Abstract
Cystic fibrosis (CF) is caused by mutations in the gene encoding the CF transmembrane conductance regulator (CFTR) protein, which has a major role as a chloride (Cl−) channel. Although perhaps all functions of CFTR are still not fully characterized, localization studies are necessary to understand the consequences of the more than 1000 mutations thus far identified. Our aim was to determine the histological localization of CFTR on respiratory and colon epithelia of human and murine origin with a panel of several antibodies produced against different CFTR epitopes, using an indirect immunofluorescence method. Our results on human tissues confirm the apical localization of CFTR in ciliated cells of the respiratory mucosa and show that in colon tissue CFTR is observed in both apical and basolateral membranes of epithelial cells from colon crypts. However, poor tissue preservation of colon biopsies after immunohistochemistry (IHC) raises doubts about the latter localization. Contrary to human, mouse colon epithelium (not biopsed) presents good tissue preservation and evidences many cylindrical surface cells with high apical expression of CFTR. For the antibodies's sensitivity, we demonstrate that MATG1061, 24-1, M3A7, and MPCT-1 give good results, allowing the histological localization of CFTR protein of both human and murine origin.
Keywords
C
Most data on CFTR expression and function have been derived from experiments on CFTR-overexpressing cells, i.e., transformed cell lines. It has been shown that wild-type (wt) CFTR is directed to the apical membrane of polarized epithelial cells via the Golgi and the trans-Golgi network (TGN), possibly interacting with many proteins during its maturation (Chang et al. 2002). Although polarized epithelial cells are probably the best models in which to study expression and interactions of CFTR, they still exhibit deviations from native tissues brought about by the transformation process.
Data from native cells/tissues are therefore still scarce and often contradictory, probably due to experimental difficulties. CFTR is generally described as a low-abundance protein in most tissues where it is endogenously expressed (Trezise and Buchwald 1991; Kartner et al. 1992). Anti-CFTR Abs capable of detecting the protein in overexpressing cell lines often are not sensitive enough to detect it in native tissues. In addition, epitopes recognized by some of these Abs frequently become inaccessible after routine tissue preservation (Kartner et al. 1992; Hanaoka et al. 1996). Nevertheless, knowledge obtained from native cells/tissues is critical to assess the impact of CFTR mutations on the cellular and subcellular localization of the protein.
The purpose of this study was, first, to compare different CFTR immunostaining protocols and, second, to determine the specificity and sensitivity of a panel of seven anti-CFTR Abs used to immunodetect CFTR in respiratory and intestinal tissues of both human and murine origin. The immunostaining technique was therefore optimized and tested for these different anti-CFTR Abs to maximize the specific CFTR signal-to-noise ratio in IHC studies.
Materials and Methods
Tissue Samples
Here we compared the histological localization of CFTR in colon and nasal epithelia of non-CF individuals and mice. Collection of human tissues was approved by the ethical committee of the hospital and informed consent was obtained from each individual.
Human Tissues. Nasal and cartilaginous bronchial mucosa have similar structures, both consisting of the association of glands, which in turn are composed of serous and mucos-secreting cells. It has therefore been widely accepted that cell biology data obtained on nasal specimens can be extrapolated to the lower airways (Verra et al. 1993; Knowles et al. 1995; Danel et al. 1996). Fresh specimens of human nasal polyps from non-CF individuals were obtained immediately after polypectomy. Polyps were rinsed with PBS, coated with OCT compound (Tissue Tek; Miles, Elkhardt, IN) and the specimens were quickly frozen in liquid nitrogen and stored at —80C until use. Alternatively, for Zamboni fixation (see below), tissues were soaked in the fixing solution immediately after collection. After ∼9 hr, tissue samples were thoroughly rinsed with PBS and frozen in liquid nitrogen as above.
Rectal biopsy specimens from non-CF individuals (n = 5) during colonoscopy were immediately immersed in DMEM/F12 medium supplemented with 250 UI/ml of penicillin, 125 μg/ml of streptomycin, and 2.5 μg/ml of amphotericin B. Then the same two protocols as above (freezing or Zamboni fixation) were applied.
Cryosections (6-8 μm thick) were obtained from frozen tissues in a cryomicrotome (Reichert-Jung; Bensheim, Germany) at −20C, placed onto silane-coated glass slides (Menzel-Glazer; Braunschweig, Germany), air-dried, and used immediately or stored at −80C until use. IHC studies were carried out either on fresh or thawed cryosections after air-drying and rehydration for 5 min in PBS.
Mouse Samples. Mice were sacrificed after anesthesia by IP injection of nesdonal. Distal colon mucosa samples of 3 mm (length) and tracheal biopsies from the last 3 mm before bronchial bifurcation were taken. Tracheal and colon samples were rapidly removed, frozen, and sectioned as above.
Hematoxylin and Eosin Staining
One cryocut per specimen was stained with hematoxylin and eosin (H&E) for histological observation. Briefly, slides with cryocuts were soaked into Mayer's hemalum solution (Merck Diagnostica; Darmstadt, Germany) for 2 min. After three or four rapid washes in water, slides were soaked in 2% (w/v) eosin (BDH; Poole, Dorset, UK) solution for 30 sec and then rinsed again with water. Cryocuts were then dehydrated by soaking in three solutions of increasing ethanol concentrations [70% (v/v), 95%, and 100%] for 2 min each and finally in xylene also for 2 min. Slides were mounted with p-xylene-bis(N-pyridinium bromide), or DPX (BDH), covered with glass coverslips, and dried for at least 1 hr before analysis.
Antibodies
For CFTR immunostaining, we tested a panel of seven antibodies to evaluate their sensitivity and specificity towards human and murine CFTR (see Table 1). MATG1061 (Transgène, Strasbourg, France; Brezillon et al. 1995) is an anti-CFTR monoclonal antibody (MAb) raised against amino acid residues 503-515 of the first nucleotide-binding domain (NBD1). MAB25031 (24-1) and MAB1660 (13-1) are MAbs (R&D Systems; Abington, UK) raised against amino acid residues 1377-1480 (C-terminus) and 590-830 (R domain), respectively (Kalin et al. 1999). M3A7 (Chemicon, Tamecula, CA; Engelhardt et al. 1992) is an MAb raised against amino acid residues 1370-1380 (C-terminus). MP-CT-1 (a kind gift from Dr. R. Dormer; University of Wales, Cardiff, UK) is a polyclonal antibody (PAb) raised against the C-terminus of CFTR (Dormer et al. 2001). Ab 169 (a kind gift from Dr. W.B. Guggino; Johns Hopkins University, Baltimore, MD) is a PAb raised against amino acid residues 724-746 of the R domain (Hoogeveen et al. 1991). CC24-R (a kind gift from Dr. H. DeJonge; Erasmus University, Rotterdam, The Netherlands) is a PAb raised against amino acid residues 693-716 of the R domain (Claass et al. 2000). Ab characteristics and dilutions used are summarized in Table 1. AE1-AE3 (DAKO; Glostrup, Denmark), an anti-cytokeratins 18/19 MAb, was used at 1:1500 dilution in 0.5% BSA/PBS. Primary MAbs were detected by a secondary FITC-conjugated anti-mouse IgG Ab (Amersham; Little Chalfont, UK) diluted 1:120 in 0.5% BSA/PBS. Primary PAbs were detected by a secondary FITC-conjugated anti-rabbit IgG Ab (Amersham) diluted 1:60 as above.
Description of anti-CFTR antibodies tested in the current study
Dilutions were performed according either to previous studies or to manufacturers's indication, except for MATG1061.
Kindly given.
NBD1, first nucleotide-binding domain.
Immunocytochemistry
A large part of the observed variability in CFTR immunostaining can be generally due to the protocol used. The latter should result in a good compromise between morphology preservation, achieved through adequate fixation, and avoiding epitope destruction, which usually occurs when too drastic fixation conditions are used. The fixation step is therefore very critical, because if it preserves the tissue from autolysis it also leads to a decrease in the signal due to concealment/disappearance of the antigenic motif (epitope). Before testing the specificity and sensitivity of the different anti-CFTR Abs, an optimization of critical steps (fixation, protease inhibition, dilution, and incubation time of primary Ab) was performed, using as a starting point protocols previously described for the immunolocalization of CFTR (Brezillon et al. 1995; Beck et al. 1999; Kalin et al. 1999; Penque et al. 2000).
In these preliminary tests, we used anti-CFTR MATG1061, previously shown to produce CFTR-specific labeling in native tissues (Kalin et al. 1999; Penque et al. 2000) to assess the integrity of CFTR epitopes and anti-cytokeratins AE1-AE3 MAbs, staining cytoskeletal elements of differentiated epithelial cells, to assess the preservation of tissue morphology. We tested the IHC detection of CFTR on both fixed and unfixed cryosections (see above). As fixing agents we used methanol or Zamboni solution, composed of 85% of solution 1 (2% v/v paraformaldehyde/0.1 M Sorensen phosphate buffer) and 15% of 51.5 mM picric acid (Stefanini et al. 1967). We then tested incubation in the presence of protease inhibitors, since the presence of proteases can also cause substantial loss of tissue morphology and signal intensity due to protein degradation. A cocktail of protease inhibitors was therefore used to decrease tissue degradation and thus improve the quality of the fluorescence signal. The composition of this cocktail was leupeptin (1 mg/ml), aprotinin (2 mg/ml), benzamidine (121 mg/ml), pefabloc (50 mg/ml), and E64 (3.5 mg/ml). IHC results were compared between slides incubated in the presence or absence of this cocktail.
The adopted optimized protocol involved fixation in methanol for 10 min at −20C before labeling with anti-CFTR Ab. Finally, incubation time with primary Ab (MATG1061) was optimized by testing two different incubation times: overnight at 4C (long) and 2 hr at room temperature in a humid chamber (short). We also tested different dilutions of the primary MATG1061 Ab in the range of 1:200-1:1000 and determined that 1:200 produced the best results. For the other Abs used here, dilutions were according to previous studies or followed those indicated by the manufacturers (see Table 1).
Incubations with secondary Abs were for 45 min, also in a humid chamber at RT. Negative controls omitting the primary Ab were always performed in parallel. Sections were washed three times for 5 min in PBS between each step and finally mounted in Vectashield anti-fading medium (Vector Labs; Burlingame, CA) containing DAPI (Sigma Chemical; St Louis, MO) for nuclear labeling. Labeled sections were stored at 4C in the dark until examined on an epifluorescence Axioskop microscope (Zeiss; Gottingen, Germany) equipped with PowerGene 810 software (PSI; Chester, UK).
Results
Optimization of Immunostaining Towards a Good Balance Between Tissue Preservation and CFTR Signal
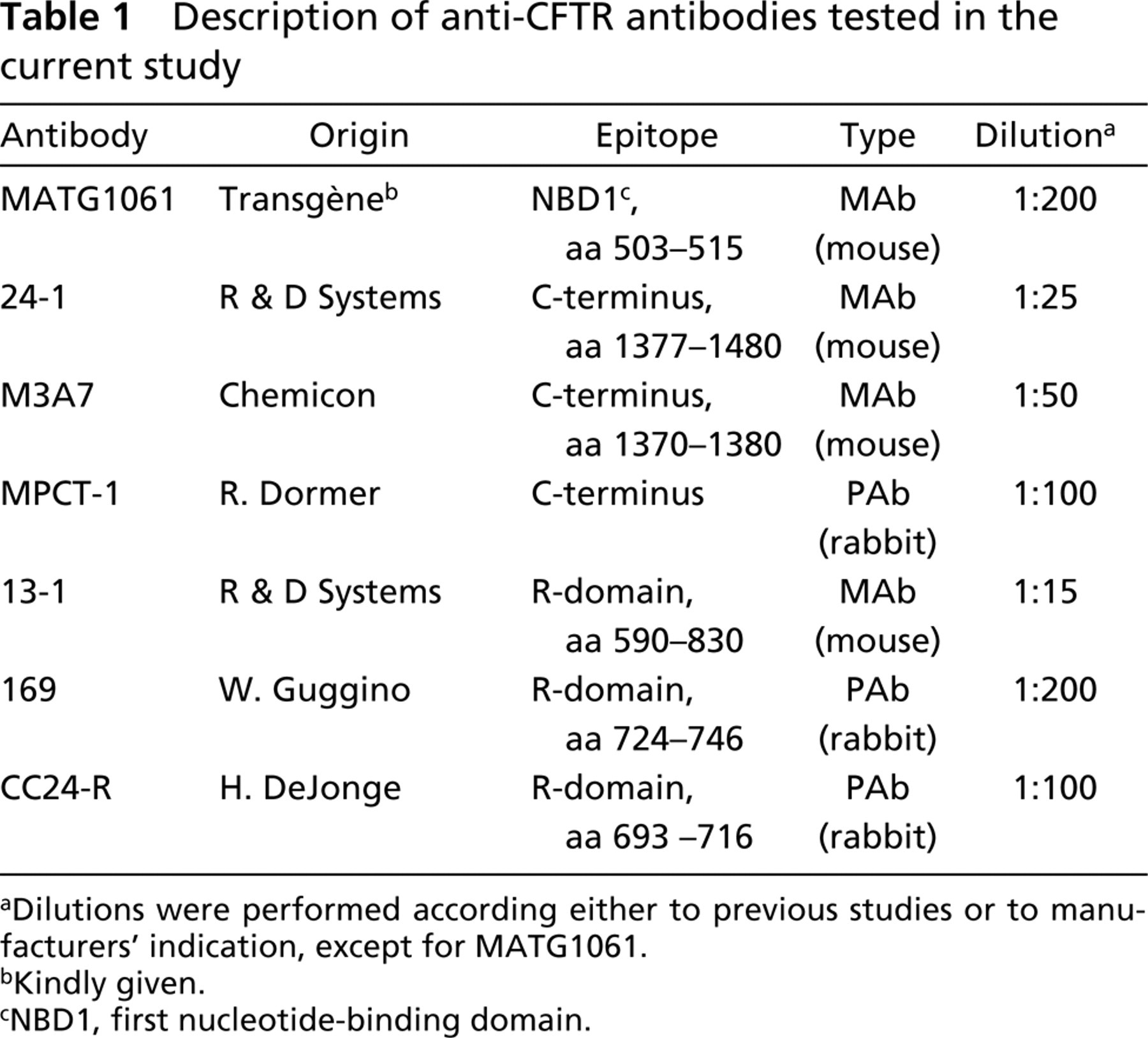
As described in Materials and Methods, a preliminary study with anti-CFTR MATG1061 was performed to optimize the general immunodetection protocol concerning the effect of fixation conditions (Stefanini et al. 1967), the presence of protease inhibitors, and the use of different incubation times (2 hr at RT vs overnight at 4C), both on preservation of tissue morphology and on the CFTR signal detected (see Figure 1). After determination of the best anti-CFTR MATG1061 Ab dilution (see Materials and Methods), we tested the time of incubation with primary Abs (both anti-CFTR and anti-cytokeratins) after methanol fixing. Overnight incubation at 4C of human intestinal tissue with either MATG1061 anti-CFTR Ab (Figure 1B) or anti-cytokeratins (Figure 1G) led to a significant loss of tissue morphology (Figures 1A and 1F) compared with a 2-hr incubation at RT (Figures 1C and 1H, respectively) without significant gain in CFTR labeling. This loss was more evident in samples incubated with the anti-CFTR Ab (Figure 1B) than with the anti-cytokeratins Ab (Figure 1G). In addition, loss of morphology was not observed in nasal samples incubated with either anti-CFTR (Figure 1L) or anticytokeratins (Figure 1P) Ab (only results after 2-hr in cubation are shown, as very similar results were obtained after overnight incubation). We therefore concluded that the short incubation time with primary Ab should be used for both tissues, because for intestinal samples tissue morphology is better preserved after short incubations (compare Figures 1B and 1C and for nasal samples equivalent labelings were observed after 2-hr or overnight incubation; only 2-hr labeling is shown for nasal tissue, Figure 1L). Indeed, the short incubation time did not diminish the CFTR signal obtained. It was previously described that Zamboni, like other paraformaldehyde fixing solutions, is not suitable for IHC detection of CFTR (Claass et al. 2000). Our results confirm these findings, as in cryocuts fixed with Zamboni's solution and probed with anti-CFTR MATG1061 MAb there was an almost complete absence of specific CFTR signal in intestinal samples (Figure 1D) and a much poorer CFTR labeling in nasal samples (Figure 1M; compare with Figure 1L showing CFTR immunostaining after methanol fixation). Indeed, under these conditions, CFTR epitopes must be unavailable for detection. However, after Zamboni fixation, some cytokeratin labeling was still observed in both intestinal (Figure 1I) and respiratory tissue (Figure 1Q), while tissue morphology was well preserved (compare with Figures 1A and 1F for intestinal tissue morphology and with Figures 1K and 1O, for respiratory tissue morphology).
Comparison of different parameters in the immunodetection of CFTR. Cryocuts from rectal biopsies (A-J) and nasal polyps (K-R) were stained with H&E (A,F,K,O) or labeled with anti-CFTR MATG1061 MAb (in green, B-E and L-N) or anti-cytokeratins 18/19 MAb (in green, G-J and P-R). Blue corresponds to DAPI nuclei staining. Four conditions were tested in rectal biopsies: methanol fixation and overnight incubation with primary Ab (B, for CFTR; G, cytokeratins); methanol fixation and 2-hr incubation with primary Ab (C, CFTR; H, cytoker-atins); Zamboni fixation and 2-hr incubation with primary Ab (D, CF TR; I, cytokeratins); no fixative agent and 2-hr incubation with primary Ab (E, CFTR; J, cytokeratins). For nasal polyps samples three fixation conditions are shown, all with 2-hr incubation with primary Ab: methanol fixation (L, CF TR; P, cytokeratins); Zamboni fixation (M, CFTR; Q, cytokeratins; no fixative agent (N, CFTR; R, cytokeratins). (E) Yellow line indicates the apical surface of the major crypt shown. Bars: A-E,G-N,P-R = 50 μm; F,O = 200 μm.
Without fixation, labeling of intestinal tissue with either anti-CFTR (Figure 1E) or anti-cytokeratins Abs (Figure 1J) was still evident but tissue morphology was only poorly preserved. Loss of morphology was not observed in unfixed nasal samples (Figures 1N and 1R). Overall, methanol fixation was chosen for the general CFTR immunostaining protocol as the best compromise between CFTR signal and tissue morphology for both intestinal and nasal tissues. No significant differences were observed between IHC sections incubated with or without a cocktail of protease inhibitors (data not shown). All the results obtained during the preliminary optimization study (Figure 1) are summarized in Table 2.
IHC Localization of CFTR with Different Antibodies
Mouse Tissues. Our objective when mouse specimens were used was to compare preservation of tissue morphology (particularly of intestinal tissue, not obtained through a biopsy procedure) and also to test the specificity of several Abs for the IHC localization of endogenously expressed murine CFTR on cryosections of different tissues. Only polyclonal (produced in rabbit) anti-CFTR Abs were tested (i.e., MPCT-1 and 169) because detection of monoclonal anti-CFTR Abs with anti-mouse IgG secondary Abs would crossreact unspecifically with sample tissues.
Trachea. With MPCT-1 PAb, intense CFTR labeling was observed in the cytoplasm and in the apical membrane of cells from the submucosal tracheal glands, as expected (white arrow in Figure 2C). Surface cells presented a characteristic apical signal (yellow arrows in Figure 2C and inset). With PAb 169, the signal observed was very faint and was detected only for the submucosal gland cells (arrow in Figure 2E). Collecting ducts were not present in the different cryosections analyzed. In the absence of primary Ab, only faint background signal was detected (Figure 2G).
Colon. Contrary to human samples, where considerable loss of morphology occurred during CFTR immunostaining (see above), mouse colon cryosections evidenced good tissue morphology when the same protocol was used (Figure 2D). Comparison of mouse intestinal sections in which CFTR was detected by immunofluorescence (Figures 2D and 2F) with similar ones stained with H&E and observed under bright-field (Figure 2B) clearly shows that morphology was still preserved in the former. Immunofluorescence signal was negative for most of the mouse colon cells, i.e., in the crypts and at the surface epithelium, when either MPCT-1 or 169 PAb was used to detect CFTR (Figures 2D and 2F, respectively). An unexpectedly intense signal was observed on a few scattered epithelial cells located on the surface epithelium (arrowhead in Figure 2D) and also on some of the most superficial cells of the crypts (not shown). This intense signal was mostly cytoplasmic, with additional apical reactivity. These peculiar “CFTR high-expressor” (CHE) cells exhibited an enterocyte-like morphology. Goblet cells were CFTR-negative. Again, no signal was observed in the absence of primary Ab (Figure 2H). Results are summarized in Table 3.
Human Tissues
It was also our objective to compare the specificity of several Abs for IHC localization of CFTR on fixed cryosections of human nasal and intestinal tissues. The optimized general immunolabeling protocol described above was used to test a panel of seven anti-CFTR Abs (Figure 3).
Results of the optimization study (testing of different fixation solutions and incubation times) using MATG1061 and AE1-AE3 on human colon and airway tissuesa
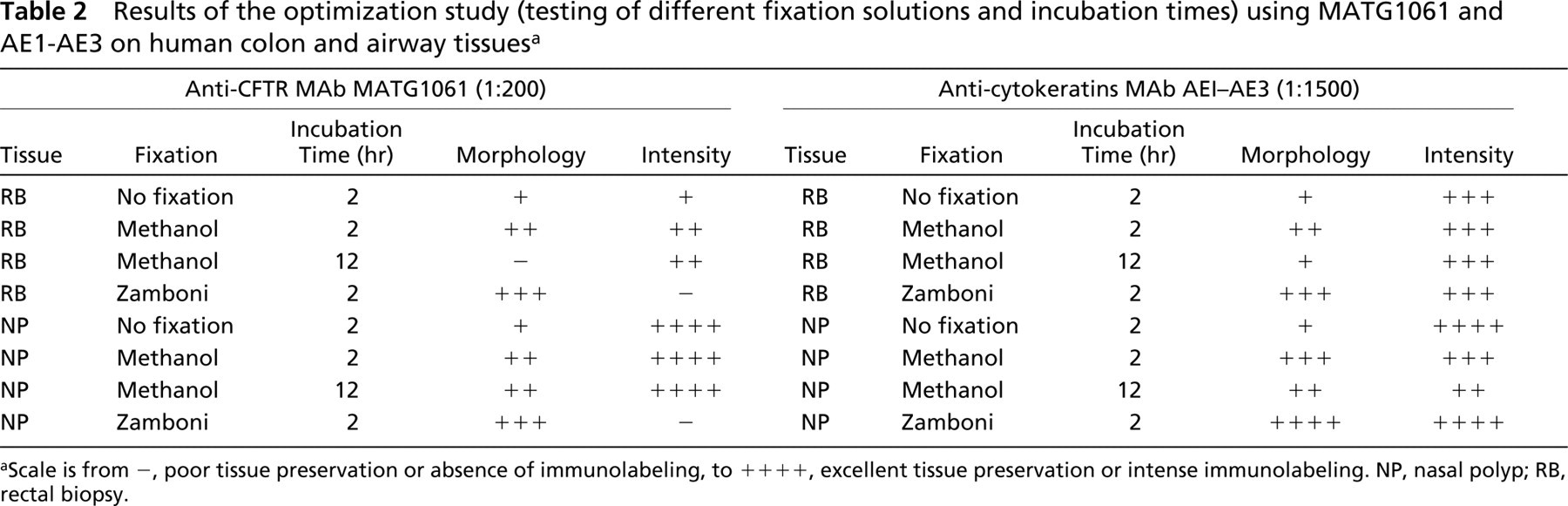
Scale is from -, poor tissue preservation or absence of immunolabeling, to ++++, excellent tissue preservation or intense immunolabeling. NP, nasal polyp; RB, rectal biopsy.
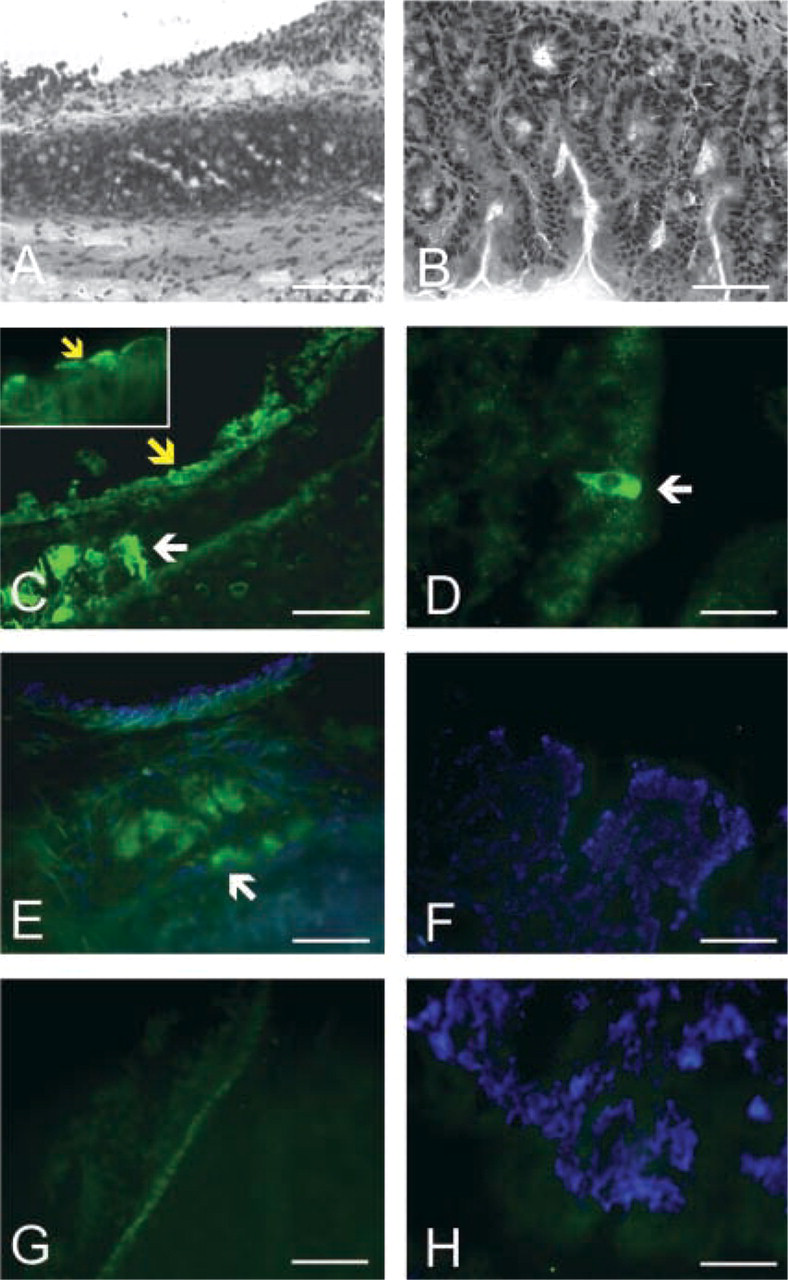
Immunohistochemical CFTR labeling in mouse tissues. Cryocuts from trachea and colon of murine origin were stained with H&E (A,B, respectively) or labeled with two different anti-CFTR PAbs (in green), MP-CT1 (C,D, respectively) and 169 (E,F, respectively). Negative controls (without primary Ab) were included (G,H).
Nasal Tissue. CFTR was clearly immunodetected on the luminal surface of pseudostratified epithelia of nasal polyps with MATG1061 and 24-1 MAbs (Figures 1L and 3A, respectively). Although cryocuts may look different in morphology, only pseudostratified surface epithelium was analyzed for CFTR immunostaining, as can be clearly observed by the monolayer of cell nuclei in cryocuts (see DAPI staining of cell nuclei Figures 3A−3C and 3G-3I).
Monoclonal M3A7 and polyclonal MPCT-1 Abs produced similar results to MATG1061, although the signal detected was weaker and somewhat more diffuse (Figures 3C and 3G, respectively). MPCT-1 Ab also immunostained basal cells to some extent (see Figure 3G). Because the latter were shown to express very low levels of CFTR or none at all (Brezillon et al. 1995; Castillon et al. 2002), also confirmed here for the other anti-CFTR Abs (see, e.g., results for 24-1 Ab in Figure 3A), labeling of basal cells by MPCT-1 can therefore be considered unspecific. No significant labeling was obtained for basal cells with M3A7 (Figure 3C) or for epithelial secretory cells for either M3A7 or MPCT-1 (Figures 3C and 3G, respectively). Glands were not present in the cryosections observed. On the other hand, all the other anti-CFTR Abs, 13-1 MAb (Figure 3B), 169 PAb (a different batch from that used by Beck et al. 1999) (Figure 3H), and CC24-R PAb (Figure 3I), failed to produce a clear specific label in the respiratory epithelium of nasal polyps.
Intestinal Specimens. IHC analysis of intestinal cryosections was very limited by the poor preservation of tissue morphology after the IHC protocol. Indeed, tissue deterioration had not occurred before the Ab incubation, as observed by the good morphology of H&E-stained sections under brightfield (Figures 1A and 1F). Therefore, no good results were obtained for human intestinal sections with any of the seven anti-CFTR Abs tested here. Diffuse apical and basolateral labeling was observed in cells from the crypts and from the surface epithelium with MATG1061 MAb (Figure 1C), 24-1 MAb (Figure 3D), 13-1 MAb (Figure 3E), and M3A7 MAb (Figure 3F). The strong unspecific background observed, however, did not allow discrimination among different cell types (e.g., goblet cells, reabsorption cells). With MPCT-1 PAb, however, apical labeling was observed when tissue morphology was preserved, a situation that sometimes but not always occurred after methanol fixation (Figure 3J). CFTR labeling was either very faint or nonspecific with 169 (Figure 3K) and CC24-R (Figure 3L) PAbs.
Discussion
The present study assessed the impact of the anti-CFTR immunostaining protocol used on the results obtained in terms of both preservation of tissue morphology and the CFTR signal detected. The other contribution resulting from this study is to define which anti-CFTR Abs are sensitive and specific to detect CFTR in human and murine histological sections of nasal and intestinal origin.
Results obtained for the immunodetection and localization of CFTR on human and mouse respiratory epithelia presented here are in agreement with those from previous studies (Puchelle et al. 1992; Whitsett et al. 1992; Engelhardt et al. 1994; Brezillon et al. 1995; Kalin et al. 1999; Penque et al. 2000; Wioland et al. 2000). It was previously described that serous cells of submucosal acini are the principal site of CFTR expression in upper respiratory tract, together with scattered cells of collecting ducts from these glands (Engelhardt et al. 1992; Jacquot et al. 1993). Here we confirm the high CFTR expression in the former (observed in the mouse trachea). We also detected here a characteristic signal at the luminal surface of both mouse tracheal and human nasal epithelial sections corresponding to the apical surface of ciliated and non-ciliated tall columnar epithelial (TCE) cells (Penque et al. 2000; Wioland et al. 2000). In spite of these expected observations, we show here that the sensitivity of different anti-CFTR Abs varies. MATG1061 MAb (Brezillon et al. 1995; Kalin et al. 1999), 24-1 MAb (Kalin et al. 1999; Wioland et al. 2000) and, to a lesser extent, M3A7 MAb and MPCT-1 PAb, are specific and sensitive for the immunodetection of CFTR in cryosections of native human tissues, as shown here for nasal polyp sections.
Immunolocalization of CFTR in respiratory and intestinal tissues of human and murine origin
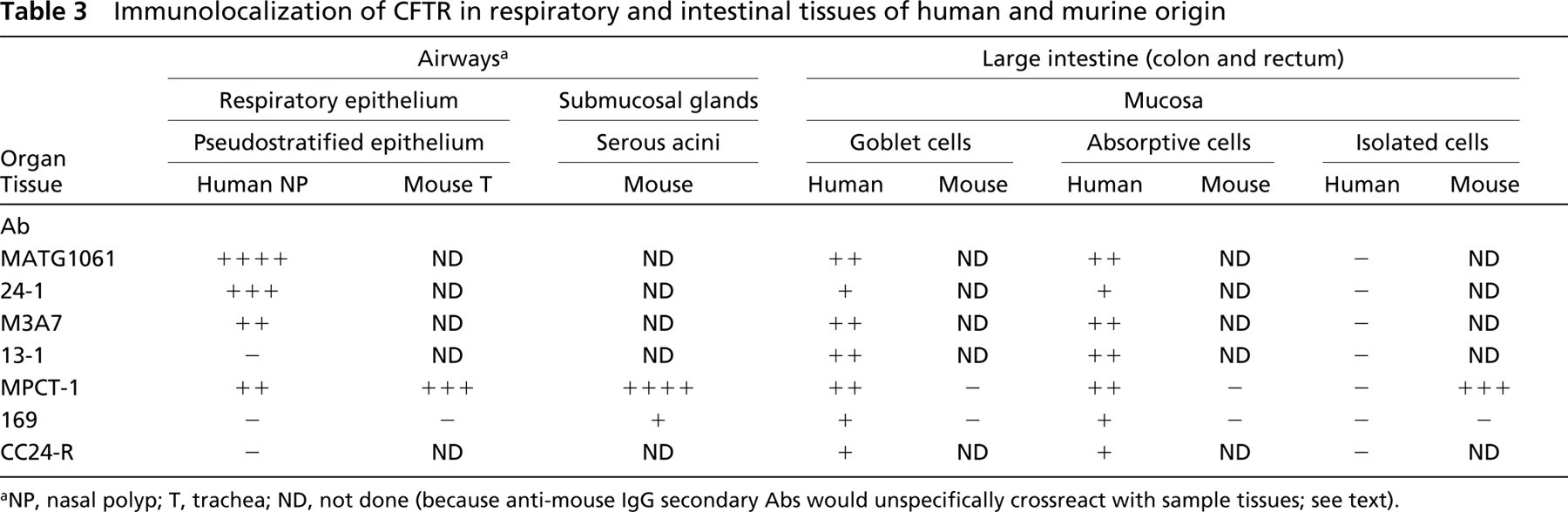
NP, nasal polyp; T, trachea; ND, not done (because anti-mouse IgG secondary Abs would unspecifically crossreact with sample tissues; see text).
Exhaustive data on CFTR localization in the intestinal tract are scarce (Crawford et al. 1991; Hoogeveen et al. 1991; Gaillard et al. 1994; Strong et al. 1994; Kalin et al. 1999; Ameen et al. 2000). As mentioned by others (French et al. 1996; Kalin et al. 1999), we confirm here that immunolocalization of CFTR in human intestinal tissues is considerably more difficult than in the airways. This difficulty may be the main reason for the contradiction in reported results. However, contradictions could also have arisen due to the relatively low expression levels of CFTR in distal intestine compared to the high expression levels in proximal (rectal) colon. On the other hand, it was described that proteolytic activity, especially high in intestinal tissues owing to the presence of bacteria (French et al. 1996), may cause loss of tissue integrity. Our results for human rectal biopsies also evidenced significant loss of morphology and, consequently, a significant increase in background, together with a drastic reduction in specific CFTR signal. This loss, however, had not occurred before the Ab incubation, as observed by the good morphology of H&E-stained sections under brightfield. Adding a protease inhibitor cocktail to the primary Ab incubation mixture did not significantly improve the preservation of colon tissue morphology. The loss in morphology for human rectal biopsies occurred only when the immunocytochemistry protocols with mild fixation or without fixation (i.e., necessary for CFTR immunostaining) were used. Indeed, morphology of rectal samples was well preserved after cytokeratin immunodetection when samples were fixed with Zamboni or after H&E staining of samples. However, under these conditions CFTR epitopes must be unavailable for detection because no specific staining was detected.
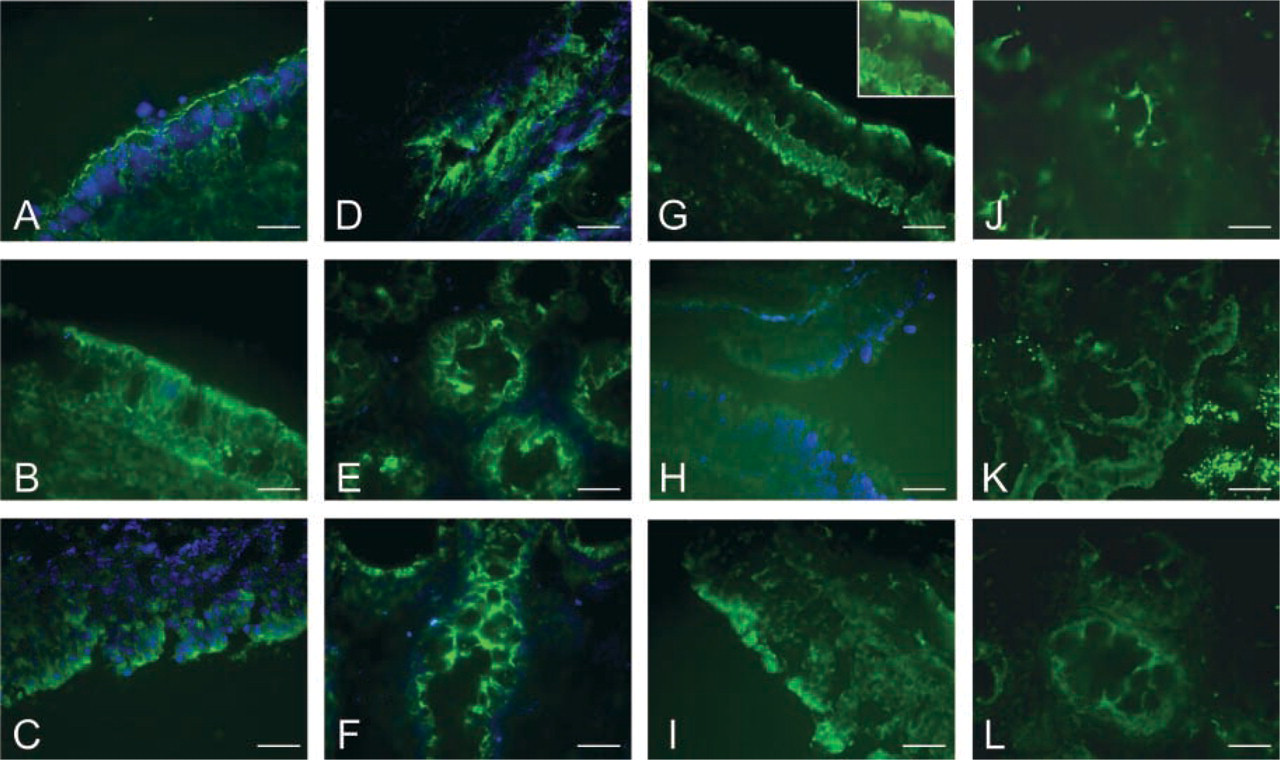
Comparison of the specificity of several anti-CFTR Abs in human samples. Cryosections from nasal polyps (A-C,G-I) and rectal biopsies (D-F,J-L) were labeled with 24-1 (A,D), 13-1 (B,E), M3A7 (C,F), MPCT-1 (G,J), 169 (H,K), and CC24-R (I,L). Bars = 50 μm.
On the other hand, in mouse colon samples (obtained by cutting off small pieces of tissue, not biopsies) morphology was much better preserved than in colon biopsies. This leads to the suggestion that additional cell shredding may be associated with the biopsy (forceps) procedure.
In our observations of human rectal mucosa sections, CFTR is detected in both the apical region and intracellularly as Kalin et al. (1999) previously described mostly for goblet cells. In the current study, the poor tissue preservation in cryofixed tissue did not allow good discrimination between enterocyte and goblet cells. Similarly, no significant differences in the intensity or localization of CFTR labeling were observed between crypt and surface epithelial cells. As mentioned by Kalin et al. (1999), results shown here suggest that the MATG1061 MAb is appropriate for immunodetection of CFTR in colon tissue.
Previous mRNA in situ expression studies (Strong et al. 1994) or protein localization experiments (Ameen et al. 1995; Kalin et al. 1999) described high levels of CFTR expression in a few enterocyte-like cells that appear scattered in villi of proximal human small intestine (duodenum, jejunum). Other authors did not observe these cells in human colon or rectal sections (Kalin et al. 1999). However, a similar distribution of these cells, was reported in rat intestine (Ameen et al. 1995). Those authors observed that immunolabeling was mostly intracellular but increased at the apical pole. They also reported that these enterocyte-like cells or CHE cells lack distinct morphological features when examined under brightfield microscopy but possess the ultrastructural and enzymatic characteristics of well-differentiated transport epithelial cells (Ameen et al. 1995). In addition, Ameen et al. (2000) reported differences in the histological distribution of CFTR between small intestine specimens of mouse and rat. In the latter, CFTR was detected along the apical domain of crypt cells (duodenum) and also at the surface of the entire length of the villi (small intestine). No CHE cells were observed by those authors in the mouse intestine. Contrary to the latter study and in agreement with Hayden and Carey (1996), we detected here a few cells in the mouse intestine with the same characteristics as CHE cells. Hayden and Carey observed CFTR in goblet cells and in uncharacteristic non-mucosal and non enteroendocrine cells of the small intestine, but not in colon. The current study is the first to show that CHE cells also appear to be present in both the surface epithelium and in the superficial part of crypts in mouse colon. Determination of the role of the CHE cells in small intestine and colon and confirmation that results can be extrapolated from mice to human may lead to further understanding of CF intestinal disease.
Footnotes
Acknowledgements
We thank the Association de Transfusion Sanguine Gaétan Salaün, the Conseil Régional de Bretagne, the Conseil Général du Finistère, and the French associations Vaincre la Mucoviscidose and ARC for financial support. The European CF Network (EU-QLK3-1999-00241) provided anti-CFTR antibodies and LD with a travel grant.
We are also indebted to R. Dormer, W. Guggino, H. De-Jonge, and Transgène for providing antibodies, and to S. Alpiarça (Gulbenkian Institute of Science; Oeiras, Portugal) and D. Gillet (EMIU 01-15, I3S, Brest) for technical assistance. We gratefully thank Prof Paulo Ramalho (Hospital de Santa Maria, Lisbon) for performing rectoscopy procedures.
