Abstract
We have adapted existing microwave irradiation (MWI) protocols and applied them to the processing and immunoelectron microscopy of both plastic-embedded and frozen sections. Rat livers were fixed by rapid MW irradiation in a mild fixation solution. Fixed liver tissue was either cryosectioned or dehydrated and embedded in Spurr's, Unicryl, or LR White resin. Frozen sections and sections of acrylic-embedded tissue were immunolabeled in the MW oven with an anti-catalase antibody, followed by gold labeling. Controls were processed conventionally at room temperature (RT). The use of MWI greatly shortened the fixation, processing, and immunolabeling times without compromising the quality of ultrastructural preservation and the specificity of labeling. The higher immunogold labeling intensity was achieved after a 15-min incubation of primary antibody and gold markers under discontinued MWI at 37C. Quantification of the immunolabeling for catalase indicated a density increase of up to fourfold in the sections immunolabeled in the MW oven over that of samples immunolabeled at RT. These studies define the general conditions of fixation and immunolabeling for both acrylic resin-embedded material and frozen sections. (J Histochem Cytochem 48:1153–1159, 2000)
Immunoelectron microscopy has been widely used to determine the intracellular distribution of proteins to gain information on their function. The procedure often requires a large number of samples and substantial processing time to determine unambiguously the localization of a given antigen under different experimental conditions. Postembedding methods applying microwave irradiation (MWI) to the fixation and immunolabeling of thin plastic-embedded sections have already been developed (Login et al. 1987, 1992, 1995; Zondervan et al. 1988; Naganuma et al. 1990).
In our experience, frozen sections are optimal substrates to carry out immunolabeling experiments. The absence of plastic embedding medium enhances the permeation of the antibody reagents into the section, leading to a higher immunolabeling density than that achieved on plastic-embedded sections. However, immunocryoelectron microscopy is time-consuming, which is a major disadvantage because the different steps from fixation of the tissues to immunolabeling of the frozen sections are usually carried out without interruption. The technique is even more time-consuming when, after immunolabeling, the frozen sections are treated with osmium tetroxide, dehydrated, and embedded in plastic according to the ultrathin plastic embedding technique (Keller et al. 1984). Consequently, we have adapted existing protocols and have used MWI to shorten the immunocryoelectron microscopy procedure.
For the studies described here, we chose peroxisomal catalase as a model for immunolabeling because the antigen is moderately abundant and withstands fixation. Furthermore, the enzyme is mostly distributed in one subcellular compartment, and the morphological features of peroxisomes are well characterized (reviewed by van den Bosch et al. 1992). Accordingly, we performed a series of experiments in which liver samples were fixed, processed, and immunolabeled both conventionally and by MWI. We then determined the average density of catalase labeling to allow comparison among different embedding polymers and immunolabeling regimens. Here we suggest protocols that substantially reduce the processing time and improve the specific immunolabeling of both acrylic resin-embedded and cryosectioned samples.
Although much has already been written about the parameters necessary for application of MW techniques to the processing and immunolabeling of samples, we recommend the articles by Leong et al. (1985), Login et al. (1987), Hjerpe et al. (1988), Giammara (1993), Kok et al. (1993), Login and Dvorak (1988, 1993), Gu (1994), Choi et al. (1995), Gu et al. (1995), Cuevas et al. (1995), Werner et al. (1996), Chicoine and Webster (1998), and Giberson et al. (1995, 1997).
Materials and Methods
The experiments reported here were carried out in a programmable model 3450 microwave oven that allows greater temperature and energy control (Ted Pella; Redding, CA). Maintaining the temperature between 25C and 42C gives the best results without damaging the samples (Gu 1994). In particular, when samples for immunocryoelectron microscopy are processed, the temperature must be critically controlled because temperatures too high could alter the integrity and antigenicity of the tissues. Temperature control can be achieved in several ways. A temperature probe with a feedback mechanism can be used to regulate the energy output of the MW to maintain the desired temperature. Better control can be achieved by placing a water load in the chamber of the MW. The water helps absorb extra energy and adds humidity in the chamber, thus slowing the evaporation of reagents. Because the energy is not evenly distributed throughout the MW chamber, a neon bulb array was used to determine a “cool” spot for greater control of temperature (Boon et al. 1990; Login and Dvorak 1993; Gu 1994; Giberson et al. 1995).
MWI Fixation of Liver Samples for both Plastic Embedding and Cryoultramicrotomy
All housing and husbandry methods were in accordance with the 1996 Guide for the Care and Use of Laboratory Animals. Livers from male rats were cut into 1-mm3 blocks that were rapidly transferred to Eppendorff tubes containing 3% formaldehyde and 0.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.2. The samples were placed in crushed ice to lower the temperature to 8C, removed from ice, and transferred to the MW oven. They were microwaved for 45 sec in a “cool” spot located by using the neon bulb display method with the temperature probe in the fixative and temperature feedback limit set to 37C.
Processing for Plastic-embedded Tissue in the Microwave
After washing at room temperature (RT), liver blocks were dehydrated in the MW oven through a graded series of aqueous ethanol solutions from 50 to 100% twice for 45 sec for each step at 45C, infiltrated with 1:1 ethanol:resin (low-viscosity Spurr's resin, Unicryl, or LR White) for 15 min, temperature set to 50C, then 100% of resin three times for 10 min at 50C with a restriction of 5 min. The tissue was transferred into BEEM capsules filled with fresh resin and polymerized as described by Giberson et al. (1995). In brief, small Parafilm squares were pressed into the lids of the BEEM capsules and snapped onto the resin-filled capsules. The capsules were then transferred into a Teflon holder (Ted Pella) that was placed in a 4-cup plastic dish, covered with water, and microwaved for 15 min to 95C and then unrestricted for 60 min. After polymerization, thin sections were cut with a Reichert Ultracut E.
Labeling of Plastic-embedded Sections in the Microwave
Immunolabeling was carried out at 37C unless otherwise specified. Briefly, the grids were floated on 30 μl of PBS containing 0.1% Tween-20 (PBST), followed by PBST containing 1% bovine serum albumin, 0.1% coldwater fish skin gelatin (Sigma Chemical; St Louis, MO) (PBST + BG), and irradiated for 5 min for each step at 37C to block nonspecific labeling with the MW oven probe in a “dummy drop” of solution. The grids were then transferred to the primary rabbit anti-catalase (Chemicon International; Temecula, CA) used at 10 μg/ml in PBST+BG and microwaved at 37C for 5, 10, or 15 min with or without a 2-min break in between irradiations. The grids were washed at RT with PBST+BG and incubated with 10-nm gold adducts of goat anti-rabbit IgG (BioCell; Cardiff, UK), used at 1:50 in blocking solution, in the same manner as the primary antibody. The grids were then washed in PBS and distilled water at RT, dried, and stained with 1% ethanolic uranyl acetate for 30 sec at 37C, then washed in distilled water and dried. Using MWI, the processing time from fixation to polymerization was substantially shortened (Table 1).
Immunolabeling and Thin Plastic Embedding of Frozen Sections in the Microwave
Cryoultramicrotomy was carried out essentially according to Tokuyasu (1980). Briefly, 1-mm3 liver blocks fixed in the MW oven as described above were cryoprotected by transferring them into a 0.5-ml drop of 2.3 M sucrose in PBS on a Parafilm-lined plastic Petri dish and microwaved for 5 min at 37C. MWI was stopped once so that the sucrose solution could be stirred. The liver blocks were placed on a cryoultramicrotome specimen pin, frozen in liquid nitrogen, and cryo-sectioned at –90C with glass knives in a Reichert Ultracut E microtome equipped with a cryochamber. The frozen sections were picked up with a drop of sucrose, transferred to carbon–Formvar-coated grids, and floated for 5 min face down on PBS to wash out the sucrose. The grids were then placed on a drop of 1% gelatin in PBS to decrease nonspecific labeling, microwaved for 45 sec at 37C, and washed by floating on PBS for 5 min at RT. The frozen sections were labeled in the MW at 37C for 5, 10, or 15 min as described above. In some experiments, the sections were irradiated continuously while others were interrupted for 2 min after every 5 min.
The immunolabeled frozen sections were then processed for ultrathin plastic embedding as previously described, except that the processing was done in the MW oven instead of at RT (Keller et al. 1984). After washing in PBS, the grids containing the frozen sections were postfixed by placing them on a drop of 1% filtered reduced osmium tetroxide and microwaved for 45 sec. They were then washed in distilled water before being placed in a Wheaton vial lid filled with 1% uranyl acetate in 50% ethanol and microwaved for 1 min. For dehydration, the uranyl was replaced with a series of 70, 90, and 100% ethanol solutions, allowing 30 sec in the MW for each step. For ultrathin embedding of the immunolabeled frozen sections, the grids were transferred to a 1:1 mixture of ethanol:LR White and microwaved at 37C for 5 min, followed by 100% LR White, and irradiated three times for 5 min with fresh LR White. The grids were placed in a Petri dish lined with a Whatman no. 50 hardened filter paper to allow excess LR White to drain before they were transferred to a fresh filter paper and microwaved for an additional 10 min to polymerize the resin. Using MWI from fixation to thin-embedding reduced the length of the procedure from roughly 7 to 2 h (Table 2). Control samples were processed at RT without any MW treatment, as described elsewhere (Keller et al. 1984).
Processing and immunolabeling of plastic-embedded tissue by MWI vs RT
Determination of Immunolabeling Density
Immunolabeled sections were viewed in a Philips CM12 TEM. Gold particles were counted in 10 randomly chosen peroxisomes in the hepatocytes. Ten micrographs of peroxisomes from liver tissue processed by each method were captured with a GATAN Retractable Multiscan digital camera. The number of gold particles in a 0.2-μm2 area of peroxisomes was determined with Digital Micrograph software and was used to extrapolate the average number of particles per μm2.
Results and Discussion
Since the pioneering work of Mayers (1970), MWI has been utilized to accelerate biological specimen processing for light and electron microscopy. Because it raises the temperature and greatly increases molecular motion (Boon et al. 1990), MWI enables chemicals such as fixatives to diffuse rapidly into cells and tissues. In addition, MW oven technology is now routinely used for antigen retrieval and to extend the range of antibodies that can be used on sections of fixed and processed tissue. It has been shown that performing the antibody reaction under controlled heating through MW irradiation results in significant improvements, such as shorter incubation times and lower antibody concentrations. MWI also facilitates the detection of antigens that are inaccessible or difficult to localize with conventional methods (Boon et al. 1990; Gu et al. 1995).
Processing and immunolabeling of frozen sections by MWI vs RT

Representative micrographs of sections from LR White-embedded rat liver fixed, processed, and immunolabeled in the MW oven with a polyclonal anti-catalase antibody are shown in Figure 1. Frozen sections similarly labeled and embedded in the MW by the ultrathin plastic embedding method are shown in Figure 2. The liver samples for both LR White and frozen sections were fixed by MWI in a primary fixative consisting of 3% formaldehyde and 0.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.2, which, in our experience, usually results in satisfactory preservation of cell structure and retention of immunoreactivity. The procedure we recommend for MW primary fixation and the subsequent immunolabeling of LR White or frozen sections is 45 sec of irradiation in the fixative solution with a limit of 37C, although shorter irradiation times on the order of 6 sec were reported to result in good preservation of the ultrastructure upon further processing in osmium and plastic embedding (Login et al. 1995). However, because different models of MW ovens are available, each tissue type and antigen must be experimented with individually to establish optimal conditions.
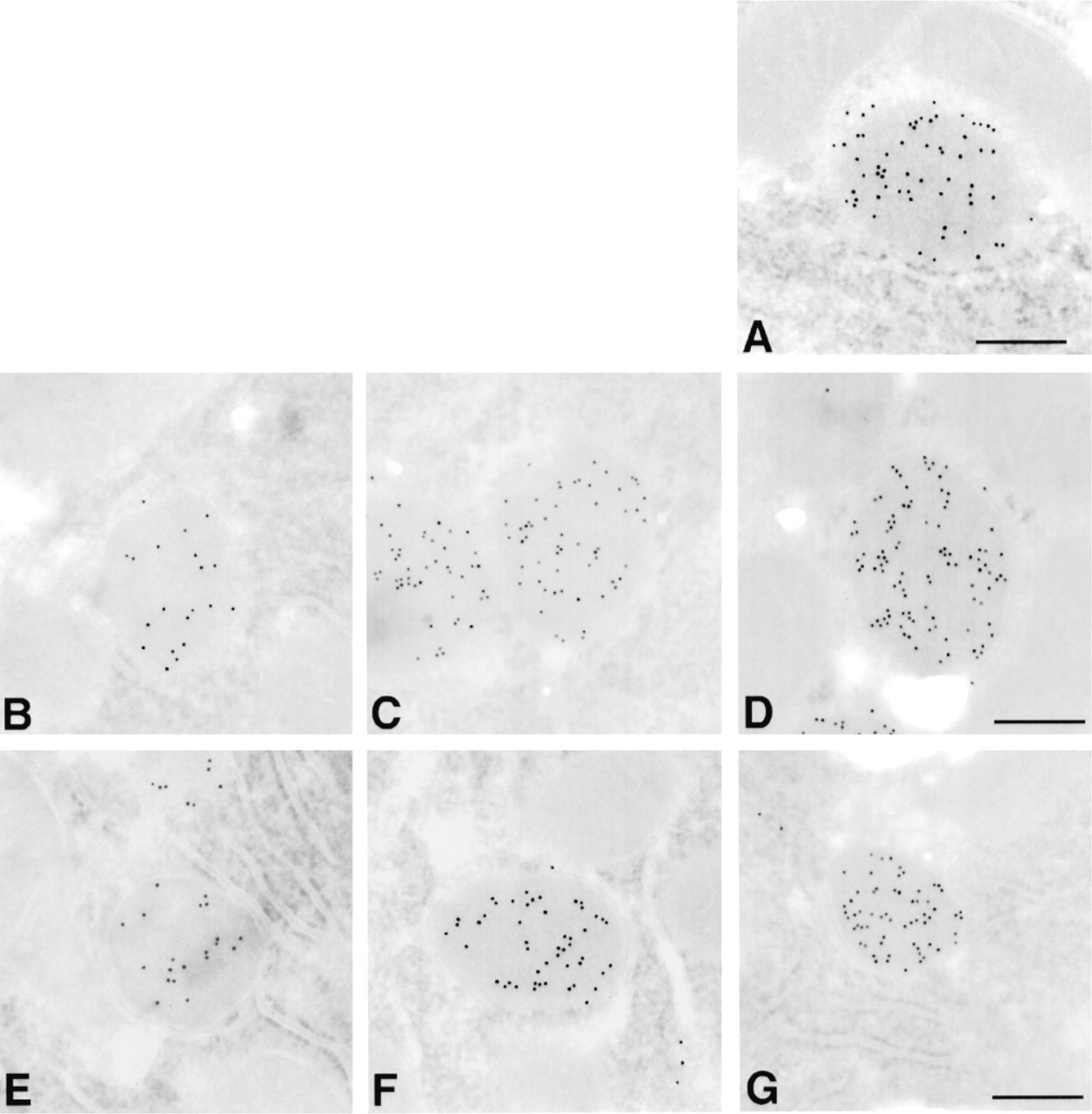
Comparison of immunolabeled LR White-embedded sections processed by microwave techniques using either continuous or interrupted irradiation. Frozen sections of rat liver were labeled for peroxisomes using rabbit anti-catalase antibody followed by 10-nm gold adducts of goat antibody to anti-rabbit IgG. (
Although good preservation of ultrastructure can be obtained in the absence of osmium, the organelle membranes are usually not contrasted in plastic-embedded samples that have not been osmicated. In the past, although cryoultramicrotomy provided high immunore-activity because of osmium treatment, dehydration, infiltration, and resin embedding omission, the ultra-structural preservation of frozen sections was often unsatisfactory. However, when the method of ultrathin embedding is carried out either at RT or in the MW as described here, frozen sections that are postosmicated after the immunolabeling step retain the detailed ultrastructural delineation of conventional osmium staining and plastic embedding (Keller et al. 1984, 1989).
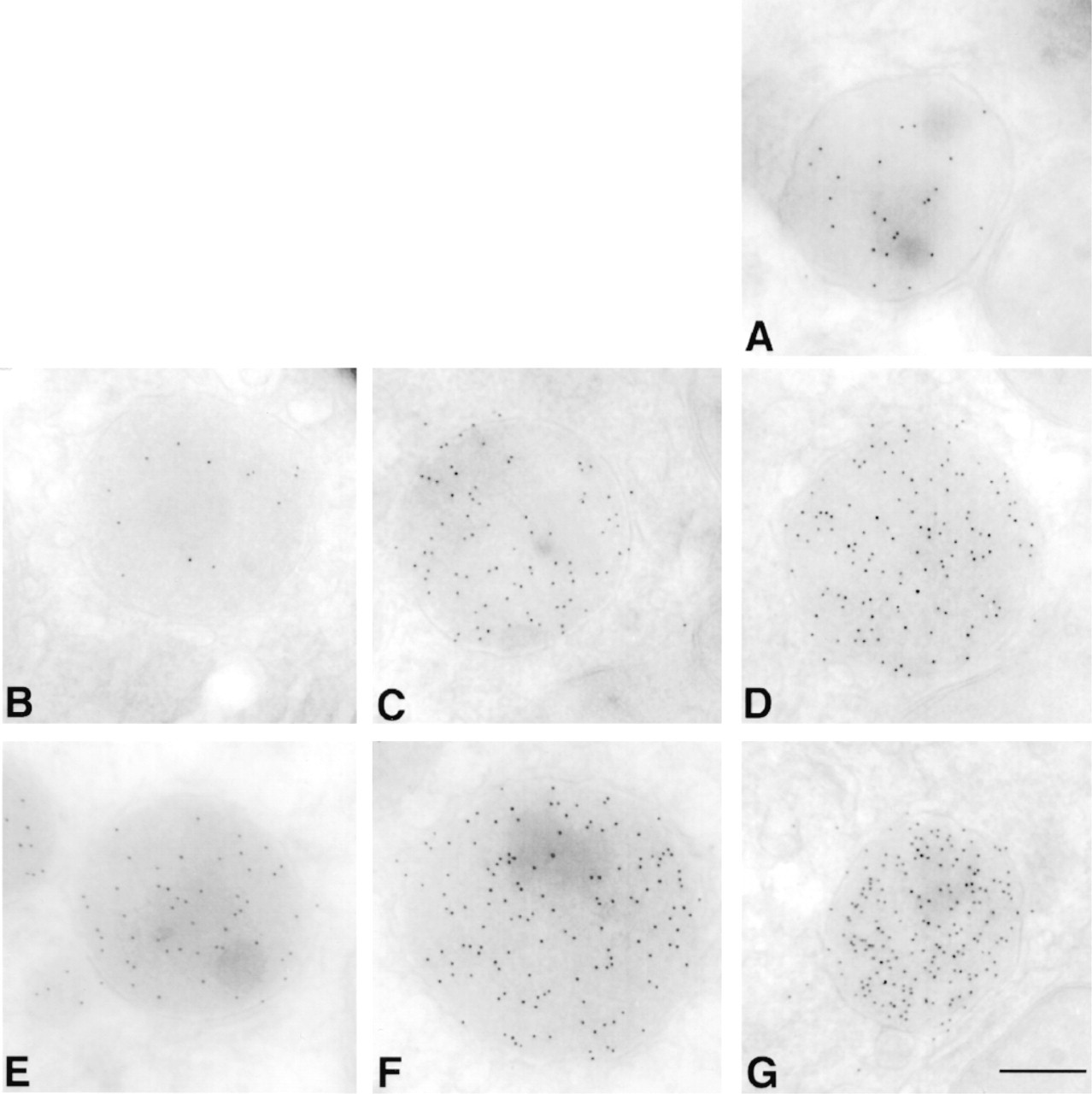
Comparison of cryoimmunolabeled sections processed by microwave techniques using either continuous or interrupted irradiation. Frozen sections of rat liver were labeled for peroxisomes using rabbit anti-catalase antibody followed by 10-nm gold adducts of a goat antibody to anti-rabbit IgG. (
Immunolabeling Density in Sections of Liver Embedded in Acrylic Resins
We found that the labeling density for peroxisomal catalase in sections from liver embedded in Spurr's resin was low, even after etching with metaperiodate. Unicryl-embedded liver samples prepared by MWI suffered from poor morphology, although the immunolabeling density was comparable to that obtained on LR White-embedded liver sections. Consequently, we did not pursue experimenting with these resins.
We did find that the immunolabeling density of LR White sections from blocks fixed and processed by MWI was substantially increased over that the samples labeled the conventional way. Whereas the immunolabeling density was 118 ± 26 particles/μm2 for the samples labeled at RT, continuous irradiation for 5, 10, or 15 min resulted in an immunolabeling density of 90 ± 10, 273 ± 24, and 448 ± 25 particles/μm2, respectively. When a 2-min break after every 5 min of MWI was allowed, the labeling density increased to 111 ± 12, 290 ± 28, and 447 ± 48 particles/μm2 after 5, 10, and 15 min, respectively (Figure 3).
Immunolabeling Density on Liver Frozen Sections
We made similar observations when we quantified the number of gold particles obtained on sections fixed and immunolabeled by the conventional method of preparation vs MWI. When frozen sections were incubated at RT for 30 min each in the primary and secondary antibodies, the labeling density was 146 ± 13 gold particles per μm2 of peroxisomal matrix. The samples irradiated continuously displayed a low labeling density at 5 min (51 ± 5 gold particles per μm2), which increases incrementally at 10 min (203 ± 20) and 15 min (287 ± 24). The labeling density increased even more dramatically when a 2-min pause was allowed in between irradiations. At 5-min irradiation with a pause, the labeling density was 140 ± 16 particles/μm2, approximately the same density achieved with conventional techniques (146 ± 13). At 10 min, the labeling density increased to 301 ± 38 particles/μm2, and at 15 minutes to 472 ± 48 particles/μm2, an almost fourfold gain over the conventional immunolabeling protocol. The longer periods of irradiation, along with a 2-min pause after every 5 min, showed a labeling density that was higher than that achieved with the conventional method and with continuous irradiation (Figure 3). In both plastic-embedded and frozen sections, the nonspecific background level remained low and was not enhanced by MWI.
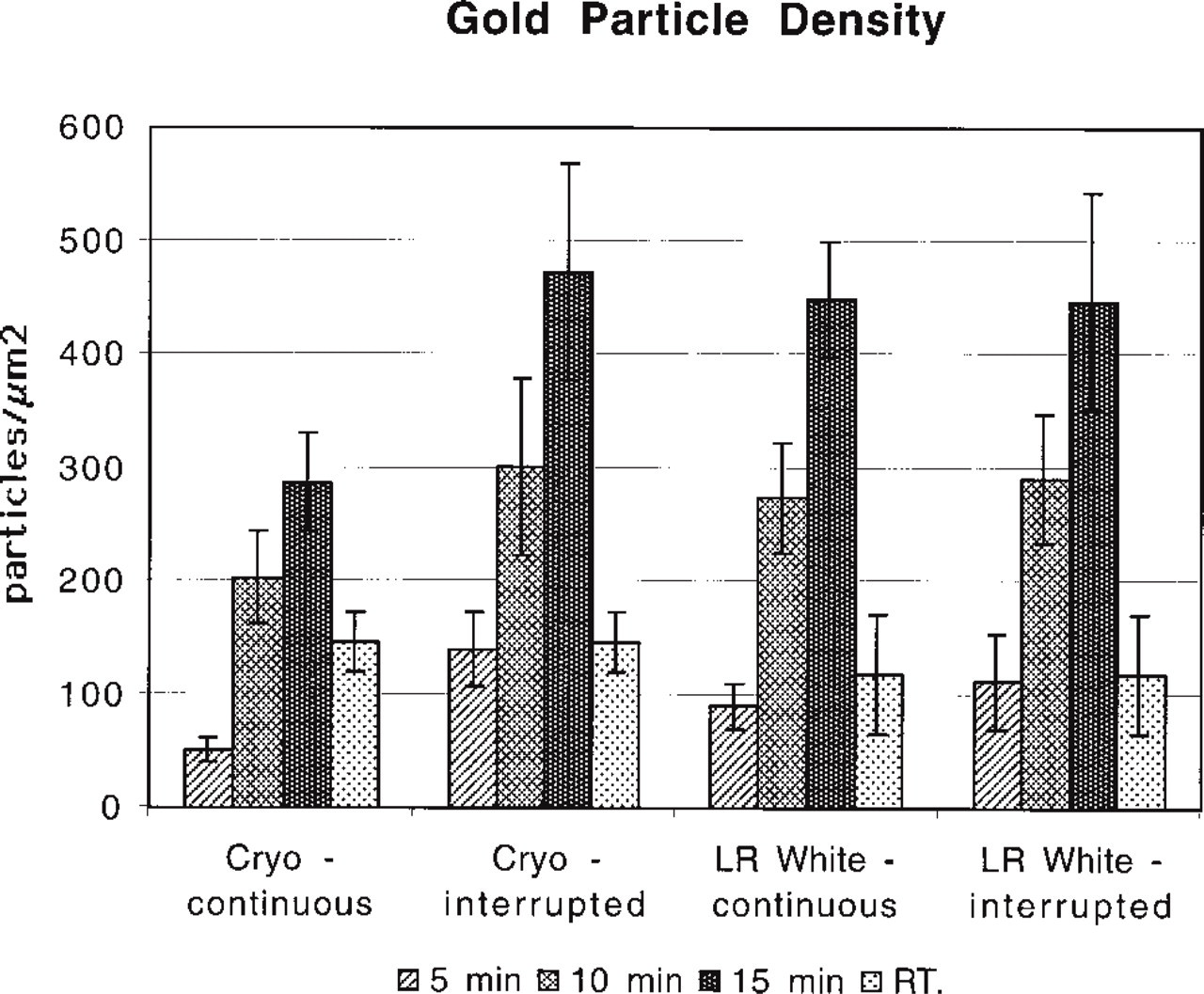
Comparison of gold labeling density on LR White-embedded and cryosectioned liver sections in continuous or interrupted irradiation mode. As microwave irradiation time increases, so does the immunolabeling density. The protocol that includes a 2-min interruption results in greater labeling density than that of continuous irradiation.
After 15 min of irradiation, the increase in labeling density plateaued in both plastic-embedded and frozen sections (data not shown). The plateau may be caused by detrimental effects of evaporation causing changes in the concentration of the antibody, molarity of the salts, and strength of the pH (Gu et al. 1995). Although MWI can increase the number of antigen-antibody interactions, higher temperatures may interfere with the stability of the binding. It is important to allow at least a 2-minute pause at RT after irradiation to allow the formation of stable antibody-antigen complexes that can withstand subsequent washings (Choi et al. 1995). The MW fixation, processing, and immunolabeling protocols that we recommend are described in Table 1.
To summarize, MWI provides an effective method for high-resolution immunoelectron microscopy that can be applied to the processing and immunolabeling not only of plastic-embedded but also of frozen sections. Because the immunolabeling density of samples in LR White (448 ± 25 particles/μm2) and frozen sections (472 ± 48 particles/μm2) was fairly similar after 15 min of MWI (Figure 3), we recommend carrying out immunolabeling experiments in LR White-embedded material when there are a large number of specimens to be immunolabeled. However, immunolabeling of frozen sections is still our method of choice, especially when the maximal retention of both cellular ultrastructure and antibody binding capacity of protein antigen in the specimen is required.
