Abstract
A number of monoclonal antibodies (MAbs) that recognize human follicular dendritic cells (FDCs) have been identified. Although some of them have already been applied individually in routine immunolabeling using formalin-fixed paraffin sections for diagnostic and experimental purposes, many antibodies are still employed only for immunolabeling using cryostat sections or particularly processed sections because they have been thought unsuitable for routine sections. A comprehensive examination re-evaluating their suitability in paraffin sections has not been reported. Accordingly, there is limited ability to examine the immunopathological contribution or diagnostic value of FDCs using routinely processed specimens or archived materials. In this study a broad panel of antibodies was systematically applied to the immunolabeling of paraffin sections of reactive tonsils or lymph nodes, in combination with advanced antigen retrieval (AR) techniques. Several antibodies, including Ki-M4p, X-11, 12B1, CNA.42, 1F8/BU32 (anti-CD21), BU38/1B12 (anti-CD23), Ber-MAC-DRC/To5 (anti-CD35), 1.4C3 (anti-CD106), NGFR5 (anti-nerve growth factor receptor p75), IIH6 (anti-CD55), 55K-2 (anti-fascin), and anti-S100 protein α-chain, were found to label FDCs in routine sections when combined with suitable AR techniques. Our results are easily adaptable for routine practice and provided useful suggestions concerning the immunopathological behavior and diversity of the particular cells.
Keywords
F
Within the past two decades, a number of monoclonal antibodies (MAbs) that recognize human FDCs have been developed to define the immunophenotype of these unique cells in cell suspensions or in tissue sections (see Table 1; reviewed in Maeda et al. 1995). Some of these have already been applied in routine immunolabeling using formalin-fixed paraffin sections for experimental and diagnostic purposes (Tanaka 1986; Tabrizchi et al. 1990; Murray et al. 1991; Nguyen et al. 1994; Dorfman and Shahsafaei 1996; Maeda et al. 1996; Raymond et al. 1997; Said et al. 1997; Bagdi et al. 2001). However, many have been employed exclusively for immunohistochemical labeling of cryostat sections of unfixed frozen materials or tissues fixed with special fixatives such as PLP (periodate-lysine-paraformaldehyde) solution (McLean and Nakane 1974). They have been considered unsuitable for immunolabeling of conventional paraffin sections because initial immunolabeling with them failed to demonstrate definitive reactivity with human FDCs in the paraffin sections. Therefore, the ability to investigate the characteristics and behavior of human FDCs using routine pathological specimens or archived materials has been limited.
Advanced techniques for antigenic retrieval (AR) and the effective enhancement of immunohistochemical sensitivity have been developed (Pileri et al. 1980, 1997; Finley and Petrusz 1982; Shi et al. 1991; Cattoretti et al. 1993; von Wasielewski et al. 1994). These are useful not only for samples subjected to prolonged formalin fixation but also for facilitating immunostaining with antibodies that were previously considered unsuitable for routinely fixed and processed materials. In the present study, using these techniques, a broad panel of MAbs already known to react with human FDCs was comprehensively tested with respect to suitability for use in conventional paraffin sections of human reactive tonsils or lymph nodes. These fundamental examinations will contribute to the expansion of immunopathological research and diagnostic value concerning FDCs, especially in human systems.
Materials and Methods
Tissues
Reactive tonsil tissue samples (n = 6) were obtained from patients undergoing therapeutic tonsillectomy due to chronic tonsillitis. Lymph nodes (n = 10) were obtained from patients undergoing surgical extirpation of unusually swollen lymph nodes for pathological examination or from patients undergoing surgical removal of the regional lymph nodes caused by gastric or colon carcinoma. These tissues were fixed in 10% formalin solution for periods from 12 hr to 3 days and then processed routinely to obtain 3-μm-thick conventional paraffin sections.
Monoclonal Antibodies
The MAbs employed in the present study are listed in Table 1. The reactivity of each was checked in advance using cryostat sections of unfixed, frozen human tonsil tissues.
Antigen Retrieval
Formalin fixation and the subsequent processes necessary to obtain paraffin sections appear to cause some masking or denaturing of antigenic epitopes, leading to weak or false-negative immunoreactivity. To remove such masking effects and recover the original immunoreactivity, several AR (unmasking) techniques have been developed (Pileri et al. 1980, 1997; Finley and Petrusz 1982; Shi et al. 1991; Cattoretti et al. 1993; von Wasielewski et al. 1994). To examine their effect in immunolabeling of human FDCs, the following procedures were performed in the present study. Initially, the paraffin sections were dewaxed in xylene, rehydrated through a series of graded ethanol solutions, and submerged in methanol containing 0.3% H2O2 to block endogenous peroxidase activity. They were then treated using one of the following methods:
1. Digestion with 0.1% trypsin (trypsin 1:250; Difco, Detroit, MI) at 37C for 30 min (regular proteolytic treatment).
2. Digestion with 0.1% trypsin at 37C for 90–120 min (prolonged proteolytic treatment).
3. Microwave (MW) irradiation (Bio-Rad Polaron H2500 MW processor; Watford, UK) with 0.1 M citrate buffer (pH 6.0) three times for 7 min.
4. MW irradiation with 1 mM EDTA buffer (pH 8.0) three times for 7 min.
5. No treatment (control).
Immunoperoxidase Labeling Using the ABC (Avidin-Biotin-Peroxidase Complex) Method
After the AR procedure, the sections were labeled using a highly sensitive immunoenzymatic technique (ABC method) as described by Hsu et al. (1981a,b). Briefly, the sections were thoroughly rinsed with PBS and incubated in 5% bovine serum albumin to prevent nonspecific binding of the antibodies. They were then incubated with each primary monoclonal antibody at 4C overnight. On the following day they were washed three times in PBS and incubated with biotinylated rabbit anti-mouse IgG+IgA+IgM antibody [HISTOFINE PO(M) kit; Nichirei, Tokyo, Japan] for 1 hr at ambient temperature. The sections were then washed three times in PBS and incubated with a pre-formed avidin and biotinylated horseradish peroxidase macromolecular complex (Vectastain Elite ABC KIT; Vector Labs, Burlingame, CA) for 1 hr at ambient temperature. After incubation, the sections were washed in PBS and in Tris-HCl buffer and the peroxidase activity was developed using 0.05% diaminobenzidine (DAB) with 0.03% H2O2 as described by Graham and Karnovsky (1966). They were counterstained with methyl green and mounted with Permount (Fisher Scientific; Fair Lawn, NJ) for light microscopic observation.
As a control, inappropriate antibodies of the same isotype were used instead of the primary antibody. In addition, control experiments were performed to determine the reactivity of the biotinylated secondary antibodies and ABC and to quantify endogenous peroxidase activity.
Results
The results of the present study are comprehensively summarized in Table 2. The staining pattern for each MAb was identical in both reactive tonsil tissue and lymph node specimens, and the results are presented together to avoid repetition. In addition, the period of fixation had no significant effect on the results of immunolabeling. In fact, no significant difference was observed between the immunolabeling of the tissue sections fixed for 12 hr (minimal duration) and those fixed for 3 days (maximal duration).
Monoclonal antibodies employed in the present study a
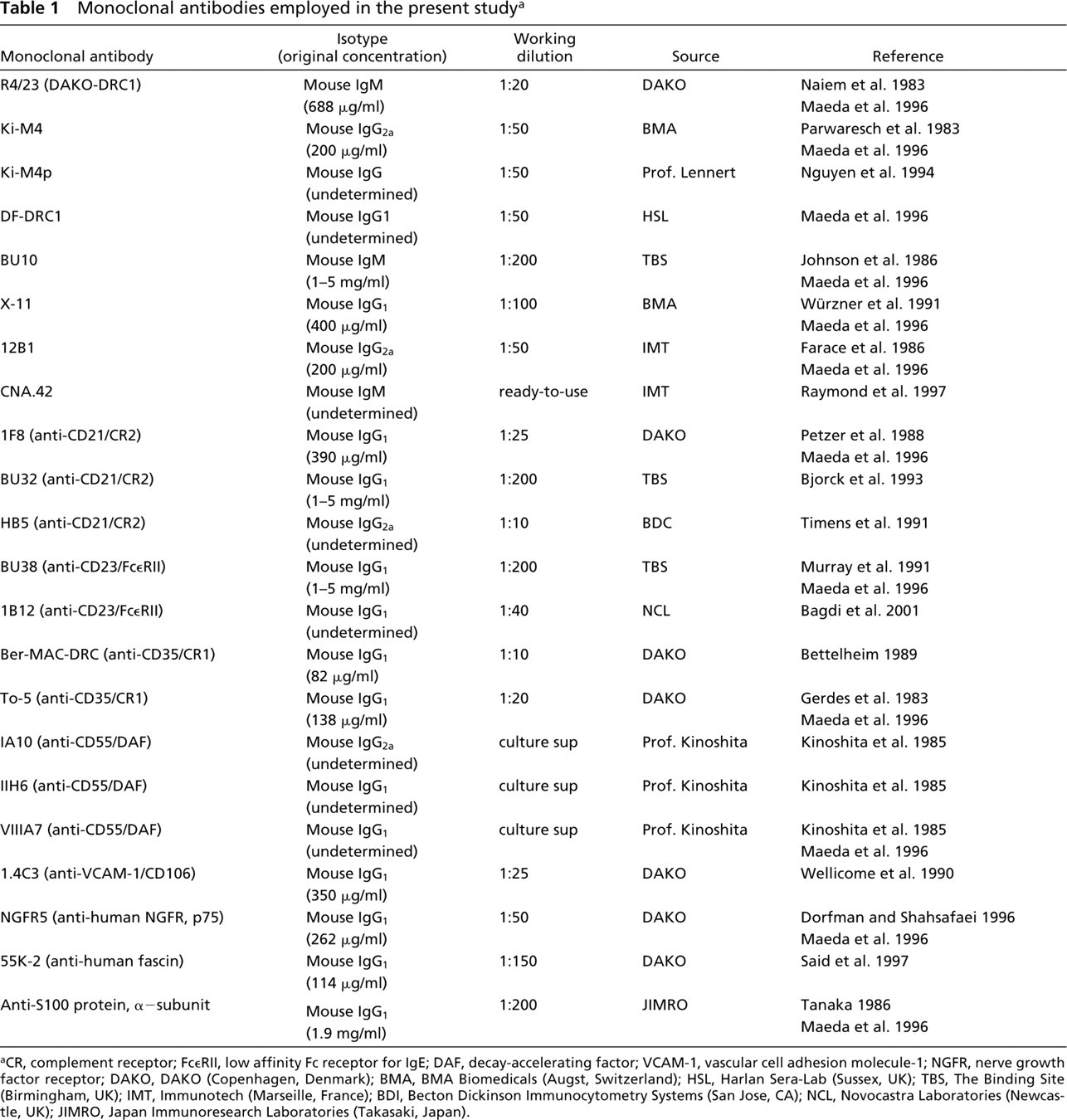
aCR, complement receptor; Fc∊RII, low affinity Fc receptor for IgE; DAF, decay-accelerating factor; VCAM-1, vascular cell adhesion molecule-1; NGFR, nerve growth factor receptor; DAKO, DAKO (Copenhagen, Denmark); BMA, BMA Biomedicals (Augst, Switzerland); HSL, Harlan Sera-Lab (Sussex, UK); TBS, The Binding Site (Birmingham, UK); IMT, Immunotech (Marseille, France); BDI, Becton Dickinson Immunocytometry Systems (San Jose, CA); NCL, Novocastra Laboratories (Newcastle, UK); JIMRO, Japan Immunoresearch Laboratories (Takasaki, Japan).
MAbs Relatively Specific for Human FDCs (R4/23, Ki-M4, Ki-M4p, DF-DRC1, BU10, X-11, 12B1, and CNA.42)
Among eight clones of MAbs known to be relatively specific markers of human FDCs, Ki-M4p, X-11, 12B1, and CNA.42 produced web-like immunolabeling within the germinal center (GC) and/or mantle zone (MZ) of lymphoid follicles (corresponding to the distribution of FDCs) on conventional paraffin sections when suitable AR had been performed (Figure 1).
Immunohistochemical reactivity of the examined monoclonal antibodies with human FDCs in formalin-fixed paraffin sections
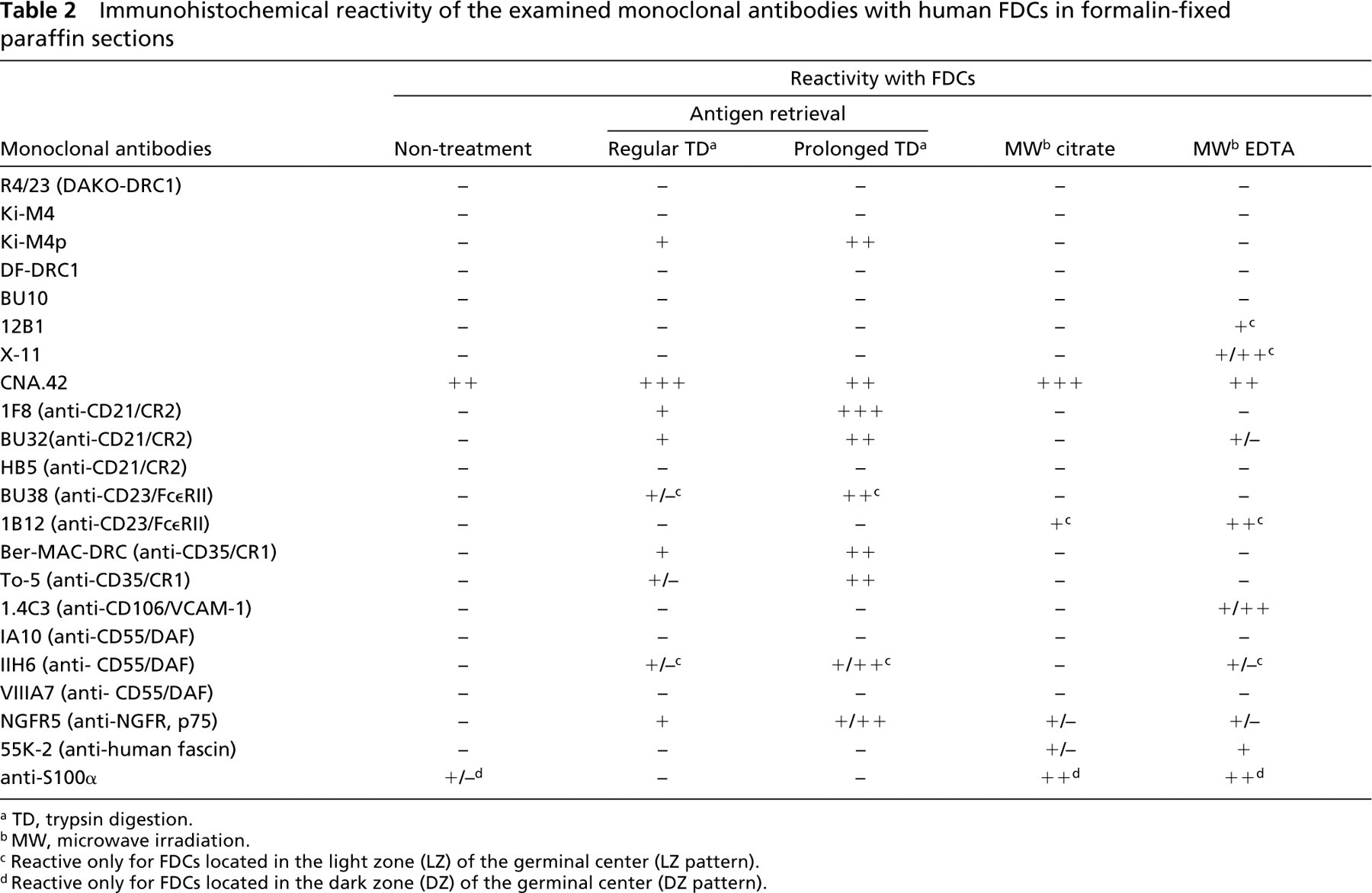
a TD, trypsin digestion.
b MW, microwave irradiation.
c Reactive only for FDCs located in the light zone (LZ) of the germinal center (LZ pattern).
d Reactive only for FDCs located in the dark zone (DZ) of the germinal center (DZ pattern).
Trypsin digestion, especially prolonged digestion (over 90 min), was significantly effective for Ki-M4p labeling, and microwave (MW) irradiation with EDTA solution was significantly effective for labeling of X-11 and 12B1. Sometimes prolonged trypsin digestion led to some deterioration in morphology in the sections. The degree of deterioration depended on the conditions of the specimens and the duration of the digestion. Nevertheless, the reactivity of the immunolabeling was well preserved and the essential purpose of the labeling was successfully accomplished even if the morphology was somewhat deteriorated.
MAb CNA.42 revealed intense reticular labeling within lymphoid follicles when all of the AR methods were applied and even when no treatment was performed.
None of the other antibodies displayed definitive reactivity in any of the tissues examined, even when the AR methods were applied (data not shown).
12B1 and X-11 reactivities were restricted to FDCs located in the light zone (LZ) of GCs (LZ pattern), whereas those of Ki-M4p and CNA.42 extended through almost the whole of the lymphoid follicle, including the MZ (Figure 1).
No cell types other than FDCs were labeled with these antibodies, except for a few plasmacytoid cells or certain mononuclear non-lymphoid cells scattered in interfollicular areas, the latter in particular being labeled with CNA.42 as described in the original paper characterizing this antibody (Raymond et al. 1997).
MAbs Against Human Complement Receptor (CR) 2/CD21 (1F8, BU32, and HB-5)
When either 1F8 or BU32 was employed as the primary antibody and trypsin digestion was performed for AR, intense labeling within lymphoid follicles was observed in every section of all tissues examined. Figure 2 shows 1F8 labelings representing them. The labelings produced a web-like reticular pattern in almost the entire area of the GCs and sometimes extended into the MZ. Prolonged (over 90 min) trypsin digestion produced more effective unmasking than regular digestion (Figure 2). A few plasma cells and immunoblastoid cells distributed in interfollicular areas were also labeled, but lymphoid cells, especially those in the MZ, were not significantly labeled with these antibodies. MW irradiation was not effective in the labeling, and neither was non-treatment (Figure 2).
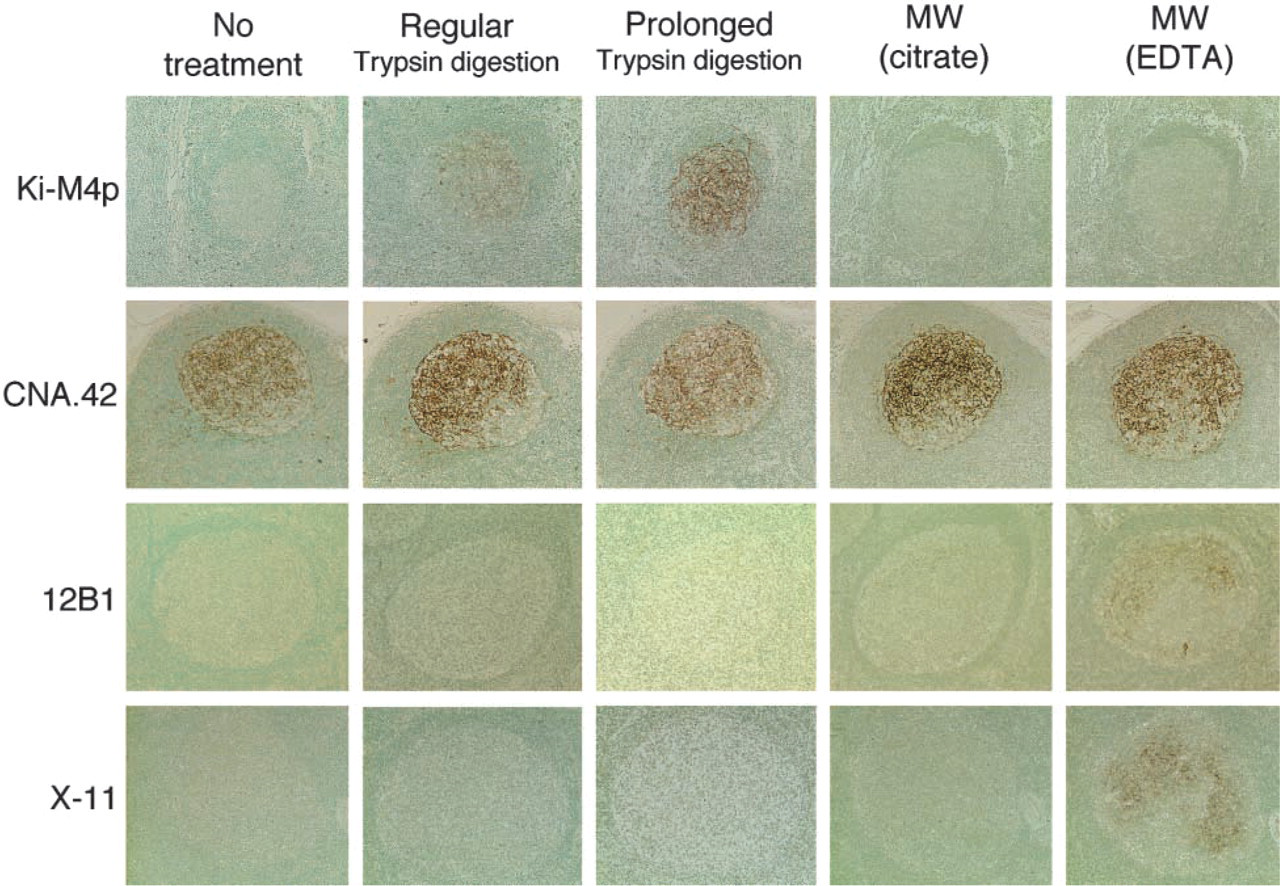
Immunohistochemical reactivity of MAbs against human FDCs (Ki-M4p, CNA.42, 12B1, and X-11) in paraffin sections of the reactive lymph nodes or tonsils in various AR conditions. Immunoperoxidase labeling using ABC method. Counterstained with methyl green. X20 objective.
In contrast, HB-5 was not effective in any of the sections, regardless of the AR performed (Figure 2).
MAbs Against Human CR1/CD35 (To5 and Ber-MAC-DRC)
To5 and Ber-MAC-DRC produced intense web-like labeling corresponding to the lymphoid follicles in paraffin sections of the tissues examined (Figure 2). The labels extended through almost the entire area of the GCs and sometimes into the MZ, similar to the labeling described above for anti-CD21. Prolonged trypsin digestion also produced more effective unmasking than regular digestion. A few plasma cells and immunoblastoid cells in interfollicular areas were also labeled. MW irradiation or no treatment produced no significant immunolabeling within lymphoid follicles in the paraffin sections.
MAbs Against Human Low-affinity Fce Receptor (Fc∊RII)/CD23 (BU38 and 1B12)
MAbs BU38 and 1B12 displayed web-like immunoreactivity within the GC of the lymphoid follicles (Figure 2). The labeling was restricted to the LZ, especially the apical LZ, of the GCs, and very faint or no labeling was observed in the dark zone (DZ) of the GCs (LZ pattern). Small lymphocytes within the MZ were also labeled with these antibodies but their labeling was less intense than those of FDCs. Interestingly, trypsin digestion, especially, prolonged treatment and produced effective unmasking for BU38, whereas MW irradiation, with EDTA in particular, was quite effective for 1B12 (Figure 2).
MAb Against Human Vascular Cell Adhesion Molecules (VCAM)-1/CD106 (1.4C3)
Anti-human VCAM-1/CD106 MAb, 1.4C3 produced web-like labeling in the GC of lymphoid follicles when MW irradiation with EDTA solution was used (Figure 3). The reactivity was exclusively the LZ pattern. The vascular endothelial cells were also labeled but very weakly. Trypsin digestion and MW irradiation with citrate buffer did not produce any reactivity, and neither did an absence of treatment.
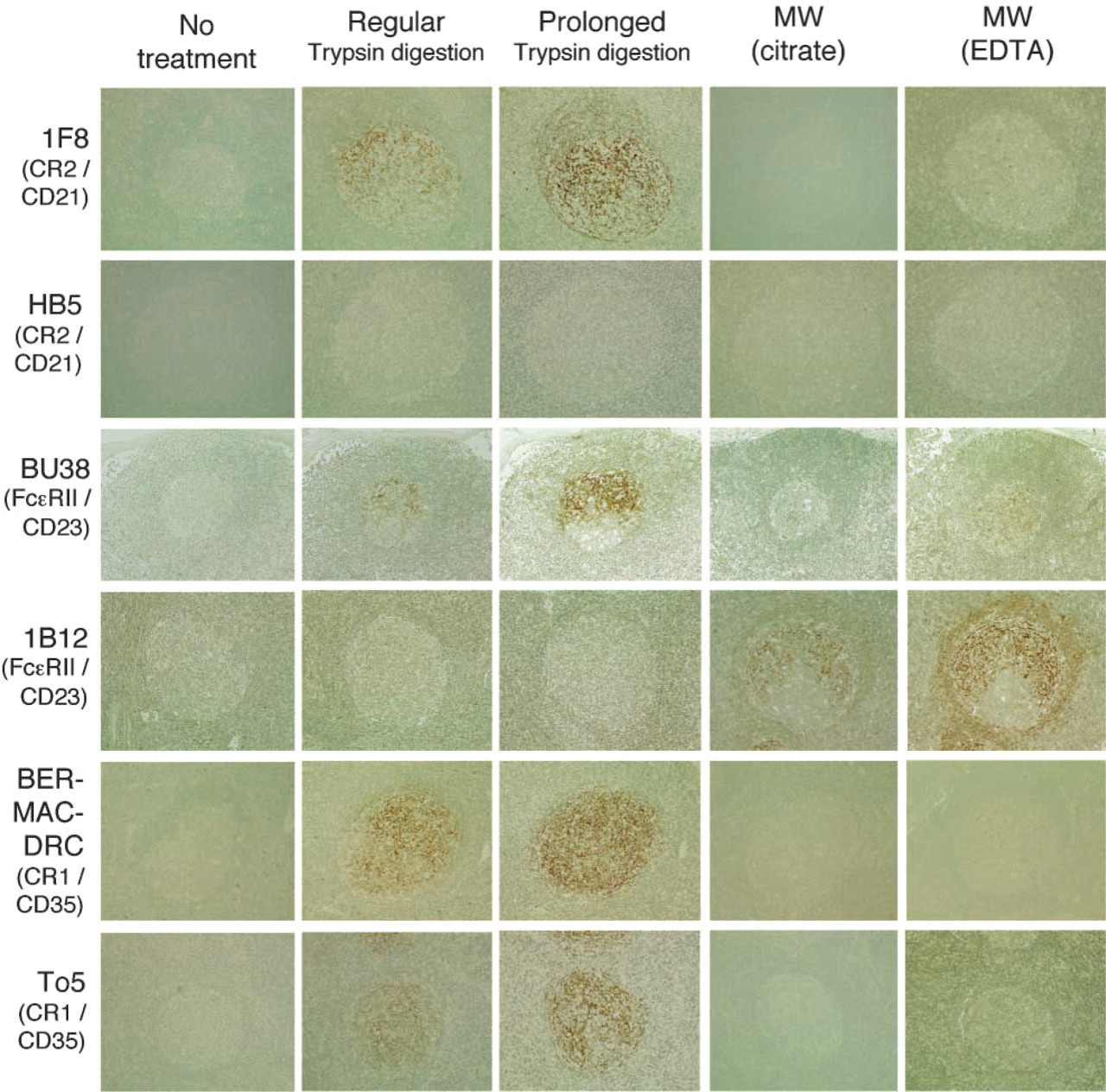
Immunohistochemical reactivity of MAbs against human CD21 (1F8 and HB5), CD23 (BU38 and1B12), and CD35 (Ber-MAC-DRC and To5) in paraffin sections of the reactive lymph nodes or tonsils in various AR conditions. Immunoperoxidase labeling using ABC method; counterstained with methyl green. X20 objective.
MAbs Against Human Decay-accelerating Factor (DAF)/CD55 (IA10, IIH6, and VIIIA7)
Among three clones of the antibodies, only IIH6 revealed definitive web-like immunoreactivity within the GC of the tissues examined (Figure 3). The labeling was of the LZ pattern and trypsin digestion, especially prolonged digestion, produced effective unmasking (Figure 3). Some plasmacytoid cells scattered in inter-follicular areas were also labeled with this antibody.
MAb Against Human Low-affinity Nerve Growth Factor Receptor (NGFR) p75 (NGFR5)
The anti-human NGFR MAb NGFR5 produced web-like immunolabeling in the GC of lymphoid follicles when trypsin digestion was performed (Figure 3). The reactivity extended through the whole of the GC and sometimes in the MZ. In addition, the basal borders of the squamous epithelium that covered the mucosal surface of the tonsils were also labeled by this antibody (data not shown). MW irradiation or an absence of treatment led to ineffective AR except for faint labeling when MW with EDTA was used.
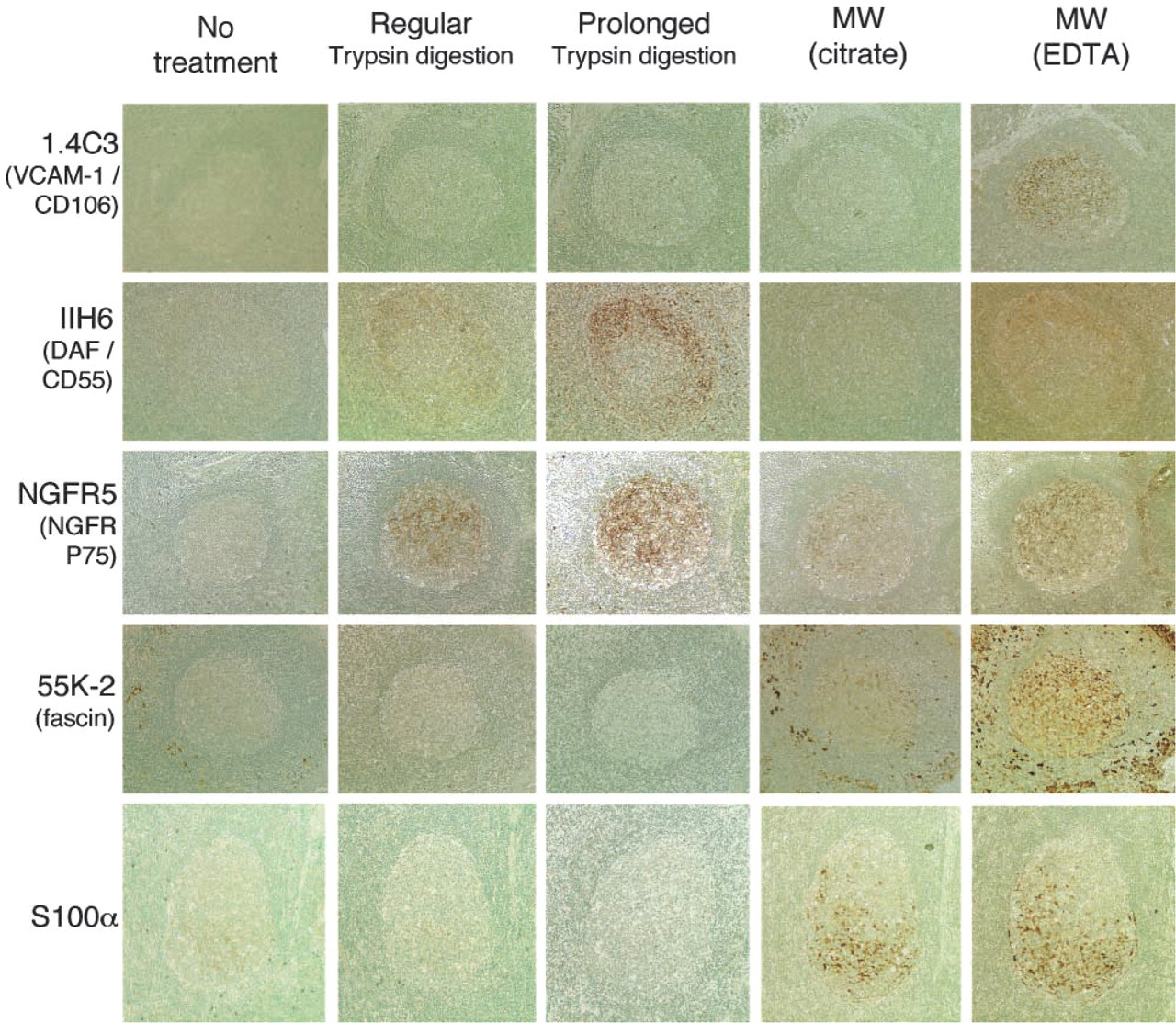
Immunohistochemical reactivity of monoclonal antibodies against human VCAM-1/CD106 (1.4C3), DAF/CD55 (IIH6), NGFR (NGFR5), fascin (55K-2), and S100 protein α-chain (S100α) in paraffin sections of reactive lymph nodes or tonsils in various AR conditions. Immunoperoxidase labeling using ABC method; counterstained with methyl green. X20 objective.
MAb Against Human Fascin (55K-2)
When MW irradiation with citrate buffer was performed or no treatment was undertaken, the MAb 55K-2 intensely labeled dendritic cells (DCs) distributed in interfollicular areas (interdigitating cells or related cells), whereas it did not produce any significant reactivity within lymphoid follicles, as shown in Figure 3. In contrast, when MW irradiation with EDTA solution was used, this antibody labeled not only the interfollicular DCs but also FDCs within GCs (Figure 3). The labeling extended through the whole of the GC and MZ but it was less intense than those of interfollicular DCs. Trypsin digestion induced loss of the immunoreactivity for both FDCs and interfollicular DCs.
MAb Against S100 Protein α-chain (S100α)
An MAb against S100α produced intense immunolabeling of the cytoplasm and nucleus of dendritic-shaped cells located predominantly in the DZ of the GC (Figure 3). MW irradiation with either citrate buffer or EDTA solution was significantly effective for antigen retrieval. The labeling disappeared when trypsin digestion was used.
Except for a few histiocytoid/stromal cells scattered in subcapsular sinus of lymph nodes or interfollicular connective tissue septa of tonsils, no other cells were labeled with S100α. There was no significant labeling of lymphoid cells with this antibody.
Discussion
Formalin fixation and the subsequent processing (e.g., dehydration, heating) of tissues for routine pathological purposes causes unpredictable alterations in antigenic molecules, which have often been held responsible for the poor reproducibility of immunohistochemical studies. Within the past two decades, highly sensitive immunolabeling methods, such as the ABC method (Hsu et al. 1981a, b), AR techniques using proteolytic enzymatic digestion (Pileri et al. 1980, 1997; Finley and Petrusz 1982), and MW irradiation (Shi et al. 1991; Cattoretti et al. 1993; von Wasielewski et al. 1994) have been described as potential tools for enhancing immunolabeling in routinely processed paraffin sections. The mechanisms by which these approaches allow the detection of most antigens in paraffin sections have not been fully defined. The proteolytic pretreatments are believed to break some of the links produced by formalin fixation and to expose a higher number of antigenic epitopes (Pileri et al. 1980). Cattoretti and Suurmeijer (1995) suggested in their review that “heat and hydrolysis may both denature and break the tissue proteins at or near the links made by formalin between adjacent amino acids” and thought it “conceivable that self-assembly of unfolded protein chains with subsequent restoration of antigenic sites occurs when the retrieval solution is allowed to cool.” In addition, Morgan et al. (1994) have reported that tight complexing of calcium ions or other divalent metal cations with proteins during formaldehyde fixation can be responsible for masking certain antigens. Therefore, the chelation or precipitation of these ions can represent a critical step in salt-mediated AR. In the present study, we have successfully established that a number of MAbs, including Ki-M4p, X-11, 12B1, CNA.42, 1F8, BU32, BU38, 1B12, To5, Ber-MAC-DRC, 1.4C3, IIH6, NGFR5, 55K-2, and anti-S100α, can label human FDCs in paraffin sections immunohistochemically when combined with a sensitive immunolabeling method and suitable AR techniques.
It has already been reported that some of these antibodies react with human FDCs, even in paraffin sections (Tanaka 1986; Murray et al. 1991; Nguyen et al. 1994; Dorfman and Shahsafaei 1996; Maeda et al. 1996; Raymond et al. 1997; Said et al. 1997; Bagdi et al. 2001). Indeed, they are actually used as markers of cells in routine diagnostic practice, especially 1F8 (anti-CD21), BU38/1B12 (anti-CD23), Ber-Mac-DRC (anti-CD35), and CNA.42. However, for reasons that follow, single or small numbers of markers are not enough to recognize the specific or related cells, including their precursor forms or transformed cells, in tissue sections: First, none of the antibodies is completely specific for FDCs but reacts with certain cell types other than FDCs, as described in the Results. Second, some of the molecules expressed on the surface of FDCs, such as CD23, are quantitatively and qualitatively changeable in various pathological conditions (Masuda et al. 1989; Bagdi et al. 2001). The present results beneficially expanded the markers of human FDCs available in paraffin sections. The broad panel of markers allows a more rigorous specificity in the definition of FDCs or related cells immunohistochemically in the tissue sections, and the realization of the possible heterogeneity or cellular differences related to either the functional phase or the maturation process.
Despite recognizing CD21/CR2, as did 1F8 and BU32, HB5 did not produce any positive labeling even if satisfactory enzymatic digestion or MW irradiation was carried out. A possible explanation for this might be that HB5 recognizes different fixation- or processing-sensitive epitopes from the other antibodies. Interestingly, antibodies against CD21 (BU32 and 1F8) produced moderate labeling of B-cells, especially the cells located in the MZ of lymphoid follicles, and strong labeling of FDCs in frozen sections. However, whereas the staining of B-cells was reduced or abolished in paraffin sections, the labeling of FDCs in paraffin sections remained as strong as in frozen sections. This phenomenon suggests that FDCs might express extremely large amounts of the CD21 antigen compared with B-cells, or that FDCs might express somewhat different epitopes. Indeed, Liu et al. (1997) reported that the CD21 molecules expressed on the surface of FDCs were a different isoform (CD21L) from those on B-cells (CD21S). This isoform might be resistant to formalin fixation and the subsequent processes involved in preparing paraffin sections.
Bagdi et al. (2001) reported that heat-induced epitope retrieval with DAKO Target Retrieval Solution (TRS) was more effective and that trypsin pretreatment was inconsistent for 1F8/CD21 labeling. In contrast to their report, the current study revealed that 1F8/CD21 produced excellent results even when trypsin digestion was performed, although heat-induced epitope retrieval with DAKO TRS was not performed. Because the antibody originally reacted with an epitope in 95-, 72-, 50-, 32-, and 28-kD trypsinized fragments of CD21 (Petzer et al. 1988), trypsin digestion should not in theory influence the antigenicity of the epitope and would be expected to be one of the effective retrieval treatments. In fact, other workers (Mori 1994; Maeda et al. 1996; Perez-Ordonez et al. 1996) have reported that proteolytic pretreatment produced effective retrieval for 1F8/CD21 labeling, as was found in this study. Because prolonged treatment significantly enhanced the immunoreactivity of FDCs for 1F8/CD21 labeling in the current study, insufficient digestion might cause inconsistent or poorly reproducible results.
Some plasmacytoid or immunoblastoid cells distributed in interfollicular areas were also labeled with 1F8 or BU32 in combination with trypsin predigestion. Such crossreactivity did not seem to be caused by nonspecific binding of the antibodies or the following ABC reagents employed because the isotype control did not demonstrate any significant reactivity with either FDCs or the plasmacytoid cells, even when sufficient trypsin digestion was performed. The labeling process, especially trypsin predigestion, might denature the conformation of immunoglobulins or related molecules to generate some common epitopes by chance.
In the present study, the lymph nodes and tonsils with hyperplastic lymphoid follicles, including a well-developed GC, were exclusively examined. The current results therefore appear to reflect the immunophenotype of well-developed or fully matured human FDCs. This information can provide an essential basis for further examination regarding the development and maturation, physiological kinetics, or pathological alteration of human FDCs. At present, based on the current observation, we are attempting to elucidate the ontogenetic development of human FDCs by systematic immunohistochemical observation of the lymph nodes from archived fetal autopsy cases (Kasajima et al. 1997).
On the basis of the present observations, preliminary criteria regarding the immunohistochemical identification of a functional human FDC can be proposed as follows: (a) the co-expression of CD21 and CD35 signifying their ability to trap and retain immune complexes (hallmark of functional FDCs), and (b) the additional expression of at least two markers recognized by other antibodies, especially Ki-M4p and CNA.42. Many cases of neoplasms possibly derived from FDCs or related cells have been reported (Perez-Ordonez et al. 1996; Fonseca et al. 1998; Perez-Ordonez and Rosai 1998; Cheuk et al. 2001). In these reports, the tumor cells shared some morphological characteristics and immunohistochemical features in common with typical FDCs. However, because definite criteria have not been established and somewhat different definitions have been adopted case by case, these cases seem to be heterogeneous and inconsistent. The above-mentioned proposed criteria may help us to establish a diagnostic policy for this kind of tumor.
Several authors have previously reported the morphological and immunophenotypical heterogeneity of FDCs (Imai et al. 1983, 1991, 1993; Szakal et al. 1985; Carbone et al. 1988; Petrasch et al. 1990; Rademakers 1992; Yoshida et al. 1993; Maeda et al. 1996). In the present study, we also noted the possible immunophenotypical diversity of FDCs. Thus, FDCs located within LZ of the GC expressed CD23, X-11, and DAF/CD55 in addition to the above-mentioned common markers, while FDCs located in the DZ of the GC showed particular cytoplasmic and nuclear labeling of anti-S100α. The biological or pathological meaning of the heterogeneity have yet to be thoroughly discussed. It may, however, reflect some difference of their functional phases, maturation stages, or cell sublineages.
Other MAbs, including Ki-FDC-1p (Tabrizchi et al. 1990), RR1 (anti-human ICAM-1/CD54) (Schriever et al. 1989; Koopman et al. 1991), antibodies against the molecules of the VLA (very late antigen) family (Koopman et al. 1991; Zutter 1991), MAS516 and 5B5 (Bosseloir et al. 1994), HJ2 (Butch et al. 1994), 7D6 (Liu et al. 1997), AS02 (Bofill et al. 2000), and 8D6 (Li et al. 2000), have also been reported to react with human FDCs. Unfortunately, they were not available for use in the present study. Further analyses can be expected to obtain more comprehensive results when these antibodies are included.
In conclusion, the present study has established that human FDCs can be detected in formalin-fixed, paraffin-embedded tissue sections by a panel of monoclonal antibodies and suitable antigenic retrieval methods. We expect that this information will provide some important bases for the development of novel approaches in immunopathological research and also in practical diagnoses of diseases.
Footnotes
Acknowledgements
Supported in part by a grant-in-aid for Scientific Research (C) from the Ministry of Education, Science, Culture and Sports of Japan (project #06670183).
We are greatly indebted to Prof Karl Lennert (University of Kiel) and Prof Taroh Kinoshita (Research Institute for Microbial Disease, Osaka University) for their generous supply of monoclonal antibodies, Ki-M4p or anti-human DAF/CD55 (IA10, IIH6 and VIIA7), respectively.
