Abstract
Tissue transglutaminase (TGase C, TGase II) is known to participate in cellular processes during morphogenesis, differentiation, and development of various prenatal tissues and organs. The expression of TGase C during myoblast proliferation and attachment to external laminae was examined by immunohistochemical (IH) localization at 5–12 weeks of developmental stages of prenatal human muscle in 23 embryos. IH detection using a monospecific antibody to TGase C showed a prominent expression of TGase C in muscle cells as stage- and spatial-specific patterns during an early embryonal period. The myoblasts of intervertebral, tongue, and limb muscles, attached to adjacent cartilaginous skeletons or fibrous fascia, showed a pronounced expression of TGase C at 5–6, 6–7, and 7–8 weeks after fertilization, respectively. The most intense activity of TGase C was observed in some cardiac myoblasts infiltrating into endocardial mesenchyme at 6–7 weeks after fertilization. Although weak staining was detected until 14 weeks after fertilization, the level of TGase C expression in all muscles was significantly decreased after 6–7 weeks, with the exception that the smooth muscle cells of blood vessels and gastrointestinal tract showed diffusely intense staining of TGase C between 5 and 12 weeks after fertilization. Western blotting analysis of the cellular extracts of pooled samples showed a single strong band at 80 kD at 6 weeks after fertilization. This band became weaker after 8–10 weeks of prenatal development. These findings of transient expression of TGase C, which coincides with the development of myoblast anchoring and differentiation, suggest that TGase C plays a role in myoblast attachment to the extracellular laminae during the early embryonal period.
T
TGases (EC 2.3.2.13, protein-glutamine:amine γ-glutamyl transferase) are a group of Ca++ ion-dependent enzymes that catalyze crosslinking of protein molecules through the formation of Ne(γ-glutamyl)lysine bond(s) between two polypeptide chains (Chung and Folk 1972; Folk and Chung 1973; Chung 1975; Greenberg et al. 1991). The Ne(γ-glutamyl)lysine is a stable isopeptide and resistant to proteolysis, thereby increasing the resistance of tissue to chemical, enzymatic, and mechanical disruption. In mammals, five different TGases were cloned and characterized as follows: a membrane-associated TGase of 92 kD, TGase 1 (TGase-K), in human keratinocytes and rat chondrosarcoma cells (Chung 1975; Rice and Green 1977; Park et al. 1988; Kim et al. 1991, 1995a,b; Candi et al. 1995; Clement et al. 1997); a ubiquitous “soluble” or “tissue” 80-kD TGase, termed TGase C (TGase 2) (Ikura et al. 1988); a soluble proenzyme activity of 77 kD, “epidermal” TGase E (TGase 3) (Park et al. 1988; Kim et al. 1990, 1993, 1995a); a soluble enzyme extracellular secretory group, from rat coagulating gland of 65 kD, TGase P (Ho et al. 1992; Jung et al. 1992; Esposito et al. 1996); and a catalytic subunit of the blood clotting factor XIIIa of 77 kD (Gorman and Folk 1980; Aeschlimann et al. 1996; Lewis et al. 1997). TGase K and TGase E enzymes are believed to be involved in the formation and assembly of the cornified envelope of the skin and its appendages and of other stratified squamous epithelia (Gorman and Folk 1980; Kim et al. 1990, 1991; Park et al. 1991). A defect in TGase K is associated with an autosomal recessive skin disease, lamellar ichthyosis (Huber et al. 1995a,b, 1997; Choate et al. 1996, 1998; Laiho et al. 1997; Petit et al. 1997; Schorderet et al. 1997). However, TGase C may have special roles in cell adhesion and programmed cell death (Cocuzzi and Chung 1986; Fesus et al. 1987, 1989, 1996; Szende et al. 1991; Cummings 1996; Szondy et al. 1997; von Zezschwitz et al. 1997). The role of TGase in enzymatic crosslinking of the structural proteins may lie in providing extensibility of the cells and empowering their resistance to chemical and physical stimuli. The expression of the TGase enzyme has a strong influence on the fate of the cells (Mufti et al. 1991; Park et al. 1991; Moore and Sartorelli 1992; Demignot et al. 1995; Adel Moallem and Hales 1996; Katoh et al. 1996a,b; Nunes et al. 1997). A possible involvement of TGase C in organogenesis was suggested by retinoid-induced teratogenesis, which is accompanied by an increased expression of TGase C during organogenesis of the limb bud of mid-gestation mouse embryos (Jiang and Kochhar 1992). Annulin, a protein that has an amino acid sequence homologous to TGase, was found to be expressed at limb segment boundaries in the grasshopper embryo and has been suggested to play a role in organogenesis (Singer et al. 1992). A transient high expression of TGase C coincides with the development of tubulo–alveolar structures suggesting that TGase C plays a role in morphogenesis in developing human salivary gland (Lee et al. 1995).
In vertebrates, three types of muscles are distinguished on the basis of appearance and location of their constituent cells: smooth, skeletal, and cardiac. Development into each type of muscle may require different cytodifferentiation and investment in tissues and organs. In all cases, however, myoblast anchorage to external laminae is required when myoblasts start to migrate distally and to differentiate into elongated muscle cells. One possible candidate for such anchorage is the formation of stable crosslinks. In this study, we examined the stage- and spatial-specific expression of TGase C, an enzyme that catalyzes the isopeptide crosslink formation in human embryonal tissue sections at the muscle attachment sites on extracellular laminae when myoblasts start to migrate distally and to differentiate into elongated muscle cells during the early embryonal period.
Materials and Methods
Twenty-three normal human embryos ranging from 5 to 12 weeks old were obtained from the Repository of the Department of Pathology (College of Medicine, Seoul National University, Seoul, Korea). These materials were collected in accordance with National Health Policy and Human Tissue Repository Regulations of this institution. The embryos were confirmed as normal, and their fertilization ages were deduced from the crown–rump length or maternal records. Guinea pig liver TGase C was purified as described previously (Folk and Chung 1985). Rabbit antibody to guinea pig liver TGase C was prepared as described (Chung and Folk 1972; Chung 1975; Folk and Chung 1985).
Immunohistology
The specimens were fixed in Bouin's solution for 3–5 hr, embedded in paraffin, and sectioned at 4 μm. The paraffin sections were deparaffinized with xylene and treated with monospecific antibodies of TGase C (Park et al. 1988; Kim et al. 1990) by an indirect immunohistochemical method using a labeled streptavidin–biotin (K681; Dako, Glostrup, Denmark) staining technique (Lee et al. 1995). Pre-immune rabbit serum was utilized for negative control staining. The immunohistochemical methods are basically the same as described in the previous study (Lee et al. 1995).
The rabbit antibody to guinea pig liver TGase C crossreacted with human TGase C (RBC enzyme). The monospecific antibody of TGase C was prepared by affinity chromatography with immobilized guinea pig liver TGase C (Park et al. 1988; Kim et al. 1990). Two ml of rabbit antiserum was added to 1 ml of the immobilized guinea pig liver TGase C attached to agarose beads (Pierce; Rockford, IL) that had been equilibrated with PBS, pH 7.2. This mixture was gently agitated overnight at 4C and packed into a column (1 × 5 cm). After the column was washed with 0.5 M NaCl in PBS, the bound immunoglobulin was eluted with 0.1 M glycine-HCl buffer, pH 3.0. The eluate was immediately brought to neutral pH by addition of 2 M Tris solution. Bovine serum albumin was added to a 0.1% final concentrations, and the immunoglobulin was dialyzed against PBS and stored frozen at −70C.
Western Blotting Analysis
Three fresh-frozen fetal tissues from 6-, 8-, 10-week-old human embryos were pooled, homogenized in PBS (1:10 w/v), and centrifuged at 30,000 × g for 30 min; the supernatant solution was concentrated 20-fold. TGase C extracted from human RBCs and TGase P extracted from human prostate gland (Lee et al. 1995) were used as positive and negative controls, respectively. An aliquot was mixed with an equal volume of the treatment buffer (2% SDS and 0.05 M PBS) and boiled for 3 min in the presence of 10 mM mercaptoethanol. Samples (30 μg) were applied to a polyacrylamide gel. SDS-PAGE analysis was carried out in an 8 × 7.5-inch gel apparatus (Novex; San Diego, CA), with use of the discontinuous Tris/glycine buffer system of Laemmli (10% crosslinked gel). The separated proteins on polyacrylamide gel were transferred electrophoretically onto a nitrocellulose strip. The strip was blocked with skim milk in Tris-HCl buffer, washed with 0.05% Tween-20, and incubated overnight with monospecific anti-TGase C in 1% gelatin solution at 4C. The strip was treated with horseradish peroxidase-conjugated protein A (1:200 dilution; BioRad, Hercules, CA). The washed strip was developed for 2–15 min with 4-chloro-1-naphthol, hydrogen peroxide reagent (Lee et al. 1995).
Results
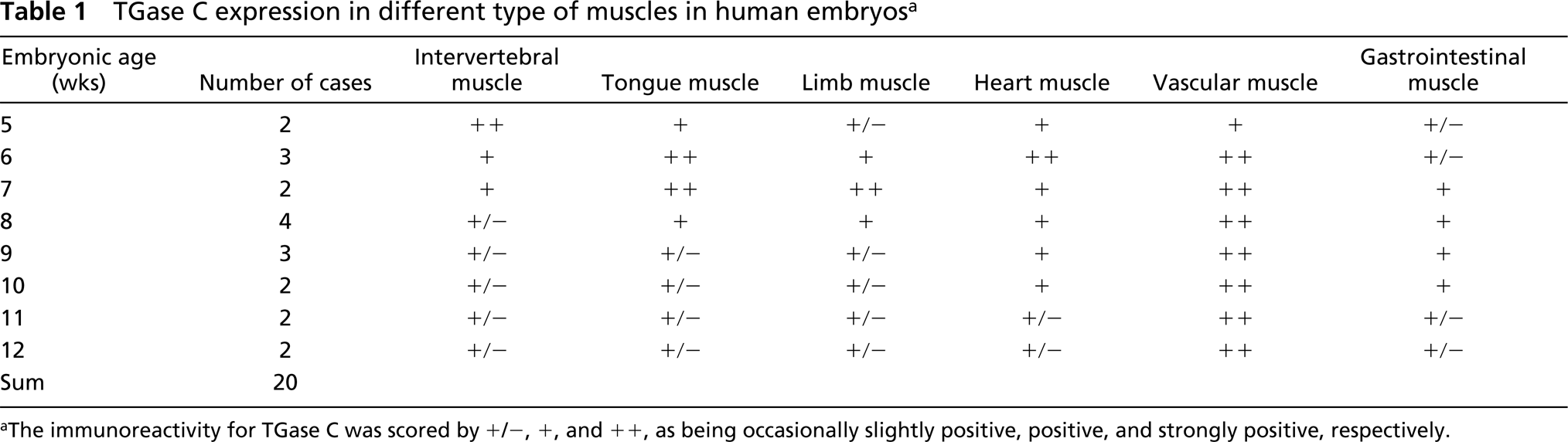
TGase C expression was detected in myoblasts of striated muscle, smooth muscle, and cardiac muscle during the early embryonal period. The expression level of TGase C in various tissues of the human embryo is shown in Table 1.
Striated Muscle
Intervertebral Muscles. Embryos between 5 and 6 weeks of age showed spindle-shaped myoblasts which were in a state of proliferation and migration towards condensed mesenchyme of vertebral columns. The cytoplasm of the myoblasts attached to the condensed mesenchyme of vertebral columns was intensely positive for TGase C antiserum (Figures 1e and 1f). As the intervertebral musculature matured, the positive reaction of TGase C decreased and became diffusely weak in whole intervertebral muscles until 12 weeks after fertilization (Figures 1g and 1h; Table 1).
Tongue and Masticatory Muscles. Five-week-old embryos showed tongue primordium in the stomodeal cavity. The tongue consisted of condensed mesenchymes in the internal and external lingual musculature and covering the stomodeal mucosa. As the lingual musculature developed at 6 weeks after fertilization, the immunostaining of TGase C was intensely positive in the cytoplasmic processes of myoblasts that were attached to the fibrous fascia of the lingual septum and intermuscular space (Figures 1a–1e). This strong positive reaction decreased rapidly at 7 weeks after fertilization. The TGase C was diffusely weak in the tongue musculature until 12 weeks after fertilization (Table 1).
Limb Muscles. Six-week-old embryos showed prominent limb buds filled with condensed mesenchyme of muscle and cartilage. From 7 weeks after fertilization, the myoblasts were differentiated and aggregated around the perichondral mesenchyme of limb cartilage. Some myoblasts attached to the perichondral mesenchyme showed a strongly positive reaction of TGase C at 7–8 weeks after fertilization, after which the immunostaining of TGase C rapidly decreased. Then the immunostaining of TGase C became diffusely weak in the whole muscle bundles of the limb musculature until 12 weeks after fertilization (Table 1).
Cardiac Muscle
Six-week-old embryos showed a weak reaction to TGase C in cardiac myoblasts. Some cardiac myoblasts infiltrating into the endocardial mesenchyme of valvular structures and into Purkinje fibers showed an intense reaction for TGase C in their cytoplasmic processes (Figures 2a–2c). This intense reaction was also found in cardiac myoblasts at 7 weeks after fertilization (Figures 2d and 2e). Thereafter, it was reduced rapidly and remained diffusely weak in the whole cardiac myoblasts of ventricle and atrium until 12 weeks after fertilization (Table 1).
Smooth Muscle
Vascular Wall. The immunostaining of TGase C was weakly positive in the vascular wall from 5 weeks after fertilization, and it gradually increased and was intensely localized in the smooth muscle of vascular vessels, including capillaries, arteries, veins (Figures 3a–3d), and lymphatics. As the perivascular smooth muscle increased in thickness, the immunostaining of TGase C became intensely positive in the whole muscle layer until 12 weeks after fertilization (Table 1).
TGase C expression in different type of muscles in human embryos a
aThe immunoreactivity for TGase C was scored by +/–, +, and + +, as being occasionally slightly positive, positive, and strongly positive, respectively.
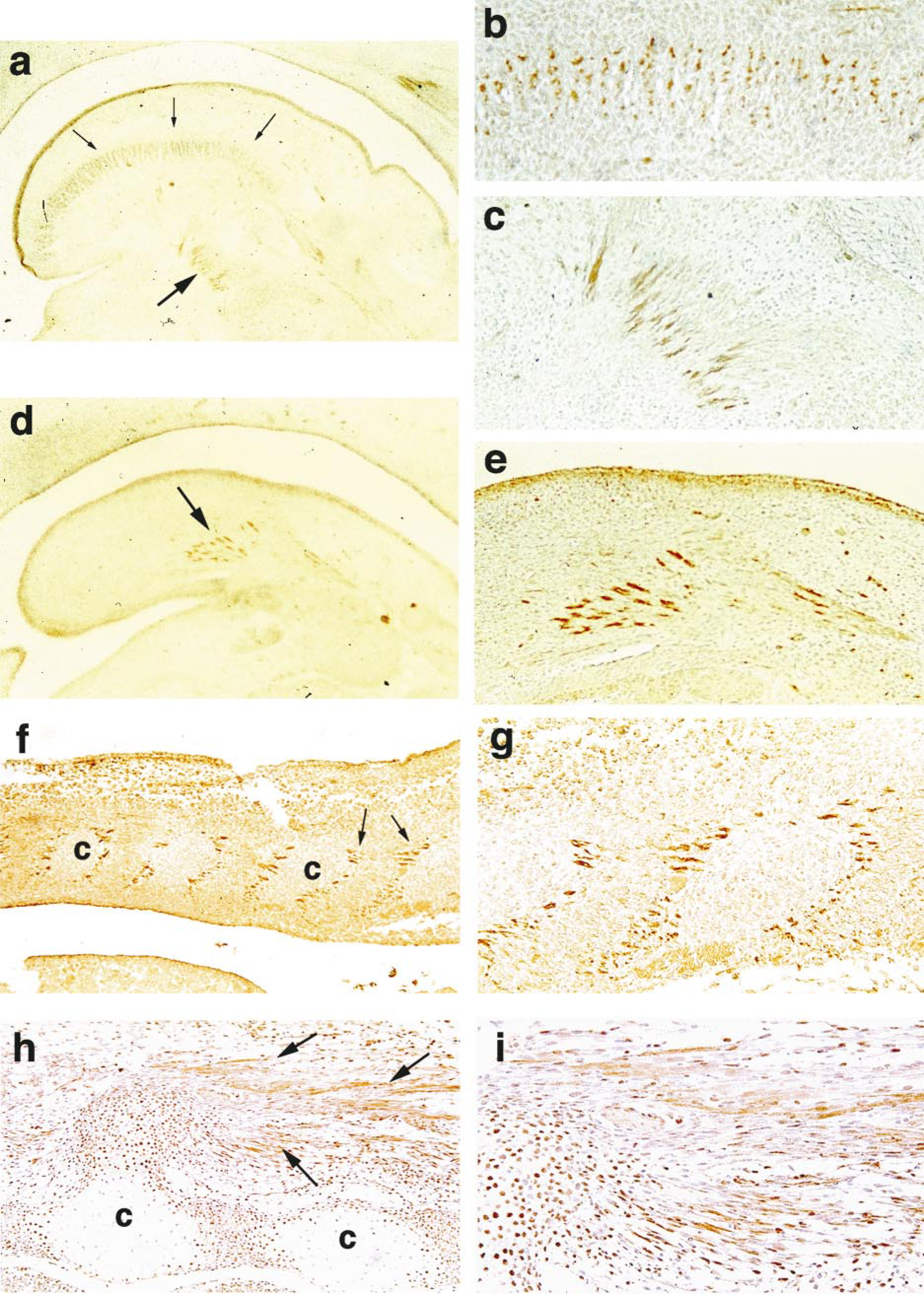
Immunostaining of TGase-C in tongue muscles of 6-week-old human embryo. (
Gastrointestinal Tract. From 6 weeks after fertilization, the mesodermal condensation of the smooth muscle of the gastrointestinal tract increased and the myoblasts became weakly positive for TGase C (Figure 3e). As the gastrointestinal tract became thickened, the cytoplasm of the myoblasts was intensely positive for TGase C (Figures 3f and 3g). The immunostaining for TGase C remained positive in the smooth muscle cells of the gastrointestinal tract until 12 weeks after fertilization (Table 1).
Western Blotting Analysis
To confirm the immunoreactivity for TGase C observed in the histologic sections, we used fresh-frozen samples to extract the soluble TGase C and to compare, by Western blotting analysis, the expression of intact TGase C at various embryonic ages. Because the designated muscles from the embryos (5–10 weeks after fertilization) were too difficult to dissect free from other tissues, we utilized the large portion of embryos containing muscle tissues as a source for TGase C. The TGase C from RBCs was used as a positive control, and TGase P from rat prostate gland was used as a neagtive control. As shown in Figure 4, a single band at 80 kD (p80) was seen in all three extracts. The relative levels of TGase C detected by the immunoblotting method grossly coincided with the immunohistochemical scoring presented in Table 1. Protein extracts from 6-, 8-, and 10-week-old human embryonal tissues showed a conspicuous band corresponding to 80 kD, which is identical to the molecular mass of human RBC TGase C. However, these Western blotting data for TGase C expression (Figure 4) do not necessarily indicate TGase C expression solely on muscle tissues, but rather (a) show the specificity of a monospecific antibody against TGase C on the extracted TGase from the embryos and (b) confirm the results of immunohistochemical staining.
Discussion
Tissue transglutaminase (TGase C) is a cytoplasmic enzyme present in many cell types, including those in the blood vessel wall. Unlike other TGases (K, E, and P), which are found in inactive zymogen, TGase C is in an active state. The finding of TGase C expression in specific cells at certain stages of human muscle development is novel and may provide an insight into TGase-dependent cellular processes in organogenesis (Greenberg et al. 1991; Lee et al. 1995). Although TGase C is essentially a cytosolic enzyme, there is increasing evidence to suggest that the enzyme can be externalized and plays a role in extracellular matrix organization. For example, TGase C reacts not only with cytoplasmic actin but also with fibronectin, an extracellular matrix protein (Bowness and Tarr 1997; Jones et al. 1997; Nemes et al. 1997). It is also suggested that the TGase C activity is related to the apoptotic activity of various tissues (Fesus et al. 1987; Fesus and Thomazy 1988; Wang and Johnson 1992; Aeschlimann et al. 1993; Amendola et al. 1996; Cummings 1996; Fesus et al. 1996; Zhang et al. 1996; Kashima et al. 1997; Melino et al. 1997; Szondy et al. 1997; von Zezschwitz et al. 1997), polyamine-crosslinked products for a protective barrier (Bradway et al. 1990; Cooper et al. 1997; Jurgensen et al. 1997; Whyard and Ablin 1997), and wound healing processes (Raghunath et al. 1996). TGase C is present in minute amounts in cells from muscle, bone and cartilaginous tissues (Aeschlimann et al. 1993, 1996; Hand et al. 1993; Kojima et al. 1993; Sorensen and Petersen 1995; Jurgensen et al. 1997). In this study, we observed that the immunostaining of TGase C was spatial- and stage-specific in myoblasts of striated and smooth muscles. The TGase C expression was quite intense in the striated myoblasts, which produce cytoplasmic processes for attachment to the skeletal tissues of the vertebral column or the lingual septae at 5–8 weeks after fertilization. Subsequently, the TGase C expression, as indicated by immunostaining in myoblasts, was rapidly reduced, and then became diffusely weak in the whole muscle cells. However, in this study we observed the intense positive reaction of TGase C in the cytoplasm of myoblasts undergoing histogenetic processes by attaching to the adjacent bony tissues. These findings are indeed suggestive of active TGase C involvement in the production of a stable matrix protein complex, a stress-resistant bonding between muscle and bony tissues. This phenomenon may be related to the histomorphogenesis of skeletal muscle during the early embryonal period.
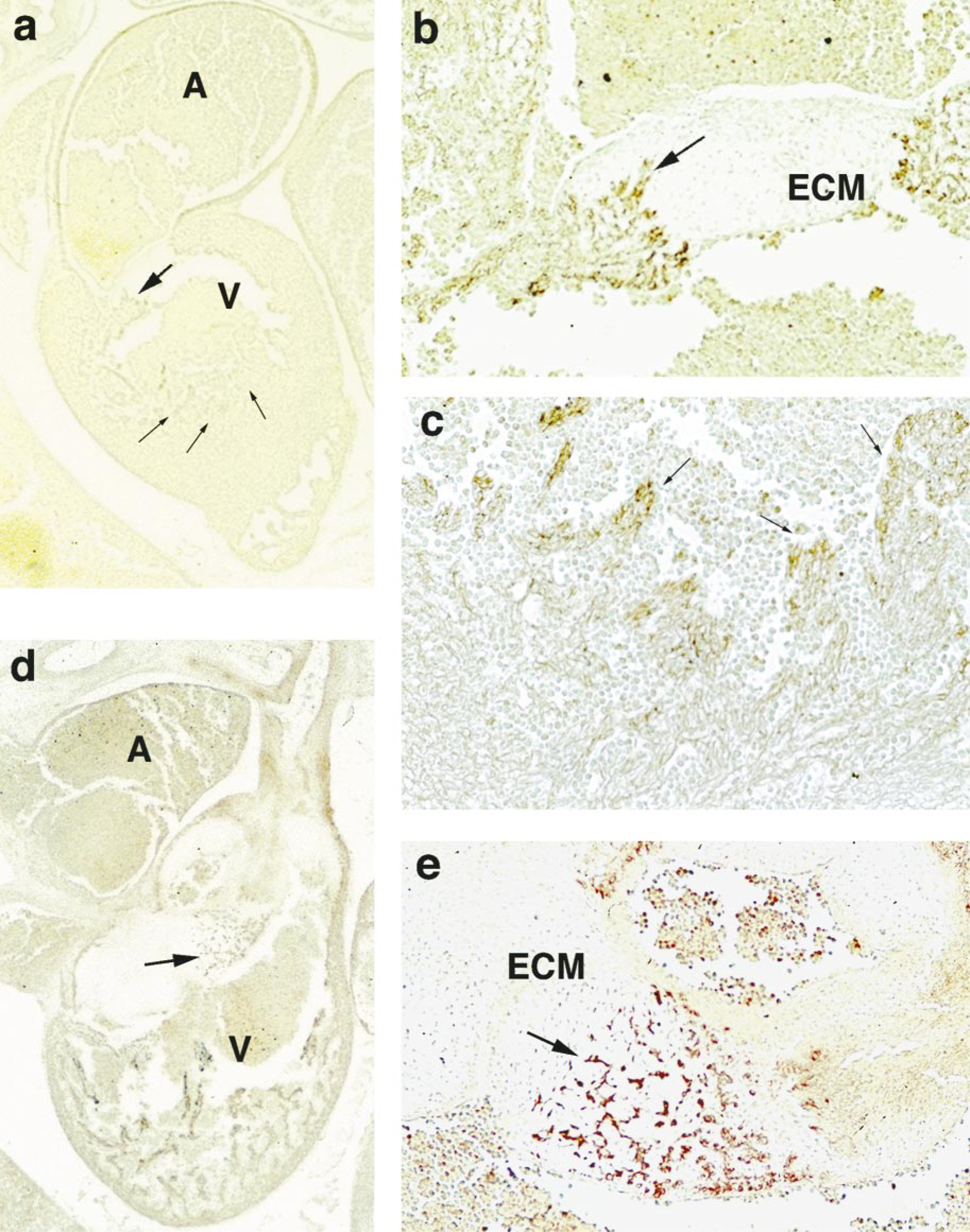
Immunostaining of TGase C in heart muscles, methyl green counterstaining. (
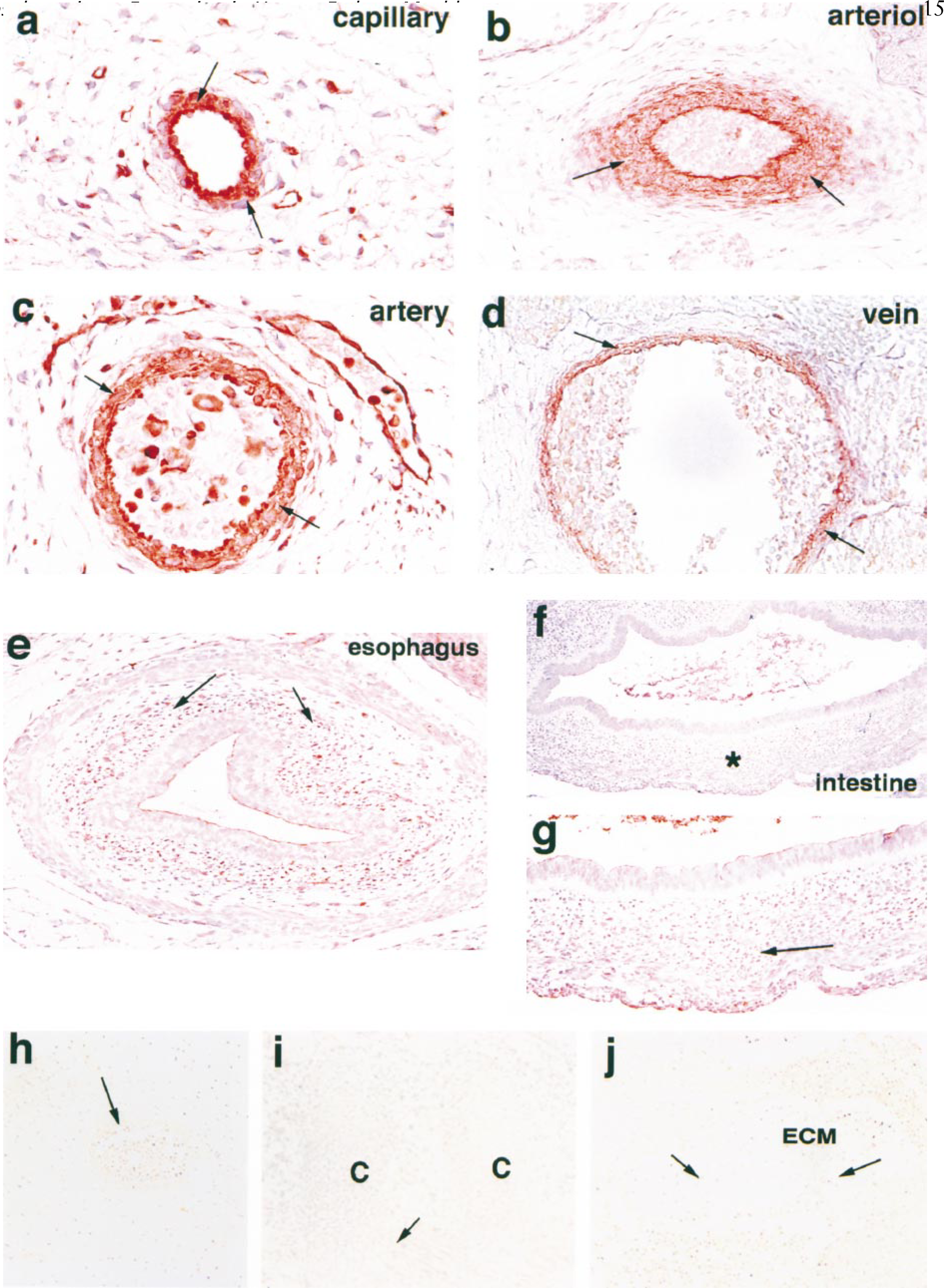
Immunostaining of TGase C in blood vessel wall, hematoxylin counterstaining. Intense positive reactions (arrows) are observed in the capillary (
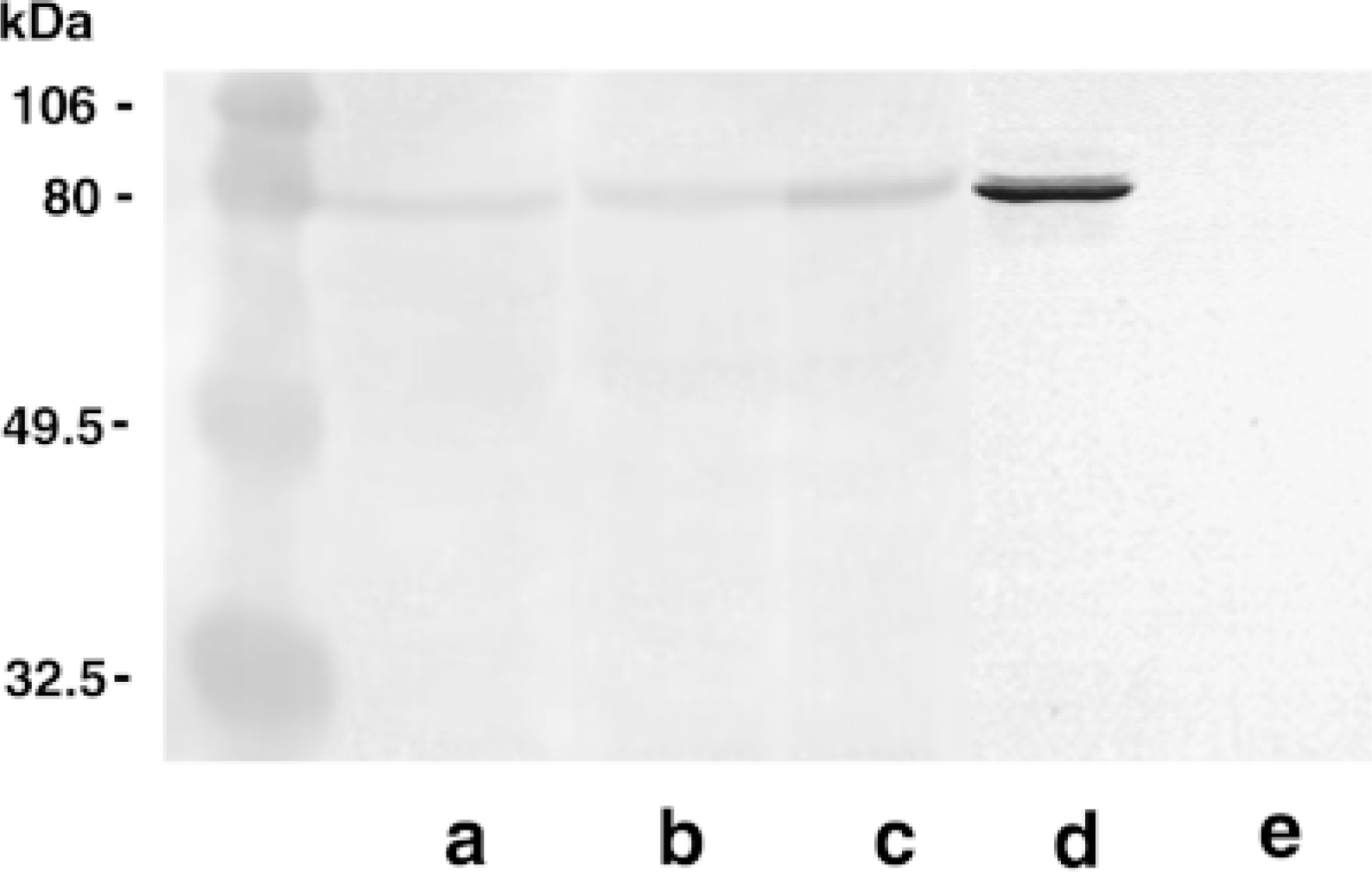
Western blotting analysis of 6-, 8-, and 10-week-old human embryo tissue extracts. Lane a, 6-week-old human embryo; Lane b, 8-week-old human embryo; Lane c, 10-week-old human embryo; Lane d, human RBC TGase C; Lane e, rat prostate TGase (TGase P). The pre-stained SDS-PAGE standards from Bio-Rad (Hercules, CA) are used as reference molecular masses: 106 kD phosphorylase B; 80 kD BSA; 49.5 kD ovalbumin; and 32.5 kD carbonic anhydrase.
A similar immune-positive reaction to TGase C was observed in cardiac myoblasts infiltrating into the endocardial mesenchyme and Purkinje fibers at 6–8 weeks after fertilization. The cardiac myoblasts were strongly anchored to the valvular structures derived from endocardial mesenchyme and also to the Purkinje fibers containing Purkinje cells, which function as a rapid conduction system for cardiac impulses. Because the arrangement of cardiac muscles is important for cardiac morphogenesis, TGase C may function here as a catalyst to bond cardiac myoblasts and adjacent functional elements in the embryonal heart.
The smooth muscles encircling the vascular wall, as well as endothelial cells of capillaries, showed a positive reaction at 5 weeks after fertilization. As the walls of blood vessels became thickened and arteriovenous structures developed, TGase C was intensely expressed in the encircling smooth muscles of arteries and veins at 6–10 weeks after fertilization. The positive reaction slightly decreased during 10–12 weeks of fertilization age, and also showed focally weak positivity at the periphery of thick arterial and venous walls.
Although the immunoreaction to TGase C was detected at 12 weeks after fertilization in this study, TGase C expression in vascular structures of the adult, especially in the endothelial cells, was reported earlier. This novel finding of transient expression of TGase C in the development of vascular smooth muscle again coincides with the morphogenetic role of TGase C proposed earlier (Lee et al. 1995, 1996). In this case, the smooth muscle myoblasts of blood vessels use TGase C to build up a tight vascular wall.
The smooth muscles of the gastrointestinal tract also showed a conspicuous positive reaction for TGase C. Usually the mesenchymal condensation appears around the gut mucosa at 6 weeks after fertilization. The circular and longitudinal smooth muscle layers are visible at 8 weeks after fertilization. The immunostaining of TGase C was most intense during the developing stage of outer layers of smooth muscle through the gastrointestinal tract. We observed the highest expression of TGase C in the myoblasts of gastrointestinal smooth muscles at 8–10 weeks after fertilization. The positive reaction was gradually reduced until 12 weeks after fertilization. In the developing gastrointestinal tract, TGase C may be utilized by smooth muscle myoblasts to build up circular and longitudinal muscle structures.
In this study, we observed the timely expression of TGase C in human embryonic tissues by a immunohistochemical method. The monospecific antibody for TGase C showed a clearly positive reaction in comparison with the pre-immune rabbit serum. Western blotting with the same TGase C antibody also showed a conspicuous band, 80 kD, from the proteins extracted from human embryonal tissues. The relative levels of TGase C detected by the immunoblotting method coincide with the immunohistochemical scoring (Table 1) in the embryonal myoblasts through the major developmental stages, i.e., from 5 to 12 weeks after fertilization. We have observed that there appears to be a stage- and spatial-specific positive reaction of TGase C associated with the development of striated, cardiac, and smooth muscle in each organ. The negative control staining using pre-immune rabbit serum in the same immunohistochemical procedure was negative. In conclusion, the embryonal myoblasts, which undergo histomorphogenetic processes at 5–8 weeks after fertilization in the human embryo, show an intense transient expression of TGase C in spatial- and stage-specific patterns. These data suggest that TGase C, a major crosslinking enzyme, is utilized by the myoblasts to form stress-resistant structures in striated, cardiac, and smooth muscle.
Footnotes
Acknowledgements
Supported by project HMP-98-M-4–0048 of the Korean Ministry of Health and Welfare.
