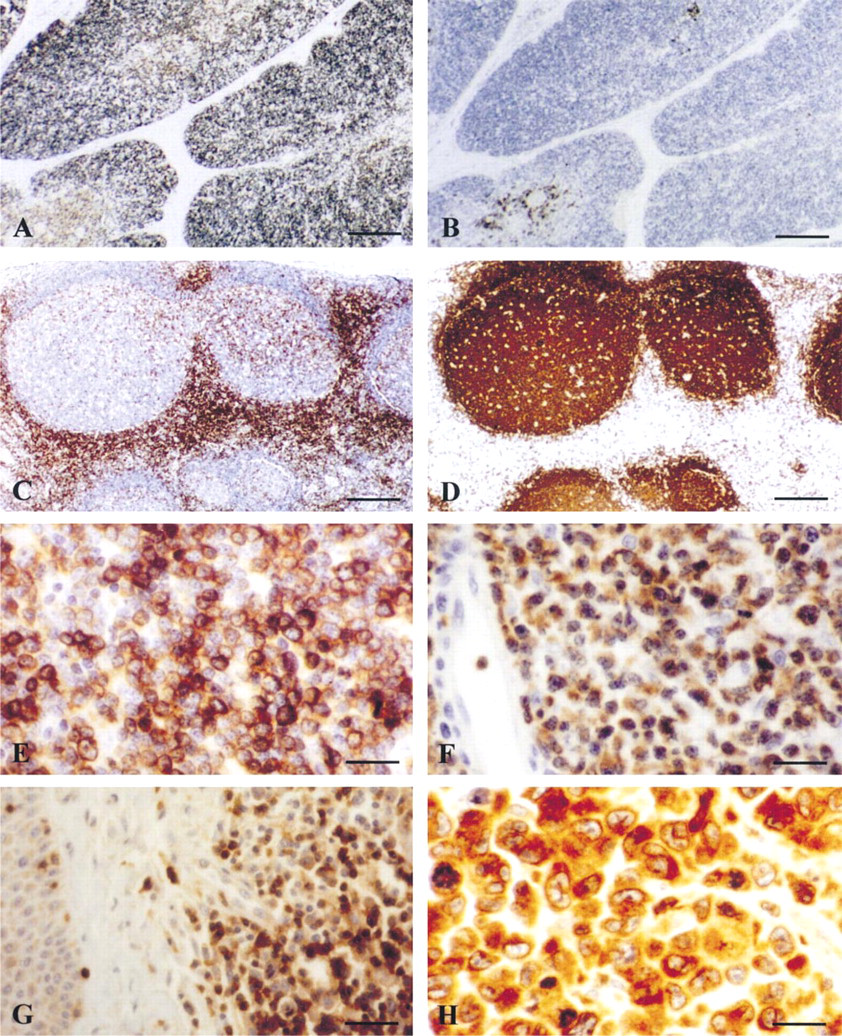
Reactivity of anti-CD∊ MAb F7.2.38 on paraffin tissue sections of normal and neoplastic human lymphoid tissues. (
The main result from this study is that MAb F7.2.38 is directed against a fixative-resistant epitope of the CD3∊ polypeptide. Therefore, it can be used to identify normal and neoplastic T-cells on paraffin-embedded tissue sections. The reactivity of the polyclonal anti-CD3∊ antibody on routinely processed biopsy specimen made it a valuable reagent for identification of T-cell lymphoid malignancies. The reactivity of MAb F7.2.38 was very similar to that of the routinely used polyclonal anti-CD3∊ antiserum. It is of interest that almost all T-cell neoplasms other than anaplastic large-cell lymphoma express CD3 antigen on the surface and/or in the cytoplasm, whereas B-cell lymphomas are invariably negative. As previously reported (Benharroch et al. 1998), ALCLs are positive for anti-CD3∊ antibody in approximately one third of cases. In addition, in this group of T-cell lymphomas the staining intensity and the percentage of positive cells are always lower than those observed in the other T-cell lymphoma subtypes. It is also of interest to note the cytoplasmic staining of three out of the five nasal type extranodal NK/T-cell lymphoma cases. As previously reported (Jaffe et al. 1996), in this category of T-cell lymphomas cytoplasmic CD3 is often detected using polyclonal antiserum to CD3∊ in paraffin sections despite the lack of CD3 detection on the cell membrane. This latter finding is likely consistent with the cytoplasmic expression of CD3∊ chain by the majority of NK-cells, from which these lymphomas originate (Lanier et al. 1992). Finally, in Hodgkin's disease, which is now considered to be a neoplasm of B-cell origin (Kanzler et al. 1996), the CD3∊ reactivity of Reed-Sternberg cells in some cases appears as a globular and not as a diffuse cytoplasmic staining, as previously reported (Cibull et al. 1989; Al Saati et al. 1997).
Until now, the polyclonal anti-CD3∊ has been the most commonly used antibody in immunohistochemistry on paraffin sections, giving specific and intense staining of reactive and neoplastic T-cells (Mason et al. 1989). It should be noted that, except for MAb F7.2.38, none of the available MAbs directed against the CD3∊ chain is suitable for use on routinely fixed and paraffin-embedded tissues. This is probably due to the fact that the vast majority of these MAbs (see Salmeron et al. 1991) are directed against conformational epitopes, especially in the extracellular domain of the CD3∊ chain, which could be easily destroyed by fixation of tissues. Interestingly, both the polyclonal anti-CD3∊ and the F7.2.38 mAb appear to recognize the cytoplasmic region of the CD3∊. This unique reactivity provides a valuable tool for functional and immunohistochemical studies. In the present study, we noted that MAb F7.2.38 worked as well as the polyclonal anti-CD3∊ in immunohistochemical methods, either manually or using automated immunostainers.
In summary, we have produced a novel MAb directed against the human CD3∊ protein, which would be very useful in studies on TCR/CD3 complex assembly as well as in the immunohistochemical diagnosis of T-cell lymphomas.
Footnotes
Acknowledgements
Supported by institutional grants from the CNRS, l'Université Paul Sabatier (UPS) Toulouse, and l'Association pour la Recherche contre le Cancer (ARC).
We thank Pr Bill Clark (UCLA) for valuable comments on the manuscript, Dr Balbino Alarcon for the kind gift of the vector pSRa-CD3∊del12, and Jeanine Boyes, Françoise Dupic, and Dr Andres Alcover for their excellent technical advice. We acknowledge the initial help of Cécile Gouaillard in this project.
