Abstract
Keywords
The endothelin (ET) family consists of the three isoforms: ET-1, vasoactive intestinal contractor (VIC)/ET-2, and ET-3. These isopeptides are composed of 21 amino acid residues and have a variety of biological roles, including involvement in vasoconstriction (Yanagisawa et al. 1988), smooth muscle contraction (Ishida et al. 1989; Mckay et al. 1991), cardiomyocyte beating (Suzuki et al. 1990), and cell growth (Lahav et al. 1996; Mazzocchi et al. 1997). The biological effects of these peptides are mediated by two distinct G-protein-coupled receptors: the endothelin A receptor (ETA) and the endothelin B receptor (ETB).
VIC is a murine-derived peptide characterized as a potent vasoactive and smooth muscle contracting compound (Ishida et al. 1989; Saida et al. 1989; Blank et al. 1991; Taniyama et al. 1993; Fang et al. 1994; Kan et al. 1994; de la Monte et al. 1995; Iwashima et al. 1997). ET-2 is a human-derived peptide characterized as a potent vasoconstrictor (Inoue et al. 1989). VIC differs from ET-2 in one amino acid of 21 residues and may be the mouse or rat counterpart of human ET-2 (Bloch et al. 1991). Structurally, VIC/ET-2 is highly homologous to ET-1. VIC differs from ET-1 in three amino acids of 21 residues; ET-2 differs from ET-1 in two amino acids. Although the biological activities and physiological roles of ET-1 and ET-3 have been well documented, little is known about the specific physiological role of VIC/ET-2.
Several studies concerning the presence of the ET system in the uterus have been reported. The presence of mRNA (O'Reilly et al. 1992) and peptide (Cameron et al. 1993) for ET-1, ET-2, and ET-3 was demonstrated in the human endometrium. Ligand binding studies using iodinated ET-1, ET-2, and ET-3 showed that binding sites are localized mostly in the endometrium, with a lower level in the myometrium, suggesting a role for the ET system in the control of human menstruation (Davenport et al. 1991). ET-1 was hypothesized to participate with oxytocin in rabbit myometrial contraction at parturition (Peri et al. 1992). Recently, we demonstrated a relatively high level of expression of VIC mRNA in mouse uterus by semiquantitative RT-PCR (Uchide et al. 1999). Although the presence in uterus of mRNA and peptide for human ET-2 and mouse VIC has been reported, detailed information on changes of expression level and cellular distribution of VIC/ET-2 throughout the various physiological stages of the uterus has not been obtained. Therefore, the physiological roles of VIC/ET-2 in the uterus remain unclear.
In this study, in an attempt to investigate the physiological roles of VIC/ET-2 in uterus in vivo, we characterized quantitative changes in VIC gene expression during the estrous cycle, pregnancy, and parturition in mouse uterus by real-time quantitative RT-PCR. The precise cellular distribution of VIC peptide and mRNA was examined using immunohistochemical and in situ hybridization techniques.
Materials and Methods
Mice
Male and female ICR mice (10 weeks old) were obtained from Nippon Clea (Tokyo, Japan). Mice were mated, and noon on the day of the vaginal plug was designated Day 0.5 of pregnancy. Pregnant mice were divided into seven groups according to gestational age (Days 7.5, 8.5, 9.5, 10.5, 12.5, 14.5, and 17.5). Non-pregnant mice were divided into three groups dependent on the stage of the estrous cycle (diestrus, proestrus, and estrus) as determined by vaginal smear. Another group of mice were examined just after parturition. Each group contained three mice (n = 3). Our experimental procedures on animal subjects were in accordance with the Guidelines on Handling of Laboratory Animals for our institution.
Cloning of cDNAs
cDNA fragments of VIC and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were amplified by PCR using primers designed in accordance with the published sequence in mouse (Saida and Mitsui 1991). The fragments were cloned using a pCR-Script SK+ Cloning Kit (Stratagene; La Jolla, CA) according to manufacturer's protocol. Recombinant plasmids were sequenced using a DyeDeoxy Terminator Cycle Sequencing Kit and Model 373A DNA Sequencer (PE Applied Biosystems; Foster City, CA). The plasmids were propagated and used as quantitative controls to estimate each gene expression level and as templates to produce riboprobes for in situ hybridization.
Poly (A)+RNA Preparation.
The excised uteri were washed well with chilled PBS, pH 7.4, on ice and weighed. One hundred mg of uterus was immediately homogenized. Total RNAs were then prepared from the homogenate using a commercial phenol and guanidine-thiocyanate solution (Isogen solution; Nippon Gene, Tokyo, Japan) according to the manufacturer's protocol. Poly(A)+ RNAs were obtained from the total RNAs by oligo(dT)-cellulose chromatography (Pharmacia; Uppsala, Sweden).
Gene Expression Analysis
We established a quantitative RT-PCR method for analysis of VIC gene expression using a real-time PCR system (PE Applied Biosystems) which is more accurate and less time-consuming than a conventional PCR system (Gibson et al. 1996; Heid et al. 1996). After RT reaction of poly(A)+RNAs, cDNAs obtained were used as templates for real-time PCR. Oligonucleotide primers specific for the amplification of mouse prepro-VIC (sense, 5′-GAGGTGACAAGAAAGGCCATC-3′; antisense, 5′-GGTACAGTAGCTAGCAACCAGCA-3′) and GAPDH (sense, 5′-CTTCACCACCATGGAGAAGGC-3′; anti-sense, 5 '-GGCATGGACTGTGGTCATGAG-3′) were designed. The amplification products were predicted to be 301 bp and 238 bp for VIC and GAPDH, respectively. Detection probes (TaqMan Probe; PE Applied Biosystems) for VIC (5′-FAM-TGGAGGAAGAGATAACACCATGAGCTGC-TAMRA-3′) and GAPDH (5′-FAM-CCTGGCCAAGGTCATCCATGACAACTTT-TAMRA-3′) were also designed. Amplification was carried out using the GeneAmp EZ RT-PCR kit (PE Applied Biosystems). Template cDNA (0.2 ng/μl) was mixed with 1 × buffer, 2.5 mM MgCl2, 300 ± dATP, dCTP, dGTP, and 600 ± dUTP, 5 U/μl rTth polymerase, 4 mM Mn(OAc)2, 100 nM TaqMan Probe, and 200 nM of each primer. All reactions were performed in the Model 7700 Sequence Detector (PE Applied Biosystems). Reaction conditions were programmed on a computer linked to the detector for 50 cycles of the amplification step (95C for 20 sec, 62C for 1 min). Analysis of data was performed on a computer by analysis software. Gene expression levels in the uterus were denoted as the “gene expression rate,” as previously described (Uchide et al. 1999). Briefly, gene expression rate was calculated using the following formula:
[Concentration of VIC mRNA in a sample/Concentration of GAPDH mRNA in a sample] × 100
Antibody for Immunohistochemistry
A rabbit antibody raised against the synthetic ET-2 peptide was used for immunohistochemistry. The IgG fraction was purified by affinity chromatography. After further purification on an ET-2-binding column followed by absorption with ET-1 and ET-3 peptide, IgG specific to ET-2 was obtained. Crossreactivity studies by ELISA showed that this antibody reacted at 100% with ET-2 and 90% with VIC but below 0.1% with ET-1 and ET-3. The absence of crossreactivity with tissue component proteins was also confirmed by Western blotting. This antibody (code no. 16152) was a gift from Immuno-Biological Laboratories (IBL; Fujioka, Japan). We used the antibody to detect VIC in mice.
Immunohistochemistry
The excised uteri were fixed with 4% (w/v) paraformaldehyde and embedded in paraffin by standard methods. Tissues sectioned at a thickness of 5 ± were deparaffined and rehydrated with graded alcohol. After washing with PBS, the sections were treated with methanol containing 0.3% hydrogen peroxide for 30 min to inactivate endogenous peroxidase activity. Immunostaining was carried out using a commercial kit (Vectastain ABC Kit; Vector Laboratories, Burlingame, CA). Briefly, the sections were washed and incubated in 10% normal goat serum in PBS for 30 min to block nonspecific immunoreactive sites. The sections were then reacted with 5 μg/ml of anti ET-2 rabbit antibody for 60 min at room temperature (RT). After washing with PBS, the sections were incubated with biotinylated goat anti-rabbit IgG for 30 min at RT, washed with PBS, and treated with streptavidin-biotin-peroxidase complex (ABC reagent) for 30 min at RT. The complex was visualized by diaminobenzidine (DAB). Counterstaining for nuclei was done with Mayer's hematoxylin. Control studies included omission of the primary antibody, its replacement with preimmune rabbit IgG, and preabsorption tests using the antibody preincubated with an excess of synthetic VIC.
Riboprobes for In Situ Hybridization
Digoxygenin-labeled sense or antisense riboprobes for in situ hybridization were synthesized by T3 or T7 polymerases from the cloned cDNA plasmid described above in the presence of digoxygenin-dUTP using a commercial kit (DIG RNA Labeling Kit; Boehringer Mannheim, Mannheim, Germany).
In Situ Hybridization
The excised uteri were immersed in PBS containing 4% (w/v) paraformaldehyde overnight at 4C to fix and then serially placed into 10%, 20%, and 30% sucrose-PBS at 4C. The tissues were frozen and embedded in a medium (OCT compound; Sakura Finetechnical, Tokyo, Japan) on dry ice and sectioned at a thickness of 10 ± using a cryostat (HM 500 OMV; Microm, Walldorf, Germany). The sections were immediately dried and stored at -80C until use. Before hybridization, the sectioned tissues were treated with 0.2 N HCl for 20 min to inactivate endogenous alkaline phosphatase, 5 μg/ml of proteinase K (Boehringer Mannheim) in PBS for 10 min at 37C, 2 mg/ml glycine in PBS for 15 min at RT for quenching, and 0.1 M triethanolamine hydrochloride, pH 8.0, containing 0.25% acetic anhydride for 15 min at RT for acetylation. After incubation in prehybridization buffer containing 50% formamide and 2 × SSC for 30 min at 50C, the sections were reacted with 1 ng/μl digoxygeninlabeled antisense riboprobe in hybridization buffer containing 50% formamide, 2 × SSC, 1 μg/μl tRNA, 1 μg/μl sonicated salmon sperm DNA, 1 μg/μl bovine serum albumin, and 10% dextran sulfate at 50C overnight. Sense riboprobe at the same concentration was used as a negative control. To lessen background signals, RNase A (20 μg/ml) treatment was carried out for 30 min at 37C. Bound digoxygenin-labeled riboprobe was detected with anti-digoxygenin antibody conjugated with alkaline-phosphatase (Boehringer Mannheim), and colored with nitroblue tetrazolium (NBT) and 5-bromo-4-chloro-3-indolylphosphate (BCIP). Counterstaining for nuclei was carried out with methyl green.
Statistical Analysis
Data were analyzed for statistical significance by ANOVA using Fisher's Protected Least Significant Difference (Fisher's PLSD) test, requiring p<0.05 for significance.
Results
VIC Gene Expression
Gene Expression During Pregnancy. To examine the expression level of VIC mRNA in the uterus during its various physiological states, we established a real-time RT-PCR system for quantification of VIC mRNA. VIC expression levels in the pregnant uterus were quantified using this technique and are shown in Figure 1 as “gene expression rates”; the values are normalized to GAPDH expression (see Materials and Methods). The gene expression rates at Days 7.5, 8.5, 9.5, and 10.5 of pregnancy (the earlier stage of pregnancy) were 0.04, 0.05, 0.05, and 0.06, respectively. During the later stage of pregnancy, at Days 12.5, 14.5, and 17.5, and just after parturition (P-0), VIC gene expression rates were 0.36, 0.20, 0.15, and 0.19, respectively. The gene expression rates in the later stage of pregnancy and after parturition were significantly p<0.05) higher (approximately fivefold) than those in the earlier stage.
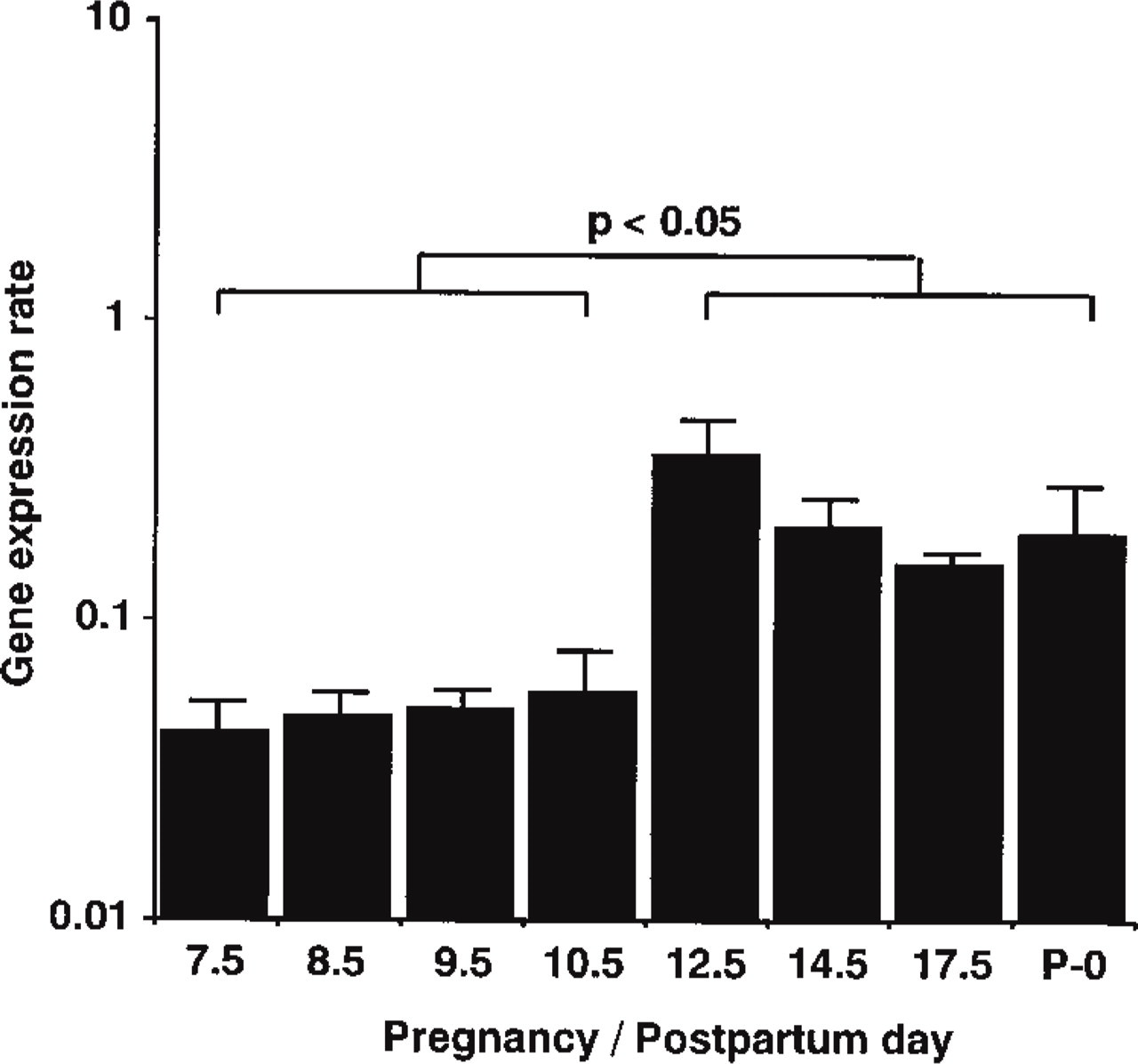
Gene expression rates of uterine VIC during pregnancy and after parturition. Gene expression rates were calculated by normalizing the expression level of VIC with that of GAPDH (see Materials and Methods). Gene expression rates in the later pregnancy stage (Days 12.5-17.5) and just after parturition (P-0) were significantly (p<0.05) higher (approximately fivefold) than those in the earlier stage (Days 7.5-10.5).
Gene Expression During the Estrous Cycle. The gene expression rates of VIC during the estrous cycle are shown in Figure 2. The gene expression rates in diestrus, proestrus, and estrus were 0.08, 0.22, and 0.35, respectively. The gene expression rates were significantly (p<0.05) higher (approximately threefold) in proestrus and estrus than in diestrus.
Immunohistochemistry
To examine the cellular distribution of VIC peptide in the uterus, immunohistochemical studies were performed using a purified IgG antibody specific to ET-2, which detects VIC but not ET-1 and ET-3 in mouse (see Materials and Methods). Several cell types were positive for immunostaining for VIC in the pregnant and non-pregnant uterus. Figure 3 shows light micrographs of the pregnant uterus at Days 10.5 and 14.5. Strong immunostaining was observed in vascular smooth muscle cells (Figure 3A), weak immunostaining in endometrial epithelium cells (Figure 3C), and no apparent staining in endometrial stromal cells (Figure 3C) and vascular endothelial cells (Figure 3B) at Day 10.5. The same immunostaining patterns were also observed in the other pregnancy stages we examined (not shown). In myometrial cells, the positive patterns of immunostaining changed between the earlier (Days 7.5-10.5) and the later pregnancy stages (Days 12.5-17.5), with an increase in the degree of staining or immunopositivity occurring between Days 10.5 and 12.5, analogous to the increase observed in the gene expression rate. At Day 10.5, myometrial cells showed some positive staining (Figure 3D). However these cells showed more intense immunostaining at Day 14.5, with almost all myometrial cells staining positively for VIC (Figure 3E). The uterus after parturition showed the same pattern of immunostaining as in the later pregnancy stage.
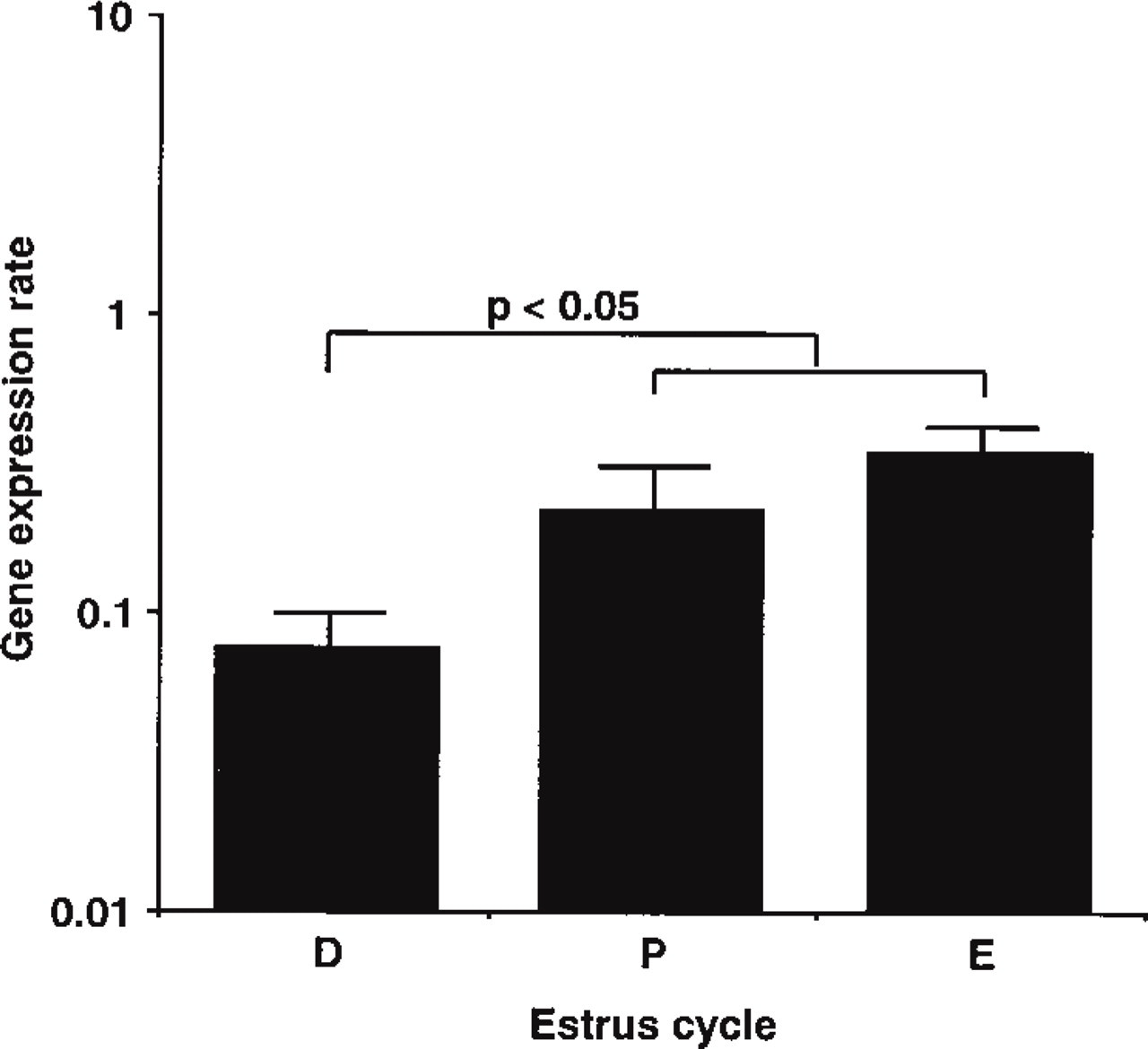
Gene expression rates of uterine VIC during the estrous cycle. Gene expression rates in proestrus (P) and estrus (E) were significantly (p<0.05) higher than in diestrus (D). Gene expression rates were calculated as described in Materials and Methods.
In non-pregnant mice in the diestrous, proestrous, and estrous uterus, specific immunostaining for VIC was dominant in vascular smooth muscle cells, faint in endometrial epithelium cells, and not apparent in stromal cells. Only in myometrial cells was a change in immunostaining observed during the estrous cycle. Myometrial cells in proestrus and estrus showed stronger positive immunostaining than those in diestrus (Figure 4).
To examine the specificity of our antibody, control sections were immunostained with preimmune rabbit IgG and antibody preabsorbed with synthetic VIC. In these experiments we observed the elimination of specific signals observed in vascular smooth muscle cells, endometrial epithelial cells, and myometrial cells (Figures 3 and 4).
In Situ Hybridization
We conducted in situ hybridization for VIC mRNA on the uterus in the proestrous and estrous stages, in the later pregnancy stage, and after parturition. In all of these stages, high expression of VIC mRNA was confirmed by real-time quantitative RT-PCR. Positive signal was located predominantly in myometrial cells (Figure 5A), was faint in endometrial epithelium cells, and was not observed in stromal cells (not shown). No signal was detected in the sections by a sense probe (Figure 5B). These findings, together with the results of the quantitative analysis for VIC mRNA and the immunohistochemical studies, demonstrate that myometrial cells in these stages strongly express VIC mRNA and produce peptide. It is also evident that cellular distribution of VIC mRNA is in accordance with that of VIC peptide.
Discussion
The uterus is a physiologically active organ that undergoes various cellular changes in response to the estrous cycle, pregnancy, and parturition. Many factors, such as steroid hormones, growth factors, and cytokines, are involved in the regulation of uterine changes. We previously detected relatively high VIC expression in mouse uterus by semiquantitative RT-PCR (Uchide et al. 1999). However, we did not discuss the role of VIC in the uterus. To clarify its role, we now have investigated VIC mRNA expression and the presence of peptide in mouse uterus under various physiological states, i.e., during the estrous cycle, pregnancy, and parturition, by real-time quantitative RT-PCR, immunohistochemistry, and in situ hybridization.
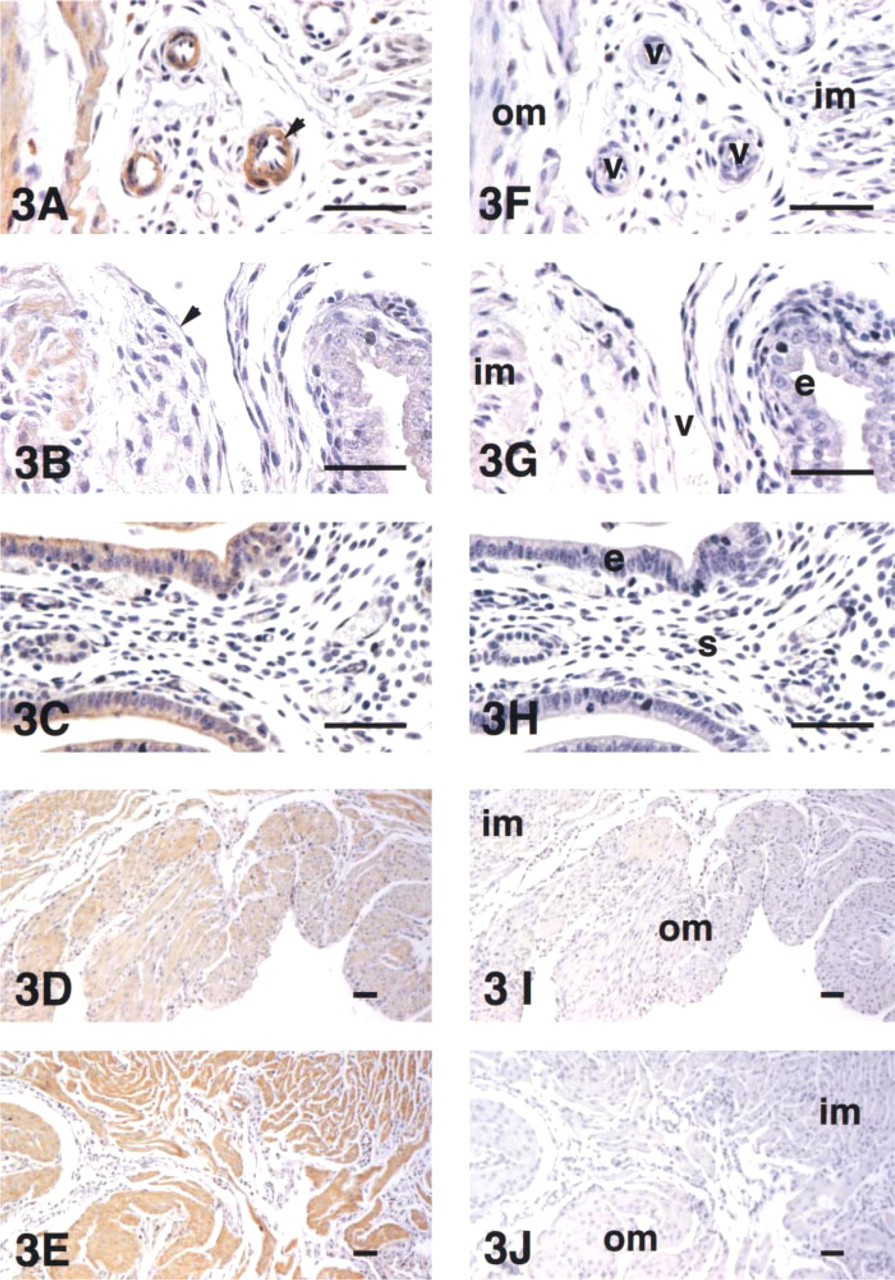
Immunolocalization of uterine VIC during pregnancy. Immunostaining patterns of vascular smooth muscle cells (
Quantitative gene expression analysis by real-time RT-PCR demonstrated that the expression level of VIC mRNA in the uterus fluctuates during pregnancy (Figure 1). In particular, we observed a significant change in gene expression between the earlier (Days 7.5-10.5) and later (Days 12.5-17.5) stages of pregnancy. Gene expression rates in the later stage showed an approximately fivefold increase compared with those in the earlier stage (Figure 1). In accordance with this increase in the gene expression rate, myometrial cells showed a stronger positive pattern of immunostaining for VIC in the later stage of pregnancy (Figure 3). In the cycling non-pregnant uterus, the gene expression rate increased as the estrous cycle advanced from diestrus to proestrus and estrus (Figure 2), and concomitantly myometrial cells in proestrus and estrus showed a stronger positivity for VIC than in diestrus (Figure 4). On the basis of these results, we concluded that the expression of VIC mRNA and peptide increases in myometrial cells in the later stage of pregnancy and in proestrus and estrus. The results of in situ hybridization (Figure 5) support this conclusion.
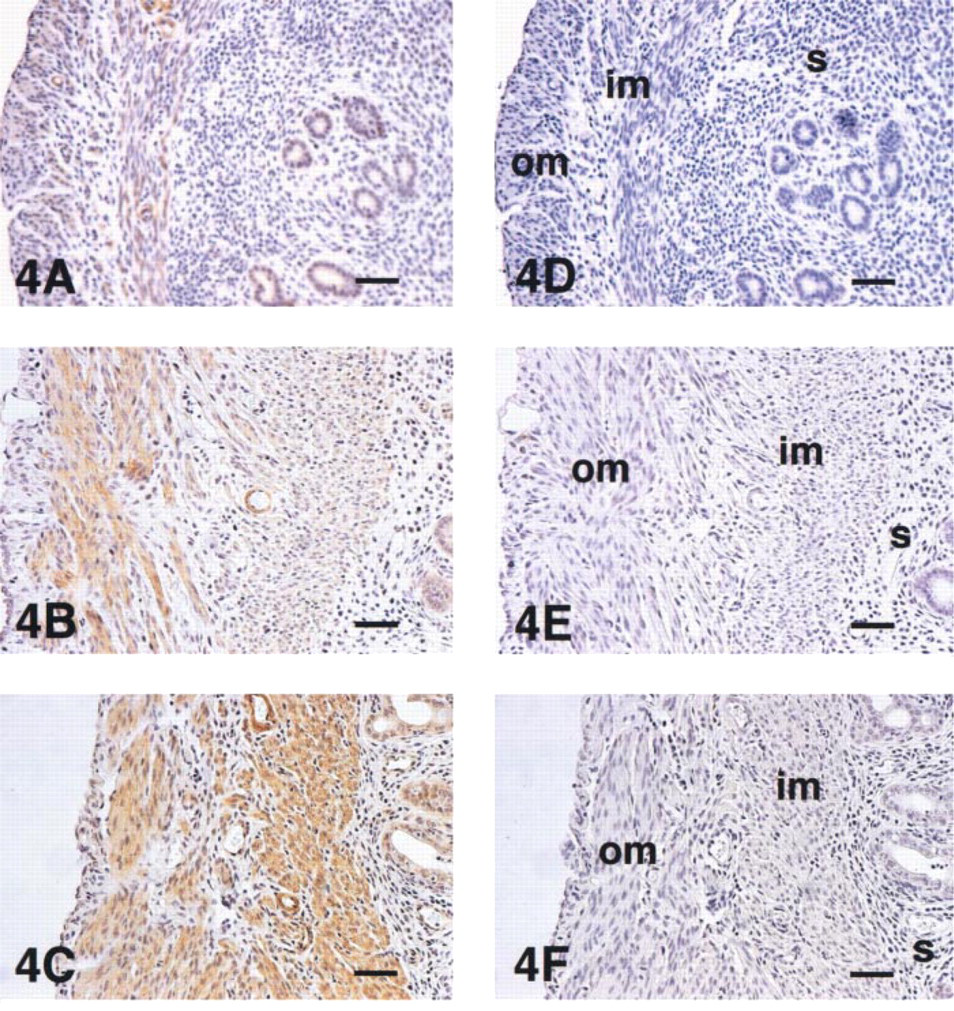
Immunolocalization of uterine VIC during the estrous cycle. Im-munopositivity of myometrial cells in proestrus (
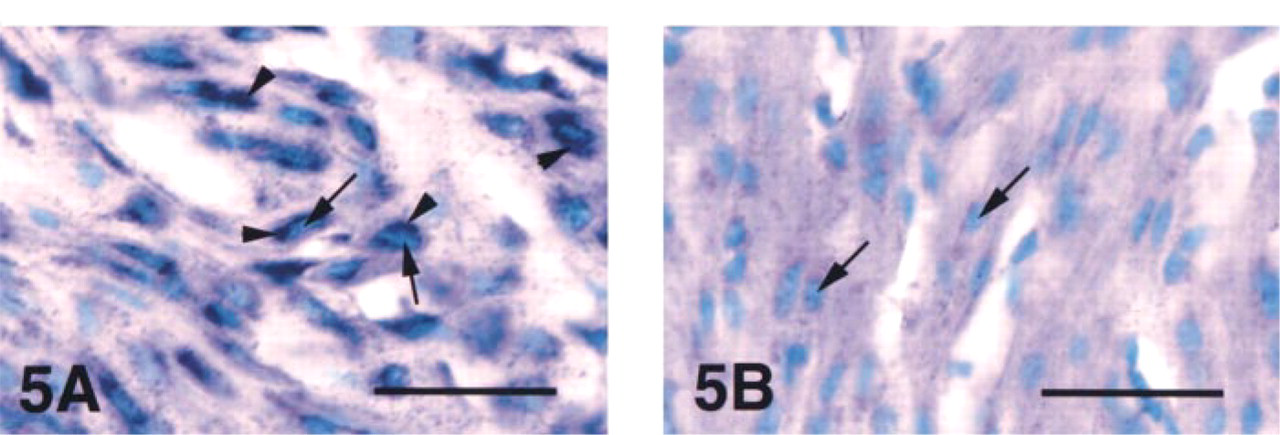
In situ hybridization of uterine VIC mRNA at day 14.5 of pregnancy. In the section hybridized with an antisense probe (
The reason for the drastic changes in VIC gene expression during pregnancy and the estrous cycle is unclear. However, the most likely explanation for the change would be hormonal regulation by the ovarian steroids estrogen or progesterone, considering that the concentration of these agents in blood changes greatly during pregnancy (McCormack and Greenwald 1974) and the estrous cycle (Greep et al. 1973). Ovarian steroids are reported to regulate gene expression and production of growth factors and cytokines in the uterus, including heparin-binding epidermal growth factor-like growth factor (Wang et al. 1994; Zhang et al. 1994, 1998), transforming growth factor-α (Nelson et al. 1992), epidermal growth factor (Huet-Hudson et al. 1990), interleukin-1, interleukin-6, and tumor necrosis factor-α (Mamata et al. 1992). In addition, the localization of ET-1 in rabbit uterus is affected by the administration of estrogen or progesterone (Maggi et al. 1991). Taking these reports into consideration, we hypothesize that the gene expression and production of VIC may be regulated by ovarian steroids, directly or indirectly; that VIC may mediate actions of ovarian steroids as local mediators, as cytokines and growth factors do; and that VIC may participate in many reproductive events including cell growth and differentiation in the uterus. Studies on the regulation of VIC gene expression by ovarian steroids using ovariectomized mouse and primary uterine cell culture are under way in our laboratory.
For the pregnant uterus, there is another possible explanation for elevated VIC gene expression in the later stage of pregnancy. The mechanical stimulation of the uterus by the growing embryos, which drastically increase in size around this period, may play a role in the regulation of VIC expression. In vascular smooth muscle cells the expression, synthesis, and secretion of angiotensin II and transforming growth factor-β are reported to be stimulated by mechanical stretching (Li et al. 1998). In the cultured cardiomyocyte, stretching stimuli increase ET-1 production by approximately threefold (Yamazaki et al. 1996). There has been, to our knowledge, no report on induction of ET family peptides in myometrial cells by mechanical stretching. However, in rabbit uterus it was confirmed that mechanical stretching stimulates growth, protein synthesis, and contractile function (Csapo et al. 1965). Similarly, considering the fact that VIC has a contractile and mitogenic action (Ishida et al. 1989; Fabregat and Rozengurt 1990), VIC expression may be induced by stretch stimuli to promote the growth of myometrial cells or to regulate uterine function (e.g., contraction and maintenance of tension against the growing embryos).
Cellular distribution of ET-1 in the uterus varies among species and among uterine physiological stages. In pregnant and parturient rabbit, a number of giant cells penetrating to the myometrium, which are proposed to be derived from trophoblastic knobs, showed intense ET-1 immunostaining, but myometrial cells did not (Peri et al. 1992). In rat, the expression of ET-1 mRNA and peptide is reported in endometrial epithelial cells of non-pregnancy and in both endometrial epithelial cells and myometrial cells of pregnancy and the early postpartum period. The expression of ET-1 mRNA in myometrial cells was strongest in the early postpartum period compared with other pregnancy stages (Kajihara et al. 1996). In our study, we demonstrated the expression of VIC mRNA and peptide in mouse endometrial epithelial cells and myometrial cells in non-pregnancy, pregnancy, and the early postpartum period. As we show here, VIC is expressed highly in non-pregnant mouse myometrial cells during proestrus and estrus. However, it has not yet been reported whether myometrial cells of non-pregnant rat express ET-1 at any point during the estrous cycle. Furthermore, mouse myometrial cells in late pregnancy and early postpartum also exhibit a high level of VIC expression. Therefore, in mouse uterus, especially in myometrial cells, VIC probably has some function, not only at parturition but also during the estrous cycle and pregnancy. VIC, which is expressed differently from ET-1, may regulate the physiological function of mouse uterus in a complementary fashion or in cooperation with ET-1.
The precise intracellular localization of VIC in myometrial cells is now unclear because we have not performed ultrastructural studies and there has been no previous report on the intracellular localization of VIC or ET-2. However, ET-1 has been demonstrated by an electron microscopic study to be in the endoplasmic reticulum (ER) and the Golgi apparatus of vascular endothelial cells and adrenal gland cells (Nakamura et al. 1990; Li et al. 1995). A sequence encoding “signal peptide,” which is necessary for insertion into the ER, also has been identified in human preproET-1 mRNA (Fabbrini et al. 1991). These findings suggest that ET-1 is synthesized in the ER and secreted through the ER-mediated secretory pathway. Furthermore, cultured muscle cells, including both vascular smooth muscle cells and cardiomyocytes, have been demonstrated through detection of the peptide in culture medium to secrete ET-1 (Kanse et al. 1991; Suzuki et al. 1993). Therefore, we postulate that VIC, a member of the ET family, probably is also synthesized in the ER of myometrial cells and secreted through the ER-mediated secretory pathway.
Both ETA and ETB receptors are expressed in myometrial cells (Bacon et al. 1995). In pregnant human myometrial cells the proportion of ETA to ETB increases (Osada et al. 1997). Uterine contraction and oscillation frequency in response to ET-1 are mediated by ETA (Tsunoda et al. 1993; Heluy et al. 1995).
However, the role of ETB in the uterus is unclear. Because VIC has almost the same selectivity and affinity for the receptors as ET-1, ETA could be critical to the effectiveness of VIC in the pregnant uterus. Detailed information on the expression and cellular distribution of ET-1 in mouse uterus has not been reported. However, given the anatomic and physiological similarity between mouse and rat uterus, VIC, like its relative ET-1, could very likely act on the uterus by means of the ETA receptor in myometrial cells. The co-localization of ligands and receptors in myometrial cells suggests that the secreted endothelins act on the same and/or adjacent cells via the receptors in an autocrine and/or paracrine manner, as previous studies have reported for various other tissues and cells (MacCumber et al. 1989; Kanse et al. 1991; FerréA et al. 1993; Ito et al. 1993; Fouassier et al. 1998).
In conclusion, we demonstrated that the expression level of uterine VIC changes during the estrous cycle and throughout pregnancy. We showed that myometrial cells in the proestrous and estrous stages, in late pregnancy, and in early postpartum dominantly express VIC mRNA and produce VIC peptide. Our present results, which reveal changes in the expression and production levels of VIC in response to various reproductive events in the uterus, suggest that VIC produced by myometrial cells may act in an autocrine/paracrine manner and play an important role in physiological functions including contraction, cell growth, and differentiation in the uterus.
Footnotes
Acknowledgements
Acknowledgments
Supported by a project grant to K.S. of Research and Development for the Elucidation of Biological Functions from the Ministry of International Trade and Industry of Japan and by a project grant of the Cooperative System for Supporting Priority Research from the Science and Technology Agency of Japan.
K.S. gratefully acknowledges the encouragement and support of Drs Syuichi Oka and Noboru Tomizuka at NIBH. T.U. and K.S. thank Drs Norio Ishida, Tomoko Niki, Junji Magae, and Yasuo Tanaka at NIBH for helpful discussion, Ms Manami Nagano at PE Applied Biosystems for the design of and advice on TaqMan probes, and IBL for the gift of antibody.
