Abstract
Keywords
Prostacyclin (PGI2) belongs to the family of prostanoids, which are labile bioactive lipids acting as local hormones (Smith 1986). Conversion of the precursor prostaglandin H2 (PGH2) to the chemically unstable prostacyclin is catalyzed by prostacyclin synthase (PGIS), a heme-thiolate protein with an apparent mass of 52 kD (Smith 1986; Hara et al. 1994). The actions of PGI2 are mediated by specific cell surface receptors called IP receptors, activation of which generally leads to elevation of intracellular cAMP through Gs-protein coupling to adenylate cyclase (Hashimoto et al. 1990; Coleman et al. 1994). Vascular endothelial cells are the major source of PGI2 (DeWitt et al. 1983), and vascular effects such as modulating tone of blood vessels and platelet suppressant actions are well described (Moncada and Vane 1978; Vane et al. 1990). In addition to the role of PGI2 in regulating vascular homeostasis, a number of studies document a role of PGI2 in pain perception (Ferreira et al. 1978; Davies et al. 1984). Various findings indicate that PGI2 is generated in response to injury or inflammation and can activate or at least sensitize peripheral afferent nerve endings (Birrell et al. 1991; Schepelmann et al. 1992; Smith et al. 1998). Moreover, high levels of IP receptors are located in the neurons of the dorsal root ganglia, indicating a role for PGI2 in facilitating pain transmission at spinal and supraspinal levels (Ferreira 1981; Malmberg and Yaksh 1992). In recent studies investigating the underlying mechanism, PGI2 has been shown to increase the release of neuropeptides such as substance P and calcitonin gene-related peptide by activating the cAMP transduction cascade (Hingtgen et al. 1995). Electrophysiological studies have revealed that the PGI2 analogue carbacyclin suppresses an outward potassium current, leading to enhanced excitability of rat sensory neurons (Nicol et al. 1997). The PGI2-induced increase in excitability and the accompanying neuropeptide release are believed to contribute to both neurogenic inflammation in the periphery and central sensitization at the level of the spinal cord, which clinically is manifested as hyperalgesia. The biological significance of these findings was recently highlighted by Murata et al. (1997), revealing that IP knockout mice show a decrease in pain perception.
Lately, there is new interest in the role of prostanoids in the CNS owing the discovery of the inducible isoenzyme of cyclo-oxygenase (COX-2). Although cyclo-oxygenase-1 (COX-1) is present under normal conditions in most cells, COX-2 is the predominant isoform in brain. In contrast to most other tissues, COX-2 is expressed under physiological conditions in forebrain neurons, particularly in neocortex, amygdala, hippocampus, and limbic cortices (Yamagata et al. 1993; Kaufmann et al. 1996). The basal expression of COX-2 in excitatory neurons has been shown to increase with synaptic activity (Kaufmann et al. 1996). Upregulation of COX-2 is reported under pathological conditions such as brain injury and ischemia or during development of Alzheimer's disease (Nakayama et al. 1998; Pasinetti and Aisen 1998; Sairanen et al. 1998). In this respect, increase of COX activity might be regarded as potentially harmful to the brain. Because upregulation of COX-2 leads to enhanced production of downstream metabolites, it is of major importance to analyze the localization and distribution of prostaglandin-synthesizing enzymes.
In this study we have investigated by immunohistochemistry and in situ hybridization the in vivo regional and cellular distribution of PGIS in various areas of the human brain, including frontal cortex, hippocampus, cerebellum, medulla oblongata, substantia nigra, and thalamus.
Materials and Methods
Postmortem Specimens
Tissue blocks from brains of three donors with no history of neurological illness were obtained from the Department of Pathology of the Robert Bosch Hospital (Stuttgart, Germany). The underlying illnesses were chronic respiratory insufficiency (female, 42 years) and illicit drug consumption (males, 33 years and 53 years). Postmortem delay time ranged from 20 to 26 hr. Tissue samples were fixed in buffered 4% formalin for 24 hr, dehydrated, and embedded in paraffin (Paraffin Plus; Shandon, Pittsburgh, PA). Sections were cut (3 ±) and mounted on glass slides. Fresh brain samples were also obtained from cortical and hippocampal surgical waste collected perioperatively from three patients with epilepsy. The removed specimens were immediately frozen in liquid nitrogen and stored at -80C before use as positive control for activity assays and Western blotting analysis.
Activity Assay of PGIS
PGIS activity in human brain tissues was determined by two independent methods. The conversion of 100 ± [14C]-PGH2 to the various [14C]-prostanoids in brain homogenates was measured by thin-layer chromatography (TLC) as described previously (Klein et al. 1994). Briefly, radioactive products were extracted at pH 3.0 into the organic solvent, gently evaporated under N2, and subsequently separated by TLC (ethyl acetate/water/iso-octane/acetic acid, 90:100:50: 20). The amount of prostanoid production was quantified with a phosphor imager system (Image Quant; Molecular Dynamics, Krefeld, Germany) and expressed as percent radioactivity of total amount of extracted radioactivity. Thereafter, the location of standard prostanoids (SPI Bio; Massy, France) was visualized with iodine vapor, and the Rf values of the metabolite standards were compared with the corresponding Rf values of the radioactive prostanoids.
Second, PGIS activity in brain preparations was measured by enzyme-linked immunoassay (EIA) using 30 ± PGH2 as substrate, according to instructions of the supplier (SPI Bio). As above, the amount of spontaneously formed 6-keto-PGF1α in buffer was subtracted from the individual samples.
Anti-PGIS Antibody
Anti-human PGIS polyclonal antibodies were used for immunohistochemical detection of PGIS (Klein et al. 1998). Western blotting analysis was performed with a monoclonal antibody directed against PGIS (5 μg/ml; Oxford Biochemical Research, Frankfurt, Germany).
Immunoprecipitation of PGIS
Extracts of 3 mg solubilized brain homogenates were precleared by addition of 40 μl of protein A/Sepharose CL-4B and the supernatant was incubated (18 hr, 4C) with 5 μg of the polyclonal antibody against PGIS (Klein et al. 1998). Immune complexes were precipitated with 30 μl of protein A CL-4B and washed with 0.5 ml SNNTE (0.5% sucrose, 1% NP-40, 0.5M NaCI, 50 mM Tris, 5 mM EDTA, pH 7.4). Protein pellets were then precipitated by centrifugation (14,000 μg, 1 min) and then resuspended in 40 μl Laemmli sample buffer/β-mercaptoethanol (9:1), heated at 95C for 5 min, and kept on ice. In addition, a sample of partially purified bovine aortic endothelial PGIS (DEAE-Sephacel column eluate) was used as positive control for Western blotting analysis. The isolation of PGIS was performed as described by Hara et al. (1994).
Western Blotting
Precipitated proteins were separated by 7.5% SDS-PAGE (30 mA, 1 hr) and blotted for 1 hr with a constant current of 200 mA onto a nitrocellulose membrane in a semidry blotting procedure (48 mM Tris/39 mM glycine/20% methanol/0.037% SDS). Proteins were visualized with a 0.1% Ponceau 5 solution in 5% acetic acid to check transfer efficiency. After destaining, the membrane was blocked with 5% milk powder in PBS/0.1% Tween-20 for 2 hr at room temperature (RT) and incubated with a monoclonal antibody against PGIS (Oxford Biochemical Research; 5 μg/ml). After washing three times with PBS/0.1% Tween-20, the membrane was further incubated with a goat anti-mouse antibody at a dilution of 1:7500 for 45 min. Antibody binding was visualized by the ECL technique (Amersham; Braunschweig, Germany).
Immunohistochemical Staining for PGIS
Immunostaining was performed with a modification of the avidin-biotin-peroxidase complex (ABC) technique (Hsu et al. 1981) as described previously (Siegle et al. 1998). Sections were stained at the same time to avoid possible inter-day variation. After deparaffinizing, sections were rehydrated and microwaved three times for 5 min for antigen retrieval. Sections were incubated with the specific polyclonal antibodies against PGIS (dilution 1:50) for 18 hr at RT in a humidified chamber, followed by incubation with a biotinylated goat anti-rabbit secondary antibody (Vector Laboratories; Burlingame, CA) diluted 1:200 for 30 min. A Vectastain ABC-Elite Kit (Vector Laboratories) was used to localize PGIS, with a chromogen of oxidized 3,3′-diaminobenzidine (DAB, 50 mg/100 ml PBS buffer/H2O2 0.03%) that was apparent as dark brown staining. Counterstaining was performed with hematoxylin for 30 sec.
To test the specificity of immunostaining, a 10-fold molar excess of the PGIS peptide was added to the anti-PGIS antibody solution 60 min before staining. No staining was observed. False-positive results originating from endogenous biotin (Wang and Pevsner 1999) were excluded because visualization of antibody binding with the APAAP technique yielded results similar to those of the ABC technique. In addition, the following control experiments were carried out (data not shown): using preimmune rabbit sera instead of the primary antibodies (in the same dilutions); omitting the primary antibody (a); omitting the secondary antibody (b); omitting the ABC complex (c); omitting a, b, and c.
Morphology
To characterize general cellular patterns, sections were stained with hematoxylin/eosin. In addition, double staining experiments for GFAP (glial fibrillary acidic protein, dilution 1:250; Progen, Heidelberg, Germany) and von Willebrandt factor (vW factor, dilution 1:400; Dako, Hamburg, Germany) and PGIS were performed. GFAP is a astroglia-associated antigen and vW factor is an endothelial cell marker. The double immunostaining was carried out with a sequential method. First the sections were stained with the polyclonal antibody against PGIS by the ABC technique. Color development was achieved by DAB/H2O2 with addition of 6.8 mg imidazole, 2 ml 1% nickel acetate, and 2.5 ml 1% CoCl2 (to each 100 ml of incubation solution), which produced a black color instead of brown. Thereafter, the sections were stained for GFAP or vW factor using an APAAP method with one repetition. The APAAP complex was developed with New Fuchsin and naphthol-AS-BI (red color) as described previously (Fritz et al. 1984).
Generation of 35S-labeled Riboprobes and In Situ Hybridization
Antisense and sense probes for the human prostacyclin mRNA were prepared as follows. RT-PCR with the depicted primer pair was performed using total human kidney RNA. A PCR fragment (420 bp) was cloned into pCR 2.1 plasmid (Invitrogen; Groningen, The Netherlands). Cloned cDNA fragments were sequenced according to the dideoxy method to confirm identity and orientation of the inserts.
In situ hybridization was performed as described previously (Breyer et al. 1994). Briefly, before hybridization, human brain sections were deparaffinizied, refixed in para-formaldehyde, treated with proteinase K (20 μg/ml), washed with PBS, refixed in 4% paraformaldehyde, and treated with triethanolamine plus acetic anhydride (0.23% v/v). Finally, sections were dehydrated with 100% ethanol. 35S-Labeled antisense and sense riboprobes synthesized from the coding region of the human PGIS cDNA were hybridized to the section at 55C for 18 hr. After hybridization, the specimens were washed once at 65C in 5 × SSC plus 10 mM β-mercaptoethanol (BME), once in 50% formamide, 2 × SSC, and 100 mM BME for 30 min. After additional washes in 10 mM Tris-HCl, 5 mM EDTA, 500 mM sodium chloride (TEN) at 37C, the sections were treated with RNase A (10 μg/ml) at 37C for 30 min, followed by another wash in TEN at 37C. Sections were then washed in 2 × SSC and twice in 0.1 × SSC at 65C. Slides were dehydrated with graded ethanol containing 300 mM ammonium acetate. Slides were then dripped in emulsion (Ilford K5; Knutsford, Cheshire, UK) diluted 1:1 with 2% glycerol and exposed for 4-5 days at 40C. After developing in Kodak D-19, slides were counterstained with hematoxylin.
Results
Western Blot Findings
Western blotting analysis was performed to demonstrate the presence of PGIS immunoprecipited with polyclonal anti-PGIS antibodies from human brain homogenates. In brain samples, monoclonal antibodies against PGIS recognized a single protein band of 52 kD (Figure 1, Lanes 2-4), consistent with the molecular weight of partially purified PGIS from bovine aortic endothelium (Figure 1, Lane 1). In general, the amount of immunoprecipited PGIS protein was unchanged in cortex of postmortem tissues (Figure 1, Lane 4) compared with the same areas of freshly obtained surgical material (Figure 1, Lanes 2 and 3, respectively).
Incubation of solubilized brain extracts with PGH2, the immediate substrate of PGIS, caused significant production of 6-keto-PGF1α, the stable degradation product of prostacyclin, as shown in Figure 1. Compared to freshly obtained material, the activity of PGIS in postmortem tissues was decreased to 31% and 67% in cortex and hippocampus, respectively. This decline in enzymatic activity was most likely related to inactivation of PGIS due to an ischemic period before death, as proposed by Zou and Bachschmid (1999). The identity of the metabolite was further validated by TLC and corresponded to a total conversion of 3.9 ± 1.3% 6-keto-PGF1α from [14C]-PGH2 in freshly obtained cortical tissues. Considerably larger amounts of PGE2 (40.3 ± 5.5%) and PGD2 (19.3 ± 2.1%) were formed as the result of spontaneous decay of PGH2 in aqueous solutions. In good agreement with the Western blotting analysis, these results clearly indicated that PGIS is expressed in notable amounts in different regions of human brain.
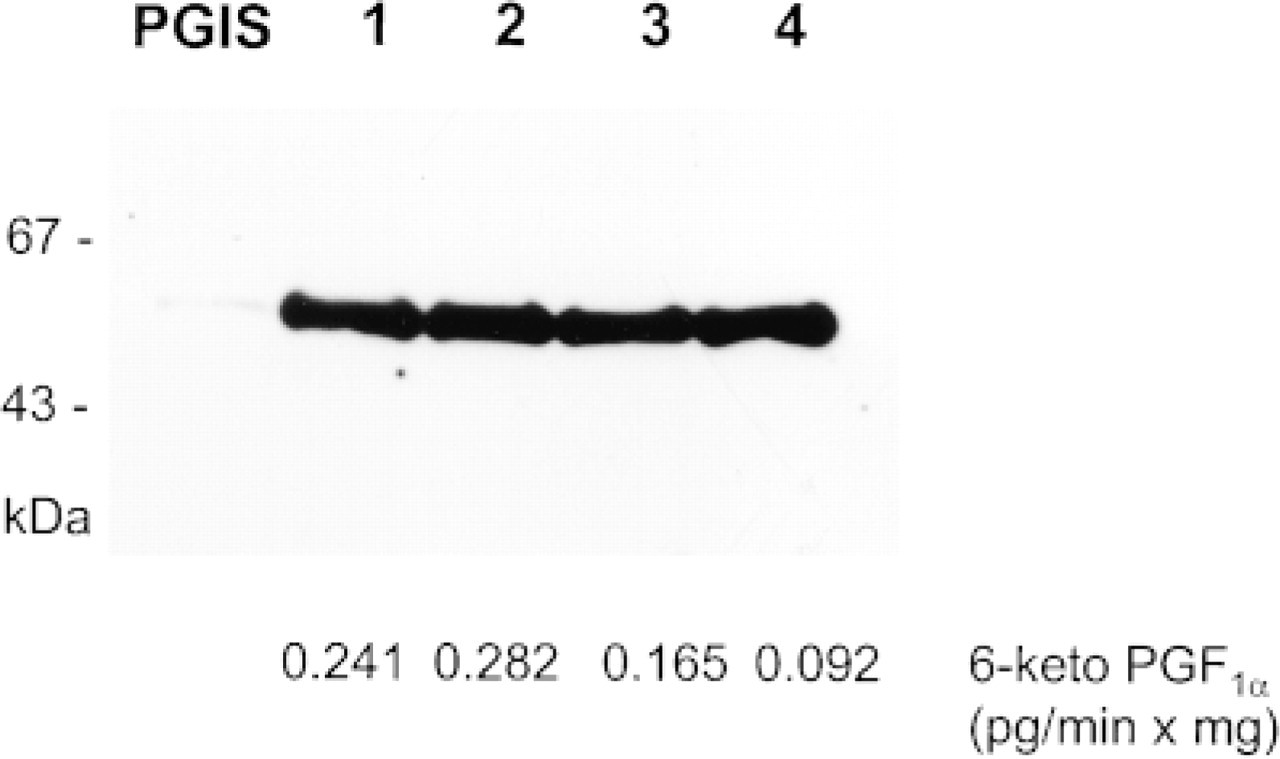
Immunopreciptitation of PGIS from human brain homogenates. PGIS was immunoprecipitated from equal amounts (3 mg) of proteins from freshly obtained cortex (Lane 2) and hippocampus (Lane 3) and from postmortem cortex (Lane 4) and were analyzed by Western blotting with a monoclonal antibody (5 μg/ml) against PGIS as described in Materials and Methods. Lane 1 shows a sample of partially purified bovine PGIS (DEAE/Sephacel column eluate) which has co-migrated exactly with the immunoprecipitated proteins from human brain. PGIS was equally expressed in all brain samples. The migration of pre-stained molecular weight standards (in kD) is shown at left. PGIS activity from corresponding samples is indicated below as formed 6-keto-PGF1α (pg/min × mg) from 30 ± PGH2 measured by EIA. Altered enzyme activity is observed in the postmortem tissues.
Immunohistochemical Findings
Incubation of brain sections with an antibody raised against a peptide homologous to human PGIS caused staining of large and small blood vessels throughout the brain. In addition, neuronal cells, glial cells of presumed microglial identity, and oligodendrocytes were found to contain PGIS immunoreactivity in most areas of the brain. No immunostaining was detected in tissue sections when any of the crucial steps of the immunostaining procedure was omitted or competitive inhibition experiments with an excess of PGIS peptide were performed. Figure 2A shows PGIS-ir Purkinje cells in cerebellum. This staining of PGIS-positive cells was completely eliminated when pre-absorbed antibody was used as primary antibody (Figure 2B). The immunoreactivity in cerebral blood vessels appeared to be mainly located in endothelial cells (Figure 3A). Figure 3B shows double immunostaining for PGIS-like immunoreactivity and vW factor, an endothelial cell marker. All of the PGIS-positive structures overlapped in staining with vW factor-positive structures. In most vessels, however, vW factor-ir cells were further surrounded by PGIS-positive cells. This staining pattern was consistent with the well known expression of PGIS in endothelial cells as well as in underlying vascular smooth muscle cells (DeWitt et al. 1983).
Immunolocalization of PGIS in frontal cortex showed widespread staining in all cortical laminae but occurred predominantly in the cortical Layers III/V and VI, in which large neurons are located (Figure 4A). In most PGIS-positive samples, intense immunostaining was detected in neurons, either pyramidal or fusiform. These cells displayed a characteristic PGIS-ir in the cytosol, sometimes extending towards dendritic processes but without nuclear staining (Figure 4B). In some cases, immunoreactivity was also localized to the neuropil. Moderate staining was abundant in the majority of small cells, which morphologically appeared to be glial cells (Figure 4C). Hence, judging from the double staining experiment with PGIS and GFAP, a cell marker for astrocytes, it can be assumed that these cells are most likely microglial cells or oligodendrocytes rather than astrocytes (Figure 4D). These findings are in contrast to those of Murphy et al. (1985), who reported that primary CNS astrocytes synthesize and release prostacyclin. Thus, immunohistochemistry in human frontal cortex suggests that both neurons and glial cells such as microglia and oligodendrocytes stain with PGIS specific antibodies.
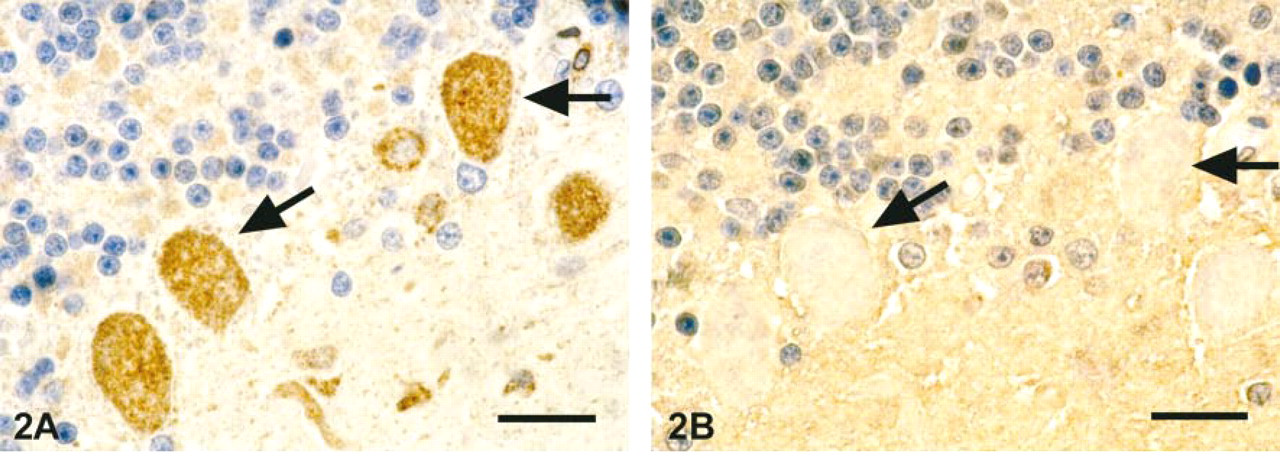
Immunohistochemical staining for PGIS with pre-absorbed antibody in human cerebellum sections. Immunostaining of formalin-fixed cerebellum tissue was performed with a modification of the ABC technique as described in Materials and Methods. PGIS expression is mainly seen in Purkinje cells, as indicated by arrows (
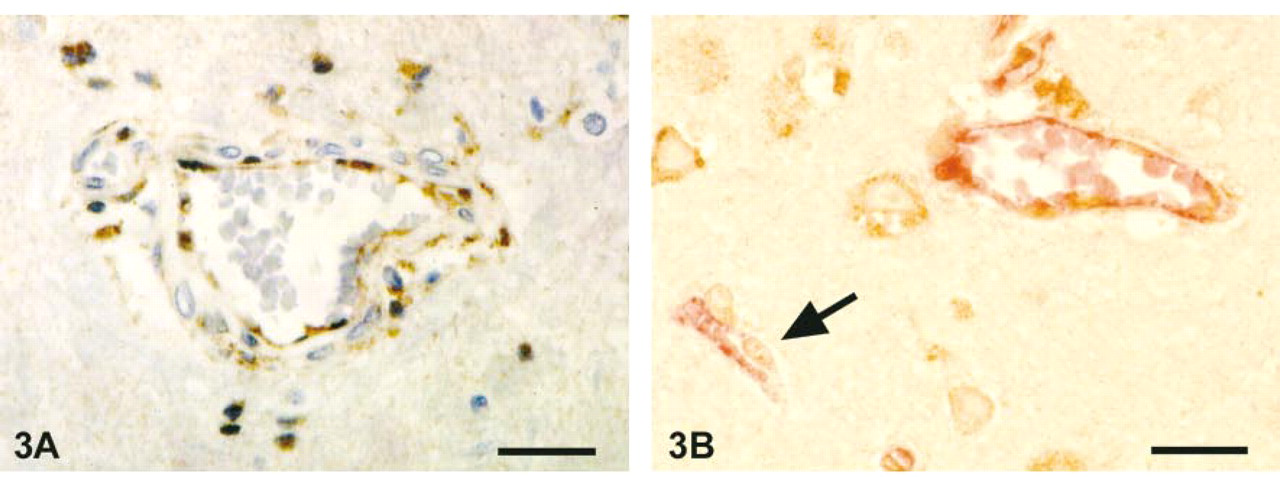
Expression of PGIS in cerebrovascular blood vessels. Endothelial cells and underlying smooth muscle cells show intensive staining for PGIS (
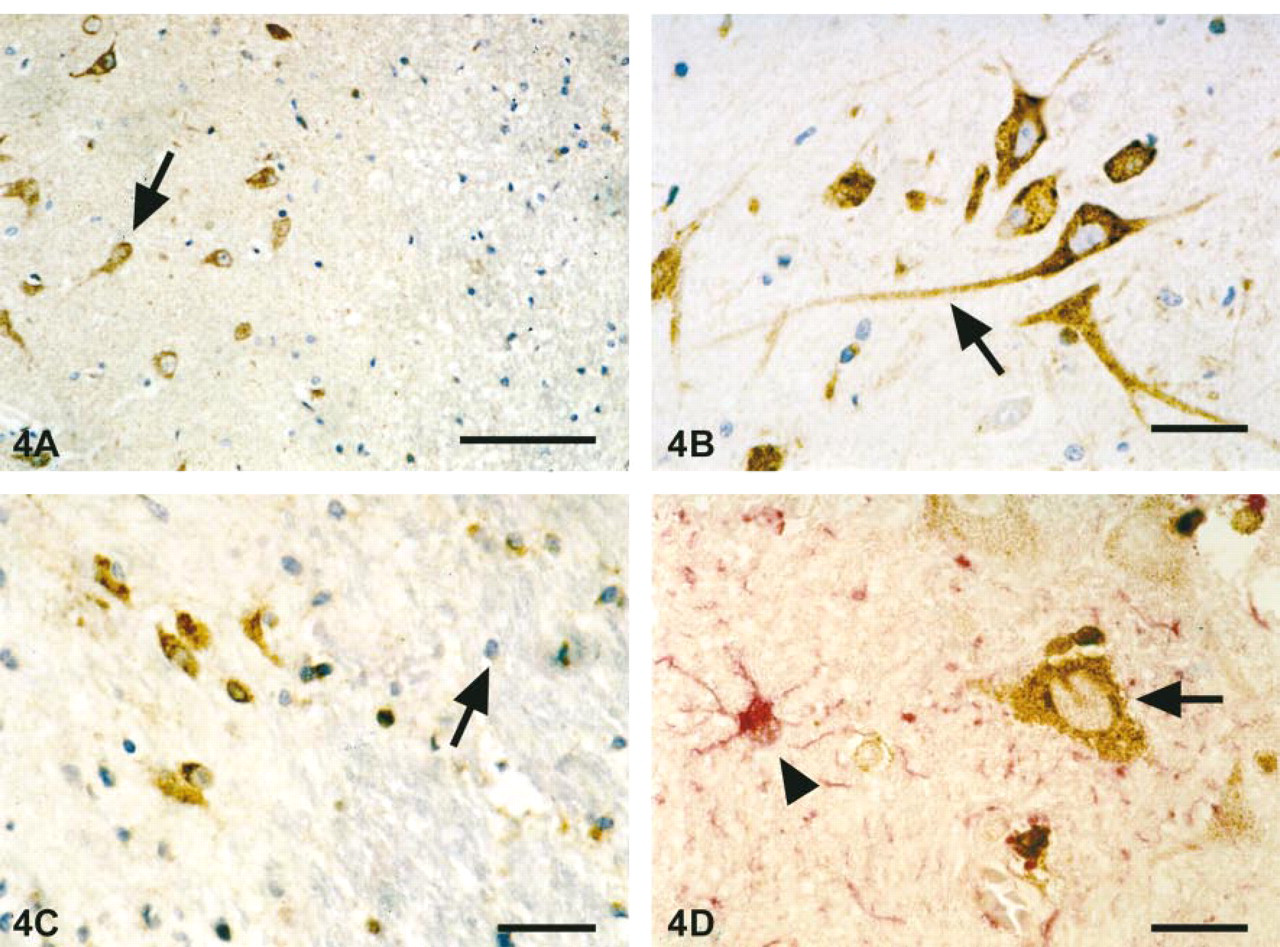
Localization of PGIS-ir protein in human cortex sections. (
The hippocampus exhibited a distinct pattern of immunostaining for PGIS. Similar to the staining of cortical tissues, the immunoreactivity for PGIS was quite extensive. A dense accumulation of PGIS-positive neurons was found in granule cells of the dentate gyrus and in the pyramidal cell layer of Ammon's horn, starting with PGIS-positive CA3 neurons in the hilus of dentate gyrus and extending into the subiculum as a broad band of slightly to moderately stained immunoreactive CA1 neurons (Figures 5A and 5B). Shorter and longer dendritic processes of these cells contained immunoreactivity (Figure 5C). Moderate staining of glial cells was detected in the molecular layer of the dentate gyrus. In general, the staining intensity among the various cells was similar, but some neurons of the gyrus dentatus appeared to express more PGIS, as shown in Figure 5D.
Immunostaining for PGIS in cerebellum was primarily located in Purkinje cell bodies and processes and in discrete cells of the molecular and granular layers. As in other regions of the brain, PGIS was expressed in neurons, whereas astrocytes in the Purkinje cell layer lacked detectable PGIS immunoreactivity (Figures 6A and 6B). The small cells positive for PGIS in the Purkinje layer were closely associated with Purkinje cells, suggesting that these cells might be Bergmann glia. In the granular layer adjacent to the Purkinje cell layer, there were also occasional PGIS-positive cells (Figure 6C). In the molecular layer, staining for PGIS was present, most likely in stellate and basket cells (Figure 6D).
Strong immunostaining was also observed in motor neurons of the medulla oblongata (Figure 7A) and in the ependymal lining of the ventricular walls (Figure 7B). Furthermore, the melanin-containing cells in the substantia nigra showed PGIS-positive staining (not shown), as did the large neurons in the thalamus (Figure 7C).
In Situ Hybridization Findings
To confirm the immunohistochemical data, we determined mRNA expression of PGIS using [35S]-cRNA. The following control experiments were performed to validate the specificity of in situ hybridization. (a) Hybridization with sense probe resulted in very low labeling with nonspecific distribution. (b) Pretreatment of sections with RNase A before hybridization completely prevented the appearance of positively stained cells (not shown). In general, the results of in situ hybridization corresponded well to the immunohistochemical findings. Expression of the PGIS gene was relatively strong in large neurons, with a distinct pattern throughout the cytoplasm, as shown in Figures 8A and 8B for neurons in the frontal cortex. In contrast, only diffuse labeling was apparent in glial cells. However, significant mRNA staining was observed in the adventitia and media of cerebrovascular blood vessels, being equally intense in both layers (Figure 8C and 8D), whereas in hippocampus the hybridization signal was most prominent over the dentate gyrus. However, the high cellularity of this structure and the scattering of the silver grains precluded a unequivocal identification of PGIS mRNA-positive cells (not shown).
Discussion
In this study, expression of authentic PGIS in human brain is evidenced by four independent methods. (a) 6-Keto-PGF1α, the stable breakdown product of PGIS activity, was detected after incubation of brain extract with the unlabeled and 14C-labeled precursor PGH2 by EIA and TLC methods. (b) Western blotting analysis using monoclonal anti-PGIS antibodies showed a band co-migrating with purified PGIS. (c) Pre-absorption of the polyclonal anti-PGIS antisera discriminated specific from nonspecific staining in brain tissues sections. Finally (d), the overall distribution the PGIS immunoreactivity matched the PGIS mRNA expression. It should be noted that PGIS mRNA was detected mainly in neurons, whereas only diffuse staining appeared in glial cells. Brain tissue samples were obtained 20-26 hr postmortem. Therefore, it is possible that the prolonged time before fixation caused degradation of mRNA and hence a decrease in in situ hybridization signals. Our major finding is that most of the brain tissue samples analyzed exhibited PGIS-ir protein and mRNA expression in neuronal cell bodies and fibers and, to a lesser extent, in glial cells. Prominent immunostaining for PGIS was observed in a number of different neuronal cell types, including pyramidal cells in neocortex and CA3 pyramidal cells in hippocampus, acetylcholine-containing motor neurons in medulla oblongata, and dopaminergic neurons in substantia nigra.
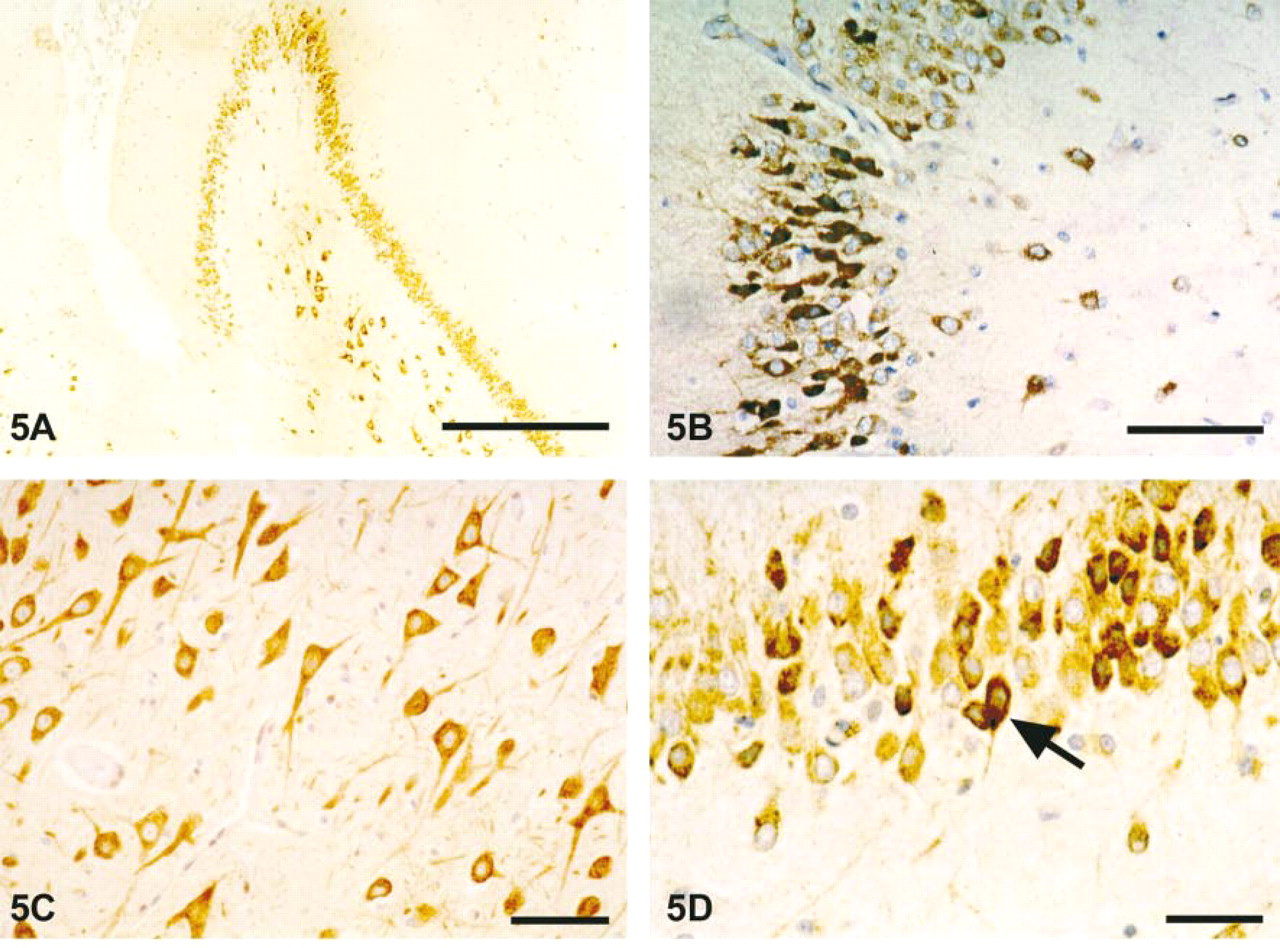
Localization of PGIS-ir protein in human hippocampal sections. (
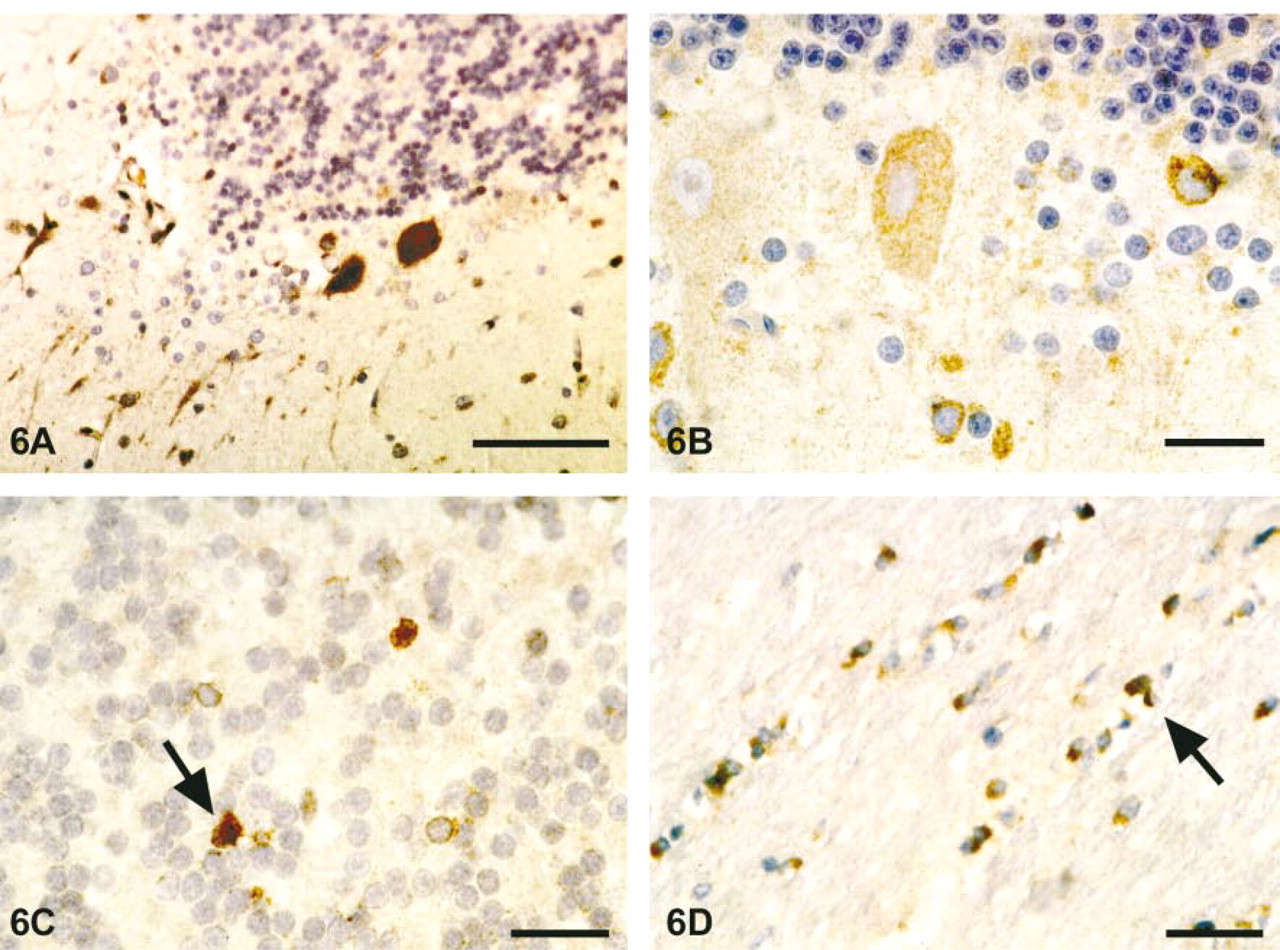
Localization of PGIS-ir protein in human cerebellar sections. (
Substantial evidence indicates that prostanoids such as PGE2, PGD2, and PGF2α play an important role in modulating neuronal functions, including those involving sleep, body temperature changes, and neuronal signaling (Hayaishi 1988; Shimizu and Wolfe 1990; Coceani and Akarsu, 1998). In contrast, knowledge about the specific functions of PGI2 in human brain is quite limited. In fact, the assumption that prostacyclin is synthesized in brain tissue in vivo is based mainly on primary cell cultures, such as astrocytes and meningeal cells (Murphy et al. 1985) and the presence and distribution of PI receptor in rodent brain (Matsumura et al. 1995; Oida et al. 1995; Takechi et al. 1996; Watanabe et al. 1999). The ability of neurons to express considerable amounts of PGIS mRNA and protein prompts the question: What might be the physiological relevance of prostacyclin in CNS? In the peripheral nervous system, prostacyclin appears to be involved in induction and maintenance of hyperalgesia by sensitization. The most convincing data have been gained by Murata and colleagues (1997) using mice lacking the prostacyclin receptor. In these animals, the acetic acid-induced writhing response and the paw edema resulting from carrageenan injection were reduced to the same levels as those found in indomethacin-treated wild-type mice. In contrast, PGE2 had almost no effect on the writhing response. Most of the reported actions of prostacyclin on peripheral terminals of nociceptive neurons has been of a facilitative nature, i.e., fibers respond to the same stimulus with a higher discharge rate. If peripheral and central sensory neurons possess similar signal transduction for PGI2, it could be speculated that prostacyclin also modulates transmission of sensory signals. Second, under pathophysiological conditions such as hyperalgesia, prostacyclin could contribute to nociception in the CNS.
Until now, only one IP receptor has been cloned (Boie et al. 1994; Nakagawa et al. 1994). In a previous study, the classical platelet IP receptor (IP1 receptor) was found to be localized in the CNS, predominantly in the nucleus tractus solitarius, spinal trigeminal nucleus caudalis and dorsal horn (Matsumura et al. 1995). More recently, a pharmacologically novel subtype of IP receptor has been proposed in rat brain which differs from the IP1 receptor, being especially abundant in hippocampus, cerebral cortex, and striatum (Takechi et al. 1996; Watanabe et al. 1999). However, it is not evident yet whether a second neuronal IP2 receptor actually exists, as there is no molecular biological proof. Because prostacyclin is a very unstable compound, it is reasonable to assume that PGIS either is co-localized with the IP receptor or at least is expressed in neighboring cells. When we compared our results with the IP receptor distribution in rat brain, we observed some similarities with the expression of PGIS in human brain. We found positive PGIS immunoreactivity in large neuronal cells in hippocampus, suggesting that PGI2 might act in an auto-crine way on these neurons. The functional significance of PGIS in pryramidal cells in hippocampus is unclear. Recent reports have indicated that PGI2 might stimulate excitatory transmission among neurons in this region (Takechi et al. 1996). On the other hand, prostacyclin could have neuroprotective effects (Cazevieille et al. 1993; Matsuda et al. 1997). Indeed, administration of isocarbacyclin, a prostacyclin analogue, has been shown to protect against delayed pyramidal cell death in the hippocampus of stroke-prone spontaneously hypertensive rat under ischemic conditions (Yamashita et al. 1996). Conversely, induction of COX-2, the key enzyme in prostanoid synthesis, has been discussed as an important factor in the etiology of neuronal cell death. New data have confirmed that increased expression of COX-2 after ischemia mainly occurs in CA1 neurons of the hippocampus that undergo necrotic cell death, probably via PGE2 synthesis (Kondo et al. 1998; Nakayama et al. 1998). However, not all neurons that show increased COX-2 synthesis after ischemia die. On the basis of these findings, we hypothesize that the close anatomic location of PGIS in CA3 neurons and the localization of low levels of PGIS in CA1 neurons synthesizing PGI2 might be involved in promoting cell survival.
Considerable evidence confirms that prostacyclin contributes to the control of cerebral blood flow (CBF) and circulation. For example, intracarotid infusion of PGI2 in baboons increased CBF (Pickard and Mackenzie 1973), whereas indomethacin has been reported to cause reduction of CBF (Pickard et al. 1980). In general, cerebral vascular cells are believed to be the predominant source of prostacyclin in CNS (Leslie and Watkins 1985) and, accordingly, we found PGIS-ir protein and mRNA to be expressed prominently in small and large blood vessels throughout the brain. This finding corresponds well to data from Tone et al. (1997), who have investigated arterial vessels in rats, in which they also observed strong hybridization signals in both cell types.
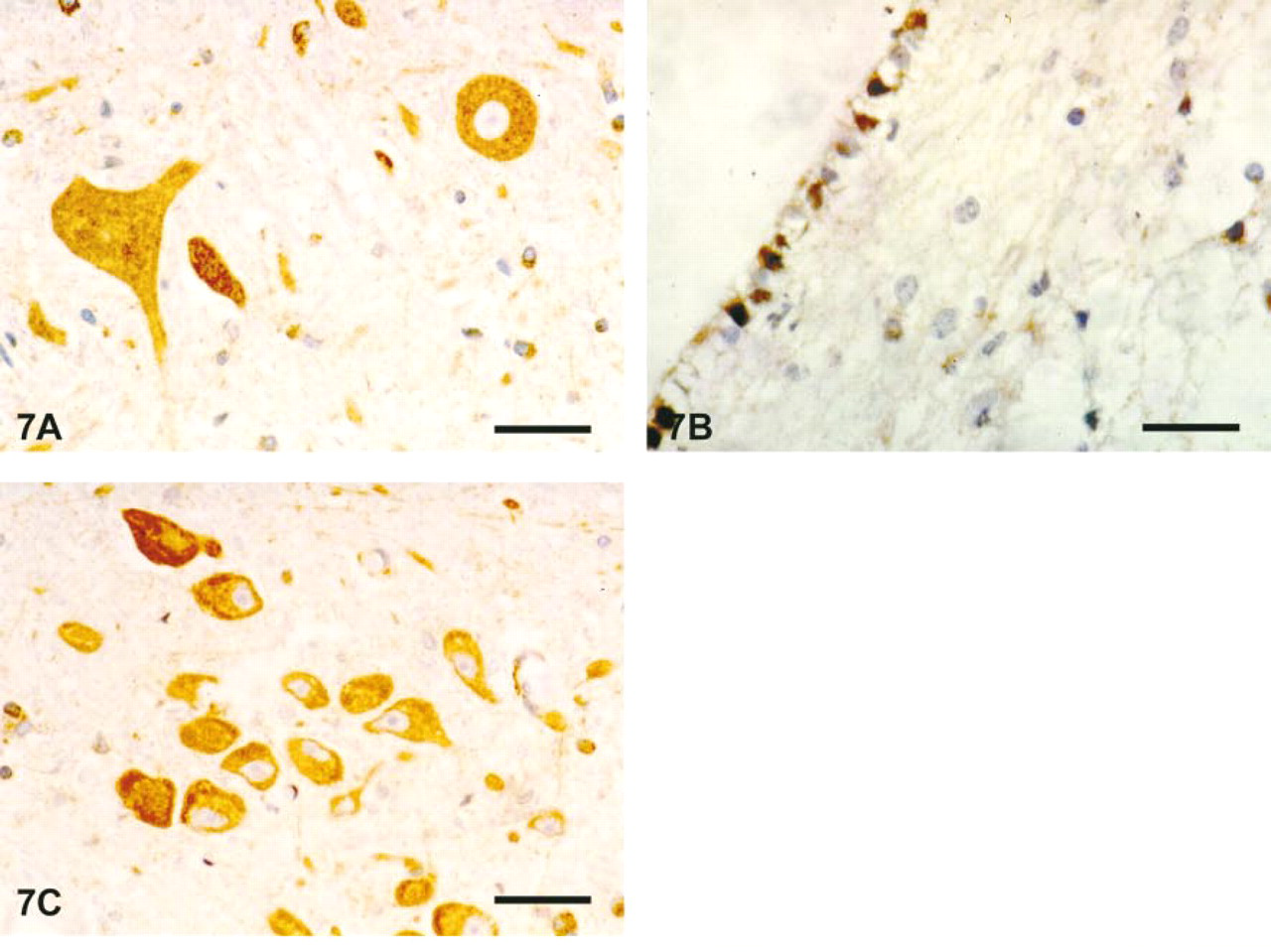
Representative photomicrographs of PGIS-ir protein in motoneurons of medulla oblongata (
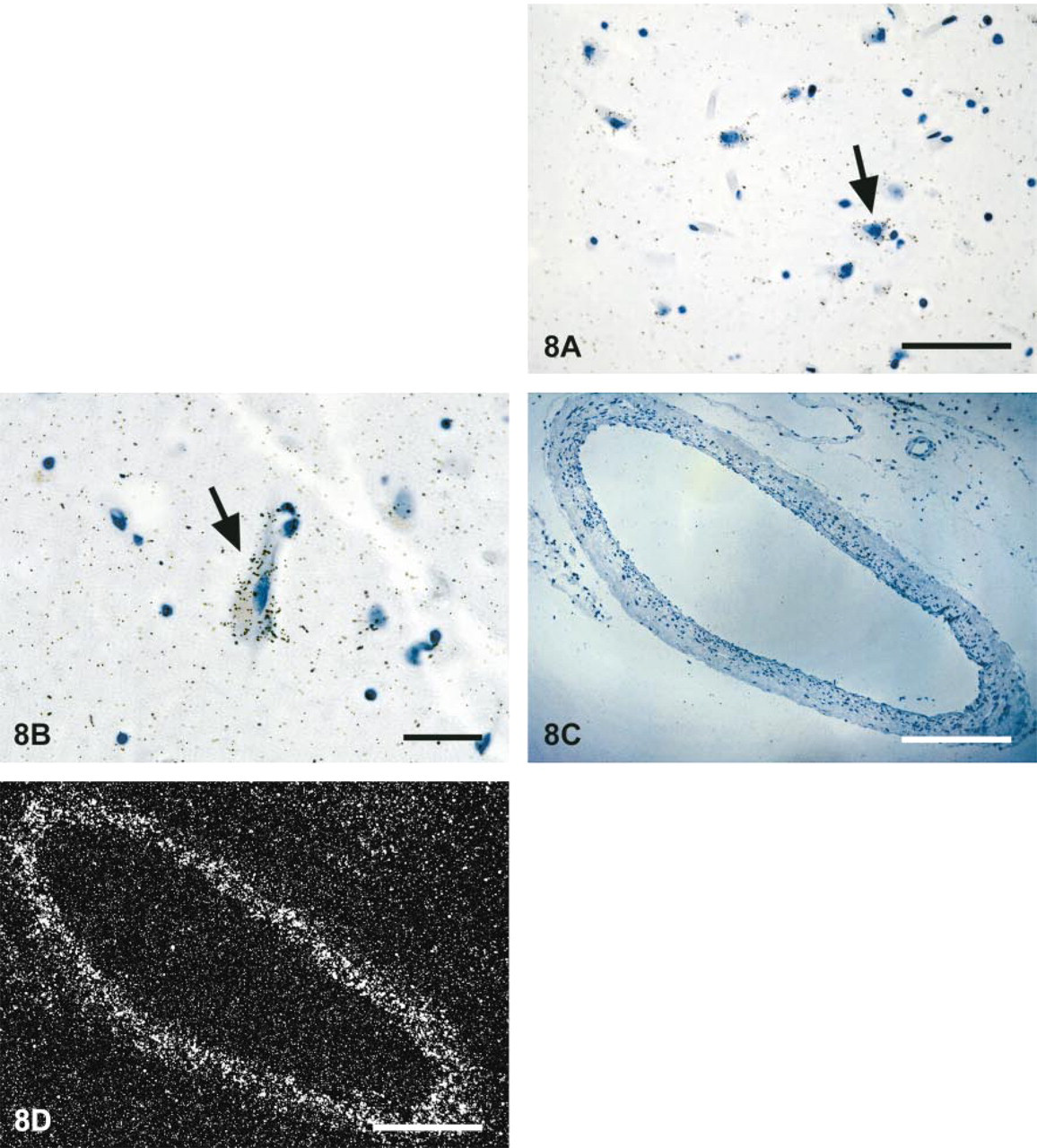
Localization of PGIS mRNA in representative human cortical sections by in situ hybridization. A 420-bp cDNA fragment of human PGIS was labeled with 35S and used for in situ hybridization of formalin-fixed sections as described in Materials and Methods. (
The results obtained in this study show a widespread expression of prostacyclin synthase in human brain tissues. Although the specific function of PGIS still remains to be established, these findings suggest that prostacyclin might play an important role in neuronal activity.
Footnotes
Acknowledgements
Acknowledgments
Supported by the Robert Bosch Foundation. We thank Dr M. Breyer for use of equipment and advice on performing in situ hybridization.
