Abstract
We examined pendrin expression in various diseased thyroid tissues by immunohistochemistry (IHC) using antiserum raised against human pendrin and by real-time quantitative RT-PCR. In normal thyroids the antiserum reacted with the apical membrane of follicular cells and its immunoreactivity was faint. In Graves’ thyroids, the IHC expression of pendrin appeared in a pattern similar to that of normal thyroids but it was more extensive and stronger, especially in areas showing marked proliferation of follicular cells. The immunoreactivities of pendrin in nodular goiters varied from case to case. In follicular adenomas, pendrin was localized in the follicle-forming parts of the tumor but was negative in trabecular parts. Pendrin was negative in all follicular carcinomas, papillary carcinomas, and in one case of medullary carcinoma. In quantitive mRNA analysis, the relative values of pendrin mRNA were significantly low in papillary carcinoma (p<0.01), whereas the values in other diseased thyroids were not significantly different from those in normal thyroids. These results suggest that pendrin may play a role in thyroid hormone production as the apical porter of chloride/iodide and investigation of pendrin leads to a better understanding of functional aspects of the iodine transportation system in thyroid diseases.
P
Materials and Methods
Tissue Samples
A series of surgical specimens from patients with thyroid disease composed of normal thyroid (18 cases), Graves’ disease (13 cases), nodular goiter (34 cases), follicular adenoma (16 cases), follicular carcinoma (four cases), papillary carcinoma (24 cases), and medullary carcinoma (two cases) were chosen to represent a wide range of thyroid pathologies from the surgical pathology files of Yamanashi Medical University Hospital and Kofu City Hospital. All specimens had been fixed in 10% neutral-buffered formalin, processed routinely, and embedded in paraffin. In addition, for real-time quantitative RT-PCR, we used 82 fresh thyroid tissues including normal thyroid (18 cases), Graves’ disease (16 cases), nodular goiter (22 cases), follicular adenoma (nine cases), papillary carcinoma (16 cases), and medullary carcinoma (one case). The thyroid specimens were diagnosed according to the World Health Organization diagnostic criteria (Hedinger et al. 1988). All normal thyroid tissues were obtained from patients who underwent subtotal or total thyroidectomy for papillary carcinoma. All Graves’ disease specimens were obtained from patients who had been treated with an antithyroid drug (propylthiouracil or methimazole) but who were still in a mid-hyperthyroid state.
Production of Antiserum to Pendrin
The three peptides spanning the human pendrin region, AAPGGRSEPPQLPEYSC (amino acids 2–18 in human pendrin), QQQHERRLQERKTLREC (amino acids 33–48 in human pendrin added to the cysteine residue at the C-terminal), and CLTEEELDVQDEAMRTLAS (amino acids 763–780 added to the cysteine residue at the N-terminal), were synthesized by BEX Co. (Tokyo, Japan). The synthesized peptides were conjugated to keyhole limpet hemocyanine (KLH) (Pierce; Rockford, IL) using m-maleimidobenzoyl-N-hydroxysuccineimide ester (MBS) (Pierce) as the coupling agent between KLH and the cysteines on the synthetic peptides. Japanese White rabbits, two female rabbits for each antigen, were injected SC with 200 μg of the synthetic peptide-carrier conjugate mixed with complete Freund's adjuvant (Difco; Detroit, MI). Subsequent boosts followed the same regimen as the primary injection at 2-week intervals. Immediately before each boost, the rabbit blood was examined using the double immunodiffusion test. According to the precipitation lines between the differently diluted seras and the antigen, a semiquantitative titer of antipeptide serum was estimated to determine the proper number of additional booster injections. At 2 weeks after the final booster injection, rabbits were bled for serum preparation. The specificity of the antiserum produced here was estimated using the double immunodiffusion test and IHC absorption test.
Double Immunodiffusion Test
A 1% agarose gel 1 mm thick was prepared on a glass slide and wells were cut into the gel. The pattern of wells is one center well and six surrounding wells. Each well was 2 mm in diameter and the distance between wells was 5 mm. A 2-μl peptide antigen conjugated with KLH (1 mg/ml concentration) was placed in the center well and the differently diluted rabbit sera (2 μl) were placed in surrounding wells (well 1, preimmune serum; well 2, immune serum, nondiluted; well 3, immune serum diluted 1:4; well 4, immune serum diluted 1:8; well 5, immune serum diluted 1:16; well 6, immune serum diluted 1:16). The gel was incubated in a moist chamber for 24 hr at room temperature and then the precipitation bands were stained with Coomassie Brilliant Blue R250 (Fluka Chemie; Buchs, Switzerland).
Immunohistochemistry
Tissue sections 3 μm thick were cut and mounted on silane-coated glass slides, deparaffinized in xylene, and rehydrated via graded ethanols to water. Deparaffinized sections were placed in plastic Coplin jars filled with 10 mM citrate buffer (pH 6.0) and incubated for 10 min at 120 in an autoclave for heat antigen retrieval (AR). After the autoclave treatment, endogenous peroxidase was quenched with 3% (v/v) H2O2 for 5 min. Indirect immunoperoxidase staining was carried out according to standard protocols. The sections on the glass slides were incubated with the antiserum raised against the synthetic peptide of pendrin, diluted 1:1000 in 1% bovine serum albumin (BSA; Itoham Foods, Hyogo, Japan) for 2 hr at RT. After washing with PBS, pH 7.2, the sections were incubated with peroxidase-conjugated goat anti-rabbit immunoglobulins (DAKO; Glostrup, Denmark) diluted 1:100 in 1% BSA, for 1 hr at RT. The sections were then washed with PBS, reacted with 0.6 nM 3–3′-diaminobenzidine tetrahydrochloride (DAB) (Dojindo; Kumamoto, Japan) in PBS, washed, and stained with hematoxylin. The immunoreactivity was evaluated using a scale from grade 0 through grade 3 (grade 0, no staining; grade 1, focal and faint; grade 2, intermediate between 1 and 3; and grade 3, diffuse and strong). For the negative control, preimmune rabbit serum was used instead of the primary antiserum.
Extraction of RNA
Total RNA was isolated from frozen thyroid tissues using the acid guanidine-phenol–chloroform RNA extraction system (ISOGEN; Nippon Gene, Toyama, Japan) according to the manufacturer's instructions. The quantity of total RNA was first determined by OD260 measurement, and the quality of total RNA was estimated by 1.5% agarose gel electrophoresis. Only RNAs showing the clear band intensity of the 28S and 18S rRNAs were selected for further analysis.
Reverse Transcription (RT)
cDNA was synthesized from total RNA using the TaqMan Reverse Transcription Reagent Kit (Applied Biosystems; Foster City, CA) according to the manufacturer's protocol. Briefly, 400 ng of total RNA was reverse-transcribed in a 20-μl volume containing 1 X TaqMan RT buffer, 5.5 mM MgCl2, 2 mM dNTP mixture, 2.5 μM random hexamers, 8 U RNase inhibitor, and 2.5 U MultiScribe Reverse Transcriptase. The thermal cycling for cDNA synthesis consisted of 10 min of hybridization of random hexamers and total RNA at 25C, 30 min of RT reaction at 60C, and 5 min of RT-inactivation at 95C. The designated concentration of samples was 20 ng cDNA/μl.
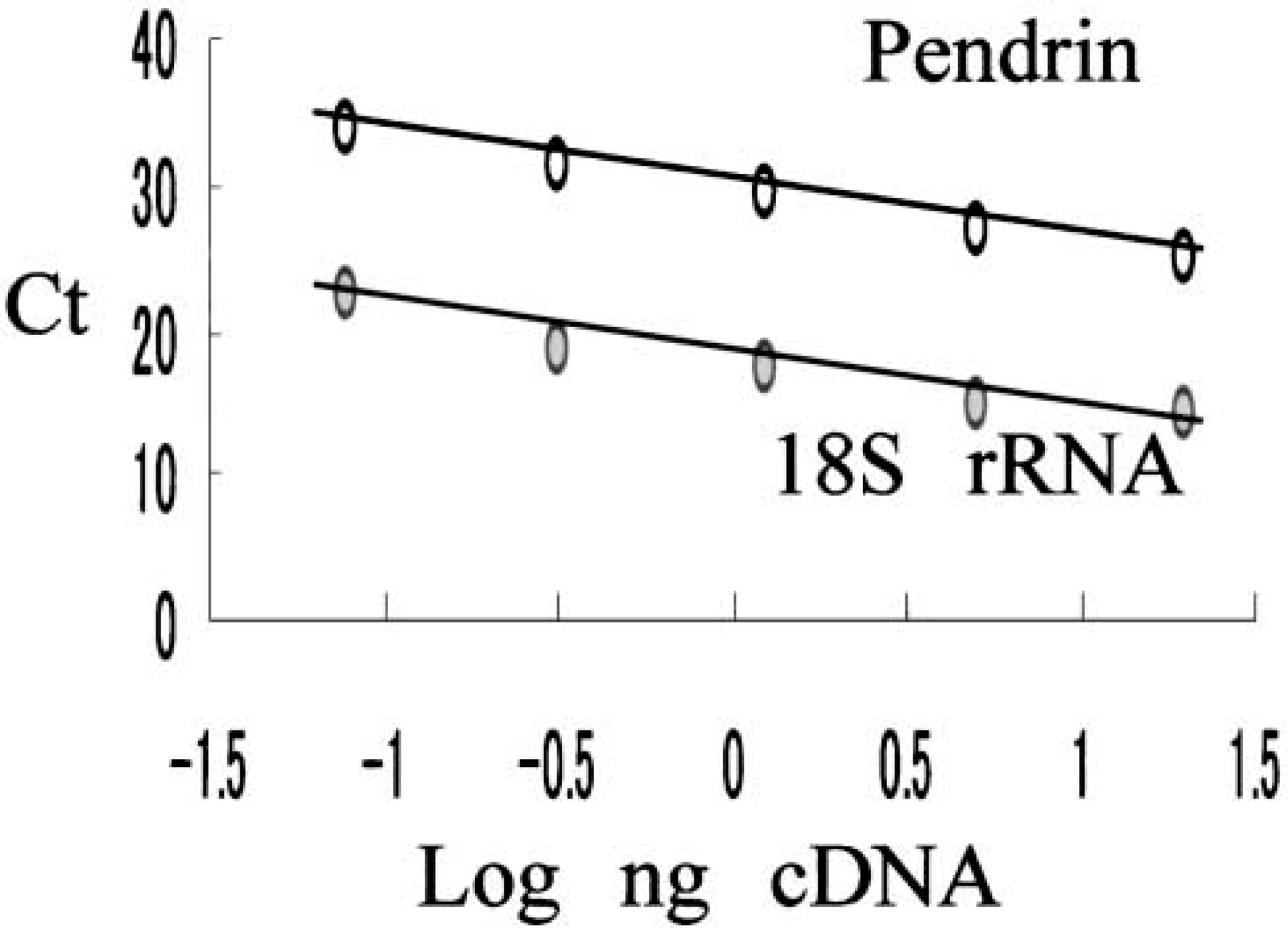
Standard curve of pendrin and 18S rRNA constructed by normal thyroid. Amount of cDNA is correlated with threshold cycle (Ct).
Real-time Quantitative PCR
Real-time quantitative PCR analysis for pendrin mRNA and 18S rRNA (endogenous RNA control) was performed using the ABI 5700 Sequence Detection System (Applied Biosystems). The sequence of oligonucleotide primers and TaqMan FAM probe for human pendrin (forward primer, 5′-CATCAAGACATATCTCAGTTGGACCT-3′; reverse primer, 5′-ACAGTTCCATTGCTGCTGGAT-3′; TaqMan probe, 5′-TCTGAGCATGGCCCCCGACG-3′) were as described previously (Bidart et al. 2000) and were synthesized by Applied Biosystems. For 18S rRNA primers and probe, the TaqMan ribosomal RNA Control Reagents Kit (Applied Biosystems) was used. A total of 1.25 ng of cDNA was amplified in 25-μl volume/well containing 1 X TaqMan universal PCR Master Mix (Applied Biosystems), 1000 nM pendrin forward primer (or 50 nM 18S rRNA forward primer), 1000 nM pendrin reverse primer (or 50 nM 18S rRNA forward primer), and 236 nM pendrin TaqMan FAM probe (or 200 nM 18S rRNA TaqMan VIC probe). DNA polymerase in TaqMan universal PCR Master Mix was activated by incubation for 10 min at 95C. PCR amplification was performed at 40 cycles consisting of 15 sec of denaturation at 95C and hybridization of primers and probe for 1 min at 60C. One normal thyroid tissue was used as the control (calibrator) for construction of the standard curve of both pendrin and 18S rRNA (Figure 1). Five differently diluted cDNAs (20 ng, 5 ng, 1.25 ng, 0.31 ng, and 0.08 ng in 25 μl volume/well) of control thyroid were used to construct standard curves. The target quantity of pendrin was determined from this standard curve and divided by the target quantity of 18S rRNA for normalization. The normalized values of pendrin were expressed as n-fold difference from the mean value of normal thyroids.
Results
Specificity of Anti-pendrin Serum
The two immune serum samples obtained from the rabbits after immunization with the synthetic peptide, amino acids 2–18 and 763–780 of pendrin, were found to react against the peptides at various dilutions (nondiluted 1:4 to 1:16) in the double immunodiffusion test, whereas preimmune sera and immune serum diluted 1:32 did not react to them. The antiserum against amino acids 33–48 of pendrin did not react in the double immunodiffusion test after three booster injections, and so we discontinued further immunization. The sera against amino acids 2–18 and 763–780 of pendrin showed identical immunoreactivities that were specifically positive at the apical membrane of follicular cells. In the IHC absorption test, specific immunoreactivities were completely inhibited by preincubation with the immune serum for each corresponding synthetic peptide (Figure 2). Further confirmation of staining specificity was carried out using pre- or nonimmune rabbit sera as a negative control for each section. Finally, we decided to use the antipeptide antiserum raised against amino acids 2–18 of the pendrin N-terminal in the following IHC study.
Immunohistochemistry
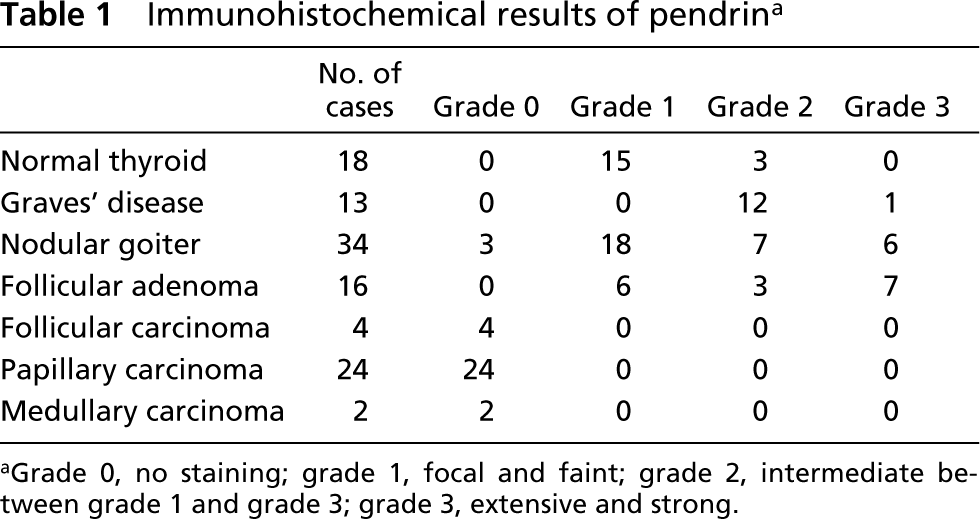
The IHC results of pendrin in normal and diseased thyroids are summarized in Table 1. In normal thyroids, pendrin was observed in all 18 cases and was localized to the apical membrane of follicular cells (Figure 3A). Most normal thyroids showed focal and faint immunoreactivity, representing grade 1. In detail, the immunoreactivity in cuboidal follicular cells of small follicles tended to be relatively stronger than that in flat follicular cells of large follicles.
In Graves’ thyroids (Figures 3C and 3D), linear immunostaining for pendrin was observed at the apical cell membrane of follicular cells. However, in areas showing marked proliferation of the follicular epithelium, pendrin positivity was characteristically strong. Pendrin immunoreactivity in all but one case was extensive and strong, representing grade 3.
The patterns of pendrin staining in hyperplastic follicles of nodular goiters were similar to those of normal thyroid follicles. Strong immunoreactivity of pendrin was observed mainly in small follicles or in benign papillae lined by the columnar epithelium (Figure 3B) and weak or negative immunoreactivity in dilated large follicles lined by flattened epithelium. Pendrin was positive in most cases but varied from case to case in its immunoreactivities.
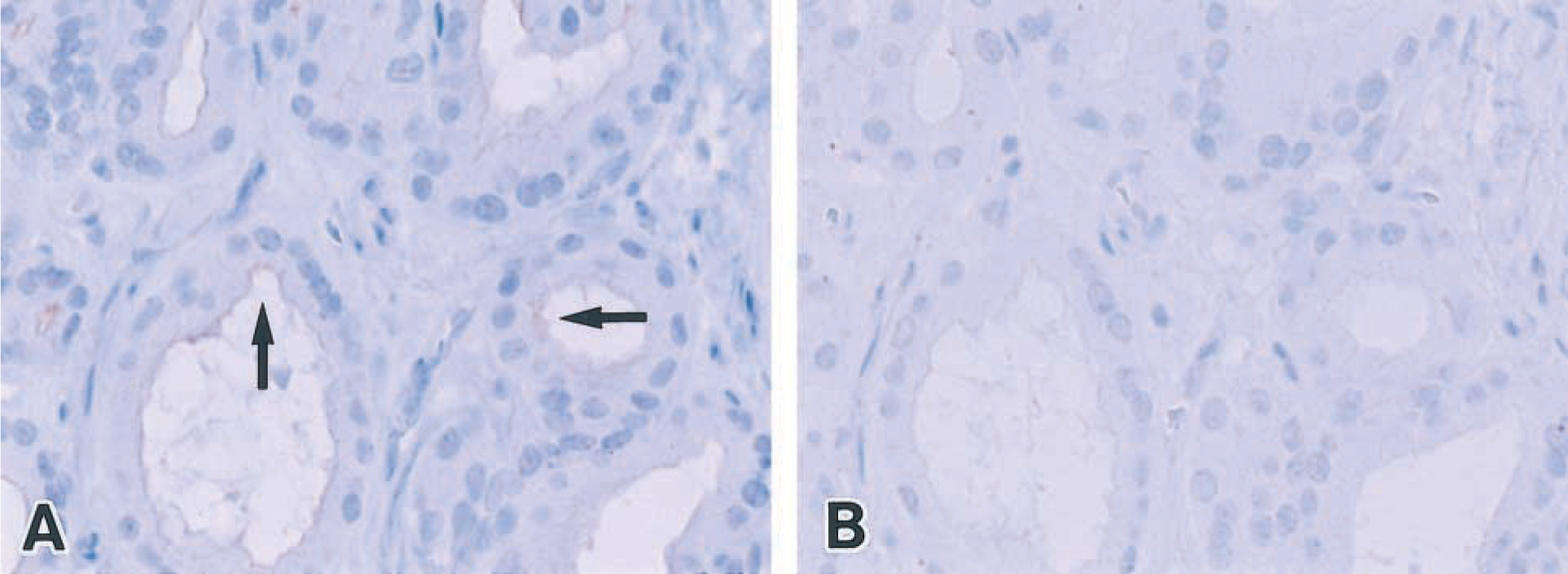
Immunohistochemical absorption test. (
In neoplastic thyroid tissues, pendrin immunostaining was demonstrated only in follicular adenomas. Pendrin was restricted to follicle-forming parts of follicular adenoma but was negative in trabecular parts (Figure 3E). No convincing positivity was detected in any follicular carcinomas or papillary carcinomas (Figure 3F). Both medullary carcinomas did not react with the pendrin antiserum.
Real-time Quantitative RT-PCR
The relative values of pendrin mRNA in normal thyroid tissues varied and were not significantly different from those in Graves’ disease tissues, nodular goiters, and follicular adenomas (Figure 4; Table 2). In papillary carcinomas, the values of pendrin mRNA were significantly lower than those in normal thyroids, Graves’ thyroids, nodular goiters, and follicular adenomas (p<0.01). In one case of medullary carcinoma, the value of pendrin mRNA was quite low. Despite using the same amount of total RNA used for PCR reaction (1.25 ng of cDNA/25 μl PCR reaction buffer), the values of 18S rRNA were higher in Graves’ disease and follicular adenomas than in normal thyroids (p<0.05) (Table 3).
Immunohistochemical results of pendrin a
aGrade 0, no staining; grade 1, focal and faint; grade 2, intermediate between grade 1 and grade 3; grade 3, extensive and strong.
Discussion
In the present IHC study, an antipeptide antiserum against human pendrin specifically reacted with the apical membrane of normal follicular cells. This finding supports the hypothesis that pendrin is an apical porter of chloride/iodide (Bidart et al 2000; Royax et al. 2000). In normal thyroids, the expression of pendrin was focal and faint and was found only in a small number of follicles. However, there was a tendency for cuboidal follicular cells of small follicles to be more frequently stained compared with flat follicular cells of large follicles. These heterogeneous immunoreactivities were also observed in nodular goiters. The heterogeneity in the follicular function can be demonstrated by IHC for other thyroid-specific proteins, e.g. thyroglobulin (Tg), tri-iodothyronin (T3), thyroxin (T4), and thyroid-specific transcriptional factor-1 (TTF-1) (Kawaoi et al. 1981,1982,1983; Tasca et al. 1985; Suzuki et al. 1998,1999; Katoh et al. 2000). Therefore, distribution of pendrin-positive cells may reflect a functional heterogeneity of thyroid follicles.
On scintigraphic scan examination, Graves’ thyroids accumulated radioiodide intensely. Strong NIS expression may contribute to this radioiodide accumulation in Graves’ thyroids. In fact, Graves’ thyroids are higher in NIS gene expression than normal thyroids (Caillou et al. 1998; Saito et al. 1998; Joba et al. 1999). In the current study, the immunoreactivities of pendrin in Graves’ thyroids were more extensive and intense than those in normal thyroids. This finding suggests that pendrin expression may contribute to iodide accumulation in conjunction with NIS.
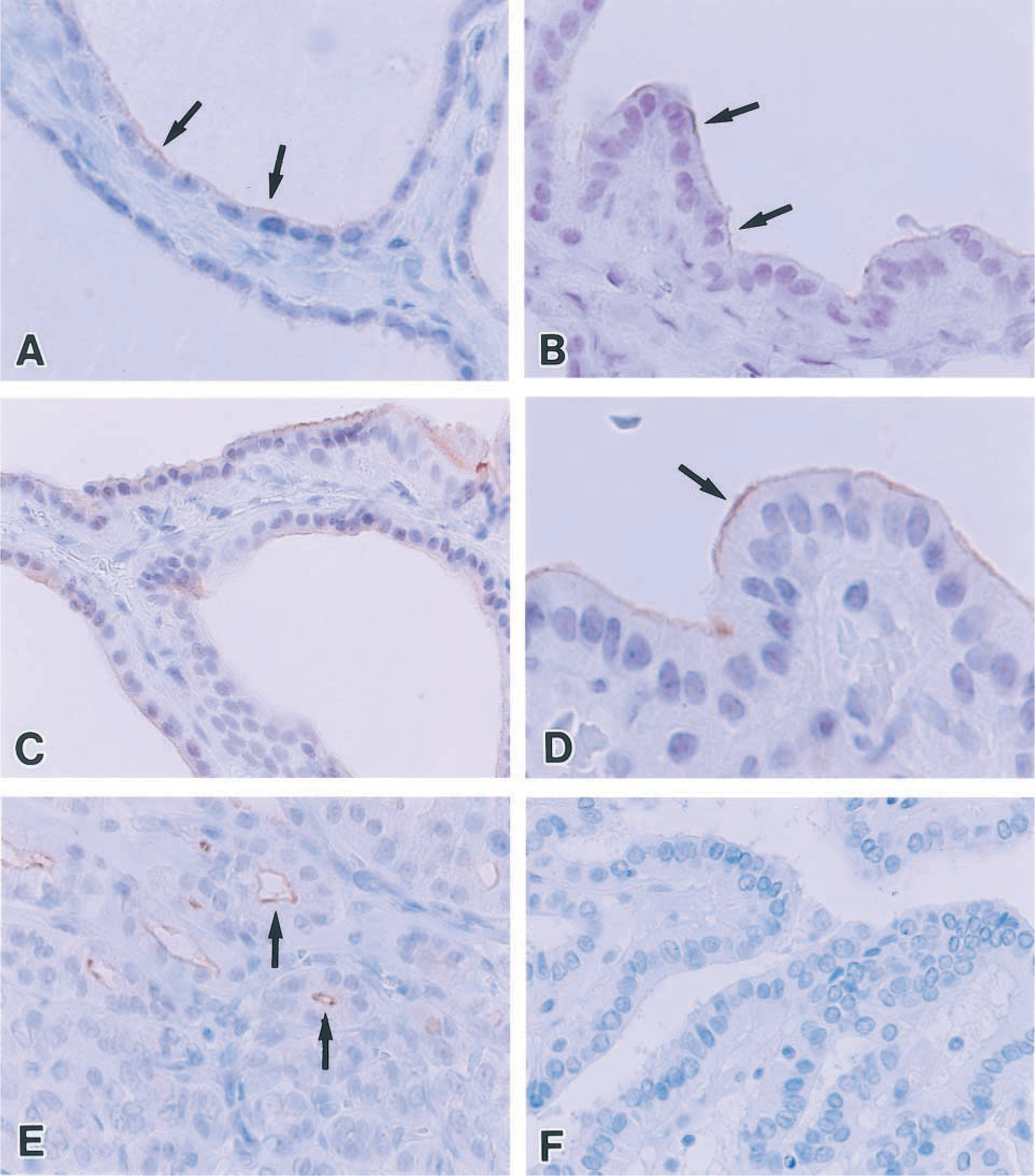
Immunohistochemical findings of pendrin. (
All 16 follicular adenomas showed immunopositivity for pendrin in association with the follicular structure. In contrast, we failed to demonstrate pendrin in all follicular and papillary carcinomas. This finding may suggest the absent or very low functional state of iodine transport in thyroid carcinoma tissue.
Real-time quantitive RT-PCR analysis revealed a low expression of pendrin mRNA in papillary carcinoma tissues. This finding is reasonable and supports our IHC results. However, the values of pendrin mRNA in Graves’ thyroids were not significantly different from those in normal tissues. In the IHC examination, the immunopositivities of pendrin were more extensive and stronger in Graves’ thyroids. Therefore, there was a discrepancy in the results between the IHC and the real-time quantitative RT-PCR analyses. Bidart et al. (2000) reported similar paradoxical results. They analyzed PDS mRNA expression by real-time RT-PCR in thyroid tissues divided into four histological groups: normal thyroid, hyperfunctioning tissues (Graves’ disease and hyperfunctioning adenoma), hypofunctioning adenoma, and carcinoma (papillary carcinoma, poorly differentiated carcinoma, and widely invasive follicular carcinoma). In their results, PDS mRNA levels by real-time RT-PCR were not significantly increased in hyperfunctioning thyroid tissues despite the stronger immunostaining of pendrin. Concerning the discrepancy in the results of IHC and real-time quantitative RT-PCR, Bidart et al. (2000) suggested that the PDS gene expression is less sensitive to the stimulatory effect of the TSH pathways and that pendrin expression is regulated at the transcriptional level. In addition to their hypothesis, it can be reasonable to suggest that this discrepancy may be due to increased proportions of 18S rRNA in total RNA in Graves’ thyroid. In fact, the values of 18S rRNA in Graves’ disease were higher than those in normal thyroids. It has been reported that TSH stimulates synthesis of rRNA as well as mRNA (Cartouzou et al. 1968). Therefore, TSH-stimulating autoantibody could alter the proportions of 18S rRNA in Graves’ disease. When we took this into account, the quantitative RT-PCR results would simply be consistent with our IHC results. However, further investigations are needed to confirm this.
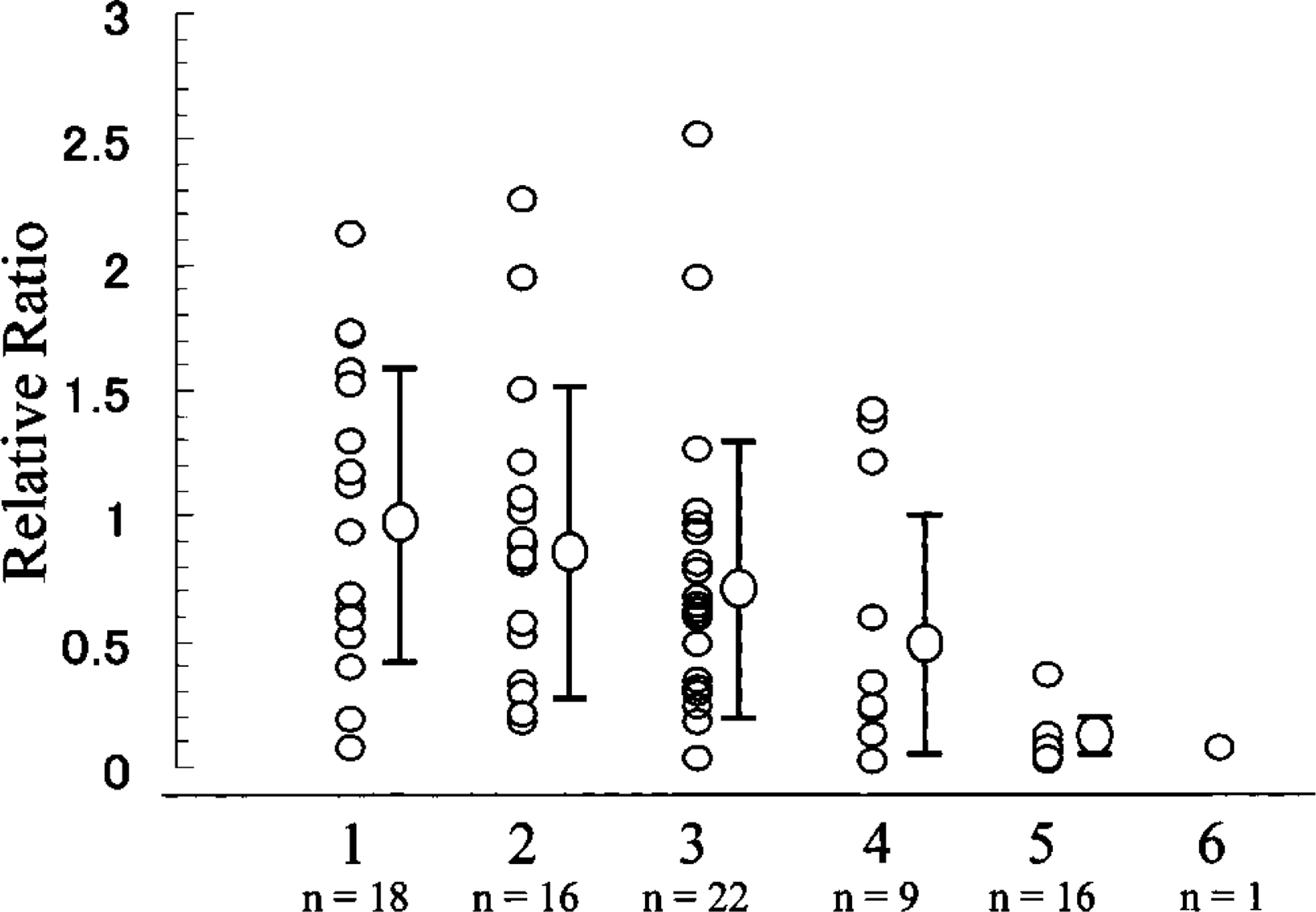
Relative quantitation of pendrin mRNA. 1, normal thyroid; 2, Graves’ thyroid; 3, nodular goiter; 4, follicular adenoma; 5, papillary carcinoma; 6, medullary carcinoma. Values are normalized with an endogenous control (18S rRNA) and are expressed as the n-fold difference relative to the mean value of normal thyroids. ∗ p<0.01 vs. normal thyroid, Graves’ disease, nodular goiter, and follicular adenoma by Student's t-test.
Relative quantitation of pendrin mRNA a
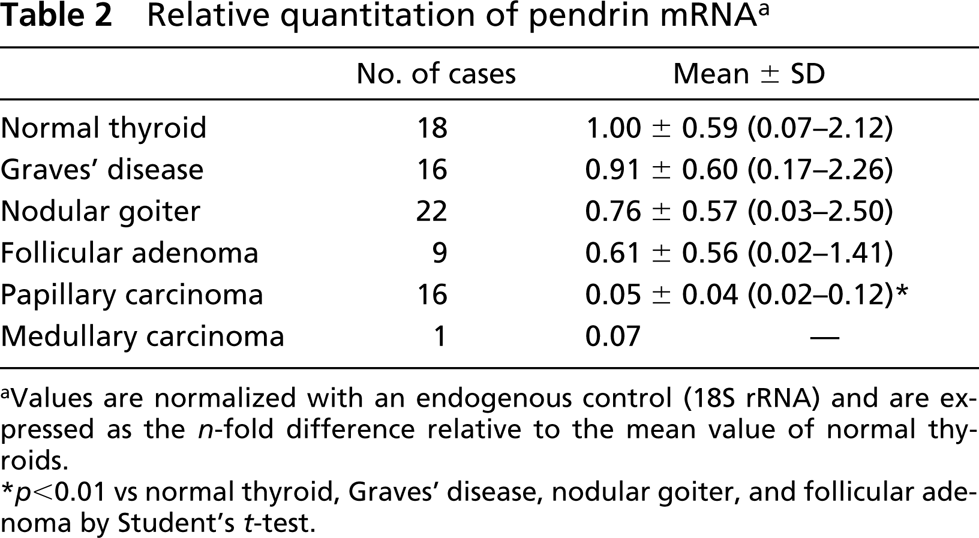
aValues are normalized with an endogenous control (18S rRNA) and are expressed as the n-fold difference relative to the mean value of normal thyroids.
∗ p<0.01 vs normal thyroid, Graves’ disease, nodular goiter, and follicular adenoma by Student's t-test.
Relative quantitation of 18S rRNA a
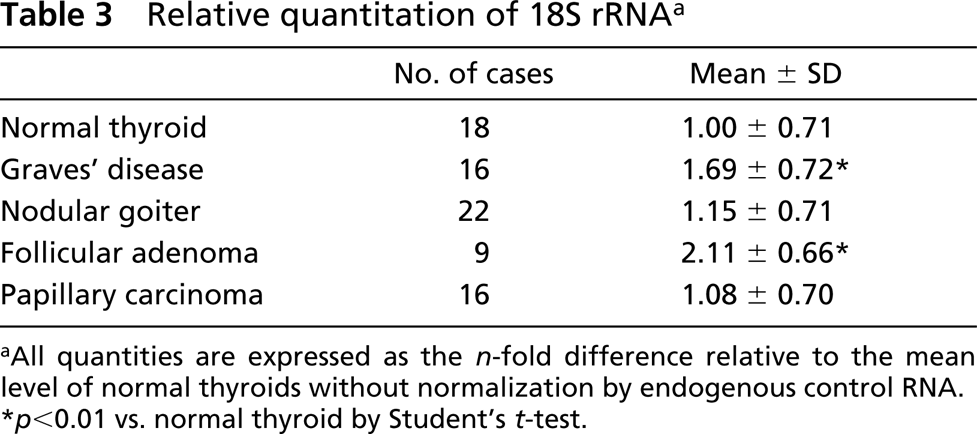
aAll quantities are expressed as the n-fold difference relative to the mean level of normal thyroids without normalization by endogenous control RNA.
∗ p<0.01 vs. normal thyroid by Student's t-test.
In conclusion, the present results suggest that pendrin may play a role in thyroid hormone production as an apical porter of chloride/iodide. Further investigations of pendrin should lead to a better understanding of the functional aspects of iodide transport in thyroid diseases.
Footnotes
Acknowledgements
We thank Prof Shigeyasu Tanaka (Department of Biology, Shizuoka University) for technical advice and helpful comments on the production of the antipeptide antiserum against human pendrin.
