Abstract
CFTR (cystic fibrosis transmembrane conductance regulator), MDR1 (multidrug resistance), and MRP1 (multidrug resistance-associated protein), members of the ABC transporter superfamily, possess multiple functions, particularly Cl–, anion, and glutathione conjugate transport and cell detoxification. They are also hypothesized to have a number of complementary functions. It is generally accepted that data obtained from nasal mucosa can be extrapolated to lower airway cell physiology. The aim of the present study was to investigate by immunohistochemistry the differential localization of CFTR, MDR1, and MRP1 in the normal mucosa of 10 human nasal turbinates. In ciliated epithelial cells, CFTR was inconstantly expressed at the apical cell surface, intense membranous labeling was observed for MDR1, and intense cytoplasmic labeling was observed for MRP1. In the glands, a higher level of expression was observed on serous cells, at the apical surface (for CFTR), on lateral membranes (for MDR1), and with an intracytoplasmic distribution (for MRP1). In conclusion, CFTR, MDR1 and MRP1 are expressed in the epithelium and glands of the nasal respiratory mucosa, but with different patterns of expression. These results suggest major roles for CFTR, MDR1, and MRP1 in serous glandular cells and a protective function for MDR1 and MRP1 in respiratory ciliated cells.
CFTR (cystic fibrosis transmembrane conductance regulator), MDR1 (multidrug resistance), and MRP1 (multidrug resistance-associated protein) are members of the ABC (ATP binding cassette) transporter superfamily, which use the energy of ATP hydrolysis to regulate or energize the transport of molecules as diverse as chloride, large organic anions, xenobiotics, or glutathione conjugates (Riordan et al. 1989; Anderson et al. 1991; Cole et al. 1992; Gottesman and Pastan 1993; Loe et al. 1996; Linsdell and Hanrahan 1998a,b; Schwiebert et al. 1999).
CFTR, the protein impaired in cystic fibrosis (CF), an inherited disease particularly affecting the conductive airways, is a chloride channel. MDR1 and MRP1 play a role in cell detoxification and multidrug resistance, and MRP1 is involved in glutathione conjugate transport. However, CFTR, MDR1, and MRP1 could share certain functions, such as control of heterologous chloride channels for CFTR and MDR1 (Hardy et al. 1995; Jilling and Kirk 1997) and transport of glutathione and glutathione conjugates or large organic anions for CFTR and MRP1 (Leier et al. 1994; Jedlitschky et al. 1996; Linsdell and Hanrahan 1998b). Moreover, in the light of certain clinical data obtained in CF patients, it has been hypothesized that a deficient CFTR protein could be complemented by other ABC proteins, such as MDR1 and/or MRP1 (Lallemand et al. 1997; Altschuler 1998). Therapeutic trials will be conducted to validate this hypothesis, and the particularly accessible nasal mucosa has been chosen to evaluate the respective expression of cftr, mdr1, and mrp1 genes (Lallemand et al. 1997). These three ABC proteins have been localized in the respiratory mucosa of bronchial airways (Engelhardt et al. 1992; Brézillon et al. 1997a; Lechapt–Zalcman et al. 1997; Bréchot et al. 1998), whereas only the epithelial localization of CFTR has been studied in the nasal respiratory mucosa, and most of these studies were performed on tissues from nasal polyps rather than on normal nasal mucosa, or on specimens collected from patients with chronic sinusitis (Puchelle et al. 1992; Brézillon et al. 1995, 1997b; Dupuit et al. 1995; Coltrera et al. 1999).
The respiratory mucosa in nasal cavities and cartilaginous bronchial airways has a similar structure, consisting of respiratory pseudostratified epithelium with ciliated, mucus-secreting, and basal cells associated with glands, mainly composed of serous and mucus-secreting cells. It has therefore been widely accepted that respiratory cell biology data obtained from investigations performed on nasal mucosa can be extrapolated to lower airway cell physiology (Knowles et al. 1981; Verra et al. 1993). For example, most therapeutic trials in CF, including gene therapy, have been conducted on nasal mucosa (Crystal et al. 1994; Knowles et al. 1995; Rubenstein and Zeitlin 1998). The potential nasal response is also a standard for evaluation of epithelial CFTR function either before or after topical or systemic treatment (Knowles et al. 1981, 1995; Rubenstein and Zeitlin 1998).
This study was therefore designed to precisely determine, by immunohistochemistry, the differential localization of CFTR, MDR1, and MRP1 transmembrane proteins in respiratory epithelium and in the glands of normal, non-polypous respiratory mucosa from human nasal turbinates.
Materials and Methods
Nasal Mucosal Samples
Mucosal samples from the respiratory segment of the nasal cavity were obtained within 30 min of resection from nasal turbinates collected from patients who underwent surgery in the Oto-Rhino-Laryngology Department of Hôpital Tenon (Paris, France). The 10 patients included in the present study (seven men and three women) underwent turbinectomy for discomfort and nasal obstruction. The patients ranged in age from 25 to 73 years (mean 45 years). Five patients were current smokers and five were non-smokers. None of the patients presented with cancer and none had received prior chemotherapy or radiotherapy. This study was conducted in accordance with the rules of the National Recommendations for Medical Research.
Fresh respiratory nasal mucosal samples were immediately fixed for 24 hr in 80% ethanol at 4C and paraffin-embedded as previously reported (Lechapt–Zalcman et al. 1997). For each specimen, a 4-μm section was stained with hematoxylin–eosin for light microscopic examination.
Immunohistochemical Detection of CFTR, MDR1, and MRP1 Proteins
Antibodies. Various specific monoclonal antibodies (MAbs) were used for immunodetection of CFTR, MDR1 and MRP1. MAb 24–1 (R&D Systems Europe; Abingdon, UK) is a mouse IgG2a directed against four C-terminal amino acids (1477–1480) of the CFTR protein (working dilution 1:200; 1 μg/ml) (Denning et al. 1992; Marshall et al. 1994; Brézillon et al. 1997b). MAb C494 (Dako; Glostrup, Denmark), a mouse IgG2a antibody, reacts with an internal epitope localized on the C-terminal domain of the human MDR1 (working dilution 1:20; 3.3 μg/ml) (Georges et al. 1990). MAb MRPr1 (Valbiotech; Paris, France) is a rat IgG2a antibody that reacts with an internal epitope of the human MRP1 protein (working dilution 1:20; 1 μg/ml) (Flens et al. 1994). All three antibodies have previously been used for immunohistochemical studies (Georges et al. 1990; Flens et al. 1994, 1996; Dupuit et al. 1995; Brézillon et al. 1997b; Chan et al. 1997; Lechapt–Zalcman et al. 1997; Bréchot et al. 1998).
Negative controls were performed by replacing the primary antibody with an immunoglobulin of the same origin and isotype, i.e., a mouse IgG2a (Dako) for C494 and 24–1 and a rat IgG2a for MRPr1(Immunotech; Marseille, France) at the same protein concentration.
Immunohistochemical Procedure. Four-micrometer paraffin sections were placed on slides pretreated with silane (3-amino-propyltriethoxysilane; Sigma Chemical, St Louis, MO) and kept overnight at 37C. The sections were dewaxed with toluene and rehydrated with distilled water through a series of alcohol solutions. Immunohistochemical visualization was performed using the sensitive Dako Envision™+ system (Sabattini et al. 1998) based on an HRP (horseradish peroxidase)-labeled polymer conjugated with secondary antibodies. Preliminary experiments showed that microwave treatment (three times for 3 min in 0.1 M citrate buffer, pH 6, 560 W) was necessary to unmask epitopes for the three proteins studied. Endogenous peroxidase activity was blocked by 20-min incubation in 0.3% H2O2 in methanol at room temperature (RT).
The sections were then rinsed in 1 × TBS–0.1% Tween-20 (TBST; Sigma) and incubated with blocking serum containing 30% normal human AB serum (Sanofi Diagnostics Pasteur; Marnes–la–Coquette, France) in TBST for 1 hr at RT, followed by 1-hr incubation with the primary MAbs diluted in 3% TBST–bovine serum albumin (Sigma)–10% normal human AB serum for 1 hr in a moist chamber at RT according to the different working solutions.
For C494 and 24–1, a secondary labeled polymer anti-mouse antibody (Envision™+; Dako) was applied for 30 min at RT.
For the MRPr1 MAb, a second rabbit anti-rat antibody (Dako) was applied for 30 min at RT, followed by incubation with the labeled polymer anti-rabbit antibody.
The sections were then exposed to a working solution containing the diaminobenzidine chromogen (Envision™+; Dako) for 5 min at RT according to the manufacturer's recommendations. Between each step, sections were rinsed three times for 5 min in TBST. The slides were counter-stained with hematoxylin (Sigma) and mounted in Eukitt (Labonord; Villeneuve d'Ascq, France).
Results
Histological examination of all samples did not reveal any inflammatory lesions or neoplastic changes. Normal pseudostratified surface epithelium, composed of ciliated cells, mucus-secreting cells, and one layer of basal cells in three samples, or with slight basal cell hyperplasia in the seven other samples, was observed (Brézillon et al. 1995; Dupuit et al. 1995).
Glands were a predominant feature of the nasal respiratory mucosa in all samples studied. They were composed of mucous, serous, and mixed secretory parts and rather short collecting ducts opening into the superficial respiratory epithelium layer.
Respiratory Epithelium
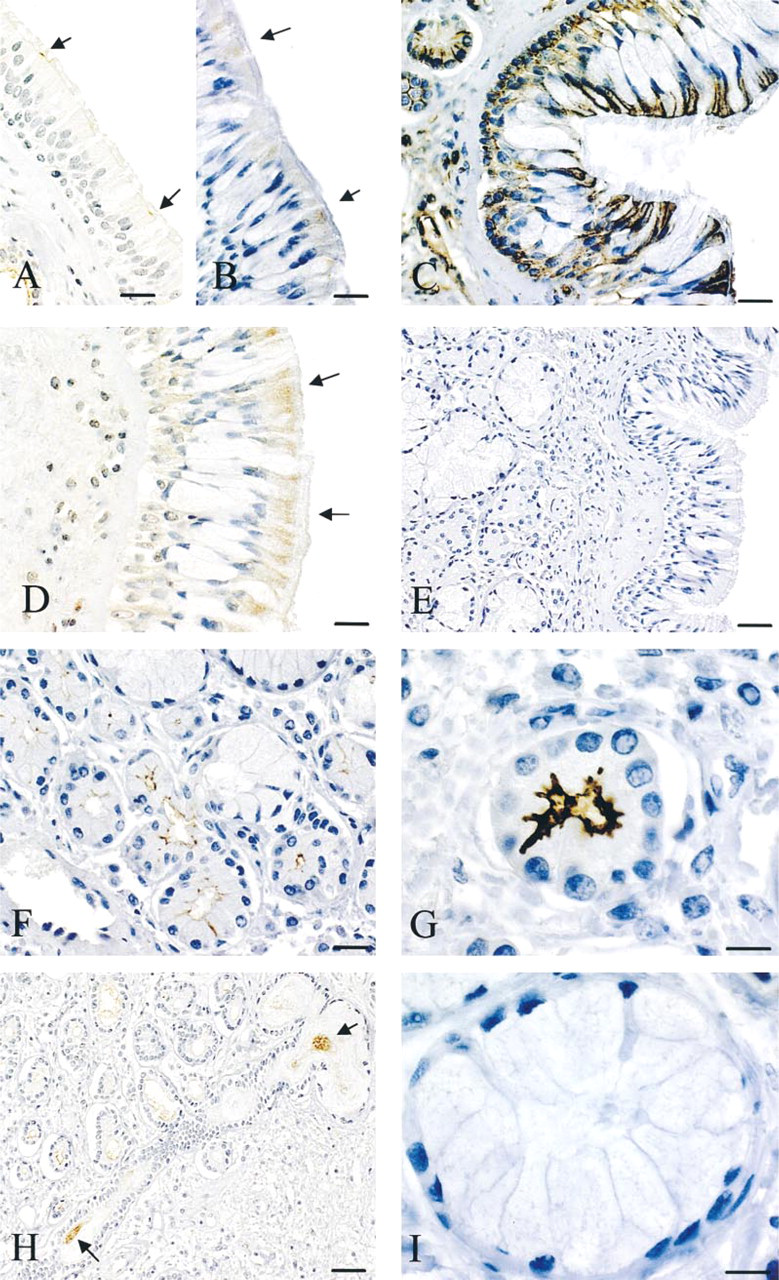
CFTR. After incubation with the anti-human CFTR MAb 24–1, epithelial labeling was always much weaker than that observed on glands, and heterogeneous results were obtained. Focal staining of the apical cell surface of ciliated cells was observed in 4/10 samples (Figure 1A). Isolated weak intracytoplasmic labeling of the supranuclear region of ciliated cells was detected in 4/10 samples and was associated with apical cell membrane labeling in 2/10 samples (Figure 1B). No staining was observed in basal or mucus-secreting cells (Figures 1A and 1B).
MDR1. In contrast to the results obtained for CFTR, intense staining with a homogeneous pattern was observed on all samples after incubation with the anti-human MDR1 MAb C494 (Figure 1C). Dense apical and basolateral staining of the plasma membrane of ciliated cells was consistently observed and was consistently associated with intracytoplasmic staining. Labeling of the cell circumference of basal cells was observed except at points of contact with the basal lamina. No staining was detected over mucus-secreting cells.
MRP1. Homogeneous labeling, different from that described above for MDR1, was observed after incubation with the anti-human MRPr1 MAb. Granular cytoplasmic staining was observed in the apical part of all ciliated cells (Figure 1D). Weak labeling was detected in the cytoplasm of basal cells. No labeling was observed over mucus-secreting cells.
Glands
CFTR. A consistent labeling pattern was observed in glands after incubation with the anti-human CFTR MAb, in contrast to that observed in the respiratory epithelium. Very dense staining was observed over all serous cells (Figures 1F–1H), with intense labeling of the apical membrane and upper part of the intercellular space with invaginations (Figure 1G). Staining was also observed on the apical surface of ciliated cells and inside the lumen of collecting ducts (Figure 1H). No staining was detected over mucous cells, and only serous cells were labeled in mixed glands (Figure 1I).
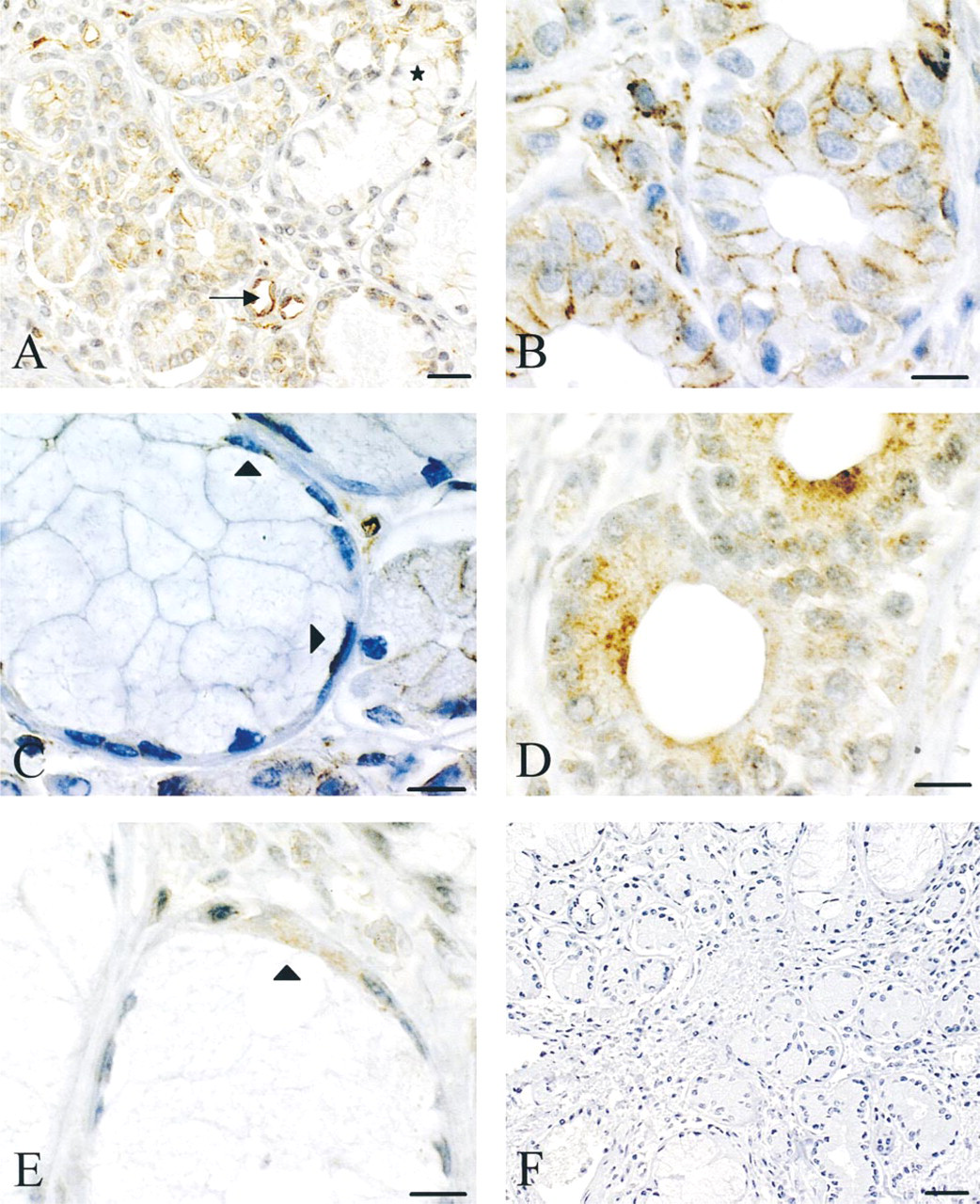
MDR1. After incubation with the C494 anti-human MDR1 MAb, a consistent labeling pattern was observed. The cell membrane limiting the intercellular space between serous cells was always intensely stained (Figures 2A and 2B), but labeling of the apical membrane was rarely observed (one sample of the 10 tested). Discrete granular cytoplasmic staining was consistently associated (Figure 2B). Lateral labeling of mucus-secreting cells was very rare (Figure 2A) and the great majority of these cells were negative, whereas myoepithelial cells were consistently stained (Figure 2C). Staining of collecting ducts was similar to that observed on serous acini. Dense staining of lymphocytes and the luminal side of endothelial cells was consistently observed (Figure 2A).
MRP1. After incubation with the MRPr1 anti-human MRP1 antibody, dense intracellular granular labeling was consistently detected in all serous cells (Figure 2D). This staining was strongly reinforced in the apical part of the cell. In contrast, no peripheral staining of the cell membrane was present. Mucus-secreting cells were negative, whereas myoepithelial cells in contact with mucus-secreting cells displayed intense cytoplasmic immunoreactivity (Figure 2E). Cells of collecting ducts were negative. Lymphocytes and endothelial cells showed weak intracytoplasmic labeling.
Controls
Staining with control isotype-matched irrelevant antibodies was negative for all samples, both for respiratory epithelium (Figure 1E) and for glands (Figure 2F).
Discussion
This study shows that immunohistochemistry clearly localizes CFTR, MDR1, and MRP1 in normal human nasal mucosa. In all samples studied, the respiratory epithelial layer had either a normal morphological appearance or exhibited only slight basal cell hyperplasia (Brézillon et al. 1995; Dupuit et al. 1995) with no inflammatory reaction.
CFTR was inconsistently detected in ciliated cells of the respiratory epithelium, either on the apical surface or with an intracytoplasmic distribution. Previous studies either failed to demonstrate the presence of CFTR in these cells (Crawford et al. 1991; Engelhardt et al. 1992) or, in contrast, showed the presence of CFTR at their apical membranes (Puchelle et al. 1992; Brézillon et al. 1995, 1997a,b; Dupuit et al. 1995). These discrepancies could be due either to the different techniques used, although the present study consistently and clearly identified the CFTR protein in serous glands, or to the fact that clear immunodetection of CFTR was reported on human nasal polyp tissue sections (Puchelle et al. 1992; Brézillon et al. 1995; Dupuit et al. 1995), nasal polyp explants (Brézillon et al. 1997b), samples collected from patients with chronic sinusitis (Coltrera et al. 1999), or bronchial biopsy specimens from lung transplant recipients (Brézillon et al. 1997a), which cannot be considered strictly “normal” tissues.
Glandular structures of the nasal mucosa are composed of mucous, serous, and mixed secretory units and myoepithelial cells (Stevens and Lowe 1997). CFTR was intensely expressed at the apical membrane of serous cells but was not detected in mucus-secreting cells, and this result is in accordance with previous data reported for bronchial mucosal glands (Engelhardt et al. 1992). The observed labeling extended from the upper part of the intercellular space into the cell. This localization could correspond to intercellular canaliculi indenting the cell membrane, producing the crypt-like extensions previously observed by electron microscopy in serous cells from bronchial glands (Meyrick and Reid 1970). The particularly marked intraluminal labeling observed on collecting duct sections could be due to the presence of CFTR in secretions, which has not been previously reported. The hypothesis of CFTR secretion is justified, because human CFTR has been shown to be secreted into the milk of transgenic animals by apocrine mechanisms (DiTullio et al. 1992) and apocrine secretions have been described in serous cells of salivary glands (Deyrup–Olsen and Luchtel 1998), which are closely related to nasal and bronchial glands. However, this hypothesis needs to be precisely investigated. Localization of CFTR at the apical cell membrane of serous cells is consistent with the recognized role of these glandular cells in the control of the surface fluid composition of the respiratory mucosa, because these cells, evaluated from human airway glands, have a wide range of functions, such as chloride secretion (Finkbeiner et al. 1994), and it is generally accepted that the fluid component of glandular secretions is predominantly provided by serous cells (Finkbeiner et al. 1994).
We (Lechapt–Zalcman et al. 1997), like others (Cordon–Cardo et al. 1990), have reported localization of MDR1 in bronchial respiratory epithelium, showing restriction of the protein distribution to the apical surface. With a more sensitive immunodetection method (Sabattini et al. 1998), MDR1 was also detected on lateral membranes of ciliated cells and at the circumference of basal cells. Similar results were obtained with the same method on bronchial epithelial cells (unpublished observations). In submucosal glands, MDR1 was predominantly detected on lateral membranes of serous cells, in myoepithelial cells, and over only a very limited number of mucus-secreting cells. MDR1 is known to transport a large variety of hydrophobic and amphiphilic substrates, as well as organic cations, and is therefore considered to be particularly effective in cell detoxification of xenobiotics, which could be its main function in the superficial nasal respiratory mucosa. The unusual lateral staining, particularly of serous cells, observed in this study suggests an efflux in these cells towards the lateral compartment, which could prevent secretion of toxins into the lumen and consequently into superficial nasal fluid, and which could also be related to the other roles of MDR1, such as cell volume regulation or transport of peptides, such as peptide growth factors (Schinkel 1997).
In the respiratory epithelium, intracellular granular staining for MRP1 was observed in all ciliated cells, particularly in the apical part of the cell. This result, at variance with the basolateral localization previously reported for bronchial respiratory cells (Bréchot et al. 1998), could be explained by the differences in tissue preparations and fixative used. In fact, we (unpublished results), like others (Nooter et al. 1996; Wright et al. 1998), detected both membrane and diffuse intracytoplasmic staining when MRP1 immunodetection was performed after fixation and paraffin embedding on ciliated epithelial cells from bronchial mucosa. In submucosal glands, MRP1 was detected only in the intracellular compartment in the apical region of serous cells, with a granular pattern that might could correspond to the many secretory granules present in this cell region. The granular cytoplasmic pattern of MRP1 could be due to a role of MRP1 in transport of compounds into intracellular compartments, as previously suggested (Flens et al. 1996). MRP1 is mainly involved in the transport of glutathione conjugates, although other functions are still debated (Loe et al. 1996). Because bronchial serous cells also secrete a mixture of antibacterial proteins (lysozyme, lactoferrin, and phosphatase), a possible role of MRP1 in regulation of the content of secretory granules can be hypothesized. However, no data in support of this hypothesis, particularly in nasal glands, are yet available.
Immunodetection of CFTR (
Immunolocalization of MDR1 (
In conclusion, although a potential crossreaction of the monoclonal antibodies used with as yet undiscovered ABC proteins cannot be formally excluded, the present study shows that CFTR, MDR1, and MRP1 are expressed in the epithelium and glands of human nasal respiratory mucosa, but with different patterns of expression. Unlike MDR1 and MRP1, CFTR is inconsistently expressed in ciliated epithelial cells. In contrast, all three ABC proteins are consistently and intensely expressed in serous cells of the glands. These results suggest major roles for CFTR, MDR1, and MRP1 in serous glandular cells, and at least a protective function for MDR1 and MRP1 in respiratory ciliated cells. The respective roles of these proteins in the various cells, particularly in serous glandular cells, either in the control of the composition of the bronchial superficial fluid layer or airway cell detoxification, need to be evaluated by cell function studies using in vitro models, such as organotypic cultures.
Footnotes
Acknowledgements
Supported by Crédits Université Pierre et Marie Curie, Paris VI, and by grant R98018 from the Association Française de Lutte contre la Mucoviscidose (AFLM). M.-A. Wioland is an AFLM fellow.
We are greatly indebted to C. Danel, C. Guernier, L. Germain, C. Prengel, and S. Ricci for constant support and to V. Gerber for expert editorial assistance.
